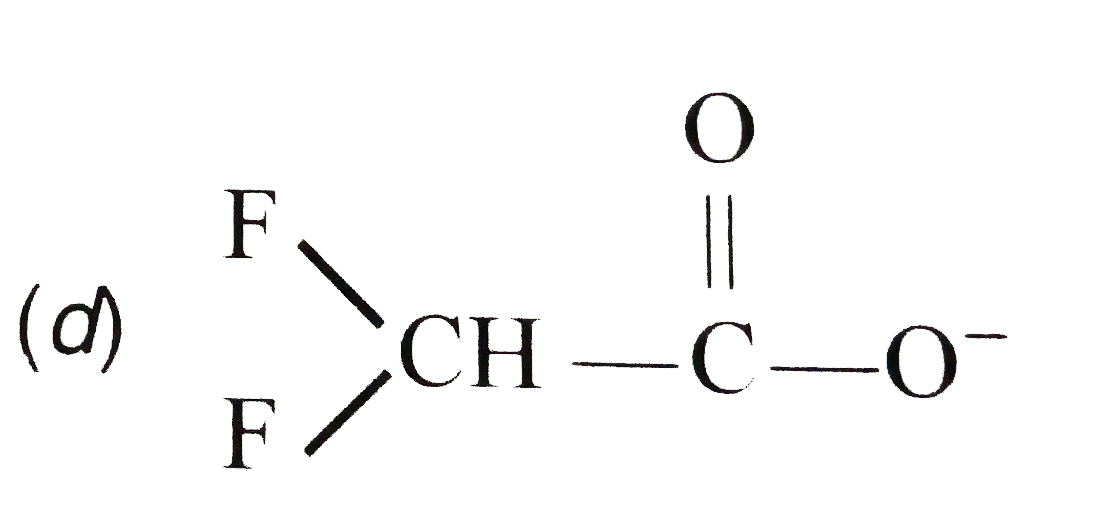Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which compound does not contain sp^(3) hybridised boron ? |
|
Answer» Product of the reaction of `H_(3)BO_(3)` and aqueoys SOLUTION of sodium HYDROXIDE |
|
| 2. |
What properties of water make it useful as a solvent ? What type of compounds can it (i) dissolve (ii) hydrolyse ? |
|
Answer» Solution :(i) Waterhas a high dielectric constant(79.39) and high DIPOLE moment (1.84D) . Because of these properties, water dissolves most of the inorganic (ionic) compounds and MANY covalent compounds. That is why water is called a UNIVERSAL solvent. Whereas ionic compounds dissolve in water due to iondipole interaction or solvation of ions, covalentcompounds such as alcohols, amines, urea, glucose, sugar, etc. dissolve in water due to H-bonding. (ii) Water can hydrolyse many oxides (metallic or non-metallic ), hydrides, carbides, nitrides, phosphides and other salts. In these reactions , `H^(+)` and `OH^(-)` ions of water interact with anions and cations respectivity leading to the formation of an acid or a base or both as shown below : `CaO(s) +H_(2)O(l) to Ca(OH)_(2)(aq)` `SO_(2)(g) + H_(2)O (l) to H_(2)SO_(3)(aq)` `CaH_(2)(s) + 2H_(2)O (l) to Ca(OH)_(2)(aq) + 2H_(2) (g)` `CaC_(2)(s) + 2H_(2)O (l) to Ca(OH)_(2)(aq) + UNDERSET("Acetylene")(HC -=CH(g))` |
|
| 3. |
Which of the following oximes is expected to exhibit both geometrical and optical isomerism |
|
Answer»

|
|
| 4. |
The reagent used to detect fluoride present in water in |
|
Answer» ALUM + CAO + `CaOCl_2` |
|
| 5. |
Why are Bohr's orbits called stationary states? |
| Answer» SOLUTION :This is because the energies of the ORBITS in which the electrons REVOLVE are FIXED. | |
| 6. |
Which of the following is not used in writing electronic configuration of an atom? |
|
Answer» AUFBAU principle |
|
| 7. |
Which of the following statements are not true ? |
|
Answer» An element with BCC structure has TWO atoms PER unit cell. |
|
| 8. |
Whatis usedin Rutherfordexperiment ? |
| Answer» Answer :B | |
| 9. |
Why are lithium salts commonly hydrated and those of the other alkali ions usually anhydrous? |
| Answer» SOLUTION :`Li^(+)`IONS are the smallest of all alkali metal ions. Due to small size, they can polarise water molecules, attach them and get hydrated easily in comparison to other alkali metal ions. This is why lithium salts are COMMONLY hydrated, e.g. LICI `2H_2O`. | |
| 10. |
Which of the following is a false statement about boric acid, H_3 BO_3? |
|
Answer» It is a strong tribasic acid. |
|
| 11. |
The velocities of two particles A and B are 0.05 and 0.02ms^(-1) respectively. The mass of B is five times the mass of A. The ratio of their de- Broglie's wavelength is |
|
Answer» `2:1` velocity of PARTICLES `B=0.02 ms^(-1)` Let the mass of particle A=X :. The mass of particle `B=5x` de-Broglie's equation is `lambda=h/(mv)` for particle A `lambda_(A)=h/(x xx0.5)` for particle B `lambda_(B)=h/(5x xx0.02)` E.q (i)//(ii) `lambda_(A)/lambda_(B)=(5x xx0.02)/(x xx0.05)` `lambda_(A)/lambda_(B)=2/1` or `2:1` |
|
| 12. |
Which of the following biphenyl is optically active? |
|
Answer»
|
|
| 13. |
The resonance effect is defined as 'the polarity produced in the molecule by the interaction of two pi-bonds or between a pi-bond and lone pair of electrons present on an adjacentatom.' The effect is transmitted through the chain. In positive resonance effect, the transfer of electrons is away from an atom or substituent group attached to the conjugated system. This electron displacement makes certain positions in the molecule of high electron densities. In negative resonance effect, the transfer of electrons is towards the atom or substituent group attached to the conjugated system. The correct statement regarding a carbonyl compound with a hydrogen atom on its alpha carbon, is |
|
Answer» a CARBONYL COMPOUND with a hydrogen atom on its alpha-carbon RAPIDLY EQUILIBRATES with its corresponding enol and this process is KNOWN as carbonylation |
|
| 14. |
The resonance effect is defined as 'the polarity produced in the molecule by the interaction of two pi-bonds or between a pi-bond and lone pair of electrons present on an adjacentatom.' The effect is transmitted through the chain. In positive resonance effect, the transfer of electrons is away from an atom or substituent group attached to the conjugated system. This electron displacement makes certain positions in the molecule of high electron densities. In negative resonance effect, the transfer of electrons is towards the atom or substituent group attached to the conjugated system. Which of the following carboxylate ions is the most stable ? |
|
Answer» `CH_(3)-overset(O)overset(||)(C)-O^(-)` |
|
| 15. |
Why are the following reactions not redox reactions ? BaCl_(2)(aq)+H_(2)SO_(4)(aq)toBaSO_(4)(s)+2HCl(aq) |
| Answer» Solution :GIVEN REACTIONS do not INVOLVE any change in OXIDATION number. | |
| 16. |
The resonance effect is defined as 'the polarity produced in the molecule by the interaction of two pi-bonds or between a pi-bond and lone pair of electrons present on an adjacentatom.' The effect is transmitted through the chain. In positive resonance effect, the transfer of electrons is away from an atom or substituent group attached to the conjugated system. This electron displacement makes certain positions in the molecule of high electron densities. In negative resonance effect, the transfer of electrons is towards the atom or substituent group attached to the conjugated system. Which of the following shows +M effect ? |
|
Answer» `- N(CH_(3))_(2)` |
|
| 17. |
The resonance effect is defined as 'the polarity produced in the molecule by the interaction of two pi-bonds or between a pi-bond and lone pair of electrons present on an adjacentatom.' The effect is transmitted through the chain. In positive resonance effect, the transfer of electrons is away from an atom or substituent group attached to the conjugated system. This electron displacement makes certain positions in the molecule of high electron densities. In negative resonance effect, the transfer of electrons is towards the atom or substituent group attached to the conjugated system. Which of the following does not show resonance effect ? |
|
Answer» Buta-1, 3-diene 
|
|
| 18. |
What is the coordination number of sodium in sodium oxide (Na_2O)? |
|
Answer» 6 |
|
| 19. |
The value of K_c for the reaction 3O_(2)(g) is 2.0 xx 10^(-50) and 25^(@)C . If equilbrium concentration of O_(2) in 25^(@)C is 1.6 xx 10^(-2) . What is the concentration of O_(3) ? |
|
Answer» Solution :`3O_(2)(g) hArrr 2O_(3)(g)` `K_(c) = ([O]^2)/([O_2]^3) or (2.0 XX 10^(-50)) = ([O_3]^2)/(1.6 xx 10^(-2))^3` or`[O_3]^2 = (2.0 xx 10^(-50)) xx (1.6 10^(-2))^3` `[O_3]^2 =(8.192 xx 10^(-56))^(1//2) = 2.86 xx 10^(-28) M` |
|
| 20. |
What is energy gap in band theory ? Compare its size in conductors, semiconductors and insulators. |
| Answer» Solution :Energy GAP in BAND theory means the gap of energy between VALENCE band and condcution band. The gap is in the order : Conductor lt SEMICONDUCTOR lt Insulator . | |
| 21. |
Which of the following statement about amorphous solids is incorrect ? |
|
Answer» They melt over a RANGE of TEMPERATURE |
|
| 22. |
What is the formula of brown ppt? |
| Answer» Answer :D | |
| 23. |
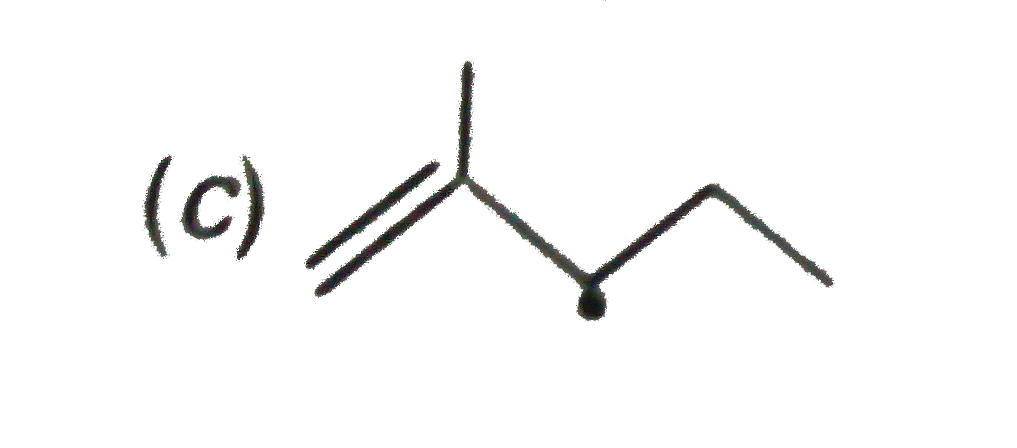
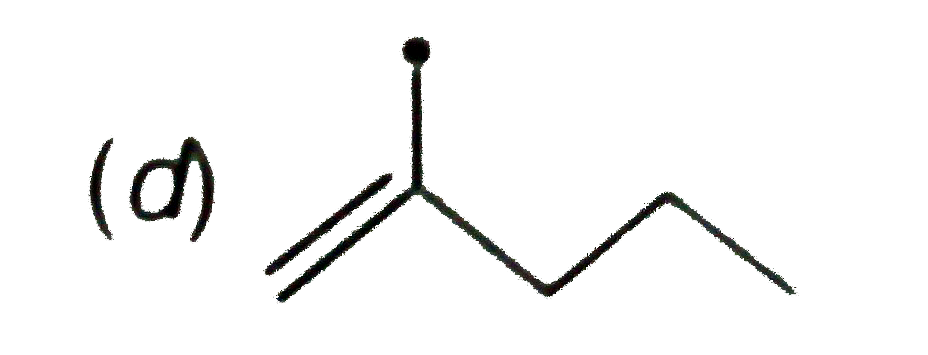
The resonance effect is defined as 'the polarity produced in the molecule by the interaction of two pi-bonds or between a pi-bond and lone pair of electrons present on an adjacentatom.' The effect is transmitted through the chain. In positive resonance effect, the transfer of electrons is away from an atom or substituent group attached to the conjugated system. This electron displacement makes certain positions in the molecule of high electron densities. In negative resonance effect, the transfer of electrons is towards the atom or substituent group attached to the conjugated system. Which of the following free radicials is the most stable ? |
|
Answer»
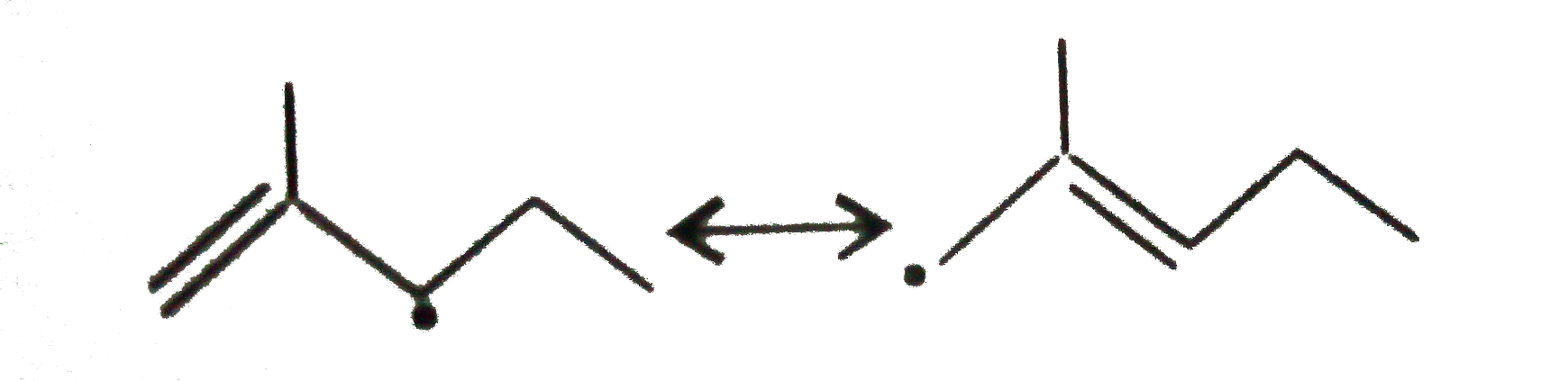
|
|
| 24. |
What is the difference between inductive effect and resonance effect? |
| Answer» SOLUTION :Due to inductive effect only the electron density of bond INCREASES near to ONE atom and DECREASES near to other atom so bond become polar. In resonance effect the POSITION of electrons of `pi` bond change which is hypothetical. Such arrangement is not real. | |
| 25. |
The resonance effect is defined as 'the polarity produced in the molecule by the interaction of two pi-bonds or between a pi-bond and lone pair of electrons present on an adjacentatom.' The effect is transmitted through the chain. In positive resonance effect, the transfer of electrons is away from an atom or substituent group attached to the conjugated system. This electron displacement makes certain positions in the molecule of high electron densities. In negative resonance effect, the transfer of electrons is towards the atom or substituent group attached to the conjugated system. Which of the following intermediates is pyramidal in shape ? |
|
Answer» `H_(3) C^(o+)` |
|
| 26. |
The resonance effect is defined as 'the polarity produced in the molecule by the interaction of two pi-bonds or between a pi-bond and lone pair of electrons present on an adjacentatom.' The effect is transmitted through the chain. In positive resonance effect, the transfer of electrons is away from an atom or substituent group attached to the conjugated system. This electron displacement makes certain positions in the molecule of high electron densities. In negative resonance effect, the transfer of electrons is towards the atom or substituent group attached to the conjugated system. What is the IUPAC name for : |
|
Answer» 5-isobutyl-2-methylheptane 4-ethyl-2, 7-dimethyloctane. |
|
| 27. |
Which is the correct order of ionic radii given below? |
|
Answer» `Ca^(2+) gt K^(+) gt Cl^(-) gt S^(2-) gt P^(3-)`  All have same (18) ELECTRONS, but nuclear charge INCREASE as a result order of IONIC radii decrease. |
|
| 28. |
Which of the following alkenes will show geometrical isomerism ? (i) Propene,(ii) But-2-ene , (iii) But-1-ene , (iv) 2, 3-Dimethylbut-2-ene |
| Answer» SOLUTION :(II) But-2-ene | |
| 29. |
Which of the following dissolves in water with a hissing sound? |
|
Answer» BLEACHING powder |
|
| 30. |
Which of the following represents largest pressure ? |
|
Answer» 1 atmosphere 1 bar `=10^(5)` Pa or N `m^(-2)`=0.987 atm 14.7 1b/sq inch =1 atmospheric pressure at SEA LEVEL Thus, 1 atm represents the largest pressure. |
|
| 31. |
What is X in the following reaction ? MgCl_(2) +2H_92) O to X+ 2HCl +H_(2)O |
| Answer» Solution :`MGCL _(2) +2H_(2)O to MgO+ 2HCl +H_(2)O` | |
| 32. |
Which of the following is an endothermic process? |
|
Answer» FIRST ELECTRON affinity of chlorine |
|
| 33. |
Write the formula of the compound of phosphorus which is obtained when conc. HNO_(3) oxidises? |
|
Answer» <P> Solution :`{:(2HNO_(3)toH_(2)O+2NO_(2)+[O]xx10),(P_(4)+10[O]toP_(4)O_(10)),(P_(4)O_(10)+6H_(2)Oto4H_(3)PO_(4)):}/{:P_(4)+20HNO_(3)tounderset("Orthophosphoric ACID")(4H_(3)PO_(4))+20NO_(2)+4H_(2)O:}` |
|
| 35. |
Which of the following is not a Lewis acid ? |
|
Answer» `AlCl_3` |
|
| 36. |
Which of the following has the most acidic hydrogen? |
|
Answer»
|
|
| 37. |
The stability and magnetic properties of a molecule can be well explained using the molecular orbital theory developed by F Hung and R.S.Mulliken.Draw the energy level diagram for the formation of the O_2 molecule. |
| Answer» SOLUTION :REFER to TEXT | |
| 38. |
Define bond order according to the M.O.theory. |
| Answer» Solution :Bond ORDER is DEFINED as ONE half of the difference between number of electrons in the bonding and the antibonding MOLECULAR orbitals. | |
| 39. |
Why a person surffering from high blood pressure is advised to take minimum quantity of common salt ? |
| Answer» Solution :Osmotic PRESSURE is directly proportional to the concentration of solutes: Our body FLUID contains a number of solutes. On TAKING large amount of salt, IONS entering into the body fluid thereby raises the concentration of solutes. As a result, osmotic pressure INCREASES which may rupture the blood cells. | |
| 40. |
What happens to the PH of water when NH_(4)Cl solid is dissolved in it and why? |
| Answer» Solution :PH Decrease: Because `NH_(4)CL` being salt of strong Acid and Weak base UNDERGOES Hydrolysis to give Acidic solution. | |
| 41. |
Whic of the following statements is incorrect ? |
|
Answer» `H_(2)O_(2)` has WEAK acidic property |
|
| 42. |
Which of the following is correct formula of ethyl acetate? |
|
Answer» `CH_(3)- UNDERSET(underset(O)(||))(C )-O - CH_(2)-CH_(3)` |
|
| 43. |
When does the transport of oxygen to different body cells stop ? |
|
Answer» SOLUTION :(i) Carbon monxide ( CO ) combines with haemoglobin ( HB) of the RBCs about 300times more readily than oxygen to form carboxy haemoglobin COMPLEX. `Hb + CO hArr HbCo` (ii) THUS, Hb will not be able to combine with oxygen to for oxyghaemoglobin complex. (iii) Hence the transport of oxygento different BODY cells does not occur ( stops ) . |
|
| 44. |
Which compound is used to remove the temporary hardness of water in Clark.s method. |
| Answer» Solution :In clark.s METHOD, the lime water `(Ca(OH)_2)` is USED to REMOVE the TEMPORARY hardness. | |
| 45. |
Underlined carbon is sp hybridised in- |
|
Answer» `CH_(3)CH=CH_(2)` |
|
| 46. |
Which elements isexpected tohave lowestfirstionizationenthalpy ? |
|
Answer» SR |
|
| 47. |
What is the action of alkali metals with oxygen (air) ? |
|
Answer» Solution :Alkali metals react with oxygen to FORM their oxides. EX. Lithium forms monoxide `4 Li + O_(2) overset(Delta)(to) 2Li_(2) O` (Lithium monoxide) SODIUM forms peroxide `2Na + O_(2) to Na_(2) O_(2)` (sodium peroxide) Others metals form super oxides as `M + O_(2) to MO_(2)` [Where M = K, Rb, Cs) |
|
| 48. |
The volume of 0.0168 mol of O_(2) obtained by decomposition of KClO_(3) and collected by displacement of water is 428 ml at a pressure of 754 mm Hg at 25^(@)C. The pressure of water vapour at 25^(@)C is |
|
Answer» 18 mm HG `=0.0168xx(0.0821xx1000xx760" ML "mm)xx(298)/(428)` 730 mm Pressure of moist gas=754 mm. Hence, pressure of water vapour =(754-730)mm=24 mm. Alternatively, volume of `0.0168" mol"` of `O_(2)` at STP `=0.0168xx22400=376.3" ml"` THUS, `V_(1)=376.3" ml", P_(1)=760" mm", T_(1)=273" K"` `V_(2)=428" ml",P_(2)=?,T_(2)=298" K"`. Calculate `P_(2)`. |
|
| 49. |
Which of the following mixtures can be seperated into its costituents by using concentrated sodium hydroxide ? |
|
Answer» `AI^(3+)` and `FE^(3+)` `Fe^(3+)+NaOHrarrFe(OH)_(3)` |
|
| 50. |
Which of the following statement is true? |
|
Answer» Allyl CATION is more STABLE than cyclo tri phenyl cation |
|