Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The bonds present in N_(2)O_(5) are |
|
Answer» IONIC |
|
| 2. |
The bond present in NaNC are |
| Answer» Answer :D | |
| 3. |
The bond order of NO_(3)^(-)ion is ………… and its shape is ………. |
| Answer» SOLUTION :1.33 PLANER TRIANGULAR | |
| 5. |
The bond order ofN_(2), N_(2)^(+) , N_(2) ^(-) andN_(2)^(2-) respectively are ……….., …………..,…………..and……………. |
| Answer» SOLUTION :3, 2.5 , 2.5 , 2 | |
| 6. |
The bond order of He^(2+) ion is- |
|
Answer» 0 |
|
| 7. |
The bond order of N-O bonds in NO_(3)^(-)ion is |
|
Answer» `0.33` 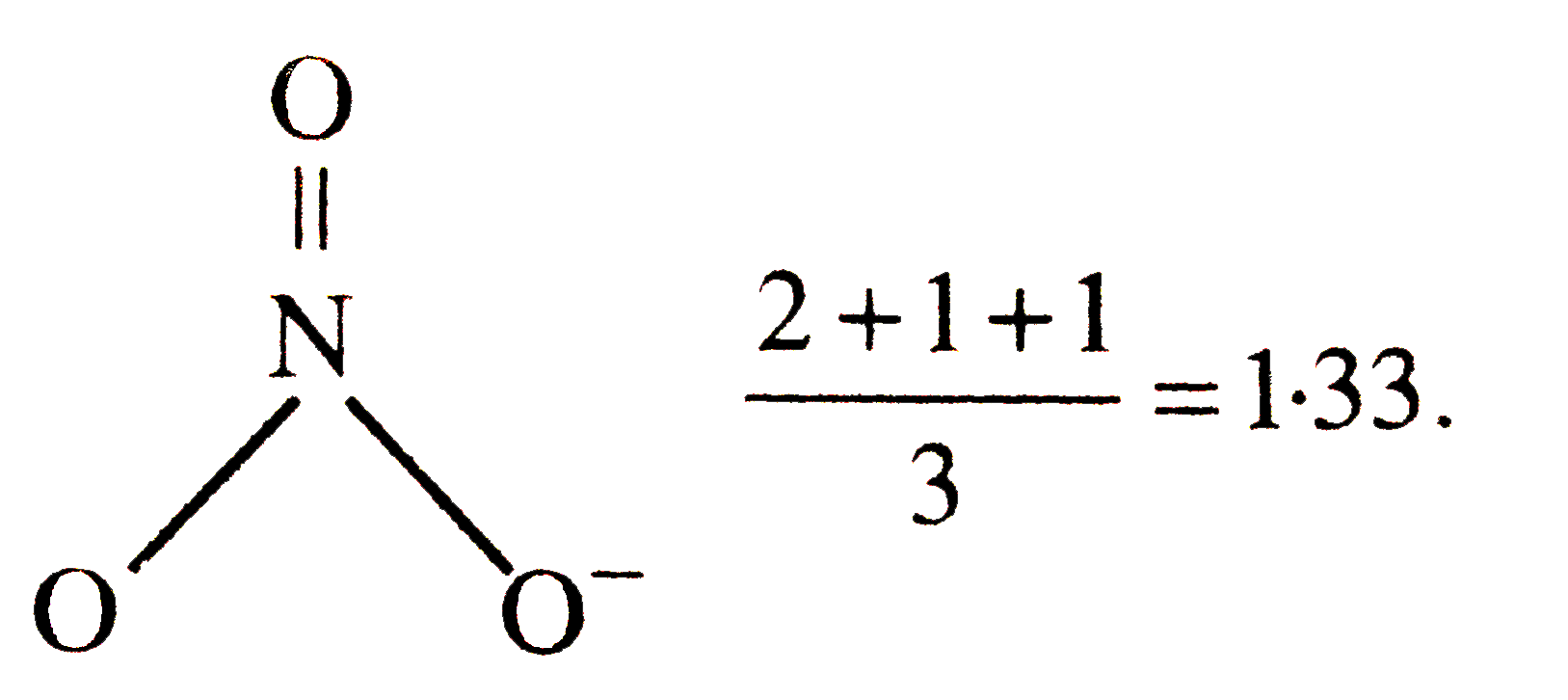 `(2 + 1 + 1 )/(3) = 1.33` . |
|
| 8. |
The bond order of individual carbon bonds in benzene is |
|
Answer» ONE |
|
| 9. |
The bond order of CO molecule on the basis of MO theory is |
|
Answer» Zero |
|
| 10. |
The bond order of a molecule is given by |
|
Answer» the DIFFERENCE between the NUMBER of ELECTRONS in BONDING and antibonding orbitals. |
|
| 11. |
The bond order in O_(2)^(2-) species is |
|
Answer» 1 |
|
| 12. |
The bond order in O_(2)^(-) species is |
|
Answer» 1 |
|
| 13. |
The bond order |
|
Answer» Can have NEGATIVE VALUE |
|
| 14. |
The bond length of two N-O bond in nitromethane is….. |
|
Answer» It is a between of SINGLE bond N-O and double bond N= O |
|
| 15. |
The bond length of H_(2) , F_(2)and HF is 74 pm, 144 pm and 92 pm respectively. Which one is most stable ? |
| Answer» SOLUTION :`H_(2)` | |
| 16. |
The bond length is affected by |
|
Answer» Hybridisation |
|
| 17. |
The bond length between central carbon atom and other carbon atom is minimun in |
|
Answer» Propene |
|
| 18. |
The bond length between C2 and C3 in acrutl aldehyde is not equal to the bond length between carbons of ethene because in acryl aldehydedouble is in conjugation , so it it shows resonance which results in an increase in the bond length betweenC2 and C3 in acryl aldehyde . Find out the correct statement (s) about the given compound ? |
|
Answer» Bond length between C2 and C3 = Bond length between C5 and C6 . |
|
| 19. |
The bond length between C2 and C3 in acrutl aldehyde is not equal to the bond length between carbons of ethene because in acryl aldehydedouble is in conjugation , so it it shows resonance which results in an increase in the bond length betweenC2 and C3 in acryl aldehyde . Which compound does not have the conjugative system to show the resonance ? |
|
Answer» `CH_(2)=CH-CH=CH-CH_(2)-CH_(3)` |
|
| 20. |
The bond length between C2 and C3 in acrutl aldehyde is not equal to the bond length between carbons of ethene because in acryl aldehydedouble is in conjugation , so it it shows resonance which results in an increase in the bond length betweenC2 and C3 in acryl aldehyde . Which compound will not show the resonance ? |
|
Answer»
|
|
| 21. |
The bond length between all the four carbon atoms is same in 1, 3 - butadine. Explain with reason. |
|
Answer» SOLUTION :`CH_(2)=CH-CH=CH_(2) harr overset(+)CH_(2)-CH=CH-overset(bar..)CH_(2)` `CH_(2)bar(...)CH bar(...)CHbar(...)CH_(2)-=overset(bar(..))CH_(2)-CH=CH-overset(+)CH_2` (Partial double bond character) `1,3 ` ,3-butadiene is a conjugated MOLECULE with four overlapping p-orbital on adjacent atoms. And A-electrons are delocalised over four atoms. This SHORTENS the bond length of central C bond Thus, the bond length between all the four C-atoms are same in 1,3-butadiene. |
|
| 22. |
Given that bond energies of N=N,H-H and N-H bonds as 945, 436 and 391 kJ/ mol respectively ,the enthalpy of the reaction N_(2)(g)+3H_(2)(g)+to2NH_(3)(g),is |
|
Answer» |
|
| 23. |
The bond enthalpies of D-D and O-O and D-O are respectively +440 ,+498 and +491.5KJ "mol"^(-1) calculation Delta H for the reaction D_(2)(g) +1/2O_(2)(g)to D_(2) O(l) |
|
Answer» SOLUTION :Heat of REACTION, =(Enthalpy of bonds DISSOCIATED )-(Enthalpy of bonds formed) `DELTA H=(440+498/2)-(2xx491.5)=689-983=-294KJ` Enthalpy of the given reaction is -294KJ. |
|
| 24. |
The bond enthalpies of D-Dand O-O and D-O are respectively, + 440, +498 and + 491.5 kJ mol^(-1). Calculate DeltaH for the reaction D_(2(g)) + 1/2 O_(2(g)) to D_2O_((l)) |
|
Answer» SOLUTION :Heat of reaction, `DeltaH `= (ENTHALPY of bonds dissoicated) - (Enthalpy of bonds FORMED) `DeltaH = (440 + 498/2) - (2 xx 491.5)` `= 689 - 983 = -294 kJ` Enthalpy of the GIVEN reaction is -294 kJ. |
|
| 25. |
The bond enthalpies of D-D and 0-0 and D - ( are respectively, + 440, + 498 and + 491.5 kJ mol^(-1). Calculate DeltaH for the reaction D _(2 (g)) + (1)/(2) O _(2(g)) to D _(2) O _((l)) |
|
Answer» SOLUTION :Heat of reaction, `DeltaH = `(Enthalpy of bonds dissociated) - (Enthalpy of bonds formed) `Delta H = (440 + (498)/(2)) - (2 xx 491 .5)` `=689-983 =-294 KJ` Enthalpy of the GIVEN reaction is `- 294 kJ .` |
|
| 26. |
The bond energy of H_(2) is foundto be 435 kJ mol^(-1). Therefore, the enthalpy of formation of hydrogen atom will be "…................" |
| Answer» SOLUTION : `435 //2 = 217.5 KJ MOL^(-1)` | |
| 27. |
The bond energy of H_(2(g)) is 436kJ/mole. The bond enthalpy of N_(2(g)) is 941.3 kJ/mole. What is the bond enthalpy of N - H if NH_3 formation energy is -46.0 kJ/mole ? |
| Answer» SOLUTION :390 kJ/mole | |
| 28. |
The bond energy of an O-H bondis 109 kcal // mol. When a mole of water is formed,then |
|
Answer» 109 kcal is released `H_(2)(g)+(1)/(2) O_(2)(g) rarr H_(2)O(L) .i.e., ( H-O-H)` `Delta_(f)H^(@) =BE(H-H) + (1)/(2) BE(O=O)-2BE(O-H)` |
|
| 29. |
The bond energy depends upon _____ |
|
Answer» 1) SIZE of the atom |
|
| 30. |
The bond energies of H - H, X - X and H - X are 104 K.Cal, 38K.Cal and 138 K.Cal respectively the electronegativity of 'X' is [sqrt(67) = 8.18] |
|
Answer» 3 |
|
| 31. |
The bond dissoication energies of X_(2), Y_92) and XY are in the ratio of 1: 0.5 : 1 . DeltaH for the formation of XY is -200 kJ mol^(-1). The bond dissociation energy ofX_(2) will be |
|
Answer» `200 kJ mol^(-1)` Given `=(1)/(2) X_(2)+(1)/(Y_(2)) rarr XY ,DELTAH = - 200 kJ mol^(-1)` `Delta_(r) H=` BE (Reactants) - BE( Products) `= [ (1)/(2) BE(X_(2))+(1)/(2) BE(Y_(2))]-BE(XY)` `:. - 200 = (a)/(2)+ ( 0.5)/( 2) -a =0.5a+ 0.25a-a =-0.25 a` `:. a=( 200)/( 0.25) = 80 kJ mol^(-1)` |
|
| 32. |
The bond dissociation of the molecules A_2 , B_2,C_2 are 498, 158, 945 kJ/ mole respectively. If so, the correct decreasing order of their bond orders is |
|
Answer» `A_2 ,B_2 ,C_2` |
|
| 33. |
The bond dissociation energy of methane and ethane are 360 kJ "mol"^(-1) and 620 kJ "mol"^(-1) respectively. Then, the bond dissociation energy of C-C bond is _____ |
|
Answer» `170 KJ "mol"^(-1)` (C-H) is equal to 90 KJ/mol |
|
| 34. |
The bond dissociation energy of B-F in BF_(3) is 646 kJ "mol"^(-1) whereas that of C-F in CF_(4)is 515 kJ "mol"^(-1).The correct reasonfor higherB-Fbond dissociationenergy as compared to thatof C-F is |
|
Answer» stronger `sigma`-bond between B and F in `BF_(3)` as compared to that between `C` and `F` in `CF_(4)` |
|
| 35. |
The bond dissociation energy needed to form the benzyl radical from toluene is................than the energy needed for the formation of methyl radical from methane. |
|
Answer» |
|
| 36. |
The bond dissociation energy of B - F in BF_(3) is 646 KJ mol^(-1) whereas that of C-F in CF_(4) is 515 kJ mol^(-1) . The correct reason for higher B - F bond dissociation energy as comparad to that of C - F is |
|
Answer» smallarsize of B-atom as compared to that of in the hybridisation can accept an electron pair from a FULL `2p_(z)` orbitals of any F-atom , formaing ` pi`-bond . Thus , B-F bondlength becomes shorter. HENCE, bond is stronger and dissociation energy is higher . |
|
| 37. |
The bond dissociation energy depends upon the nature of the bond and nature of the molecule. If any molecule more than 1 bonds of similar nature are present then the bond energy reported is the average bond energy. If enthalpy of hydrogenation of C_(6)H_(6(l)) "into" C_(6)H_(12(l)) " is " -205 kJ and resonance energy of C_(6)H_(6(l)) is -152kJ//mol then enthalpy of hydrogenation of is ? Answer Delta H_("vap") " of " C_(6)H_(6(l)), C_(6)H_(10(l)), C_(6)H_(12(l)) all are equal: |
|
Answer» `-535.5` kJ/mol `=("Heat of Hydrogenation of Benzene + Resonance ENERGY")/(3)` `= (-205 + (-152))/(3) = -199kJ` |
|
| 38. |
The bond dissociation energy depends upon the nature of the bond and nature of the molecule. If any molecule more than 1 bonds of similar nature are present then the bond energy reported is the average bond energy. Determine C-C and C-H bond enthalpy (in kJ/mol). Given: Delta_(f)H^(0) (C_(2)H_(6),g)= -85kJ//mol, Delta_(f) H^(0) (C_(3)H_(8), g)= -104kJ//mole, Delta_("sub")H^(0) (C,s)= 718kJ//mol, B.E. (H-H)= 436 kJ/mol, |
|
Answer» 414345 `3C_(("graphite")) + 4H_(2(g)) rarr C_(3)H_(8(g)), Delta H = - 104` kJ/mole …(2) `C_(("graphite")) rarr C_((g)), Delta H = 718` kJ/mole …(3) `H_(2) rarr 2H, Delta H = 436` kJ/mole ...(4) To get expression for heat of reaction in terms of Bond ENERGY we have to use the expression with all the components are in gaseous state. So, from Eq. (1) and (3) `2C_((g)) + 3H_(2(g)) rarr C_(2)H_(6(g)), Delta H = - 1521 kJ` ...(5) From Eq.(2) and (3) `3C_((g)) + 4H_(2(g)) rarr C_(3)H_(8(g)) , Delta H = - 2258kJ` ...(6) We know that heat of reaction `Sigma` B.E of reactants `= Sigma` B.E of products. So, `-1521= 3 (H- H) - [6 C-H + C-C]` ...(7) `-2258 = 4(H-H) - [8C- H + 2C -C]` ..(8) On solving the above two equations. C- H bond energy = 414 C- C bond energy = 345 |
|
| 39. |
The bond dissociation energies of X_(2), Y_(2) and XY are in the ratio of1 : 0.5 : 1. DeltaH for the formation of XY is -200 kJ mol^(-1).The bond dissociation energy of X_(2) will be |
|
Answer» `200 kJ"MOL"^(-1)` Then B.E. Of `Y_(2) = 0.5 x kJ mol^(-1)` B.E of XY = x kJ `mol^(-1)` `1/2 X_(2) + 1/2Y_(2) to XY DeltaH = -200 kJ mol^(-1)` `DeltaH = Sigma`B.E (reactants) - `Sigma`B.E(PRODUCTS) `DeltaH = 1/2 B.E.(X_(2)) + 1/2 B.E. (Y_(2)) - B.E.(XY)` ` -200 = 1/2(x) + 1/2 (0.5 x) - 1(x)` `-200 = -0.25x` `therefore x = (200)/(0.25) = 800 kJ"mol"^(-1)`. |
|
| 40. |
The bond dissociation energies of X_2, Y_2 and XY are in the ratio of 1: 0.5 : 1. Delta Hfor the formation of XY is -200 kJ "mol"^(-1) . The bond dissociation energy of X_2will be |
|
Answer» `400 kJ "mol"^(-1)` |
|
| 41. |
The bond dissociation energies of gaseous H_(2), Cl_(2) and HCl are 104, 58 and 103 kCal/mole respectively. If magnitude of heat of formation of HCl = 11x, x=? |
|
Answer» As all are in gaseous state `DELTA H = Sigma` B.E of Reactions `- Sigma` B.E. of PRODUCTS `= (1)/(2) (104) + (1)/(2) (58) - 103 = -22` |
|
| 42. |
The bond dissociation energies forCl_(2),I_(2) and ICIare 242.3,151.0 and211.3 kJ //mole respectively . The enthalpyof sublimation of iodine is62.8 kJ // mole.What is the standard enthalpy of formation ofICI(g) nearly equal to ? |
|
Answer» `- 211.3 kJ //`MOLE We are given`:` (i) `Cl_(2)(g)rarr 2Cl(g), DeltaH=242.3 kJ` (II) `I_(2)(g) rarr 2I(g),DeltaH = 151 kJ` (iii) `ICI (g) rarr (i) + Cl(g), DeltaH =211.3 kJ` (iv) `I_(2)(s) rarrI_(2)(g), DeltaH =62.8 kJ` `(1)/(2) (ii) (1)/(2) (i)- (iii) +(1)/(2) ` gives the required EQUATION with `DeltaH =(1)/(2) (151) +(1)/(2) (292.3) -211.3 + (1)/(2) (62.8) = 16.75kJ` |
|
| 43. |
The bond dissociatio of the molecules A_(2),B_(2),C_(2) are 498, 158, 945 kJ/mole respectively. If So, the correct decreasing order of their bond orders is |
|
Answer» `A_(2),B_(2),C_(2)` |
|
| 44. |
The bond between two identical non-metal atoms has a pair of electrons |
|
Answer» unequally SHARED between the TWO |
|
| 45. |
The bond between chlorine and bromine in BrCl is |
|
Answer» ionic |
|
| 46. |
The bond between carbon atom (1) and carbon atom (2) in the compound N -= overset(1)(C )-overset(2)(C )H= overset(3)(C )H_(2) invoves the hybrids as….. |
|
Answer» `SP and sp^(2)` |
|
| 47. |
The bond between carbon atom (1) and carbon atom (2) in compoundN-=overset(1)C-overset(2)CH=CH_2involves the hybridization 1) spa and sp? |
|
Answer» `SP^(2)" and "sp^(2)` |
|
| 48. |
The bond between B and C will be |
|
Answer» Ionic |
|
| 49. |
The bond between B and C will be ... |
|
Answer» ionic The bond between B and C will be covalent. Both Band Care non-metal atoms. B represents PHOSPHORUS and C represent chlorine. |
|
| 50. |
The bond angle in H_2 O is 105^@but in H_2 S is 92^@ . Explain the difference. |
|
Answer» Solution :The larger size of the S atom compared with O atom, minimises the elcectron REPULSIONS and allows the BONDS in `H_(2)S` to be more purely p-type. Moreover O in `H_(2)O` is `sp^(3)` hybridised but pure p-orbitals are USED by S of `H_(2)S` |
|






