Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The compound in which C uses only sp^(3) hybrid orbitals for bond formation is |
|
Answer» HCOOH |
|
| 2. |
The compound having the highest melting point and lowest boiling point is a) n-Hexane |
|
Answer» n-Hexane |
|
| 3. |
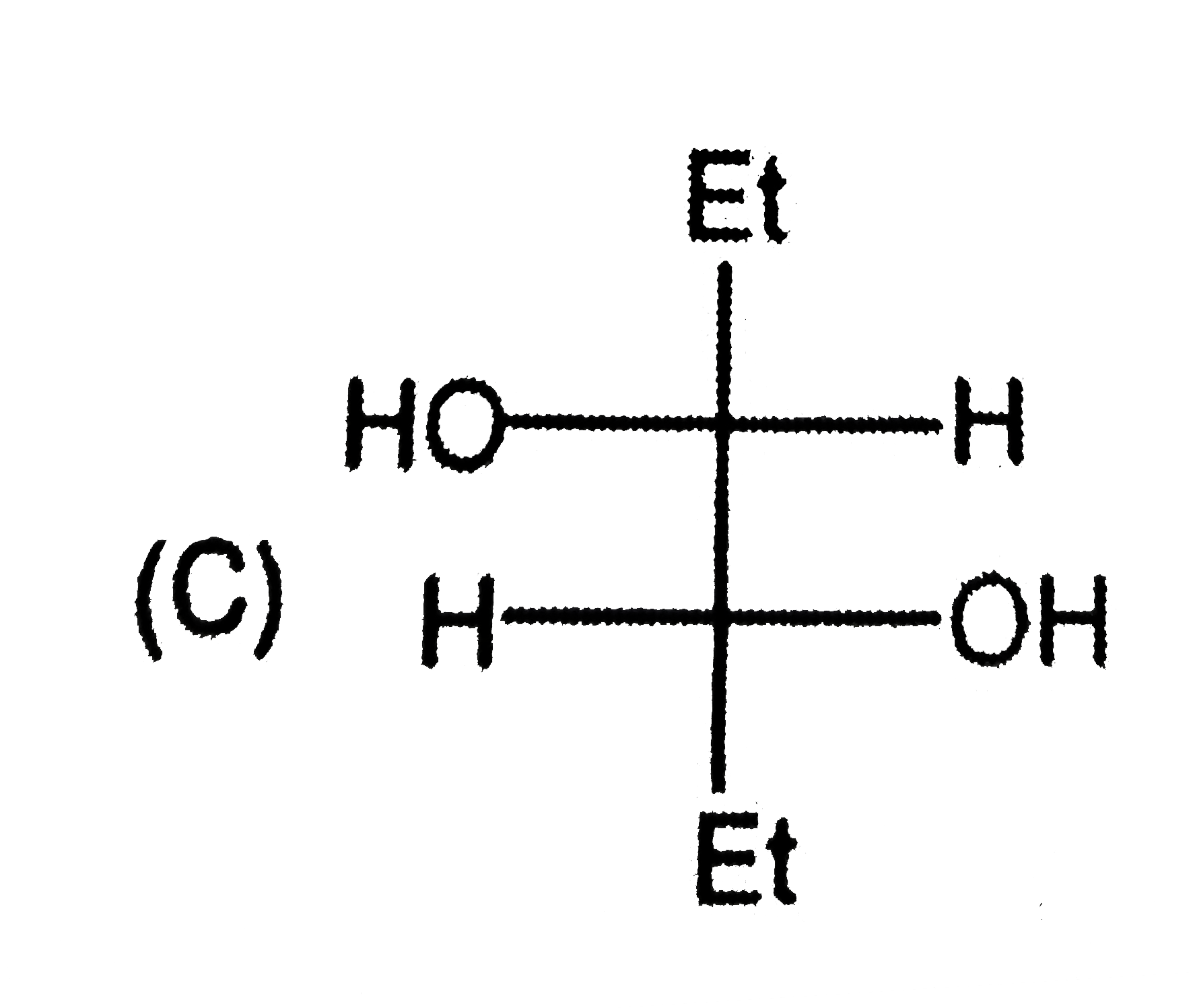
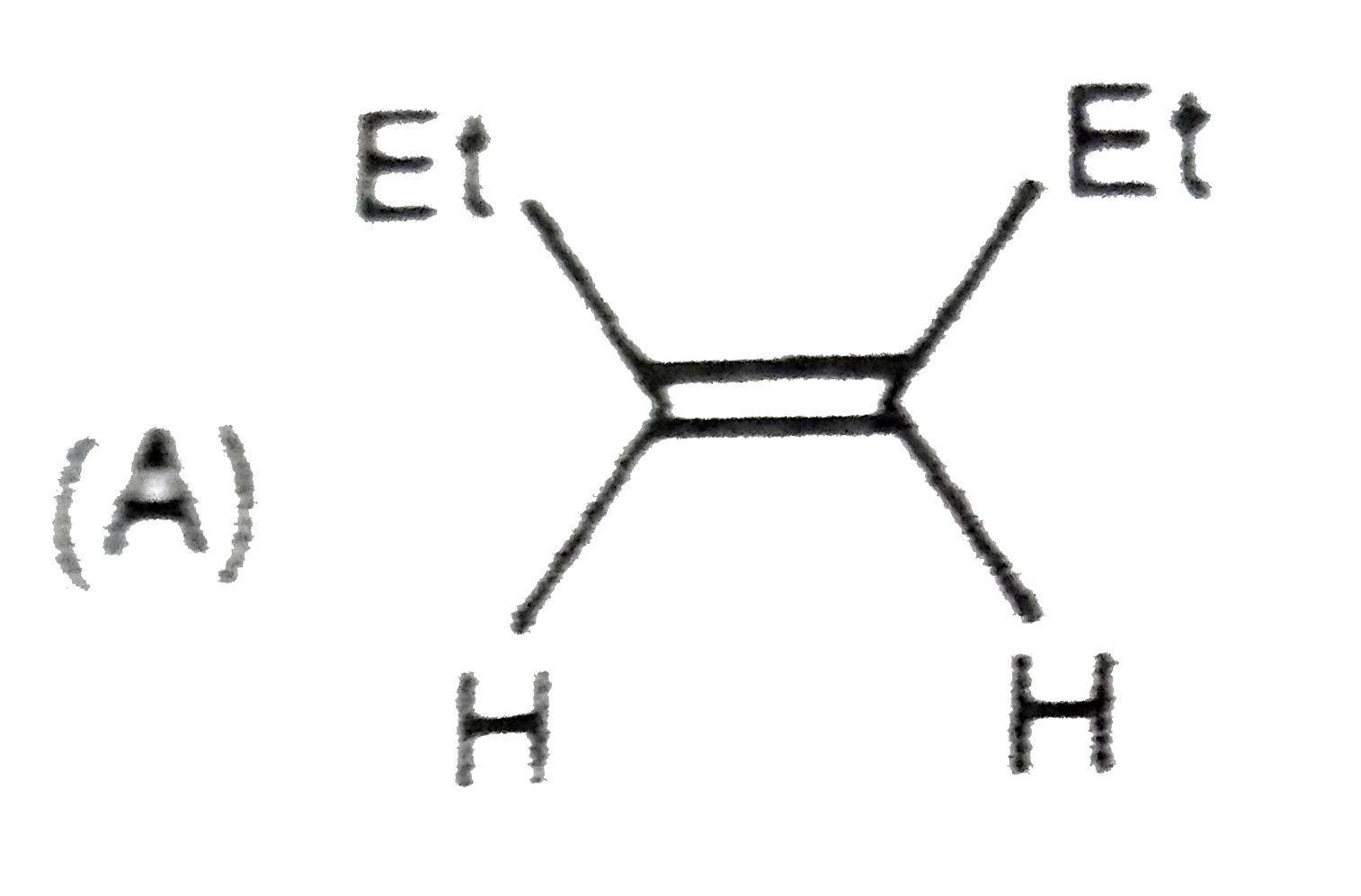
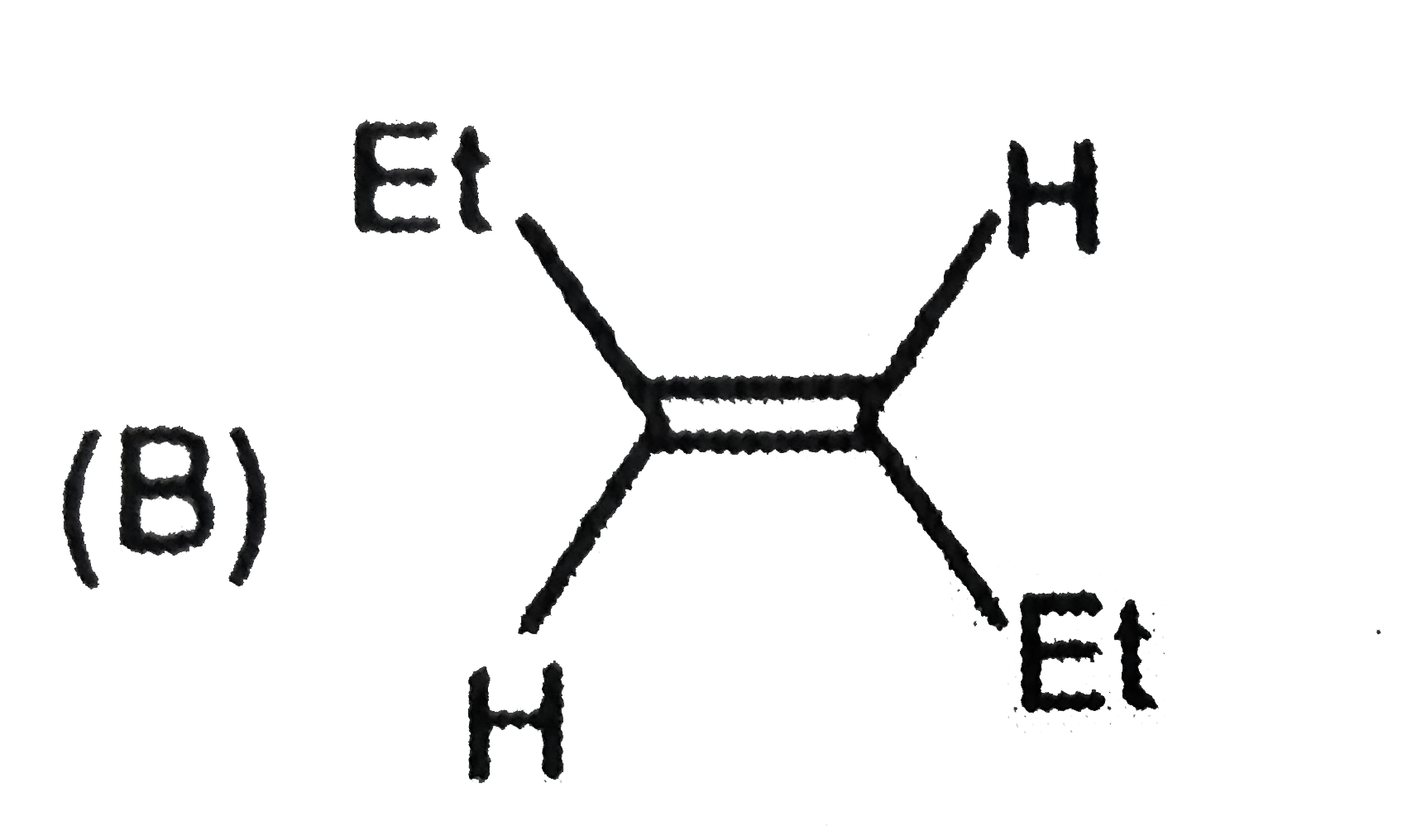
The compound having longest C-Cl bond is |
|
Answer»

|
|
| 4. |
Which of the following has highest dipole moment? |
|
Answer» `UNDERSET(CH_3) underset(|) OVERSET(Cl) overset(|)C= underset(Cl) underset(|) overset(CH_3) overset(|)C` |
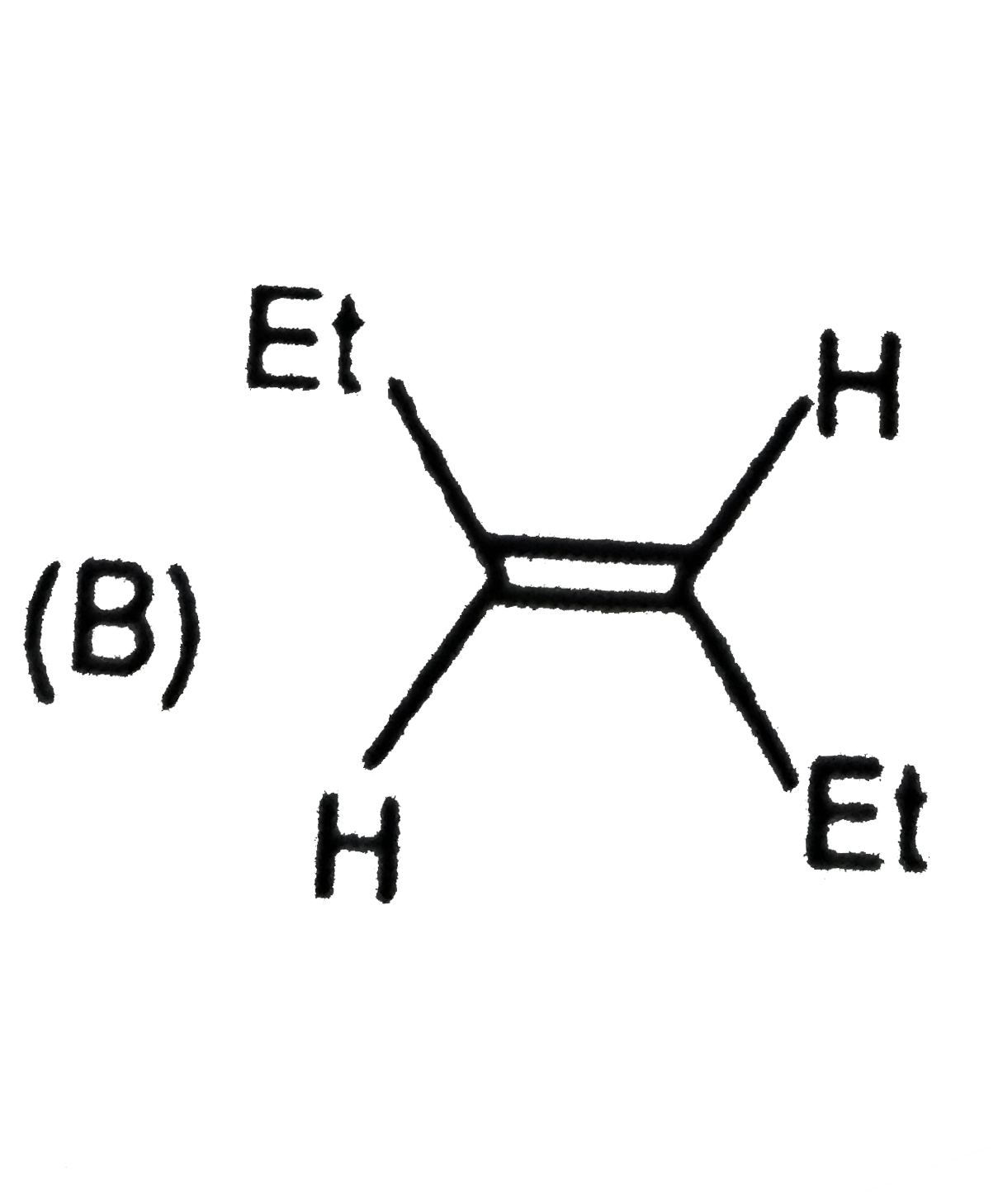
|
| 5. |
The compound having both sp and sp^(2) hybridised carbon atoms is…… |
|
Answer» propene |
|
| 7. |
The compound formed when heavy water reacts with CaC_2 is |
|
Answer» acetylene |
|
| 8. |
The compound formed when heavy water reacts with Ca_3P_2is 'X'. Its molecular mass is |
|
Answer» 34 |
|
| 9. |
The compound formed when enamel on the surface of the teeth reacts with Fions |
|
Answer» `CaF_(2)` |
|
| 10. |
The compound formed in the brown ring test has the formula [Fe(H_(2)O)_(5)NO]SO_(4). The oxidation state of iron in it is |
|
Answer» `+1` `H_2O` is NEUTRAL ligand . |
|
| 11. |
The compound formed in the BoraxBead Test of Cu^(2+) ions in the oxidising flame is: |
|
Answer» CU |
|
| 13. |
The compound formed as a result of oxidation of ethyl benzene by KMnO_(4) is |
| Answer» Answer :D | |
| 14. |
The compound 'D' is used as nitrogeneous fertilizer. |
|
Answer» Urea |
|
| 15. |
The compound(E) is : |
|
Answer» TWO MOLES of |
|
| 17. |
The compound CuCl has ZnS stucture and the edge length of its unit cell is 500 pm. Calculate its density ( Atomic masses : Cu = 63 , Cl= 35.5 , Avogadro 's constant =6.02 xx 10^(23)"mol"^(-1) ? |
|
Answer» |
|
| 18. |
The compound CuCl has ZnS structure and the edge length of its unit cell is 500 pm. Calculate its density Atomic masses Cu=63, CI=35.5. Avogadro's constant =6.02 xx 10^23 mol^(-1)) |
|
Answer» |
|
| 19. |
The compound CuCl has Zns (cubic) structure. Its density is 3.4 cm^(-3) What is the length of the edge of the unit cell ? (At masses Cu =63.5, Cl=35.5). |
|
Answer» |
|
| 20. |
The compound CuCl has ZnS (cubic) Structure. Its density is3.4 g cm^(-3) , what is the length of the edge of the unit cell ?(At masses Cu= 63. 5, Cl = 35.5) |
|
Answer» |
|
| 21. |
The compound CsAuCl_3 is found to be diamagnetic. Gold (Au) in this compound is in (there could be a covalent bond between two gold atoms) |
|
Answer» AU oxidation state only |
|
| 22. |
The compound, CH_(3)CH_(2)-overset(CH_(3))overset("|")"C "=CH-CH_(3) on reaction with NaIO_(4) in the presence of KMnO_(4) gives |
|
Answer» `CH_(3)COCH_(3)+CH_(3)CHO` 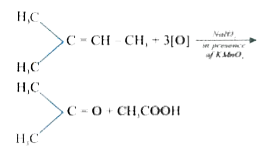
|
|
| 23. |
The compound CH_(3)-underset(CH_(2)-CH_(3))underset("|")(""C=CH-CH_(3)) is |
|
Answer» 2-ethyl-2-butene |
|
| 24. |
The compound , , may be named as : |
|
Answer» 2-ethyl-2-methyl oxirane |
|
| 25. |
The compound C_7H_8undergoes the following reactions :C_7H_8 overset(3Cl_2//Delta)(to) A overset(Br_2//Fe)(to) B overset(Zn//HCl)(to)C The product C is: 1) m- bromotoluene2) o-bromotoluene3)3-bromo-2, 4, 6-trichlorotoluene4) p-bromotoluene |
|
Answer» m-bromotoluene |
|
| 26. |
The compound C_(4)H_(10)O_(4) reacts with acetic anhydride to give a derivative with the formula C_(12)H_(18)O_(8). How many (OH) groups are present in the original substance |
|
Answer» 2 `= 4 XX 12 + 10 xx 4 xx 6 = 122` M. mass of polyacetate `=12 xx 12 + 18 xx 16 = 290` `:.` No. of OH GROUP `= (290-122)/(42) = 4` |
|
| 27. |
The compound C_(4)H_(10)O can show…… |
|
Answer» metamerism (ii) `underset("ethoxy ETHANE")(CH_(3)-CH_(2)-O-CH_(2)-CH_(3))` (III) `underset("methyl isopropyl ether")((CH_(3))_(2)-CH-O-CH_(3))` (iv) `underset("1-butanol")(CH_(3)-CH_(2)-CH_(2)-CH_(2)-OH)` (v) `underset("2-butanol")(CH_(3)-CH_(2)-underset(underset(OH)(|))(C H)-CH_(3))` (vi) `underset("2-methyl-propanol")(CH_(3)-underset(underset(CH_(3))(|))(CH)-CH_(2)-OH)` (vii) `underset("2-methyl 2-propanol")(CH_(3)-underset(underset(OH)(|))overset(overset(CH_(3))(|))(C )-CH_(3))` (i), (ii), (iii) metamerism (iv), (v), (vi), (vii) are position isomers (i), (ii), (iii) and (iv), (v), (vi), (vii) are functional group isomers |
|
| 28. |
The compound 'C' is used to make urea and Thio urea. |
| Answer» Solution :`CaNCN+H_(2)SO_(4)tounderset(C )(NH_(2)CN+CaSO_(4)` | |
| 30. |
The compound Ba_(2)Cu_(3)O_(7), which shows super conductivity, has copper in oxidation state ......... Assume that the rare earth element yttrium is in its usual +3 oxidation state. |
| Answer» SOLUTION :`+(7//3)` | |
| 31. |
The compound 'B' is having oxidation state of: |
|
Answer» zero |
|
| 32. |
The compound B formed in the following sequence of reaction CH_(3)CH_(2)CH_(2)OH overset(PCl_(5))rarrA overset(alc.KOH)rarrB is |
|
Answer» Propyne |
|
| 33. |
The compound AgF_(2) is ustable however if formed the comound acts as a very strong oxidising agents why ? |
|
Answer» Solution :In `AgF_(2)` oxidtion state of Ag is +2 which is highly UNSTABLE THEREFORE it quickly ACCEPTS an electrons form the more STABLE +1 OXIDATION state`Ag^(2+)+E^(-)rarr Ag^(+)` therefore `AgF_(2)` if formed will act as a strongoxidising agent |
|
| 34. |
The compound AgF_(2) is unstable compound. However, if formed, the compound acts as a very strong oxidising agent. Why? |
|
Answer» Solution :In `AgF_(2)`, OXIDATION number of Ag is +2 which is very unstable. Therefore it rapidly gains electron and COMES into +1 oxidation state. `Ag^(+2)+e^(-)TOAG^(+)` Therefore, `AgF_(2)` is strong OXIDIZING agnet. |
|
| 35. |
The compound AgF_(2) is unstable compound. However, if formed, the compound acts as a very strong oxidising agent. Why ? |
|
Answer» Solution :In `AgF_(2)` the O.S. of AG is +2 which is a HIGHLY unstable STATE of silver. Therefore, it quickly undergoes reduction by taking an electron and attains a stable O.S. of +1. `underset(("unstable"))underset(O.S.=+2)(Ag^(2+))+e^(-)tounderset(("Stable"))underset(O.S.=+1)(Ag^(+))` This is why `AgF_(2)`, if formed, ACTS as a very STRONG oxidizing agent. |
|
| 36. |
The compound AgF_2 is unstable. However, if formed, the compound acts as a very strong oxiding agent. Why? |
| Answer» Solution :The O.N of silver is +2 in `AgF_2` which is quit UNSTABLE. It readily ACCEPTS an electron to form stable +1 state. Hence good o0XIdising AGENT. | |
| 37. |
The compound 'A' when treated with methyl alcohols and few drops of H_(2)SO_(4) give smell of wintegreen. The compound 'A' is |
|
Answer» Succinic ACID 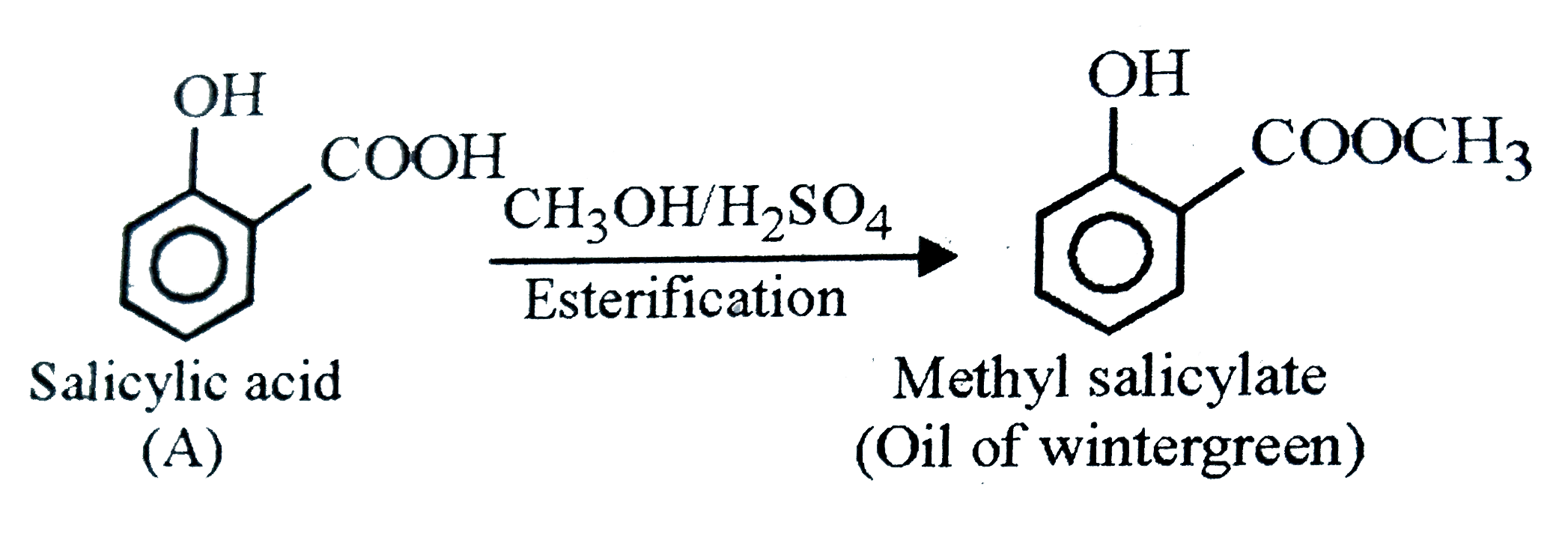
|
|
| 38. |
The compound 'A' is a colourless solid. It gives following tests: Test-I: The addition of compound 'A' solution to a solution of KI, followed by acidification with dilute H_(2)SO_(4), results in the formation of brown coloured compound 'B'. Test-II: The acidified KMnO_(4) solution is decolourized, by a solution of compound 'A', but no gas is evolved. Compound 'A' and 'B' may be: |
|
Answer» `Na_(2)S_(2)O_(3)` and S |
|
| 39. |
The compound 'A' is a colourless solid. It gives following tests: Test-I: The addition of compound 'A' solution to a solution of KI, followed by acidification with dilute H_(2)SO_(4), results in the formation of brown coloured compound 'B'. Test-II: The acidified KMnO_(4) solution is decolourized, by a solution of compound 'A', but no gas is evolved. Which of the following reaction(s) is/are correctly matched about Test-I and Test-II? |
|
Answer» `2NaNO_(2)+3KI +2H_(2)SO_(4) rarr KI_(3) +2NO +2H_(2)O +2SO_(4)^(2-)+2K^(+) +2Na^(+)` |
|
| 40. |
The compound 1, 2-butadiene has …. Hybridised carbon atoms |
|
Answer» SP |
|
| 41. |
The compouhnd formed when alcoholic solution of ethylene dibromide is heated with granualted zinc is |
|
Answer» ethene |
|
| 42. |
The composition ofthe equilibrium mixture (Cl_(2)hArr2Cl), which is attained at 1200^(@)C is determined by measuring the rate of effusion through a pin-hole. It is observed that at 1.80 mm Hg pressure, the mixture effuses 1.16 times as fast as Krypton effuses under the same conditions. Calculate the fraction of chlorine molecules dissociated into atoms (Atomic weight of Kr=84) |
|
Answer» Solution :`(r_("mix"))/(r_(Kr))=sqrt((M_(Kr))/(M_("mix"))),` i.e., `1.16=sqrt((84)/(M_("mix")))"or" M_("mix")=62.43` If x moles of `Cl_(2)` dissociate at equilibrium, then `{:(,Cl_(2),hArr,2Cl),("At.eqm".,1-x,,2x):}` Average , molecular MASS of the MIXTURE`=((1-x)xx71+2x xx35.5)/((1-x)+2x)=(71)/(1+x)` `:.(71)/(1+x)=62.43` WHICHGIVES x=0.137. Hence, fraction dissociated=0.137 |
|
| 43. |
The composition of mica is |
|
Answer» `NaAlSiO_4.3H_(2)O` |
|
| 44. |
The composition of LPG is butane and isobutane. The amount of oxygen that would be required for combustion of 1 kg of LPG will be approximately. |
|
Answer» 4.5kg |
|
| 45. |
The composition of magnalium Alloy is |
|
Answer» `Mg(1-15%) , Al (85-99%)` |
|
| 46. |
The % composition of four hydro carbons is as follows: {:((i),(ii) ,(iii),(iv),),(75,80,85.7,91.3,%C),(2520,14.3,8.7,%H,):} The date illustrates the law of |
|
Answer» CONSTANT proportion |
|
| 47. |
The composition of compound A is 40% X and 60Y. The composition of compound B is 25% X and 75%Y. According to the law of multiple Proportions the ratio of the weight of element Y in compounds A and B is: |
|
Answer» `1:2` |
|
| 48. |
The composition of compound A is 40% X and 60%Y. The composition of compound B is 25% X and 75%Y. According to the law of multiple Proportions the ratio of the weight of element Y in compounds A and B is |
|
Answer» `1:2` |
|
| 49. |
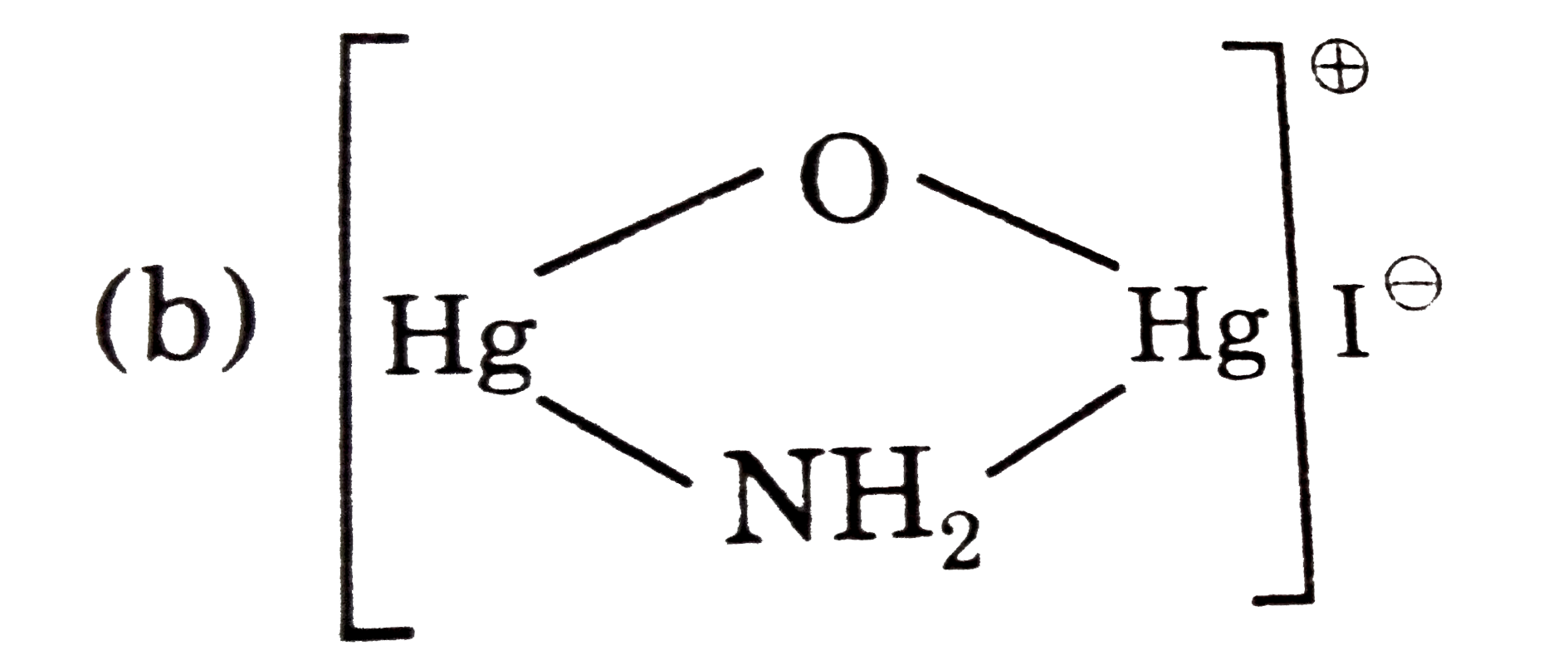
The composition of C is/are: |
|
Answer» `HgO.HgNH_(2)I` (C) `rarr HgO.Hg(NH_(2))I` (D) `rarr CrO_(2)CI_(2)`, (E) `rarr Na_(2)CrO_(4)` |
|
| 50. |
The composition of a sample of wustite is Fe_(0.93)O_(1.00). What percentage of the iron is present in the form of Fe(III) ? |
|
Answer» SOLUTION :The composition is `Fe_0.93 O_1.00` instead and FeO because some `Fe^(2+)` ions have been REPLACED by `Fe^(+)` ions. Let us FIRST calculate the number of `Fe^(2+)` and `Fe^(3+)` ions present. The formula `Fe_0.93 O_1.00` implies that 93 Fe ATOMS are combined with 100 O-atoms. Out of 93 Fe atoms , suppoe Fe atoms present as `Fe^(3+)=x`. Then `Fe^(2+)=93-x`.As the compound is neutral, total charge on `Fe^(2+)` and `Fe^(3+)` ions =total charge on `O^(2-)` ions. Thus, `3 xx x +2 (93-x)=2 xx 100` or 3x+ 186-2x =200 or x=14 , i.e., `Fe^(3+)=14` Hence, `Fe^(2+)` =93-14=79 Thus, out of 93 Fe atoms , Fe present as `Fe^(3+)`=14 `therefore` % age of Fe present of Fe (III) =`14/93xx100=15%` |
|