Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The bond angle H-O-H in ice is closest to |
| Answer» Answer :D | |
| 2. |
The bond angle formed by different hybrid orbitals are in the order : |
|
Answer» ` sp^(2) GT sp^(3) gt sp^(3)` |
|
| 3. |
The bond angle between two hybrid orgials is105^(@) . Calculate the percentege of s-character of the hybrid orbital .Given that cos75^(@) = 0 .2588. |
|
Answer» Solution : ` cos alpha = - (1)/(m) ` where m = hybridisation index ` therefore - (1)/(m) cos 105^(@) = cos (180 - 75 ) = - cos ^(@) = - 0.2588 orm = (1)/(0.2588) = 3.86` % s-character ` = (1)/(1+m) xx10- = (1)/(1+3.86) XX100 = 20.58%` Alternatively , s-character decreases as the bond angle decreases. For example , `{:(" Hybrid orbital" ""sp^(3)"" sp^(2) ""sp),("s-character " ""25% ""33.3% ""50%),(" Bond angle """ 109.5^(@) "" 120^(@) ""180^(@)):}` THUS, when bond angle decreases below ` 109.5^(2)`, the s-character will dcrease accordingly. Although the decrease is not linear , even then approsimate value can be calculated as follows : Decrease in angle= `120 - 109.5^(@) = 10.5^(@)` Decrease in s-character =` 33.3 - 25 = 83 ` ACTUAL decrease in bond angle=` 109.5^(@) - 105^(@) = 4.5^(@)` ` therefore ` EXPECTED decrease in s-character `= (8.3)/(10.5) xx4.5 = 3.56%` Thus, s-character should decrease by about 3.56% HENCE, s-character= ` 25 - 3.56 = 21 .44%` |
|
| 4. |
The bond angle and dipole moment of water respectively are |
|
Answer» `109.5^(@), 1*84 ` D |
|
| 5. |
The boiling points of two miscible liquids, which do not form azeotropic mixture, are close to each other. Their separation is best carried out by : |
|
Answer» Vacuum distillation |
|
| 6. |
The boiling point of water = ............""^(@)F. |
|
Answer» 210 |
|
| 7. |
The boiling point of water in pressure cooker is |
|
Answer» `100^@C` |
|
| 8. |
The boiling point of water at (i) sea shore (ii) 5000 ft. height is 373 K and 370 K respectively ? Why ? |
| Answer» Solution :The boiling is the TEMPERATURE at which vapour pressure is EQUAL to ATMOSPHERE pressure. At HEIGHT atmosphere pressure is less so boiling point is also lower. | |
| 9. |
The boiling point of water at a pressure of 50 atm is 538 K.Compare the theoretical efficiencies of a stem engine operating between the boiling point of water at 50 atm pressure , assuming the temperature of the sink to be 35^(@)C in each case. |
|
Answer» Solution :Boiling point of water at 50 atmpressure `(T_b)=265^@C` =265+273=538 K (i)Boiling point of water at 1 atm PRESSURE `(T_b)=100^@C` =100+273=373 K % Efficiency of STEAM engine `=[(T_1-T_2)/T_1]xx100` `=[(538-373)/538]xx100` =0.3066 x 100 = 30.66% (ii) Boiling point of water at 50 atm pressure `(T_b)=35^@C` =35+273 =328 K % Efficiency of steam engine =`[(T_1-T_2)/T_1]xx100` `=[(538-328)/538]xx100=(210xx100)/538` =39.03 % |
|
| 10. |
The boiling point of water (100^(@)C) is much higher than that of HF(19.5^(@)C), even though they have similar molecular masses. Explain. |
Answer» SOLUTION :Each water MOLECULE is INVOLVED in intermolecular H-bonding with four other water molecules but each HF molecule is involved in intermolecular H-bonding with two other HF molecules. Therefore, the degree of molecular association in water is much HIGHER than that in HF. 
|
|
| 11. |
The boiling point of p-nitrophenol is higher than that of o-nitrophenol because |
|
Answer» `NO_2` group at para position behaves in a different way from that at ORTHO position |
|
| 12. |
The boiling point of H_(2)O is higher than that of H_(2)S . Explain. |
| Answer» SOLUTION :DUE to EXTENSIVE intermolecular H-bonding in `H_(2)O` , the b.p. of `H_(2)O` is much HIGHER than that of `H_(2)S`. | |
| 13. |
The boiling point of inert gases increases with the increase in atomic number because "_______________". |
|
Answer» there is ABSENCE of intermolecular FORCE of attraction between the molecules |
|
| 14. |
The boiling point of glycerol is more than propanal because of: |
|
Answer» Hybridisation |
|
| 15. |
The boiling point of glycerol is 563K. Before boiling point it is decomposed. So, which is the suitable technique for purification of it? |
|
Answer» Partitial DISTILLATION |
|
| 16. |
The boiling point of chorofrom ,CHCl_(3), is 61.7^(@)C and its enthalpy of vaporization is 31.4 KJ-mol^(-1) Calculate the molar entropy of vaporization forchlorofrom : |
|
Answer» `10.7Jxxmol^(-1).K^(-1)` |
|
| 17. |
The boiling point D_(2)O is greater than H_(2)O it if because |
|
Answer» `D_2O` has a lower ionic PRODUCT |
|
| 19. |
The boiling point of aniline is 457k. At which temperature aniline boils in simple distillation and in vepour distillation? |
| Answer» SOLUTION :In simple distillation at 457K and in vapour distillation boils at LOWER temperature 373K | |
| 20. |
The boiling point difference the two liquids is not much then..........method used to separate them |
|
Answer» SIMPLE DISTILLATION |
|
| 21. |
The Bohr's energy of a stationary state of hydrogen atom is given as E_(n)=(-2pi^(2)me^(4))/(n^(2)h^(2)). Putting the values of m and e for n^(th) energy level which is not the correct value? |
|
Answer» `E_(N)=(-21.8xx10^(-19))/(n^(2))J atom^(-1)` |
|
| 22. |
The Bohr's radius of Li^(2+) of 2^(nd) orbit is…………. |
|
Answer» Solution :0.7053A `r_n=((0.529xx2^(3)n^(2)))/(Z)A,n=2,Z=3(For Li^(2+))` `R=(0.259xx2^(2)=(0.529xx4)/(3))=0.7053A` |
|
| 23. |
The BOD values of four samples of water A,B,C and D are 165 ppm. 120ppm, 20ppm and 5ppm respectively . The most polluted and least polluted water sample are |
|
Answer» A & B |
|
| 25. |
The blue compound formed in the positive test for nitrogen with Lassaigne solution of an organic compound is |
|
Answer» `Na_(4)[FE(CN)_(5)(NOS)]` |
|
| 26. |
The blue colour of potassium solution in liquid ammonia is due to the presence of - |
|
Answer» solvated ELECTRON |
|
| 27. |
The blood red colour in the combination test of nitrogen and sulphur in organic compound is the to the formation of |
|
Answer» FERRIC THIOCYANATE |
|
| 28. |
The blood red colour in the combination test of nitrogen and sulphur in organic compound is due to the formation of: |
|
Answer» FERRIC sulpho cyanide |
|
| 29. |
The blocking of capillaries due to sudden release of bubbles ofN_2 gas in blood is known as |
|
Answer» BENDS |
|
| 30. |
The bleaching action of H_(2)O_(2) is due to the following reaction |
|
Answer» `PbS+4H_(2)O_(2) RARR PbSO_(4)+4H_(2)O` |
|
| 31. |
The bimolecular reaction is represented by : |
| Answer» Answer :D | |
| 32. |
The bicarbonate of alkali metal which does not exist in the solid state is ………… . |
| Answer» SOLUTION :`LiHCO_(3)` | |
| 33. |
the biggest particulate matter is |
| Answer» Solution :Fly ash | |
| 34. |
The best sequence to use to form propylbenzene from benzene is |
|
Answer» Friedel-Crafts ALKYLATION USING 1-chloropropane and aluminum trichloride. |
|
| 35. |
The best sequence of reactions for the following conversion is |
|
Answer» (i)1 mol `Br_2//FeBr_3 " " (II)KMnO_4,"HEAT" " " (iii)HNO_3+H_2SO_4` |
|
| 36. |
The best sequence of reactions for preparation of the following compound from benzene is |
|
Answer» (i)`CH_3`COCl/`AlCl_3` (II)OLEUM (iii)`(CH_3)_2`CH-Cl(1 mole ) /`AlCl_3` |
|
| 37. |
The best route for the preparation of BeF_(2) is : |
|
Answer» thermal decomposition of `(NH_(4))_(2)" BeF_(4)` |
|
| 38. |
The best representation of Lewis structure can be selected by using following guidelines . Which one is correct ? |
|
Answer» A structure in which all FORMAL charges are zero preferred over the ONE with charges . |
|
| 39. |
The best reagents for the ring alkylation of phenol are |
|
Answer» `C_(2)H_(5)CL` and anhy. `AlCl_(3)` |
|
| 40. |
The best reagent to convert pent-3-en-2-ol into pent-3-in-2-on is |
|
Answer» Acidic permanganate |
|
| 41. |
The best reagent to convert pent-2-en-2-ol into pen-2-ene-2-one is |
|
Answer» PYRIDINIUM chloro-chromate |
|
| 42. |
The best reagent for converting an alcohol into the corresponding chloride is |
|
Answer» `PCl_(3)` |
|
| 43. |
The best reagent for converting 2-phenylpropanmide into 2-phenylpropanamine is ………………. |
|
Answer» excess `H_(2)` |
|
| 44. |
The best reagent for converting, 2-phenylpropanamide into 1-phenylethanamine is_____________ |
|
Answer» Excess `H_(2)//PT` |
|
| 45. |
The best method to separate the mixture of ortho and para nitrophenol (1:1) is |
|
Answer» steam distillation 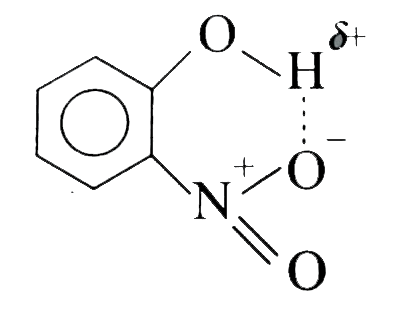 Thus, the effects of chelation are lower solubility and greater volatility in the `o-nitrophenol`, thereby enabling it to be separated from its `m-` and `p-isomers` by steam distillation. Howerver, `o-isomer` may also be separated from the `p-isomer` by crystallization or by CHROMATOGRAPHY. |
|
| 46. |
The best method for the separation of naphthalene and benzoic acid from their mixture is |
|
Answer» sublimation |
|
| 47. |
The best method for the preparation of 2,2-dimethylbutane is via the reaction of |
|
Answer» `Me_3CBr` and `MeCH_2Br` in Na/ether 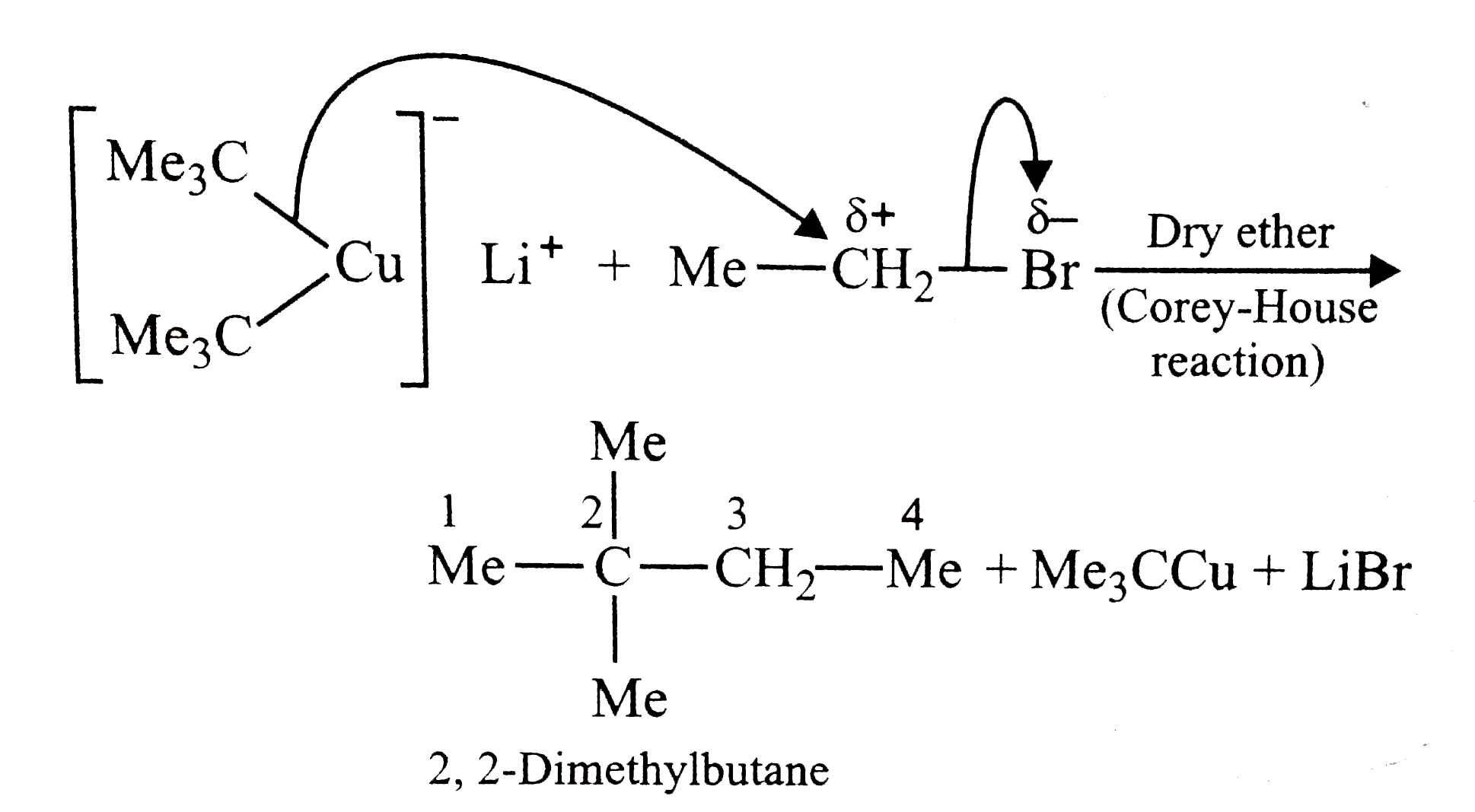
|
|
| 48. |
The best method for locating the position of double bond in an unknown alkene is ............... |
| Answer» SOLUTION :OZONOLYSIS | |
| 49. |
The best leaving group produces a _________ base that is a (n) ________ anion. |
|
Answer» WEAK, STABLE |
|