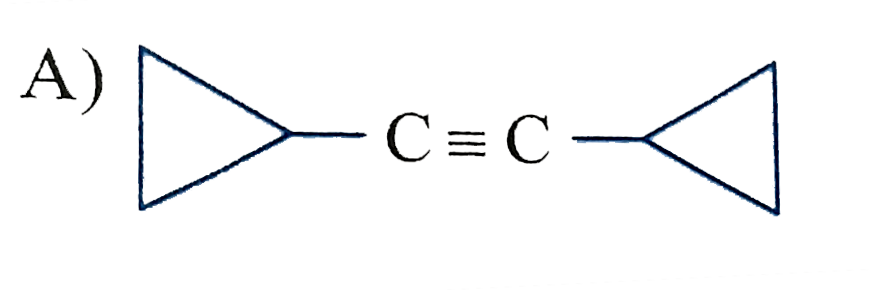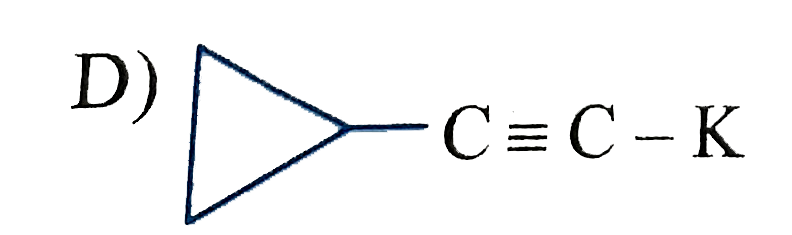Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What do you understand by nitration ? Give an example. |
| Answer» SOLUTION : Nitration is a GENERAL class of chemical processes for the introduction of a nitro group into an organic compound. The term ALSO is applied INCORRECTLY to the different process of forming nitrate esters between alcohols and nitric acid (as occurs in the synthesis of NITROGLYCERIN). | |
| 2. |
Writeelectromagneticspectrum. |
Answer» SOLUTION :THEREARE manytypesof electromagneticradiationswhichdifferfromoneanotherinwavelength.Theseconstitute What is calledelectromagneticspectrum .Thefigureof electromagenticspectrumis assunder :
|
|
| 3. |
Which is the most stable carbocation among the following ? |
|
Answer» Iso-propyl |
|
| 4. |
What is the chief product of reaction between beta-butylene chloride and alc. KOH/NaNH_(2) |
|
Answer» 1,2-butadiene |
|
| 5. |
Which of the following compounnds will not show resonance ? |
|
Answer»
 Since nitrogen can.t form FIVE BONDS: (More than OCTET) |
|
| 6. |
Which elements are semi metals ? |
| Answer» Solution :SILICON, Germanium, ARSENIC, Antimony and TELURIUM. | |
| 7. |
Which of the following has zero dipole moment? |
|
Answer» cis-2-butene |
|
| 8. |
What is dissociation constant of a weak base (K_(b)). |
|
Answer» Solution :The dissociation constant of a weak BASE is the ratio of the product of molar CONCENTRATIONS of the products of ionisation to the molar CONCENTRATION of the un-ionised base at equilibrium. `BOHhArrB^(+)+OH^(-)` `thereforeK_(B)=([B^(+)][OH^(-)])/([BOH])` |
|
| 9. |
The solution of which one of the following will be least effective in coagulation of Fe(OH)_(3) sol ? |
|
Answer» `K_(4)[FE(CN)_(6)]` |
|
| 10. |
While one of the following reactions does not correspond to the preparation of "synthetic gasoline" during the Fischer-Tropsch process? |
|
Answer» `CO+3H_(2) RARR CH_(4)+H_(2)O` |
|
| 11. |
Write the resonance structure of carbonate ion (CO_(3)^(-2)) |
Answer» SOLUTION :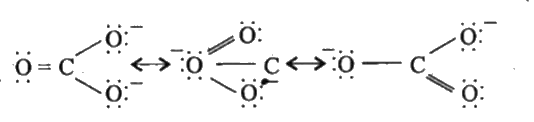
|
|
| 12. |
underset(heat)overset((CH_(3))_(3)COK^(+),DMSO)to Identify the product. |
|
Answer»
|
|
| 13. |
The solubility of Sr (OH)_(2) at 298K is 19.23 g/L of solution. Calculate the concentrations of strontium and hydroxyl ions and the pH of the solution. (Atomic mass of Sr = 87.6) |
|
Answer» Solution :Molar MASS of Sr `(OH)_(2) = 87.6+34=121.6 g "MOL" ^(-1)` Solubility of Sr `(OH)_(2) ` in moles `L^(-1)=(19.23g L^(-1))/(121.6g "mol"^(-1))=0.1581M` Assuming complete DISSOCIATION , Sr`(OH)_(2)rarrSr^(2)+2OH^(-)` `:. [Sr^(2+)]=0.1581 M, [OH^(-)]=2xx0.1581=0.3162`M `pOH=-log 0.3162=0.5, :. pH = 14-0.5=13.5` |
|
| 14. |
The recommended concentration of fluoride ion in drinking water is up to 1 ppm as fluoride ion is required to make teeth enamel harder by converting [3 Ca_3(PO_4)_2. Ca(OH)_2] to |
|
Answer» `[CaF_2]` |
|
| 15. |
Wrong statement of the following is |
|
Answer» Beryllium chloride has a CHAIN structure in the solid state |
|
| 16. |
Which species will undergo electrophilic substitution reaction most easily? |
|
Answer» Ph-OH |
|
| 17. |
Which of the following compound will be suitable for Kjeldahls method for nitrogen estimation? |
|
Answer»
|
|
| 18. |
Draw the structure of 4-cyclopropyl-4-ethyl-2-heptene |
|
Answer» |
|
| 19. |
Which of the following will increase the BOD of water supply? |
|
Answer» `CO_(2)` |
|
| 20. |
What happens when a ferromagnetic or anti-ferromagnetic or a ferrimagnetic solid is heated ? |
| Answer» SOLUTION :It CHANGES into PARAMAGNETIC at some TEMPERATURE. | |
| 21. |
When there are two electrons in the same orbital they have the spins |
|
Answer» `+(1)/(2), +(1)/(2)` |
|
| 22. |
What is limiting reagent ? |
|
Answer» |
|
| 23. |
When the rate of formation of reactants is equal to the rate of formation of products, this is known as, |
|
Answer» Chemical REACTION |
|
| 24. |
Write the Lewis dot structure of the nitrite ion (NO_(2)^(Θ)) . |
|
Answer» Solution :Solution. Step 1. N = 2,5, O = 2,6, - ve charge = 1 . ` therefore ` Total no of valence electrons in `NO_(2)^(-) = 5 + 2xx 6 + 1 = 18 = 9 ` pairs . Step 2. Skeletal structure of `NO_(2)^(-)` will be ON O Step 3. Puttin a single bond (ONE shared PAIR) between O and N and N and O, we have `[O:N:O]^(-)` Step 4. Putting the remaining7 pairs of electrons as lone pairs, 3 on each O and 1 on N (so that octets of O are completed), wehave `H:overset(. .)C:overset(. .)N:` Step 5. As octet of N is not complete, multiple bonding is required . As N is short of only one pair of elecrons . one pair from O should be shifted as shared pair between O and N or N and O. i.e., a double bond should EXIST. THUS, the structure should be  (called resonating structures, discussed later). |
|
| 25. |
Which of the following conditions shows the polluted environment. |
|
Answer» pH of rain water is 5.6. The amount of oxygen required by BACTERIA to break the organic matter present in a certain volume of sample of water is known as Biochemical Oxygen Demand. Clean water would have BOD value of 5 ppm whereas highly polluted could have BOD value of 17 ppm or more. Normally rain water has pH of 6. When water reacts with carbon dioxide in the atmosphere `H^+` on formed. When the pH of the rain water drops below 5.6, it is called acid rain. |
|
| 27. |
Which orbital isused by oxygen atom to form a sigma bond with other oxyge atom in O_(2) molecule? |
|
Answer» PURE p-ORBITAL |
|
| 28. |
Which of the following is propargyl group ? |
|
Answer» `-CH_2 - C -=CH` 
|
|
| 29. |
What are non-stoichiometric compounds? |
| Answer» Solution :If the actual RATIO of the CATIONS and ANIONS is not same as represented by the ideal chemical formula of the compound, it is called a non-stoichiometric compound. | |
| 30. |
Twenty millilitres of a gaseous hydrocarbon required 400 ml of air for complete combustion. The air contains 20% by volume of oxygen. The volume of gaseousmixture after explosion and cooling was found to be 380 ml.Volume of residual nitrogen is |
|
Answer» 300ml |
|
| 31. |
Twenty millilitres of a gaseous hydrocarbon required 400 ml of air for complete combustion. The air contains 20% by volume of oxygen. The volume of gaseousmixture after explosion and cooling was found to be 380 ml.Volume of O_(2) used is |
|
Answer» 70ml |
|
| 32. |
Twenty millilitres of a gaseous hydrocarbon required 400 ml of air for complete combustion. The air contains 20% by volume of oxygen. The volume of gaseousmixture after explosion and cooling was found to be 380 ml.Formula of the hydrocarbon is |
|
Answer» `C_3H_5` |
|
| 33. |
Twenty millilitres of a gaseous hydrocarbon required 400 ml of air for complete combustion. The air contains 20% by volume of oxygen. The volume of gaseousmixture after explosion and cooling was found to be 380 ml.Volume of CO_(2) is |
|
Answer» 40ml |
|
| 34. |
Write the structure of the following compounds.NH_(3) |
|
Answer» Solution :A = N ATOM , B = H atoms ` (##SUR_CHE_XI_V02_QP_19_E01_043_S01.png" width="80%"> |
|
| 35. |
The structure of triphenylmethyl cation is given below. This is very stable and some of its salts can be stored for months. Exaplin the cause of high stability of this cation |
Answer» SOLUTION :Due to resonance, the +ve charge can MOVE at both the o- and p-position of each benzene ring. As a result, three resonance STRUCTURES (I, II and III) are possible for each benzene ring as SHOWN below. Total ten resonance structures are possible. This, (I) is MAIN and other nine are resonance structure 
|
|
| 36. |
The species : H_2O,HCO_3^(-) , HSO_4^(-)and NH_3 can act both as Bronsted acids and bases. For each case give the corresponding conjugate acid and base. |
|
Answer» SOLUTION :CONJUGATE acid : `H_2O` of `H_3O^(+) , HCO_3^(-)` of `H_2CO_3 , HSO_4^(-)` of `H_2SO_4` and `NH_3` of `NH_4^(+)` Conjugate BASE : `H_2O` of `OH^(-) , HCO_3^(-)` of `CO_3^(2-) , HSO_4^(-)` of `SO_4^(2-)` and `NH_3` of `NH_2^(-)` |
|
| 37. |
The theory of resonance was developed primarily by Pauling in the 1930s. According to this theory , many molecules and ions are best described by writing two or more Lewis structure and considering the realmolecule of ion to be a composite of these structures .They are also sometimes referred to as resonance structures or resonance contributors . We show that the real molecule or ion is a resonance hybrid of the various contributing structures by interconnecting them with double headed arrows . Resonance structures are not in equilibrium with each other . . Stability order of the following resonatin structure will be |
|
Answer» `I GT II gt III gt IV` |
|
| 38. |
The theory of resonance was developed primarily by Pauling in the 1930s. According to this theory , many molecules and ions are best described by writing two or more Lewis structure and considering the realmolecule of ion to be a composite of these structures .They are also sometimes referred to as resonance structures or resonance contributors . We show that the real molecule or ion is a resonance hybrid of the various contributing structures by interconnecting them with double headed arrows . Resonance structures are not in equilibrium with each other . . Find out the stability order of intermediate . |
|
Answer» `I GT II gt III gt IV` |
|
| 39. |
(Total number of monochloro structural products) |
|
Answer» 2 
|
|
| 40. |
Which pair of species have same percentage of carbon? |
|
Answer» `CH_(3)COOH` |
|
| 41. |
Why are pentahalides more covalent than trihalides ? |
|
Answer» Solution :The elements of group 15 have five electrons (two in the s-and three in the p-orbitals) in their respective VALENCE SHELLS. Since it is difficult ot loss all the three electrons of form `E^(3+)` or even more difficult to lose all the five valence electrons (two s- and three p-) to form `E^(5+)` ions, therefore, higher elements have no TENDENCY to form ionic COMPOUNDS. Instead they form COVALENT compounds by sharing of electrons. Since elements in the +5 oxidation state have less tendency to loss electrons than in the +3 oxidation state, therefore, elements in the +5 oxidation state are more covalent than in the +3 oxidation state. In other words, pentahalides are more covalent than trihalides. |
|
| 42. |
When CO_(2)is passed through the solution of slaked lime it forms.... |
|
Answer» PARTIALLY soluble `CaCO_(3)` |
|
| 43. |
Which is the best description of the behaviour of bromine in the reaction given below H_(2)O+Br_(2) rarr HOBr+HBr |
|
Answer» Oxidised only |
|
| 44. |
What is the harmful effect of water having pH value less than 6.5 ? |
|
Answer» It decreases the effect of chlorination. |
|
| 45. |
Which one of the following is hardest compound of the following. |
|
Answer» Boran carbide |
|
| 46. |
Which of the following groups of ions makes the water hard ? |
|
Answer» Sodium and BICARBONATE |
|
| 47. |
What is the relationship between pK_(a)andpK_(b) values where K_(a)andK_(b) represent ionization constants of the acid and its conjugate base respectively? |
| Answer» SOLUTION :`pK_(a)+pK_(B)=pK_(W)=14`. | |
| 48. |
Which of the following is/are true with respect to carbon -12? |
|
Answer» relative atomic MASS is 12 u |
|
| 49. |
The difference in molecular weights of two consecutive members of a homologous series is |
|
Answer» |
|
| 50. |
Which of the following is least reactive towards electrophillic addition |
|
Answer»
 is LEAST reactive TOWARDS ELECTROPHILIC addition * Because `underset(NO_2)(C)H=CH_2` is electroedanating group equile is ELECTRONE group , So the electron density is less in this compound while. is LEAST reactive TOWARDS ELECTROPHILIC addition * Because `underset(NO_2)(C)H=CH_2` is electroedanating group equile is ELECTRONE group , So the electron density is less in this compound while.
|
|