Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The reduction of ester with sodium and alcohol to form alcohols is called |
|
Answer» Rosenmaund reduction |
|
| 2. |
The solubility on any substance in water may be classified as : Molecular ability: AB(s/l/g) overset(aq)iffAB(aq) Ionic solubility : AB (s/l/g) overset(aq)iff A^(+)(aq)+B^(-)(aq) For a substance AB(s) the following thermodynamic informations are available at 300K: Molecular solubility: Delta H^(@)=+20kcal/mol, DeltaS^(@)=+40kcal/K-mole Ionic solubility: DeltaH^(@)=-25 kcal/ mol, DeltaS^(@)=-50 cal/K-mole When thetemperature of aqueous solution AB(s) is increased from 00K then the extent of : |
|
Answer» MOLECULAR as WELL as IONIC SOLUBILITY increases |
|
| 3. |
What transition of Li^(2+) spectrum will have the same wavelength asthat of the second line of Balmer series in He^(+) spectrum ? |
|
Answer» |
|
| 4. |
The solubility on any substance in water may be classified as : Molecular ability: AB(s/l/g) overset(aq)iffAB(aq) Ionic solubility : AB (s/l/g) overset(aq)iff A^(+)(aq)+B^(-)(aq) For a substance AB(s) the following thermodynamic informations are available at 300K: Molecular solubility: Delta H^(@)=+20kcal/mol, DeltaS^(@)=+40kcal/K-mole Ionic solubility: DeltaH^(@)=-25 kcal/ mol, DeltaS^(@)=-50 cal/K-mole Which of the following statements is correct for the solubillity of AB(s) in water at 300K? |
|
Answer» It UNDERGOES MOLECULAR solubility. |
|
| 5. |
Write the electronic configuration of Hydrogen molecule. Calculate its bond order and mention its magnetic property. |
|
Answer» Solution :Electronic CONFIGURATION `-SIGMA 1 S^(2)` BOND ORDER `=(2-0)/2=1` Diamagnetic PROPERTY. |
|
| 7. |
When two or more isotopic atoms are connected to the asymmetric chiral carbon atom of the same element, the priority sequence is : |
|
Answer» ISOTOPE of higher MASS preceeds over the lower |
|
| 8. |
Which of the statement are true? |
|
Answer» Law of constant compositon is true for all types of compounds. b. At 1 atm, `25^(@)C` molar volume `= 22.7 L` c. `Mw = 2 xx VD`, Due ot EXISTANCE of isotopes. |
|
| 9. |
Which of the following compounds is not chiral ? |
|
Answer» 1-Chloropentane 
|
|
| 10. |
What is the inert pair effect ? |
|
Answer» Solution :In the elements of 4th, 5TH, and 6th periodof the p-block elements which COME after d-block elements, the electrons present in the interveningd- and f-orbitals do not shield the s-electron of the valence shell effecitively. As a result, `ns^(2)`-electrons remains more tightly held by the nucleusand hence do not participate in bonding. This is called INERT pair effect. |
|
| 11. |
Weight of H_(2)O_(2) present in 1000ml of 2.24 vol H_(2)O_(2) is |
|
Answer» `3.2` G |
|
| 12. |
The variation of the boiling points of the hydrogen halides is in the order HPgt HI gt HBr gt HCl. What explains the higher boiling point of hydrogen fluoride? |
|
Answer» The bond energy of HF molecules is greater than in other hydrogen halides. Due to strong H-bonding HF molecule, boiling point is highest for HF :`HF gt HI gt HBR gt HI` |
|
| 14. |
Which of the following species is expected to have maximum enthalpy in an electrophilic aromatic substitution reaction ? |
|
Answer» SPECIES (II) |
|
| 15. |
Which of the following is a linear equations? |
|
Answer» `I_(3)^(-)` |
|
| 16. |
The sulphide ore which is red in colour is |
| Answer» Answer :D | |
| 18. |
Which one of the following has linear shape? |
|
Answer» `PbCl_(2)` |
|
| 19. |
The standard electrode potential corresponding to the reaction Au^(3+)(aq)+3e^(-)rarrAu(s) is 1.50 V predict if gold can be dissolved in 1M HCI solutoin and on passing hydrogen gas through god salt solution metallic gold will be precipitated or not |
|
Answer» Solution :Consider the half reaction `2H^(+)(aq)+32e^(-)rarrH_(2)(g),E^(@)=0.0 V` `AU^(3+)(aq)+3e^(-)rarrAu(s),E^(@)=1.50 V` since `E^(@)` for `Au^(3+)//Au` is higher than that `H^(+)//1/2 H_(2)(0.0 v)` therefore `Au^(3+)` can be more EASILY reduced than `H^(+)` ion this implies that `Au^(3+)` IONS can be reduced to metallic gold by `H_(2)` gas but `H^(+)` ions cannot oxidise metallic gold to `Au^(3+)` ionsin other words metallic gold does not dissolve in 1 M HCI INSTEAD `H_(2)` gas can reduce gold salt to metallic gold |
|
| 20. |
What is the product of each reaction |
|
Answer» (i) `Ph-CH_(2)-OH` (II) `(##RES_CHM_ROHR_E01_005_A01##)` |
|
| 21. |
Which of the following statements is incorrect ? |
|
Answer» In equilibrium mixture of ice and water KEPT in perfectly insulated flask, mass of ice and water does not change with time. |
|
| 22. |
What is the valency and oxidation number of nitrogen in nitrogen pentoxide ? |
| Answer» SOLUTION :BASED on the oxide theory, valency of N in `N_2O_5` is 5 (But the actual valency of N in `N_2O_5` is the number of bonds of formed by N=4) OXIDATION number of N in `N_(2)O_(5) = 5` | |
| 23. |
Which of the following compounds shall not produce propene by reaction with HBr followed by elimination reaction ? |
|
Answer»
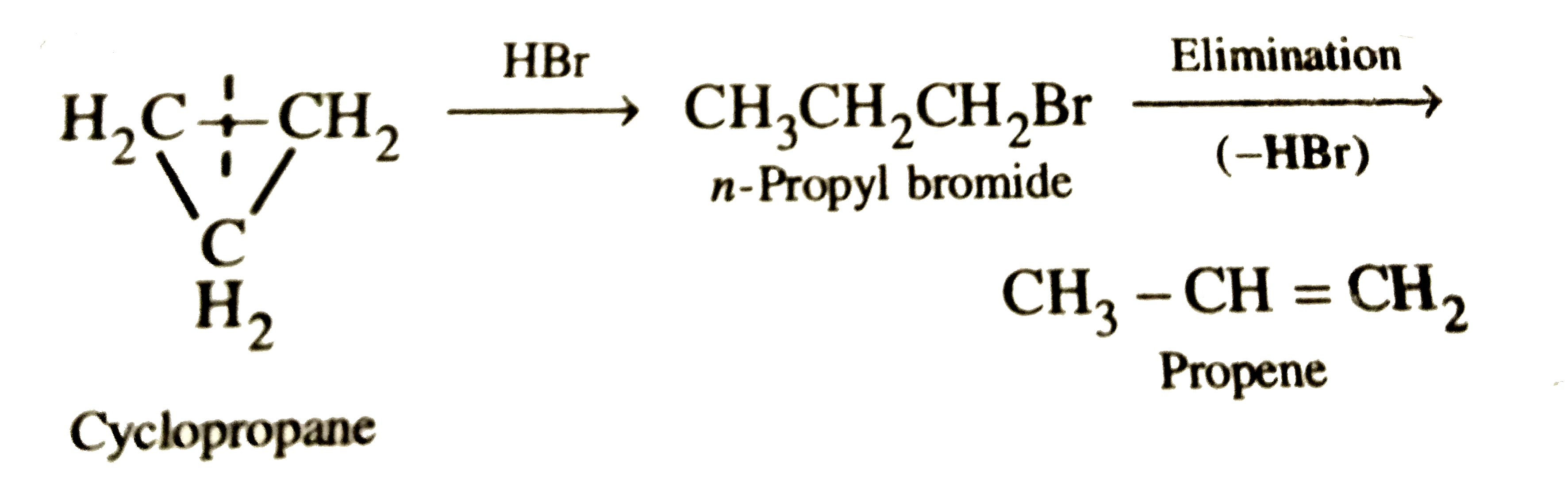 (b)`UNDERSET"n-Propyl alcohol"(CH_3-CH_2-CH_2OH)underset(-H_2O)overset"HBr"to underset"n-Propyl bromide"(CH_3CH_2CH_2Br) underset"(-HBr)"overset"Elimination"to underset"Propene"(CH_3-CH=CH_2` (d)`CH_3CH_2CH_2Br` gives `CH_3CH=CH_2`. Thus, `H_2C=C=O` does not give `CH_3CH=CH_2` . i.e., OPTION (c ) is CORRECT. |
|
| 24. |
What is IUPAC (modern) and common method for nomenclature of organic compounds? |
|
Answer» Solution :(A) Nomenclature by IUPAC method: Organic chemistry deals with MILLIONS of compounds. In order to clearly identify them, a systematic method of naming has been developd and is known as the IUPAC system of nomenclature. IUPAC means International Union of Pure and Applied Chemistry. Some time IUPAC name are lengthy and complicated so common name are used. In this systematic nomenclature, the names are correlated with the structure such that the reader or listener can deduce the structure from the name. (B) Trivial nomenclature method: Organic compounds were ASSIGNED names based on their origin or certain properties. e.g. Citric acid is named so because it is FOUND in citrus fruits. The acid found in red ant is named formic acid snce the Latin word for ant is formica. These names are traditional and are CONSIDERED as trivial or common names. Some common names are followed even today. For example, Buckminster fullerene is a common name given to the newly discovered `C_(60)` cluster (a form of carbon) noting its structural similarity to the geodesic domes popularised by the famous architect R. Buckminster FULLER (C ) Common or Trivial Names of Some organic Compounds: 
|
|
| 25. |
Which of the following reagents can be used to differentiate 1^(@) and 3^(@) alcohols ? |
|
Answer» pcc<BR>`K_(2)Cr_(2)O_(7)//H^(o+)` |
|
| 26. |
Which of the following constitute irreversible colloidal system in water as dispersion medium? |
| Answer» Answer :D | |
| 28. |
What is the total number of sigma and pi bonds in the following molecules ?(a)C_(2) H_(3) Cl(b) CH_(2) Cl_(2) (c) H_(3) C - overset(H)overset(|)C = overset(H)overset(|)C - C - H |
|
Answer» Solution : (a) is`(c) H - overset(H)overset(|)C = overset(H)overset(|)C - CL`(5 ` sigma ` bonds ,one `PI` bond ) (B) is `H- overset(H)overset(|)underset(Cl)underset(|)C - Cl` (only 4 ` sigma ` bonds ) (c)is `H- overset(H)overset(|)underset(H)underset(|)C -overset(H)overset(|)C -=overset(H)overset(|)C- C-=C - H`(10 `sigma` bonds ,`3 p i` bonds) |
|
| 29. |
What is the effect of added inert gas on the reaction at equilibrium at constant volume. |
| Answer» Solution :When an inert gas (i.e, a gas which does not REACT with any other species involved in equilibrium) is added to an equilibrium system at constant VOLUME, the total number of moles of gases present in the container increases, that is, the total pressure of gases increases =. The partial pressure of the reactants and the products or the molar concentration of the substance involved in the REACTION REMAINS unchanged. Hence at constant volume, ADDITION of inert gas has no effect on equilibrium. | |
| 30. |
Which of the following compounds is Z-isomer |
|
Answer»
|
|
| 31. |
The values of X and Y in the following redox reaction are XCl_(2) +6OH rarr ClO_3^(-) +YCl^(-) +3H_(2)O |
|
Answer» `X = 2, Y= 4` |
|
| 32. |
What is conjugate system ? Give examples and its effect |
| Answer» Solution :Conjugate system: The presence of ALTERNATE SINGLE and double bonds in an open chain or cyclic system is termed as a conjugated system. E.g. 1, 3-butadiene, aniline and NITROBENZENE etc. contain conjugate SYSTEMS. In such system the `pi`-ELECTRONS are delocalised and the system develops polarity. | |
| 33. |
Which set of conditions represents easiest way to liquefy a gas ? |
|
Answer» LOW TEMPERATURE and HIGH PRESSURE |
|
| 34. |
Which are correct statements for Be and Al ? |
|
Answer» Both are RENDERED passive by conc. `HNO_(3)` |
|
| 35. |
Which of the following hydrogen halide is liquidat room temperature ? |
|
Answer» HF |
|
| 36. |
When 1-pentyne (A) is treated with 4N alcoholic KOH at 175^@C, it is converted slowly into an equilibrium mixture of 1.3% 1-pentyne(A), 95.2% 2-pentyne(B) and 3.5% of 1,2 pentadiene (C) the equilibrium was maintained at 175^@C, calculate DeltaG^0 for the following equilibria. B hArr A "" DeltaG_1^0=? B hArr C "" DeltaG_2^0= ? |
|
Answer» Solution :`T=175^@C`=175+273 =448 K CONCENTRATION of 1 pentyne [A] =1.3% Concentration of 2-pentyne [B] =95.2% Concentration of 1,2 - pentadiene [C] =3.5% At EQUILIBRIUM `B hArr A` `95.2 % "" 1.3% RARR` `K_1=3.5/95.2`=0.0136 `B hArrC ` `95.2%"" 3.5% rArr` `K_2=1.3/95.2`=0.0367 `rArr DeltaG_1^0=-2.303 RT log K_1` `DeltaG_1^0`=-2.303 x 8.314 x 448 x log 0.0136 `DeltaG_1^0` =+16010 J `DeltaG_1^0`=+16 kJ `rArr DeltaG_2^0=-2.303 RT log K_2` `DeltaG_2^0`=-2.303 x 8.314 x 448 x log 0.0367 `DeltaG_2^0`=+12312 J `DeltaG_2^0` =+12.312 kJ |
|
| 37. |
The value of DeltaH for cooling 2 moles of an ideal monoatomic gas from 125^@C to 225^@C at constant pressure will be [given C_P=5/2R ] _____ |
|
Answer» `-250R` |
|
| 38. |
Which one of the following is a diatomic molecule? |
| Answer» SOLUTION :Hydrogen | |
| 39. |
Which of the following will give yellow precipitateon shaking with an aqueous solution of NaOH followed by acidification with dil. HNO_(3) and addition of AgNO_(3) solution? |
|
Answer»
|
|
| 40. |
Which of the following is not correct in the case of Be and Al ? |
|
Answer» both are RENDERED PASSIVE by conc.`HNO_3` |
|
| 41. |
The species having trigonal planar shape is |
|
Answer» `CH_(3)^(+)` |
|
| 43. |
Which of the following sets of quantum numbers is not allowed |
|
Answer» `n=1, l=0 , m=0 ,`s=+1/2` |
|
| 44. |
Which of the following is more reactive thentoward Nu^(-) Addition? |
|
Answer» |
|
| 45. |
Which of the following is more covalent : SbCl_(5)orSbCl_(3) ? |
| Answer» SOLUTION :Higher the oxidation STATE, more COVALENT is the compound, i.e., `SbCl_(5)` is more covalent than `SbCl_(3)`. | |
| 46. |
What is slaking of lime? |
|
Answer» SOLUTION :The addition of limited amount of WATER breaks the LUMP of lime. This process is called slaking of lime and the product is slaked lime `CaO + H_(2)O to CA(OH)_(2)` |
|
| 47. |
Which one of the following is the weakest base ? |
|
Answer» LiOH |
|
| 48. |
What are the chief pollutants of the atmosphere which are most likely to deplete the ozone layer? |
|
Answer» SULPHUR DIOXIDE |
|
| 49. |
Write the IUPAC names of the following alkanes: (i) (CH_(3))_(2)CHCH_(2)CH_(3) (ii) (CH_(3))_(2)CH-CH(CH_(3))_(2) (iii) (CH_(3)CH_(2))_(2)CH-CH(CH_(3))_(2) ltbr (iv) CH_(3)CH_(2)underset(CH_(3))underset(|)(CH)CH_(3)underset((CH_(2))_(2)CH_(3))underset(|)(CHCHCH_(3)CH)(CH_(3))_(2) (v) (CH_(3))_(2)CHCH_(2)CHCH_(3)CH(CH_(3))_(2) (vi) (CH_(3))_(3)C CH_(2)C(CH_(3))_(2) (vii) (viii) Isobutane (ix) Neopentane (x) Isooctane (xi) (xii) (xiii) (xiv) (xv) 4-tert-Butyloctane Strategy: Open up the structure if brancehes are present. Select the longest possible continous C chain carrying the maximum number of branches as the parent chain. Number the parent chain usingthe smallest possible number or lowest set of locants for the substituents (s). Assign the name and position number to each and every substituents. Write the name as a single word. Use hyphens to seperate numbers and letters. Do not leave any spaces. |
Answer» Solution :(i) 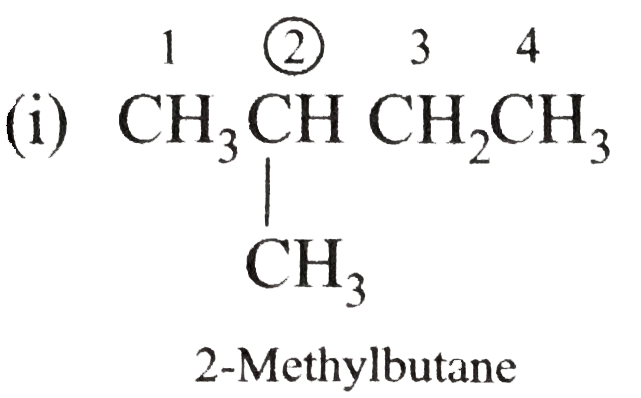 We have numbered the LONGEST chain beginning with the end of the chain nearer the substituents. (II) 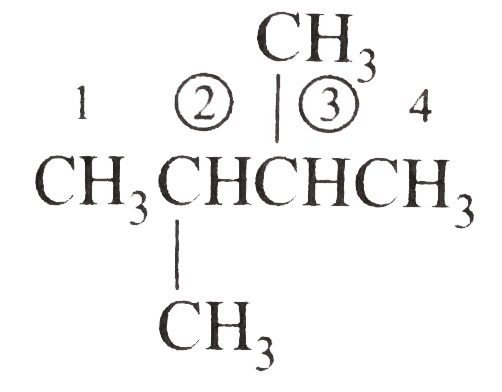 The presence to two identical substituents is INDICATED by the prefix `di`. Every substituent is given a number corresponding to its location on the longest chain. Normally, students make the mistake of not using the prefix `di` (iii) 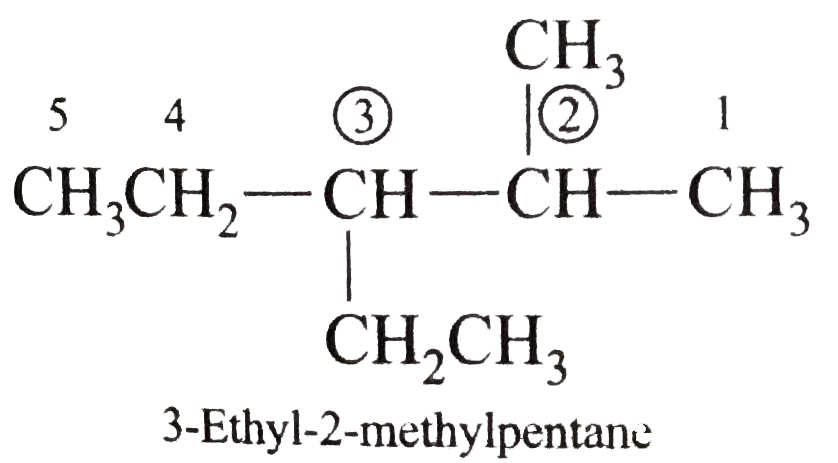 The names of DIFFERENT substituents arel listed alphabetically (iv) 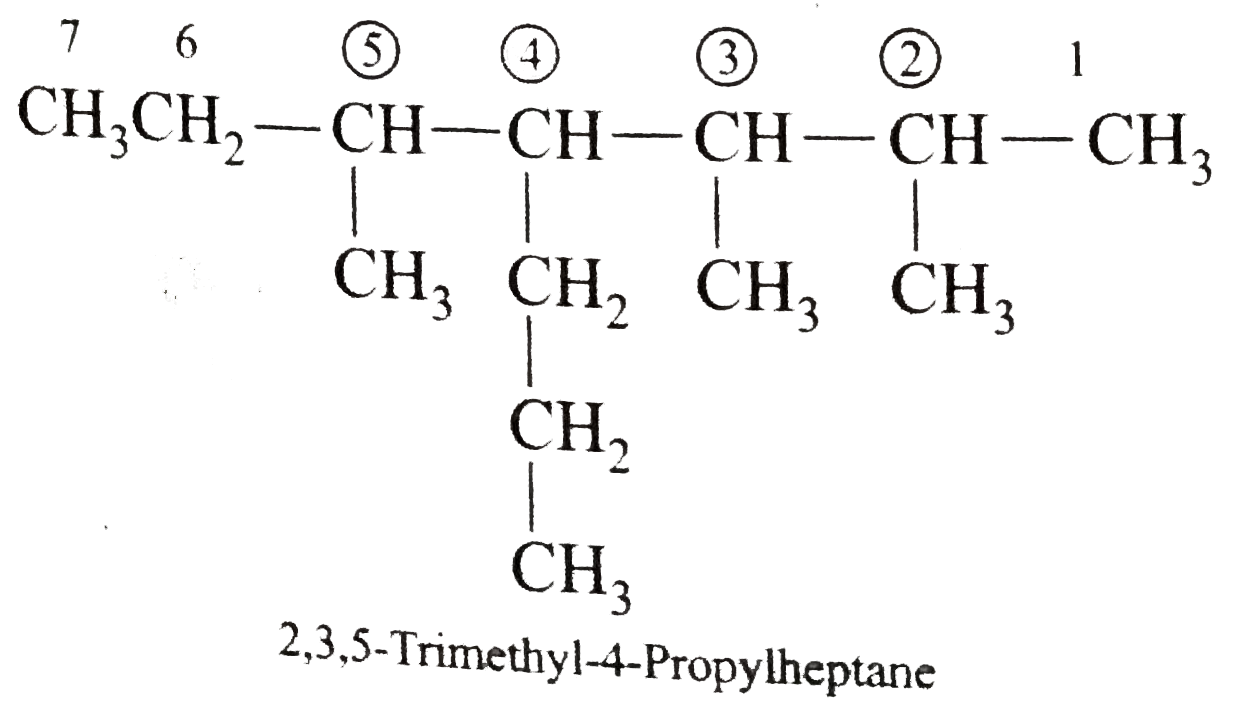 The selected parent chain carries the maximum number of branches. Commas are used to separate numbers from each other. (v) 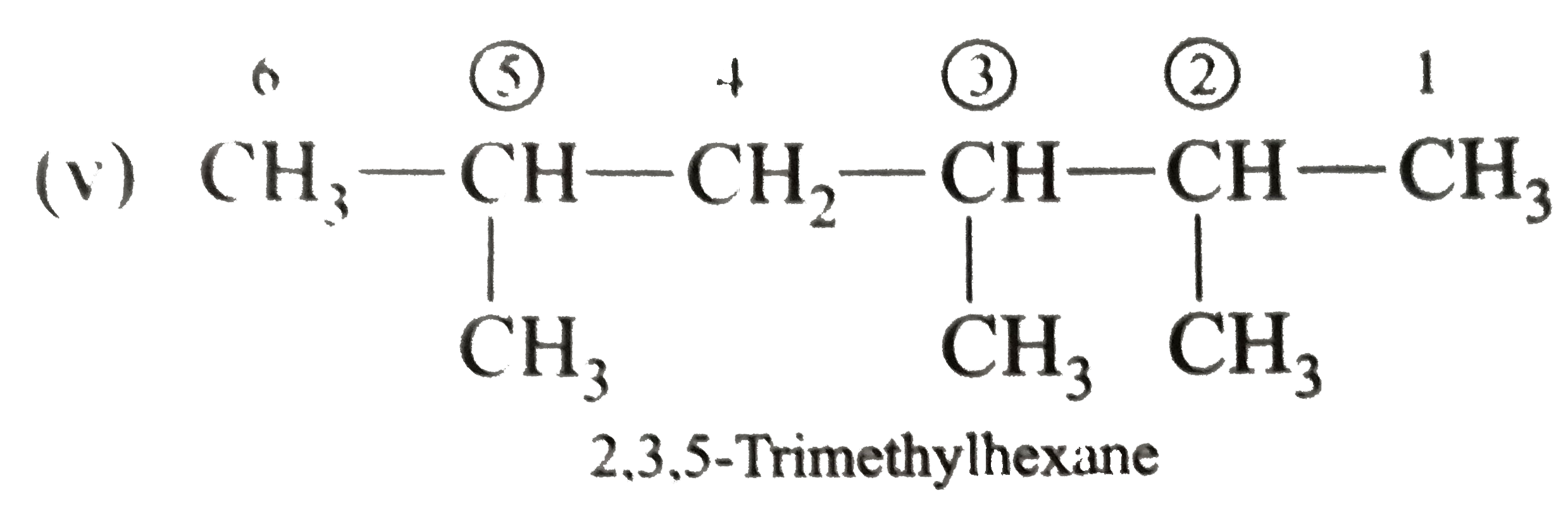 The numerical prefix tri indicates the presence of threee methyl group on the parent chain. (vi) 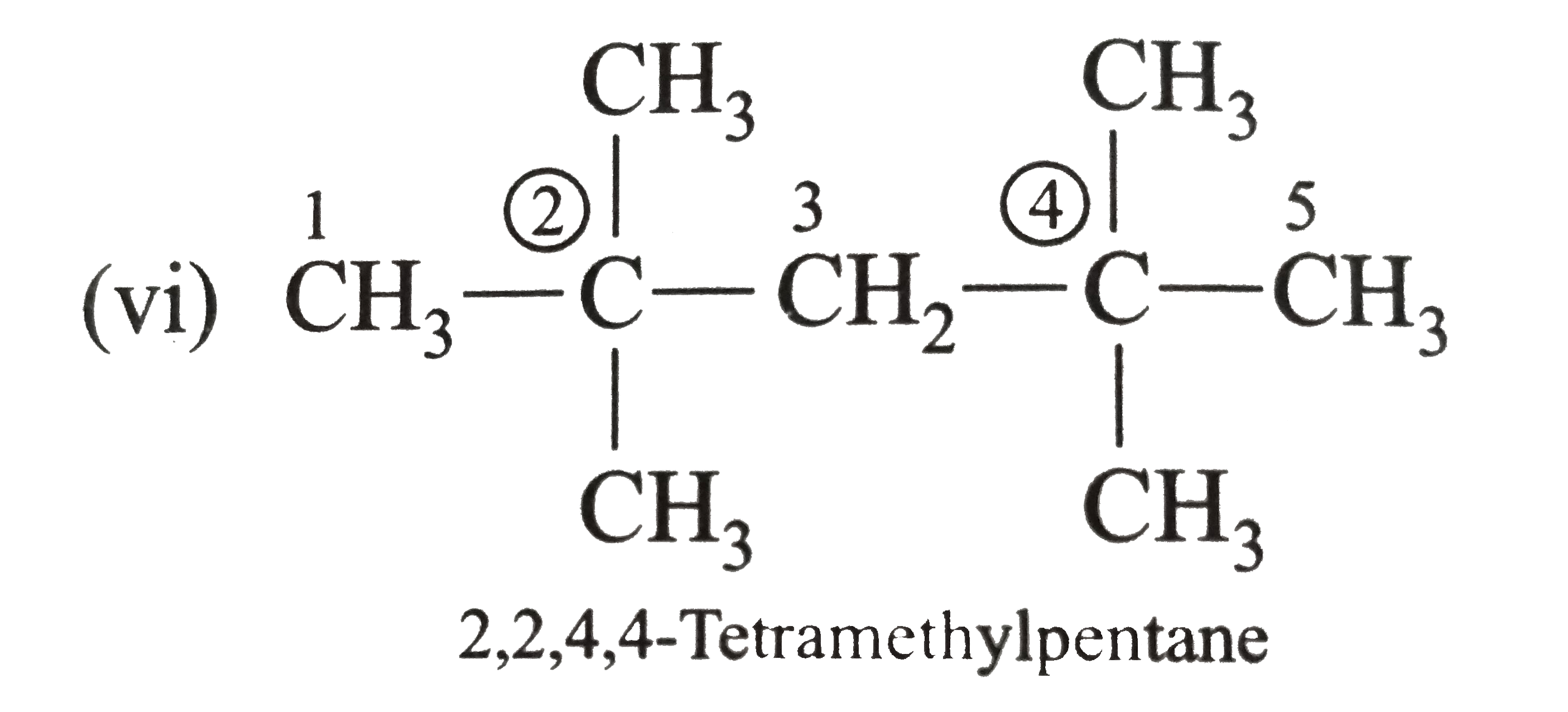 The numerical prefix tetra indicates the presence of four methyl groups on the parents chain. (vii) 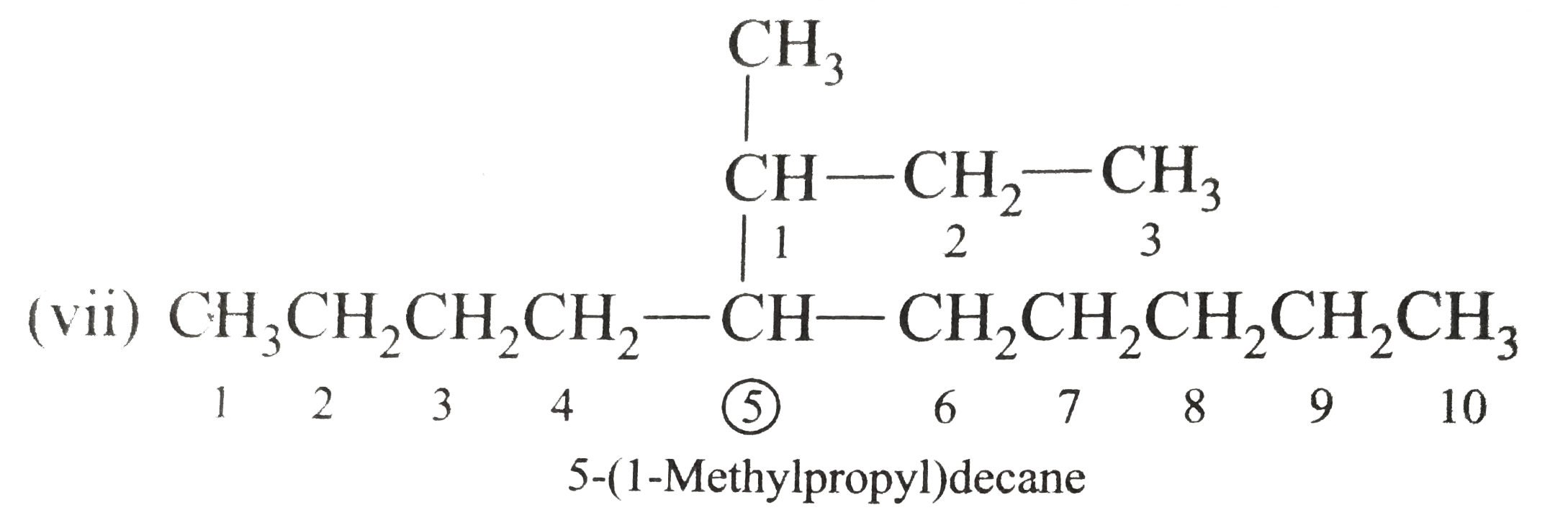 The name of complex substitutent is ALWAYS enclosed within parentheses. 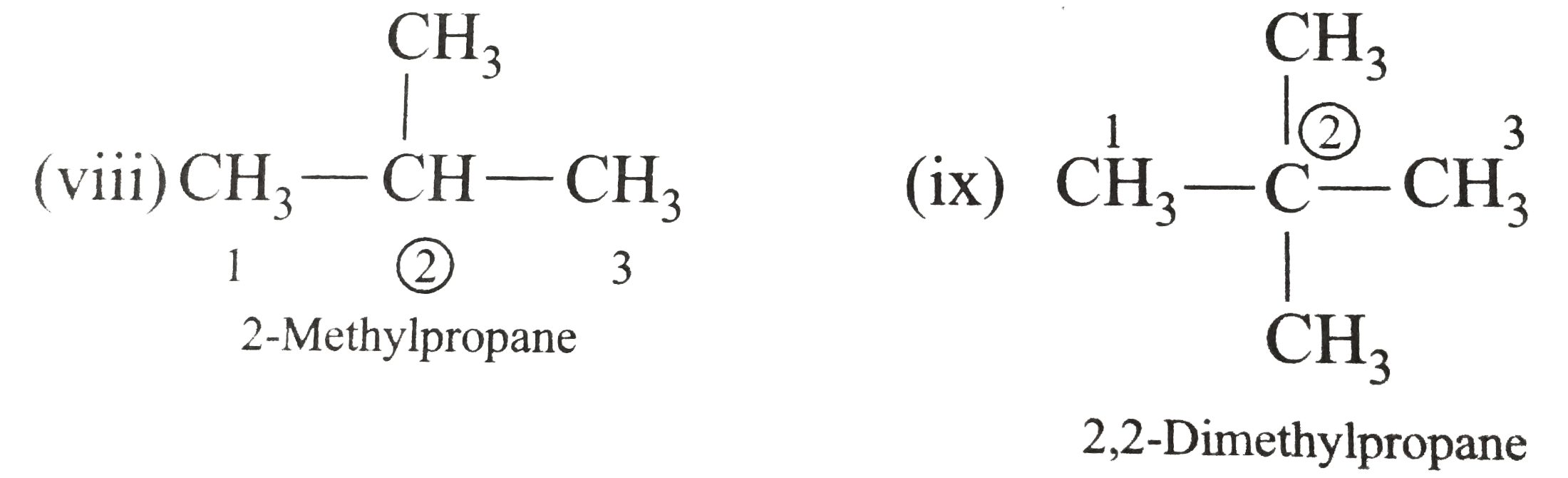 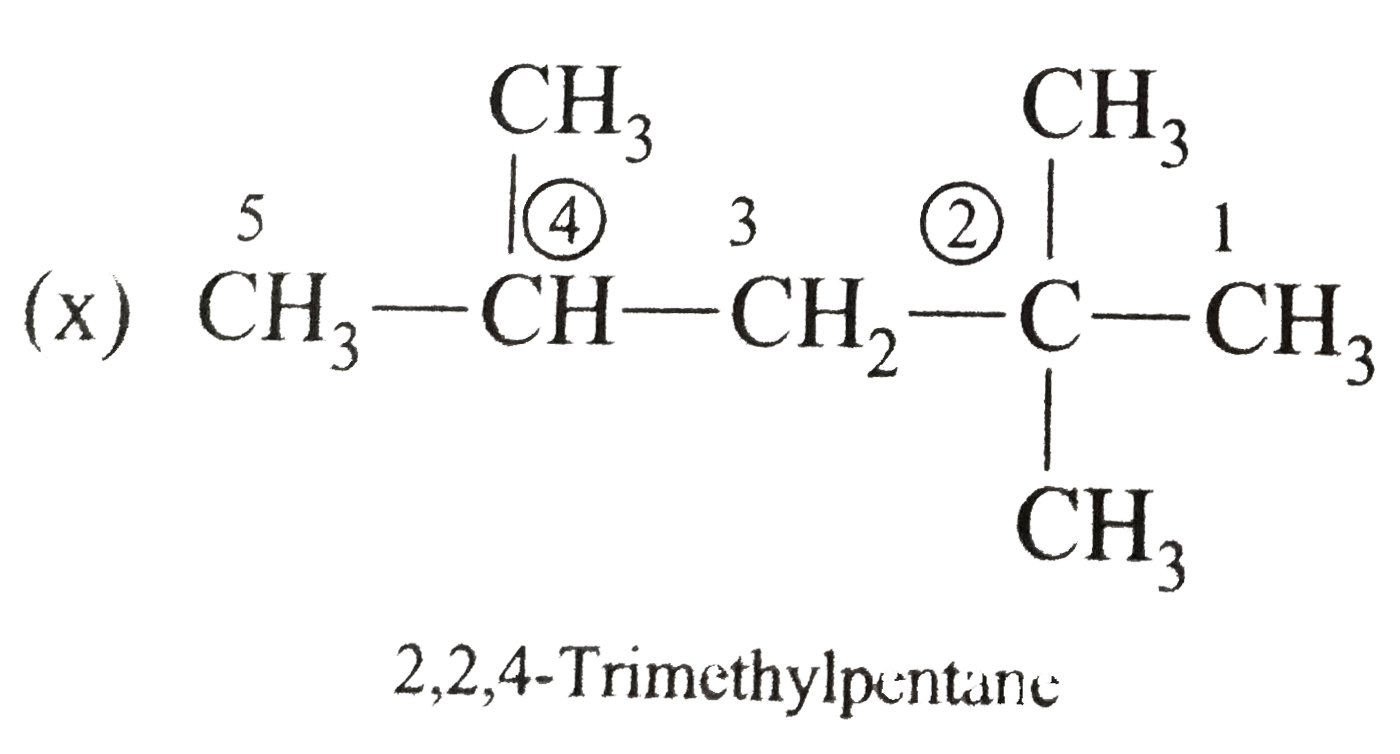 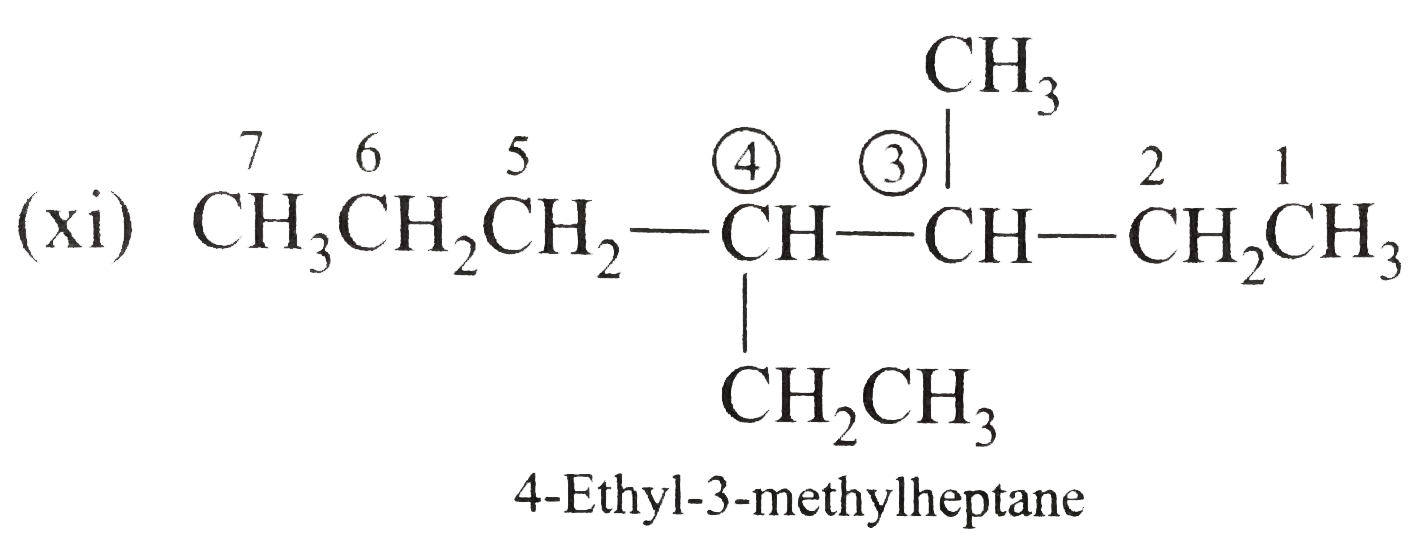 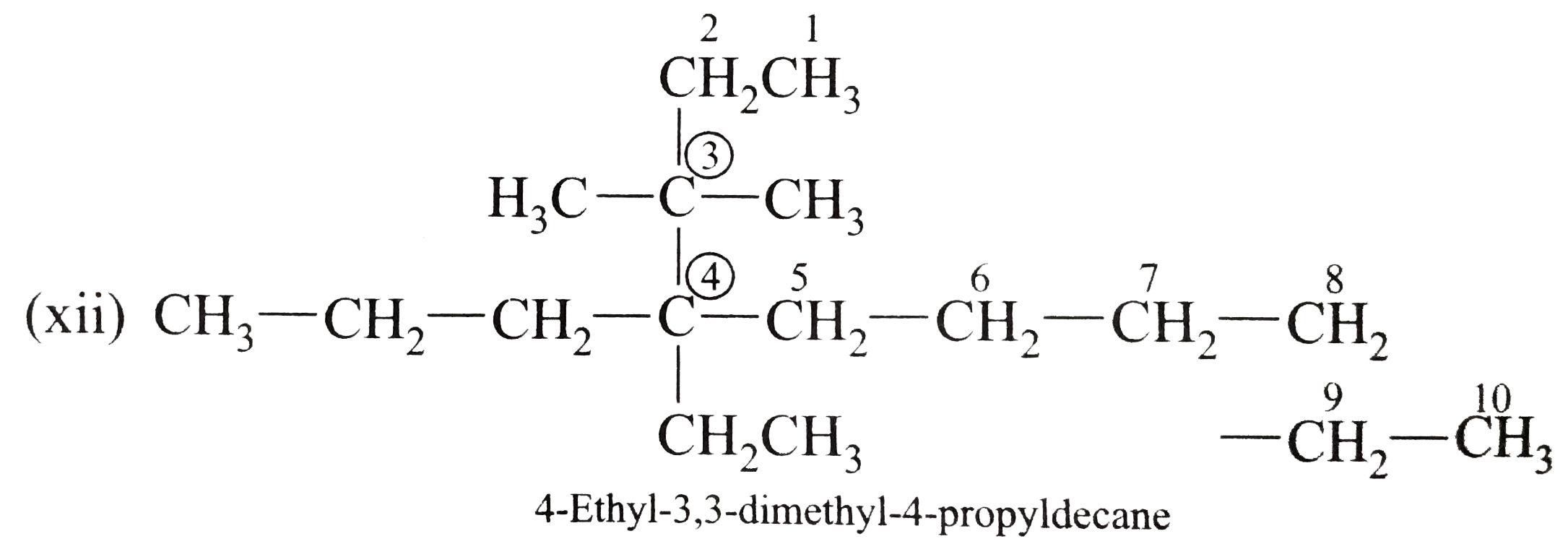 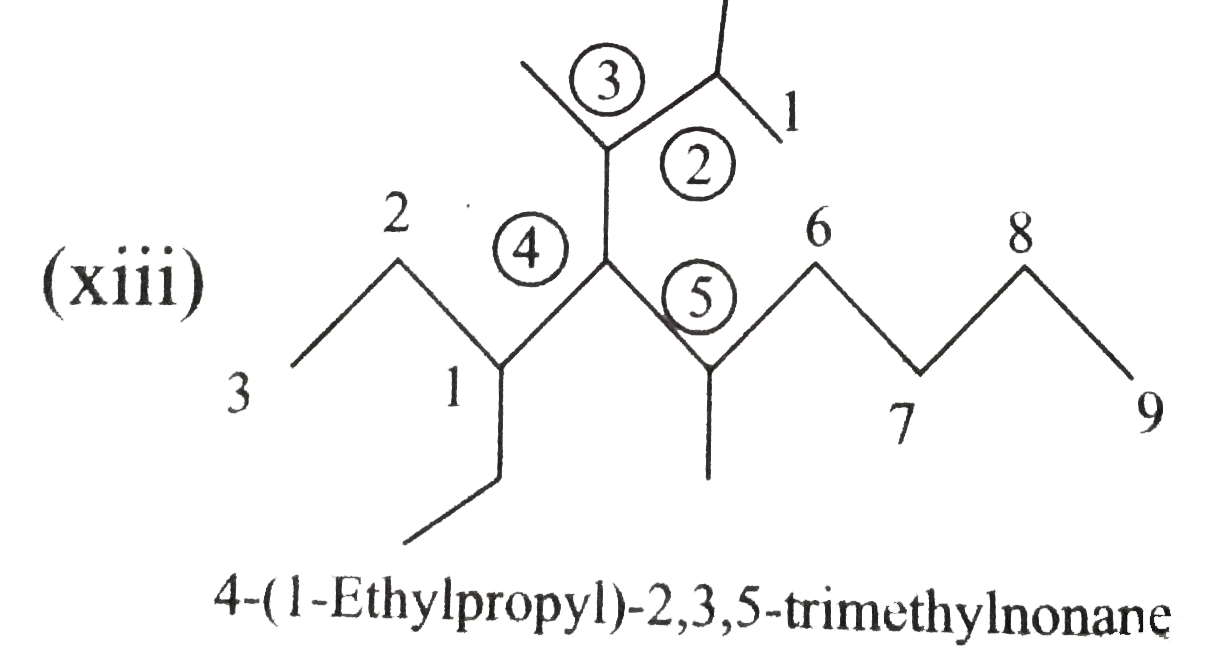 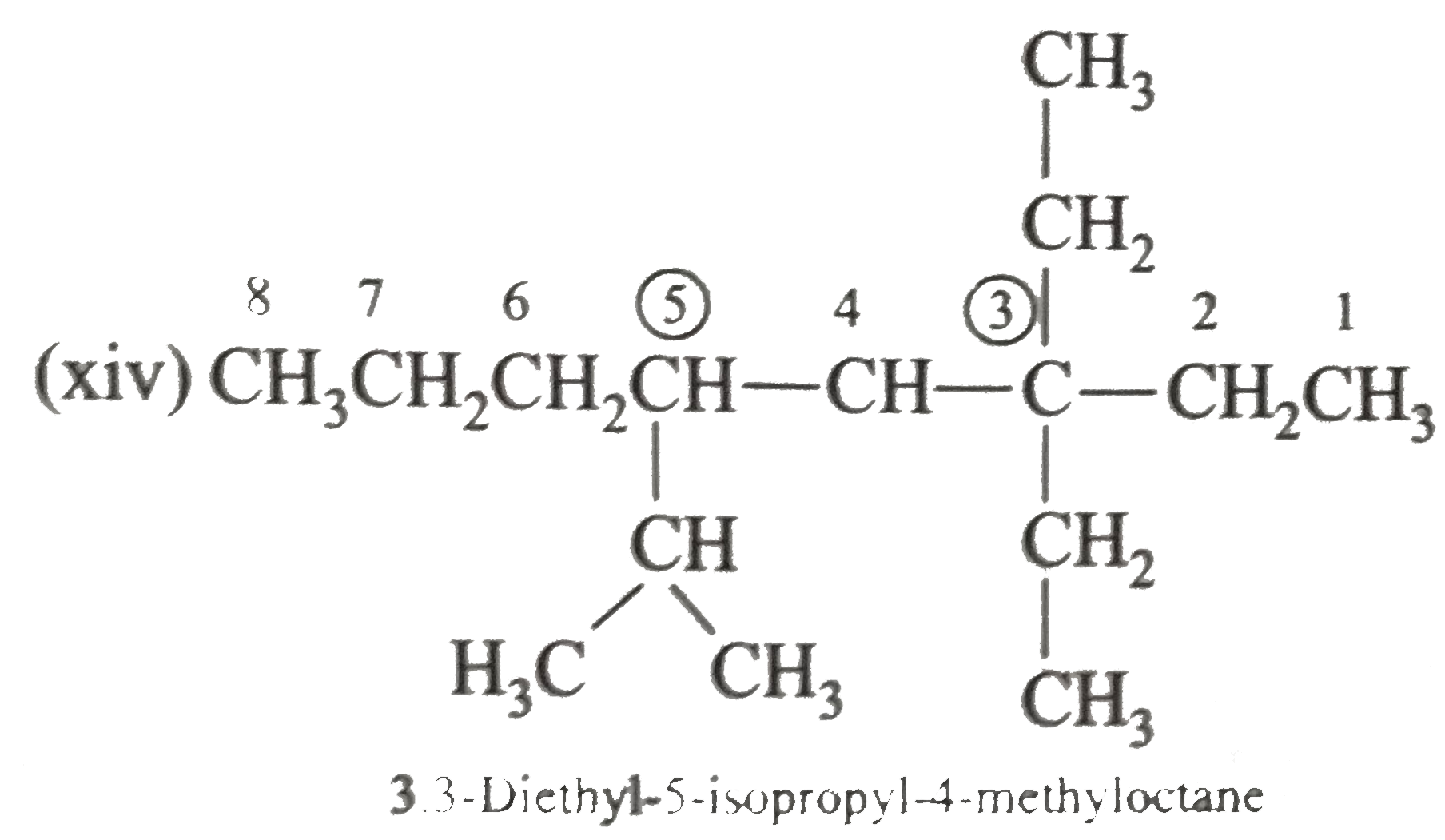 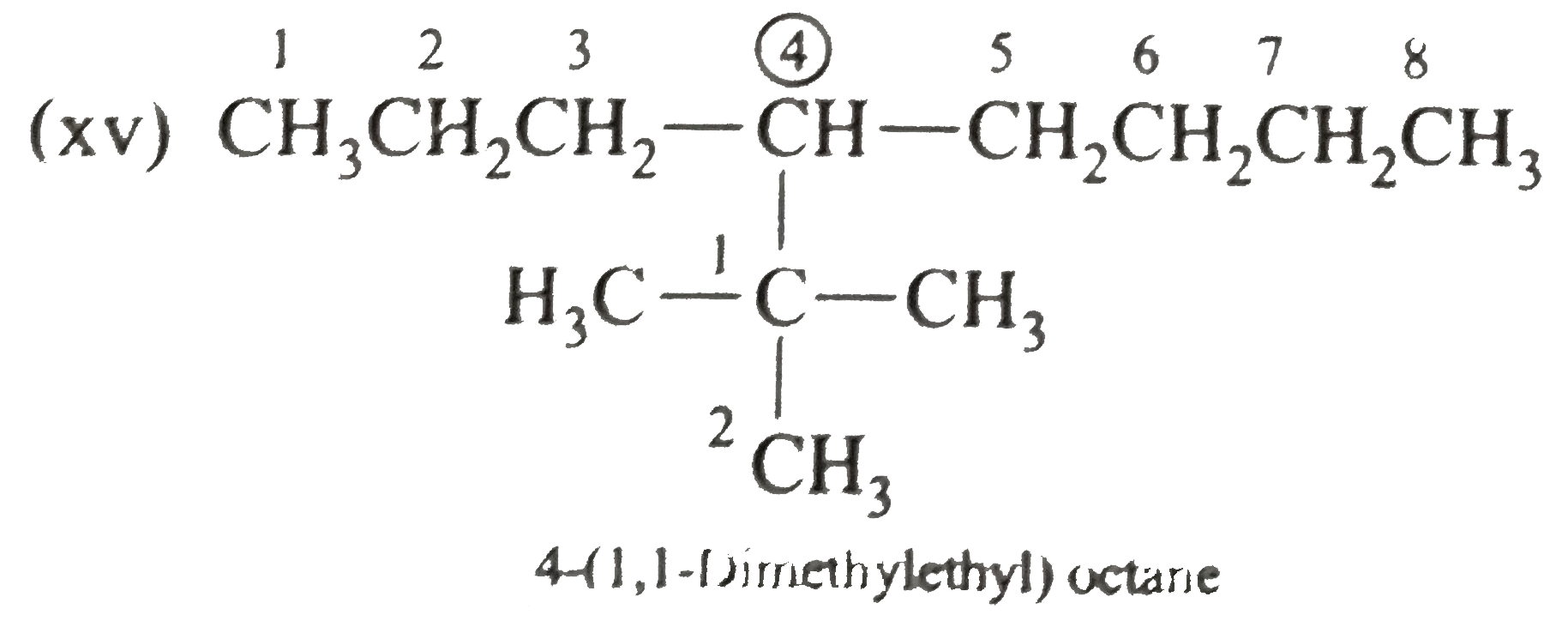 In deciding the alphabetical order for isopropyl, isobutyl, sec-butyl, and tert-butyl, we should disregard structure-defining prefixes that are written initalics and are spearated from the name by a hypen. 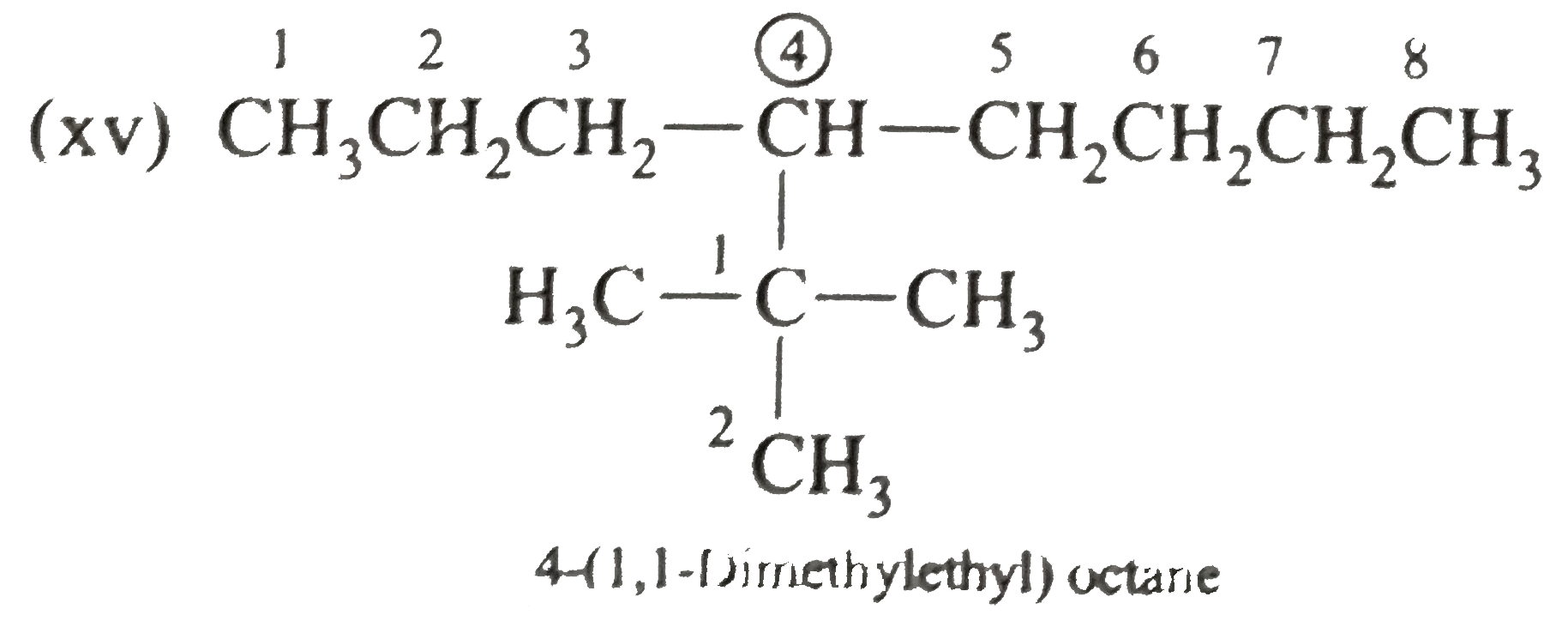
|
|
| 50. |
Which state of triply ionized beryliium (Be^(3+)) has the same orbital radius as that of the grounnd state of hydrogen atom ? |
|
Answer» Solution :For H-like particles, `r_(n) = 0529 XX (n^(2))/(Z)` For ground state of H-atom, `Z = 1, n = 1, r_(1) = 0.529 Å` For `Be^(3+) (Z = 4)`, if nth orbit has the same radius, then `0.529 xx (n^(2))/(4) = 0.529 or n^(2) = 4 " i.e., " n = 2` THUS, second orbit of `Be^(3+)` ion has same radius as that of ground state (1ST orbit) of H-atoms |
|