Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Whichelementdo youthinkwouldhave beennamed by (i)LawrenceBerkeleyLaboratory (ii) Seaborg's group |
| Answer» Solution :(i) LAWRENCIUM(Z= 103) and BERKELIUM(Z= 97)(ii) SEABORGIUM (Z=106) | |
| 2. |
Which is not correct? |
|
Answer» `GeCl_(2)` is more stable than `GeCl_(4)` |
|
| 3. |
What of the following relationship is correct? |
|
Answer» `DELTA G^(0) = -RT ln K` |
|
| 4. |
What weight of flurine is present in one gram of a fluoride tooth paste, forhans (Na_(3)PO_(4)F)? |
|
Answer» |
|
| 5. |
What type of structures are exhibited by (a)BaCl_2 , (b)Na_2O |
| Answer» SOLUTION :(a)FLUORITE TYPE STRUCTURE (B)Antifluorite type structure. | |
| 6. |
To Ag_(2)CrO_(4) solution over its own percipitate CrO_(4)^(2-) ions are added . This results in |
|
Answer» INCREASE in `AG^(+) ` concentration |
|
| 7. |
What is the monomer unit present in teflon ? |
| Answer» SOLUTION :`-CF_2 - CF_2 -` | |
| 8. |
What is the increasing order of acidic strength of the following acid ? Give reason HI, HCl, HF, HBr. |
|
Answer» SOLUTION :`HF lt lt HCl lt lt HBr lt lt HI` Acidic strength INCREASES `to` (`because` x is increases) F, Cl, Br, I are in same group in PERIODIC table. Its size of atom is in increasing order So, strength of H - A bond is DECREASE and the strength of these acids are increase. |
|
| 9. |
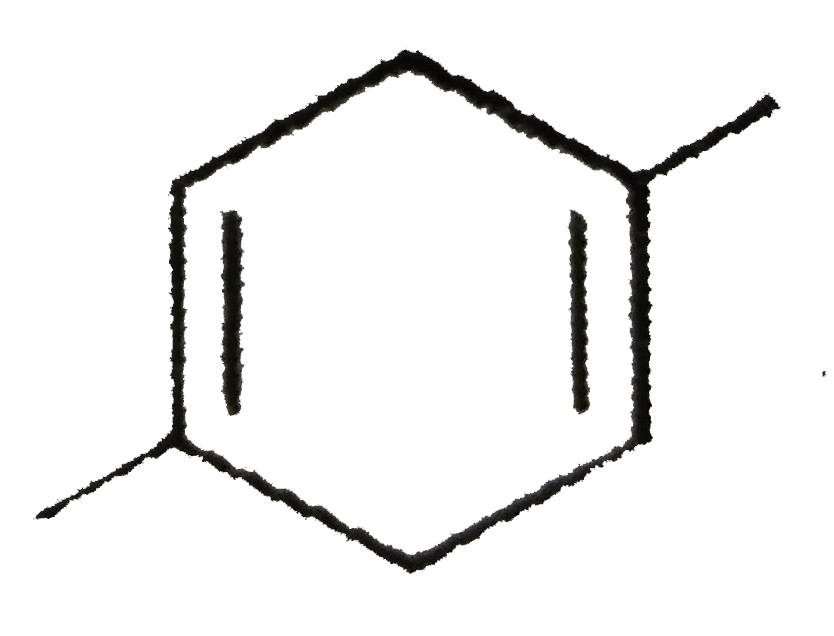
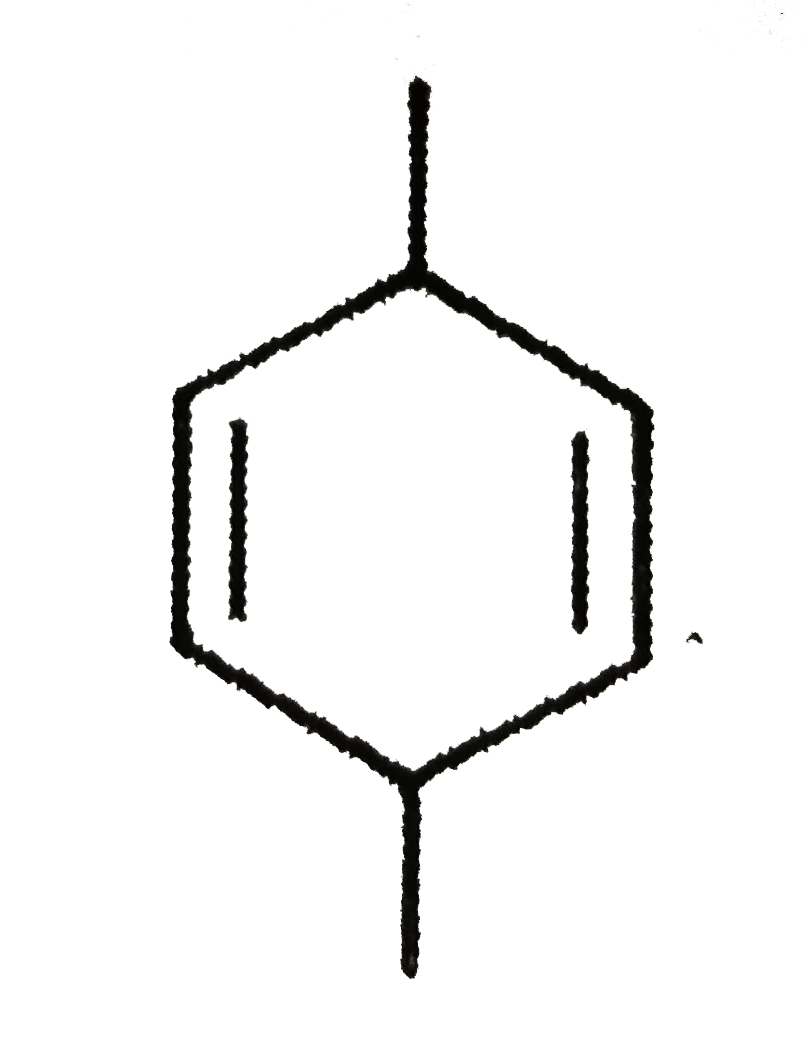
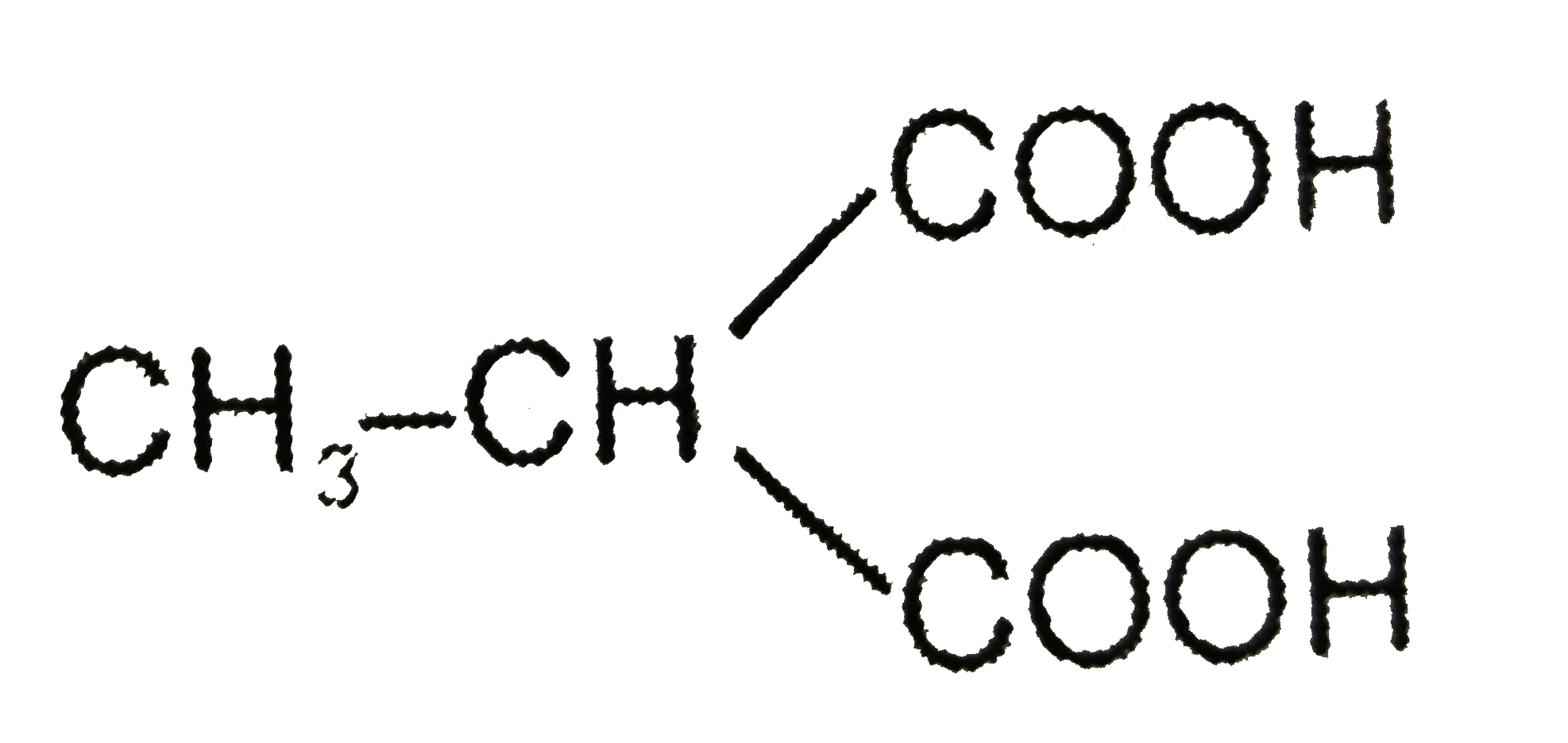
Which of the following compounds should be least soluble in water? |
|
Answer» Phenol |
|
| 10. |
The stability of the following alkali metal chlorides follows the order |
|
Answer» `LICL GT KCl gt NACL gt CsCl ` |
|
| 11. |
Which anion unit is observed in Muscovite ? |
|
Answer» `(Si_2O_5^(2-))_n` |
|
| 12. |
What is a spontaneous process? For the equilibrium A+2BhArrC DeltaH is +400kJ and DeltaS is +200JK^(-1) Calculate the temperature above the reaction becomes spontaneous? |
|
Answer» SOLUTION :The PROCESS that takes place on its own without the ASSISTANCE of external agency is called SPONTANEOUS process. `DeltaG=DeltaB-TDeltaS` At EQUILIBRIUM `DeltaG=0` `TDeltaS=DeltaH` `T=(DeltaH)/(DeltaS)=(400J)/(200J//k)=(cancel(400)xx10^(3)cancel(J))/(cancel(360_(1))cancel(J)//k)` `T=2000K` |
|
| 13. |
Use the data given below the find the type of cubic lattice to which the crystal of iron belongs :a/pm= 286,p/g"cm"^(-3)= 7.86 |
|
Answer» |
|
| 14. |
When treated with H_(2)O_(2) aqueous KMnO_(4) in acidic medium gives finally |
|
Answer» `Mn^(2+)` |
|
| 15. |
The standard molar heat for formation of ethane, carbondioxide and water are respectively, -21.1,-94.1 and -68.3 Kacal "mol"^(-1). What is the standard molar heat of combustion of ethane ? |
|
Answer» Solution :`2C_("(graphite)") + 3H_(2(s)) to C_2H_(6(g)) , DeltaH_1 = -21.1 K CAL` `C_(("graphite")) + O_(2(g)) to CO_(2) , DeltaH_2 = -94.1 kcal` `H_(2(g)) + 1/2O_(2) to H_2O_((L)) , DeltaH_2 = -68.3 k cal` The required equations is `C_(2)H_(6(g)) + 7/2 O_(2(g)) to 2CO_(2(g))+3H_(2)O_((l)) , DeltaH = ?` `DeltaH= 2DeltaH_(2) + 3DeltaH_(3) - DeltaH_1` `DeltaH = 2(-94.1) + 3(-68.3) - (-21.1) = -372 kcal` |
|
| 16. |
Which of the following pairs represents isomers of each other |
|
Answer»
 MOLECULE FORMULA is same for both the COMPOUNDS `M.F : C_4H_8` |
|
| 17. |
Which of the following electronic configuration corresponds to an inert gas? |
|
Answer» `1s^(2)2S^(2)2p^(5)` |
|
| 18. |
To a solution of acetic acid, solid sodium acetate is added greadually. When x mole of salt is added, the pH has a certain value. If y mole of salt is added, the pH is this time changes by 0.6 units to previous pH. What is the ratio of x and y ? If the solution is diluted after addition of y mole salt, what will be the change in pH ? Given that y gt x. |
|
Answer» |
|
| 19. |
Which is a stronger reducing agent, SbH_(3)orBiH_(3), and why? |
| Answer» SOLUTION :As we move down the group 15 elements from Sb to Bi, the atomic size increases and hence the strength of the BOND it makes with hydrogen decreases, i.e., Bi-H bond is WEAKER than Sb-H bond. In other works, `BiH_(3)` is thermally less stable than `SbH_(3)`. Therefore, on heating `BiH_(3)` has a greater TENDENCY to liberate hydrogen than `SbH_(3)` and hence `BiH_(3)` is a stronger reducing agent than `SbH_(3)`. | |
| 20. |
Which is the correct name of the following structure? Why? CH_(3)-CH_(2)- underset(underset(CH_(2)-CH_(3))(|))(CH)-CH_(2)-CH_(2)-underset(underset(CH_(3))(|))(CH)-CH_(2)-CH_(3) (i) 3-ethyl-6-methyloctane (ii) 3-methyl-6-ethyloctane (iii) 6-methyl-3-ethyloctane |
|
Answer» Solution :(i) NAME is correct because, form both the order of carbon is same, so, methyl GROUP has 6 no. and ETHYL group has 3 no. as PER alphabetical order. Name (iii) is wrong- because ethyl must be FIRST than methyl |
|
| 21. |
Unimely and unusual rains are due to |
|
Answer» GLOBAL warming |
|
| 22. |
Which of the following species in not diamagnetic ? |
|
Answer» `N_(2)` |
|
| 23. |
Write the resonance structures of SO_3, NO_2 and NO_3^- |
Answer» SOLUTION :
|
|
| 24. |
What happens when Borax is heated |
|
Answer» |
|
| 25. |
Which of the following gas is insoluble in water? |
|
Answer» `SO_(2)` |
|
| 26. |
What is quantum mechanicalmodelof atomgive its important features. |
|
Answer» Solution :Quantum MechanicalModel of ATOM : Thismodelof atom is the pictureof thestructureofthe atomwhichemerges from the applicationof theschrodingerequationto ATOMS (i) the energyof electronin atomsisquantizedforexamplewhen electronsare boundto thenucleusin atoms (ii) theexistenceof quantizedelectronicenergylevelsis adirectresult ofthe wavelikepropertiesof electronsand are allowedsolutions ofSchrodinger wave equation. (iii) boththeexactpositionandexactvelocityofdeterminesimultaneously(Heisenberguncertaintyprinciple ) .THEPATH of anelectronin an atom thereforecannever bedeterminedor knonwn accurately (iv)An atomicorbitalis thewavefunction w foran ELECTRON in an atom (v ) Theprobabilityof findingan electron at apointwithinan atonis proportional to thesquareof theorbitalwavefunctioni.e.,wdensityandisalwayspositive From the valueof `w^(2)` at differentpointwithinan atom it ispossiblepredict the regionaroundthe nucleuswhereelectronwill mostprobablybe found. |
|
| 27. |
Which of the following elements does not show disproportionation tendency ? |
| Answer» Solution :Being the most electronegative ELEMENT F can only be reduced and HENCE it always SHOW an oxidation number of -1 futher due to the absence of d orbitals it cannot be oxidised and hence it does not show +ve oxidation number in other words F cannot be simultanusly oxidised as well as reduced and hence does not show disproportionation reaction thusoption c correct | |
| 28. |
Which is incorrect about 'green house effect' |
|
Answer» It is due to HIGH concentration of `CO_(2)` in atmosphere |
|
| 29. |
Urine has a pH of 6.0 . If a patient eliminates 1300 ml of urine per day, how many gram equivalents of the acid he eliminates per day ? |
|
Answer» Thus, 1000 ml of the urine contain acid `=10^(-6)G` EQ. `:.` 1300 ml of the urine will contain acid `=1.3 XX 10^(-6) ` g eq. |
|
| 30. |
Which one of the following is a molecular crystal? |
|
Answer» ROCK salt |
|
| 31. |
Write electronic configuration of oxygen molecule and calclate its bond order. |
| Answer» Solution :`KK^(**)sigma2S^(2)SIGMA^(**)2 S^(2) PI 2 PX^(2)=pi2py^(2)py^(**)2pix^(1)=pi^(**)2PY^(1)` BOND Order`=(N_(b)-N_(A))/r=(8-4)/2=2` | |
| 32. |
What is the basic difference in approach between Mendeleev's periodic table and modern periodic table? |
| Answer» Solution :The Main BASIC differnce between Mendeleev.s PERIODIC table and MODERN periodic table is that FIRST one is constructed on the basis of atomic weight and the later is constructed on THEBASIS of atomic number. | |
| 33. |
Write of the following have identical bond order ? CN^(-), O_(2)^(-) , NO^(+) and CN^(+) |
|
Answer» Solution :Total no. of electrons in `CN^(-) = 6 + 7 + 1 = 14` Total no. of electrons in ` O_(2)^(-) + 8 + 8+ 1 = 17` Total no. of electrons in ` NO^(+) + 7 + 8 -1 = 14` Total no. of electrons in `CN^(+) = 6 + 7 - 1 = 12 ` As ` CN^(-) and NO^(+)` have the same BOND order . |
|
| 34. |
Which is correct statement? |
|
Answer» Alkanes undergo ozonolysis |
|
| 35. |
What is another name for photo-chemical Smog ? |
|
Answer» LOS ANGELES SMOG |
|
| 36. |
When KBr is treated with conc. H_(2)SO_(4) reddish-brown gas is evolved. The gas is |
|
Answer» `B_2O_3` |
|
| 37. |
True statements is/are |
|
Answer» STRUCTURE of X is 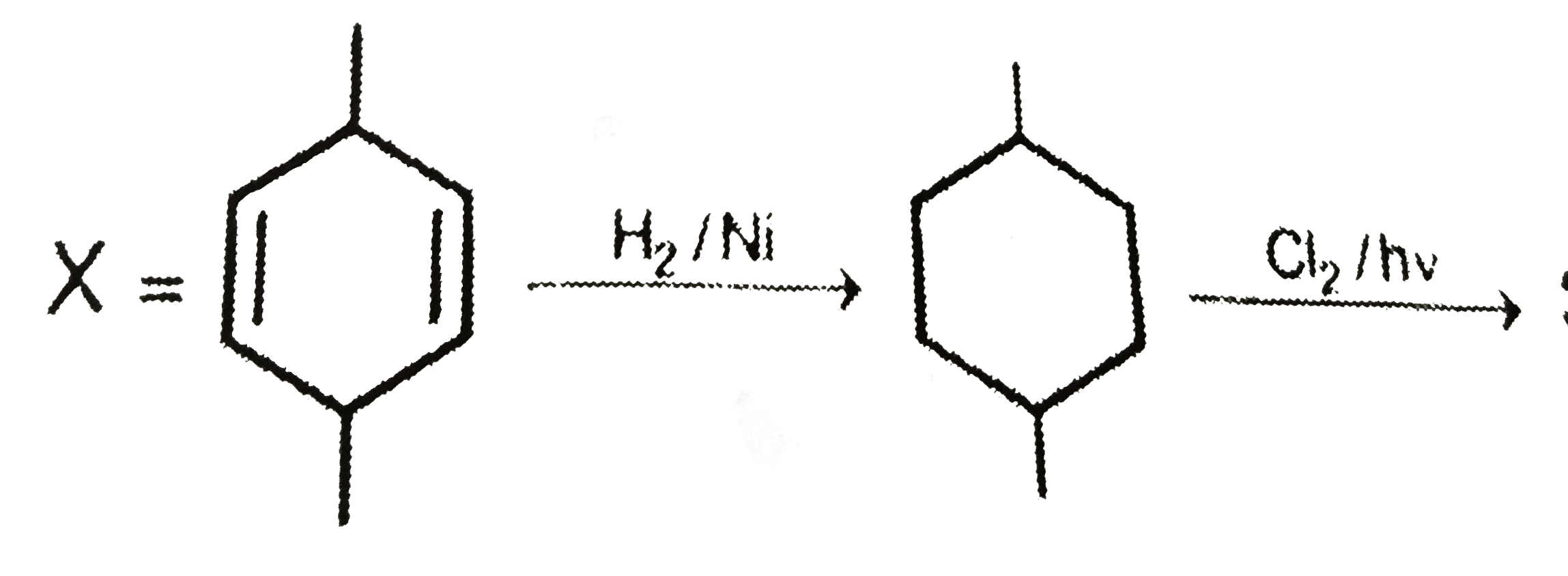
|
|
| 38. |
The solubility of solid silver chromate. Ag_(2) CrO_(4) . is determined in three solvents. Substance K_(sp)of Ag_(2) CrO_(4) = 9 xx 10^(-12) (I) pure water(ii) 0.1 M AgNO_(3) "" (iii) 0.1 M Na_(2) CrO_(4) Predict the relative solubility of Ag_(2) CrO_(4)in the three solvents: |
|
Answer» `I = II = III` |
|
| 39. |
What is isomerism? Which are the types of isomerism? |
Answer» Solution :The phenomenon of existence of two or more COMPOUNDS possessing the same MOLECULAR formula but different properties is KNOWN as isomerism. Such compounds are called as ISOMERS. The FOLLOWING flow chart shows the different types of isomerism 
|
|
| 40. |
Which is an extensive property of the system? |
|
Answer» Volume |
|
| 41. |
Which of the following pairs of d-orbitals will have electron density along the axes ? |
|
Answer» `d_(z)^(2),d_(XY)` |
|
| 42. |
Which of the following metal can be extracted by smelting ? |
|
Answer» Al |
|
| 43. |
Which of the following statement is correct for a galvanic cell? |
|
Answer» REDUCTION occurs at cathode |
|
| 44. |
Which are true ? |
|
Answer» A gas can be liquified above its `T_C` `implies` more liquifiable. |
|
| 45. |
When XO_(2) is fused with an alkali metal hydroxide in presence of an oxidizing agent such as KNO_(3), a dark green product is formed which disproportioates in acidic solution to afford a dark purple solution. X is: |
|
Answer» Mn |
|
| 46. |
You are provided with a set of compounds. What is the number of compounds that are expected to show geometrical isomerism. |
|
Answer» |
|
| 47. |
Write the polymerication of alkene ? |
|
Answer» Solution :(a) definition : Polythene is obtained by the combination of large number of ethene moelcules at HIGH temperature, high pressure and in the presence of a CATALYST. The large MOLECULES thus obtained are called polymers. This reaction is known as polymerisation. (b) Example :(i) Polymerization of ethelene, forms polyethelene. `underset("Ethene")(nCH_(2)=CH_(2))underset("High pressure, catalyst")overset(473-673, O_(2) " High temperature")rarr underset("Polyehene")((-CH_(2)-CH_(2)-)_(n))` (ii) POlymerization of propelene, yields propelene polymer. `underset("Ethene")(nCH_(3)-CH=CH_(2))underset("High pressure, catalyst")overset("High temperature")rarr underset("Polypropene")((-overset(CH_(3))overset(|)(CH_(2))-CH_(2)-)_(n))` (c) Uses : POlymers are used for the MANUFACTURE of plastic bags, squeeze bottles, refrigerator dishes, toys, pipes, radio and T.V. cabinets etc. Polypropeneis used for the manufacture of milk crates, plastic buckets and other MOULDED articles. Limitations : Though these materials have now become common, excessive use of polyethene and polypropylene is a matter of great concern for all of us. |
|
| 48. |
Which reagent can produce salicylic acid from sodium phenoxide under suitable conditions? |
|
Answer» `K_(2)Cr_(2)O_(7)//H^(+)` |
|
| 49. |
Two elements X and Y combine to form a compound XY. Under what conditions the bond formedbetween them will be ionic ? |
|
Answer» If the difference in ELECTRONEGATIVITIES of X and Y is 1.7. |
|
| 50. |
When H_(2)O_(2) is treated with a solution of titanium dioxide in conc. H_(2)SO_(2) an orange colouredc compound is formed. The oxidation Bumber of the metal in that compound is |
| Answer» SOLUTION :The compound is `H_(2)TiO_(4)` | |