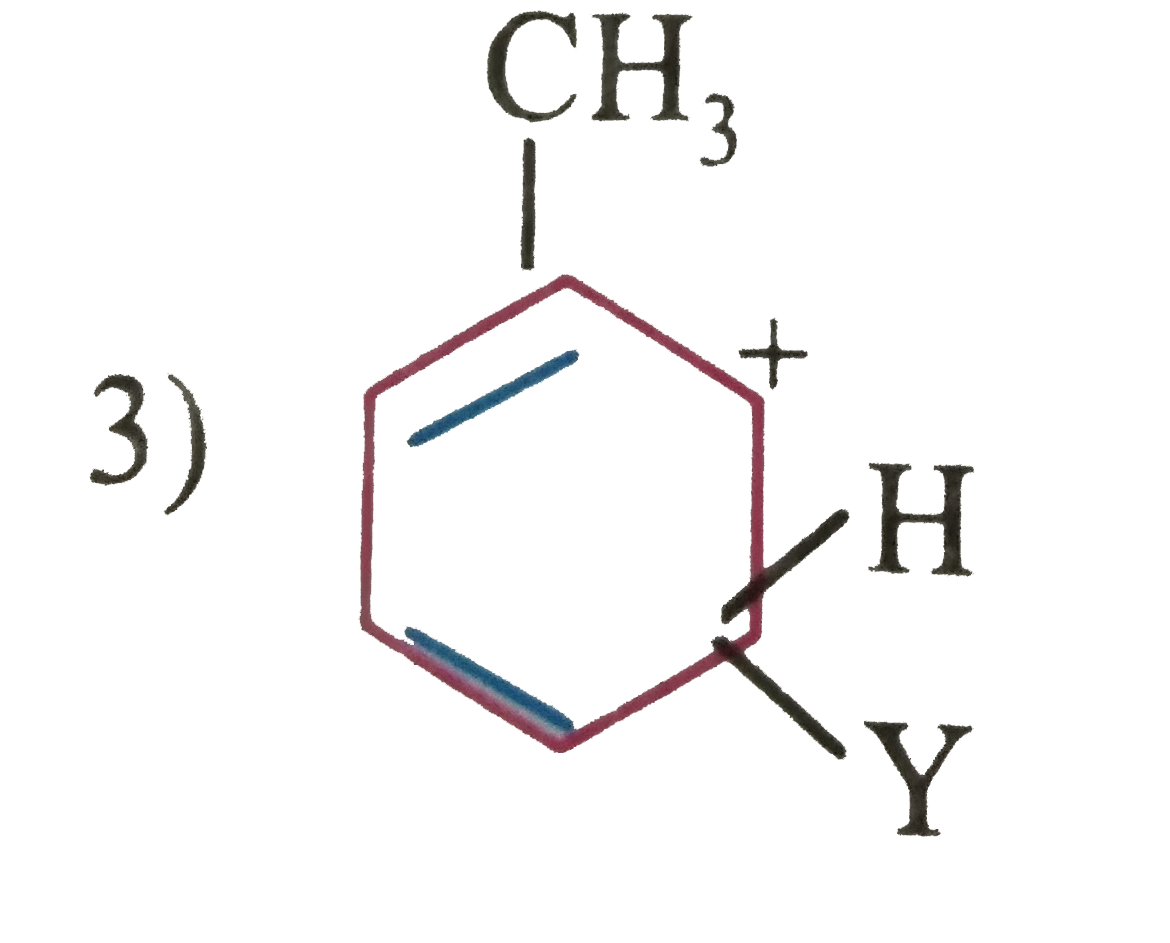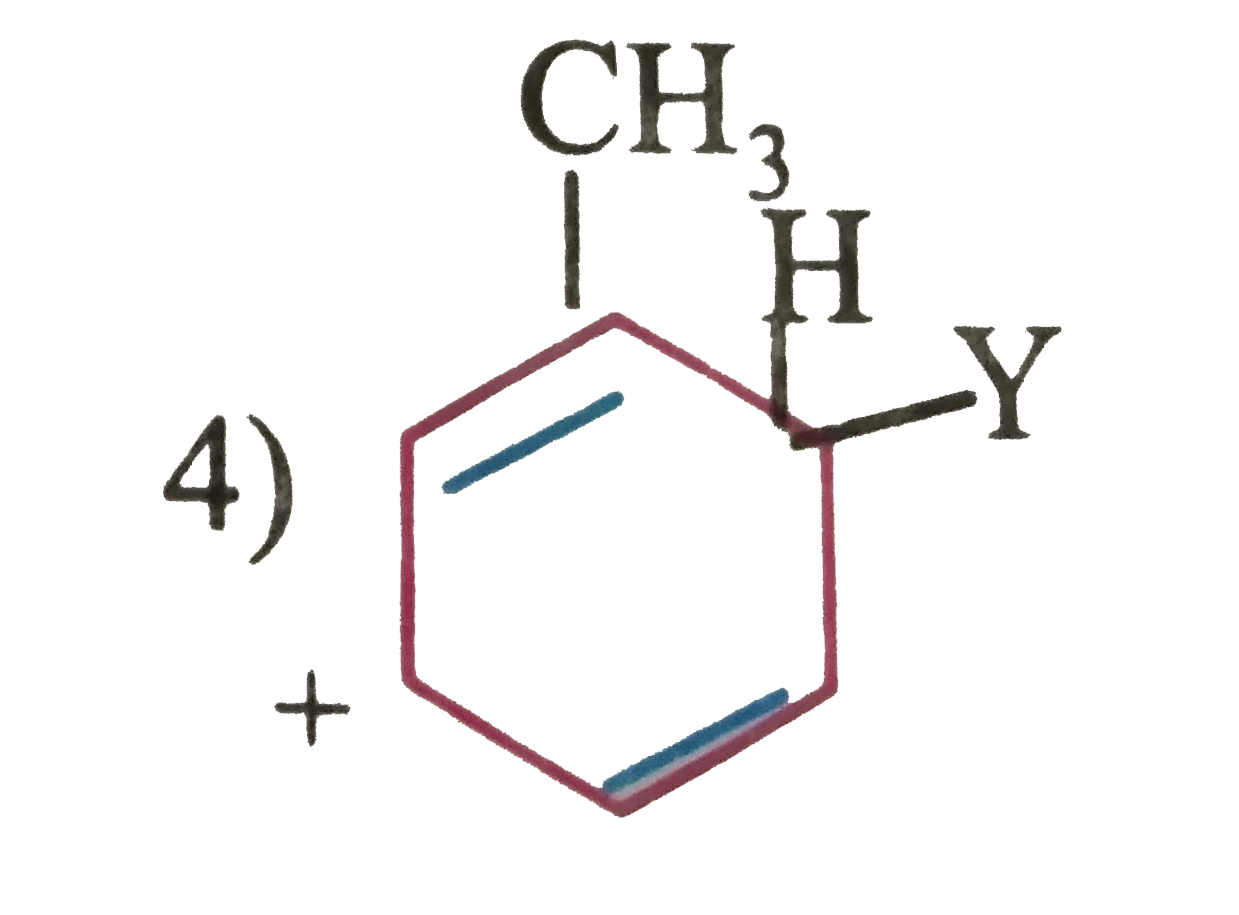Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the pH of a solution whose H^(+) ion concentration is 2xx10^(-5)g ions/litre ? |
|
Answer» |
|
| 2. |
Which element of the following possesses half filled valence shell with electron ? |
|
Answer» C |
|
| 3. |
Which of the following statement is(are) correct ? |
|
Answer» Fluorine has the highest electro negativity |
|
| 5. |
Which of the following statement is correct |
|
Answer» `X^(+)` ion is LARGER than `X^(-)` |
|
| 6. |
Write the products A & B for the following reaction. Cl-CH_(2)-CH_(2)-Cloverset(KOH)underset(-HCl)(to)Aoverset(KOH)underset(-HCl)(to)B |
|
Answer» Solution :`underset(((1,2,"dichloro ethane")))(CL-CH_(2)-CH_(2)-Cl)overset(al.KOH)underset(-HCl)(to)underset((A))underset(("chloro ethane"))(CH_(2))` `=CH-Cloverset(al.KOH)underset(-HCl)(to)underset((B))underset("ETHYNE")(CH=CH)` 
|
|
| 7. |
The solubility product by Lead sulphate, PbSO_4 is 1.3 xx 10^(-8) calculate its solubility in pure water. The molecular mass of PbSO_4 = 303 g "mol"^(-1) |
| Answer» SOLUTION :`3.45xx10^(-2) G L^(-1)` | |
| 8. |
Which of the following is used to visualize live cells? |
|
Answer» Chlorine |
|
| 9. |
Which gas has high speed between H_(2) and O_(2) ? Why ? |
| Answer» Solution :SPEED of `H_(2)` GAS increases than `O_(2)` because its MOLECULAR MASS increases. | |
| 10. |
Which one is a set of isolectronic species ? |
|
Answer» `N_(2), CO_(2), CN^(-)` `CN^(-) = 6 + 7 + 1 = 14 e^(-)`. Hence, (a) is not `N = 7E^(-), H_(2)S = 2 + 16 = 18e^(-)` `CO = 6 + 8 = 14 e^(-)`. Hence (B) is not `N_(2) = 14 e^(-), CO =6 + 8 = 14e^(-), CN^(-) = 6 + 7 + 1` `= 14e^(-)`. Hence , (c) is a SET isolectronic species |
|
| 11. |
Which of the following is correct IUPAC names are correct for the compound?HOOC - CH_2 - CH_2 - oversetunderset(|)(COOH)(C )H - CH_2 - CH_2 - COOH |
|
Answer» Pentane-1,3,5-tricarboxylic ACID |
|
| 12. |
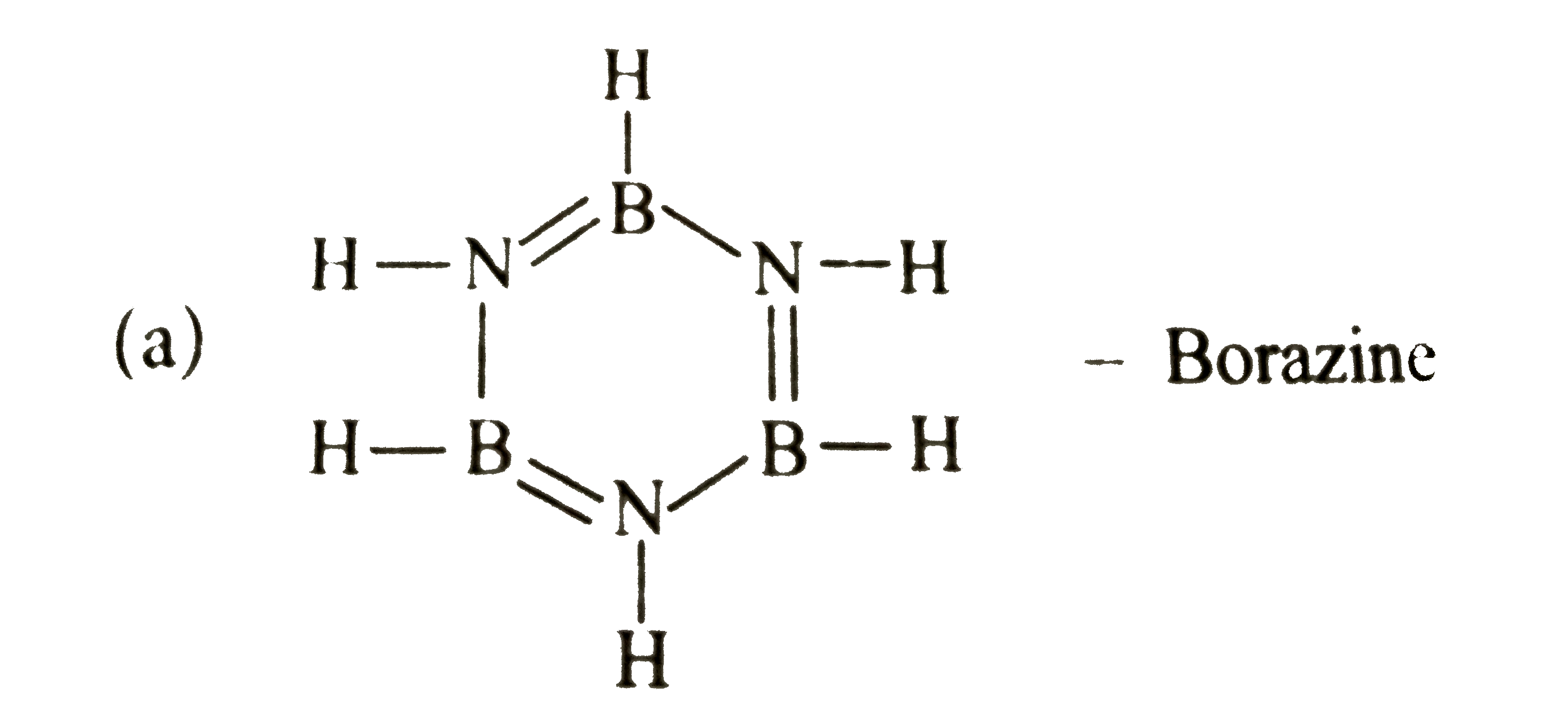
Which of the following species is aromatic? |
|
Answer»
|
|
| 13. |
What does atomic radius and ionic radius really mean to you? |
|
Answer» Solution :Atomic radius : Atomic radius is ONE HALF of the distance between the nuclei of two covalently bonded atoms of the same element in a molecule. It CASE of metals, the atomic radius is called metallic radius. It corresponds to one half of the distance between two adjacent atoms in a crystal lattice. Ionic Radius : Ionic radius means the size of the ion, a cation or anion. This GIVES the effective distance from the nucleus of the ions upto which it has an influence in the ionic bond. The size of the cation is always smaller that of the parent atom while the size of the anion is always larger than that of the parent atom. |
|
| 14. |
From the following data CH_(3)OH(l)+(3)/(2)O_(2)(g) rarr CO_(2)(g) +2H_(2)O(l) Delta_(r) H^(@)=-726 kJ mol^(-1) H_(2)(g)+(1)/(2)O_(2)(g) rarr H_(2)O(l), Delta _(r) H^(@)=-286 kJ mol^(-1) C("graphite") +O_(2)(g) rarr CO_(2)(g), Delta _(r) H^(@)=-393 kJ mol^(-1) The standard enthalpy of formation of CH_(3)OH(l)" in "kJ mol^(-1) is |
|
Answer» `Z = X +y ` |
|
| 15. |
The standard enthalpies of formation of C_2H_5OH_((l)), CO_(2(g)) and H_2O_((l)) are -277 , -393.5 and -285.5 "kJ mol"^(-1) respectively . Calculate the standard enthalpy change for the reaction C_2H_5OH_((l)) +3O_(2(g)) to 2CO_(2(g)) +3H_2O_((l)) The enthalpy of formation of O_(2(g)) in the standard state is zero , by definition. |
|
Answer» SOLUTION :The standard enthalpy change for the combustion of ethanol can be calculated from the standard enthalpies of formation of `C_2H_5OH_((L)), CO_(2(g))` and `H_2O_((l))` The enthalpies of formation are -277,-393.5 and -285.5 kJ `"MOL"^(-1)` respectively. `C_3H_5OH_((l))+3O_(2(g)) to 2CO_(2(g)) +3H_2O_((l))` `DeltaH_r^0=[(DeltaH_f^0)_"products"-(DeltaH_f^0)_"reactants"]` `DeltaH_r^0=[2(DeltaH_f^0)_(CO_2)+3(DeltaH_f^0)_(H_2O)]-[1(DeltaH_f^0)_(C_2H_5OH)+3(DeltaH_f^0)_(O_2)]` `DeltaH_r^0=["2 mol (-393.5) kJ mol"^(-1) + "3 mol (-285.5 ) kJ mol"^(-1)]-["1 mol (-277) kJ mol"^(-1) +"3 mol (0) kJ mol"^(-1)]` =[-787-856.5]-[-277] =-1643.5+277 `DeltaH_r^0` =-1366.5 KJ |
|
| 16. |
Time taken for an electron to complete one revolution in the Bohr orbit of hydrogen atom is |
|
Answer» `(4pi^(2) mr^(2))/(nh)` No. of revolution per sec `= ("Velocity")/("Circumference of the orbit")` `= (v)/(2pi r) = (nh)/(2pi mr) xx (1)/(2pi r) = (nh)/(4pi^(2) mr^(2))` `:.` TIME taken for one revolution `= (4pi^(2) mr^(2))/(nh)` |
|
| 17. |
Which of the following is not use of CaO ? |
|
Answer» As BLEACHING powder |
|
| 19. |
Which of the following elements has the lowest melting point? |
|
Answer» NaCl |
|
| 20. |
Which statement is wrong ? |
|
Answer» The STABILITY of hydride of group 15 increases as MOVING from top to bottom |
|
| 21. |
When Ag_(2)CO_(3) is heated then product will be: |
|
Answer» `Ag_(2)O` |
|
| 22. |
What is the state of hybridisation of carbon in diamond. |
| Answer» SOLUTION :Diamond :Each CARBON in diamond is `sp^3` hybridised and is BOUND to four other carbon atoms. | |
| 23. |
The subsidiary quantum number decides _________. |
|
Answer» the shape of the orbital |
|
| 24. |
Which of the following reaction involves the liberation of energy? |
|
Answer» `Na_(s) rarrNa_(g)^(+)` |
|
| 25. |
What will be the molar volume of nitrogen and argon at 273.15 K and 1 atm? |
| Answer» SOLUTION :At 273.15 K 1 atm PRESSURE, every gas has MOLAR volume=22.4 L . | |
| 26. |
Which of the following is not a reducing agent ? |
|
Answer» `SO_(2)` |
|
| 27. |
Which of the following statements is not true regarding the cis and trans isomers of an alkene |
|
Answer» They are configurational isomers |
|
| 28. |
Under what conditions of tenperature and pressure the formation of atomic hydrogen from molecular hydrogen will be favoured. |
|
Answer» HIGH TEMPERATURE and high PRESSURE |
|
| 29. |
What is the maximum concentration of Mg^(+2)that can be introduced into a solution containing0.1 M NH_3 and 0.01 M NH_4^(+ )without causing precipitation of Mg (OH)_2 ?K_b of NH_3 = 10^(6)K_(sp)of Mg(OH)_2 =1.2 xx 10^(-12) |
|
Answer» ` 0.012 M` `rArr IPlt KSP rArr [Mg^(+2) ] [OH^(-)] ^(2) LT 1.2 XX 10 ^(_12) ` ` [OH^(-) ] =(K_bxx [NH_3])/([NH_4^(+) ])=(10 ^(-6)xx 0.1)/( 0.01)= 10 ^(-5) ` ` rArr [Mg^(+2) ] lt(1.2xx 10 ^(-12))/( 10 ^(-10) )= 1.2 xx 10^(-2) ` |
|
| 30. |
You are given a mixture of acetone and methyl alcohol. How will you separate them ? |
|
Answer» |
|
| 31. |
Which of the following is the structure of (S)-Pentan-2-ol is ? |
|
Answer»
 the arrow is CLOCKWISE but least PRIORITY GROUP is on horizontal line of fischer projection. the arrow is CLOCKWISE but least PRIORITY GROUP is on horizontal line of fischer projection.
|
|
| 32. |
Vapour pressure of a pure liquid A is 10.0torr at 27^(@)C. The vapour pressure is lowered to 9.0 torr on dissolving one gram of B in 20g of A. If the molar mass of A is 200 g mol^(-1) then calculate the molar mass of B. |
|
Answer» Solution :`P _(A)^(@) = 10` torr,` P_("solution") = 9` torr, `W_(A) = 20g, W_(B) =1G, M _(A) =200 g mol ^(-1), M _(B) =` ? `(Delta P )/(P _(A) ^(@))= (W_(B) xx M _(A))/(M _(B) xx W _(A))` ` (10 -9)/(10) = (1XX 20 0)/( M _(B) xx 20)` `M _(B) = (200)/(20) xx 10 =100 g mol ^(-1)` |
|
| 33. |
Which of the following metal/metals can be used for the extraction of Ti from TiO_2 |
| Answer» Solution :Na or Mg is USED for the EXTRACTION of TI from `TiO_2` . | |
| 34. |
Which elements of group 1 and 2 are differ from the other elements of same group ? Give brief on diagonal relationship. |
|
Answer» Solution :Lithium and beryllium, the FIRST ELEMENTS of Group 1 and Group 2 respectively exhibit some properties which are different from those of the other members of the RESPECTIVE group. In these anomalous properties they resemble the second element of the following group. Thus, lithium shows similarities to magnesium and beryllium to aluminium in many of their properties. This TYPE of diagonal similarity is commonly referred to as diagonal relationship in the periodic table. The diagonal relationship is due to the similarity in ionic sizes and /or charge/ radius ratio of the elements. Monovalent sodium and potassium ions and divalent magnesium and calcium ions are found in large proportions in biological fluids. These ions perform important biological functions such as maintenance of ion balance and nerve impulse conduction. |
|
| 35. |
What happens when heavy water is added to calcium cardide ? |
| Answer» SOLUTION :Deuteroacetylene is formed , `CaC_(2) + 2D_(2)O to Ca(OD)_(2) + DC -=CD` | |
| 36. |
Which of the following pair of element and ore is incorrect? |
|
Answer» K - Sylvine (D) Be (BERYLLIUM) : Oxide bromalite : BEO (45% BeO) |
|
| 37. |
Which of the following reaction is used to prepare hydrocarbon with even number of carbon ? (i) Wurtz reaction (ii) Decarboxylation reaction (iii) Kolbe's electrolysis (iv) Reduction (v) Hydrogenation |
| Answer» Solution :(i) WURTZ reaction and (ii) Kolbe.s ELECTROLYSIS method is used to PREPARED hydrocarbon with EVEN number of carbon. | |
| 39. |
Which layer is present in upper region of stratosphere? |
|
Answer» `O_3` |
|
| 40. |
Under ambient conditions the total number of gases released as other products in the first step of the reaction scheme shown below is:{:(KMnO_(4)overset("strongly")underset("heated")rarrP+"other product"),(""darr"disproportionation in" H^(+)//H_(2)O),("""Product(s)"):} |
|
Answer» |
|
| 41. |
The total number of acyclic structural isomers possible for compound with molecular formula C_(4)H_(10)O is |
|
Answer» 9 (i) `CH_(3)CH_(2)CH_(2)CH_(2)OH`, (II) `(CH_(3))_(2)CHCH_(2)OH` (iii) `CH_(3)CH_(2)CHOHCH_(3)`, (iv) `(CH_(3))_(3)COH` and three are ethers : (v) `CH_(3)CH_(2)OCH_(2)CH_(3)`, (vi) `CH_(3)OCH_(2)CH_(2)CH_(3)` and (VII) `CH_(3)OCH(CH_(3))_(2)` |
|
| 42. |
Which of the following has maximum number of significant figures ? (i) 0.00453 (ii) 4.8046 (iii) 5.643 |
|
Answer» |
|
| 43. |
Which metal can displace hydrogen from dilute acids from the following data. E_(zn//zn^(2+))=-076V.E_(Cu//Cu^(2+))=0.343V |
|
Answer» |
|
| 44. |
what is the oxidation number of (i) C in CH_(2)O (i) Pt in [Pt (C_(2)H_(4))CI_(3)]^(-) |
|
Answer» SOLUTION :(i) x +2 (+1)+1(-2)=0 or x=0 (II) x+0 +3 (-1)=-1 or x=+2 please note that O.N of `C_(2)H_(4)` n(neutral ) is ZERO |
|
| 45. |
Which of the following is a correct pair? |
|
Answer» `BeCl_2` LINEAR |
|
| 46. |
What mass of CaO will be required to remove the hardness of 1000 litres of water containing 1.62g of Ca(HCO_3)_2 per litre? |
|
Answer» Solution :`underset("162 g")(Ca(HCO_(3))_(2))+underset("56g")(CaO) to 2CaCO_(3)+H_(2)O+CO_(2)` `5.6 xx 10^(2)g" be CAUSE solution has 1620 g"Ca(HCO_(3))_(2)`. |
|
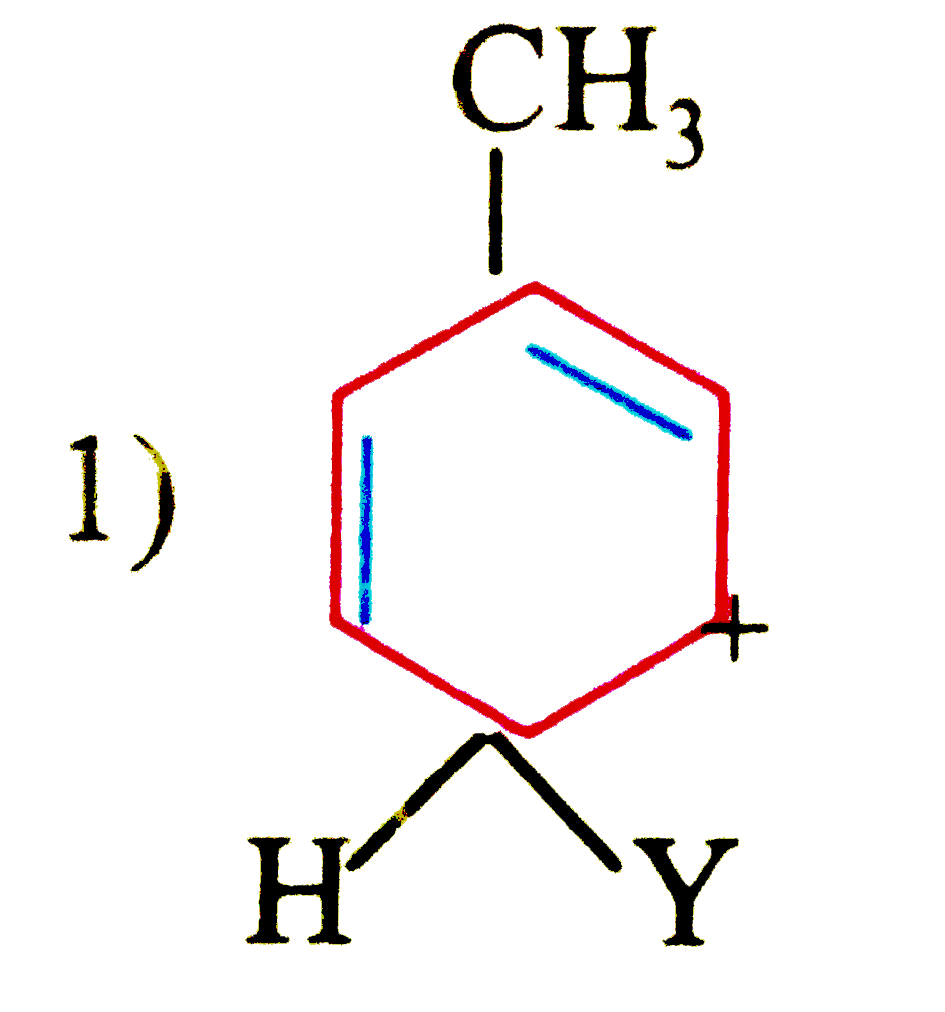
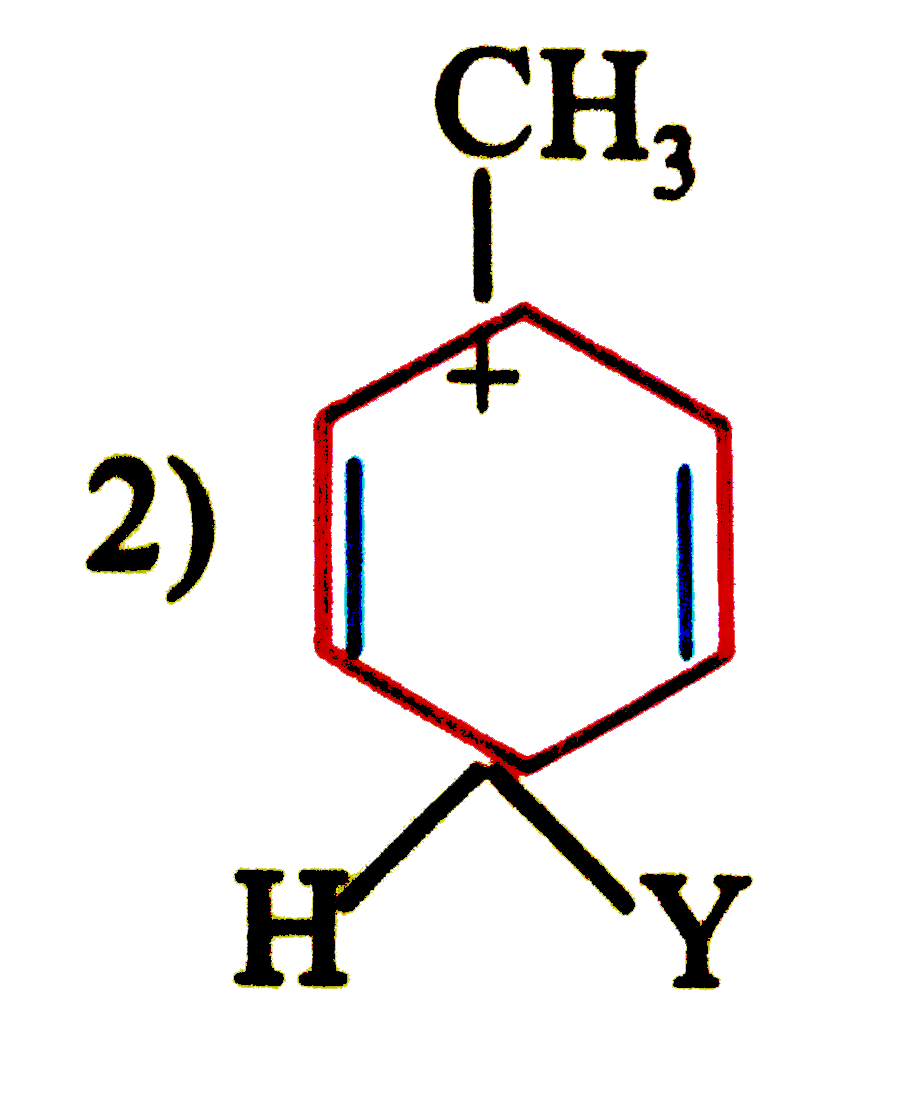
| 47. |
which of the following carbon is expected to be least stable. |
|
Answer»
|
|
| 48. |
Which one has highest melting point in given alkali chlorides ? |
| Answer» SOLUTION :NaCl | |
| 49. |
Which of the following compounds will react with NaNH_(2)? |
|
Answer» `CH_(3)--= CH` |
|