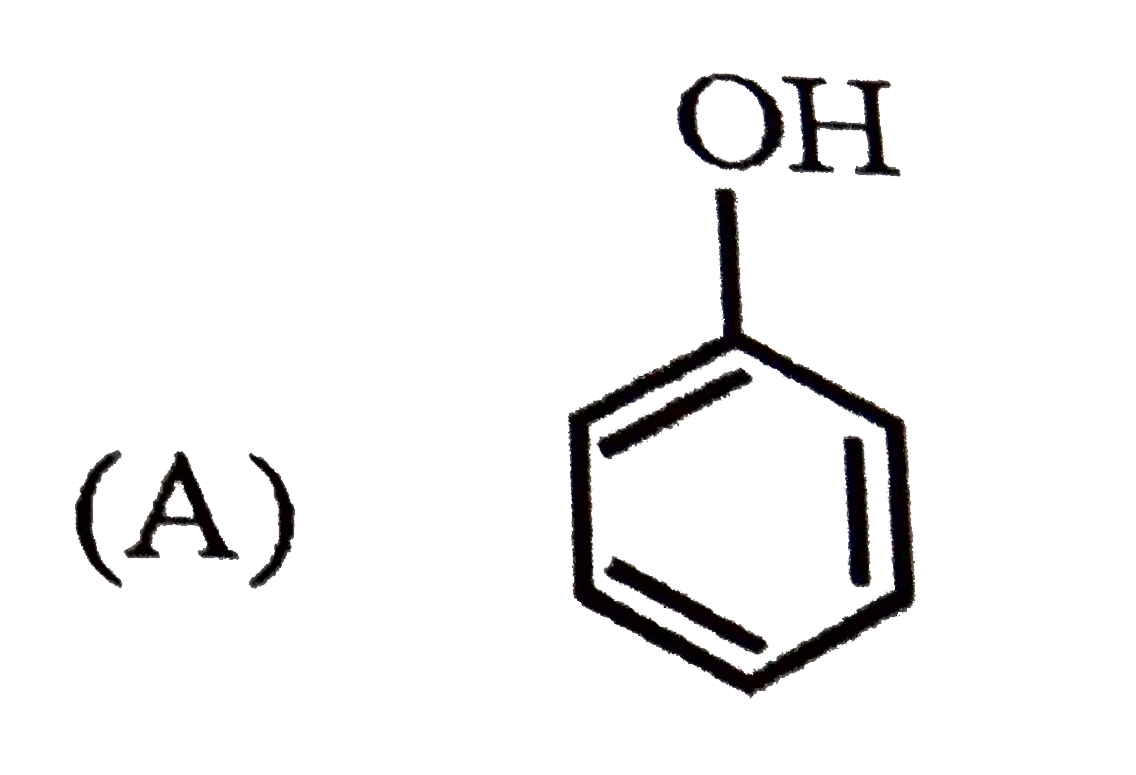Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which statement is not true about potash alum |
|
Answer» It's EMPIRICAL formula is `KAl (SO_(4))_(2). 12H_(2)O` |
|
| 2. |
Which compounds are found as impurity in impure sodium chloride ? |
| Answer» SOLUTION : Compounds like sodium sulphate, calcium sulphate, calcium chloride and MAGNESIUM chloride are found to be a IMPURITY in IMPURE sodium chloride. | |
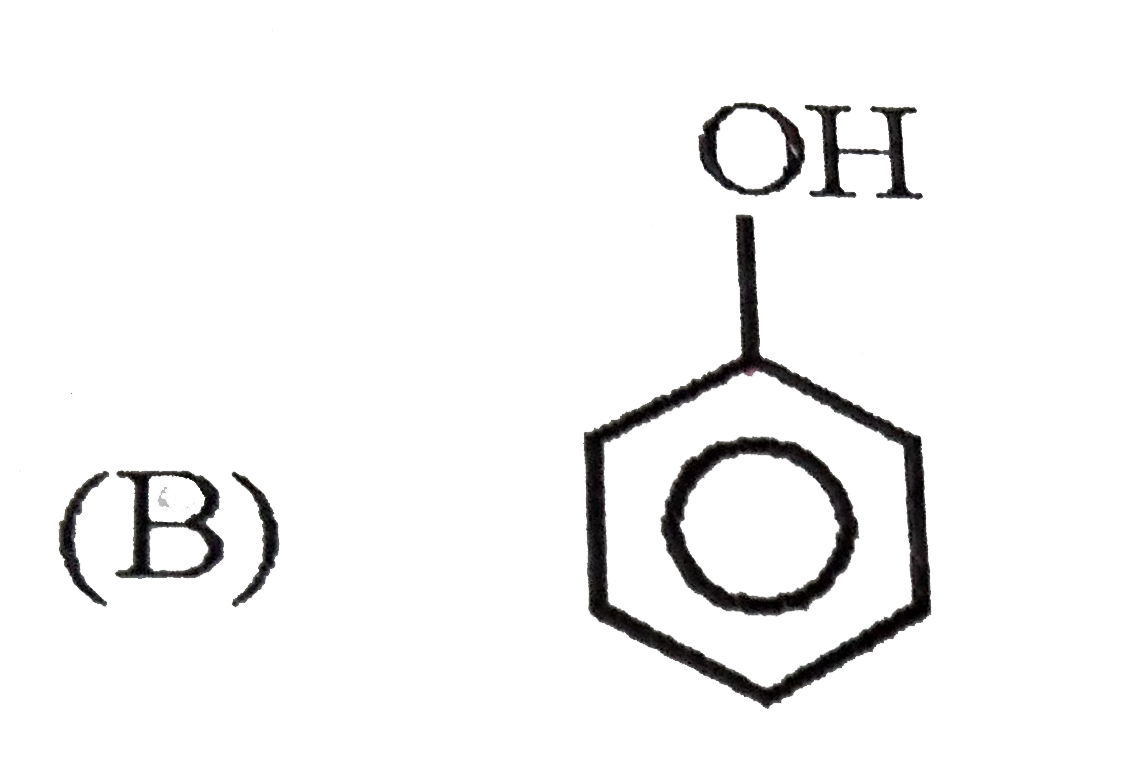
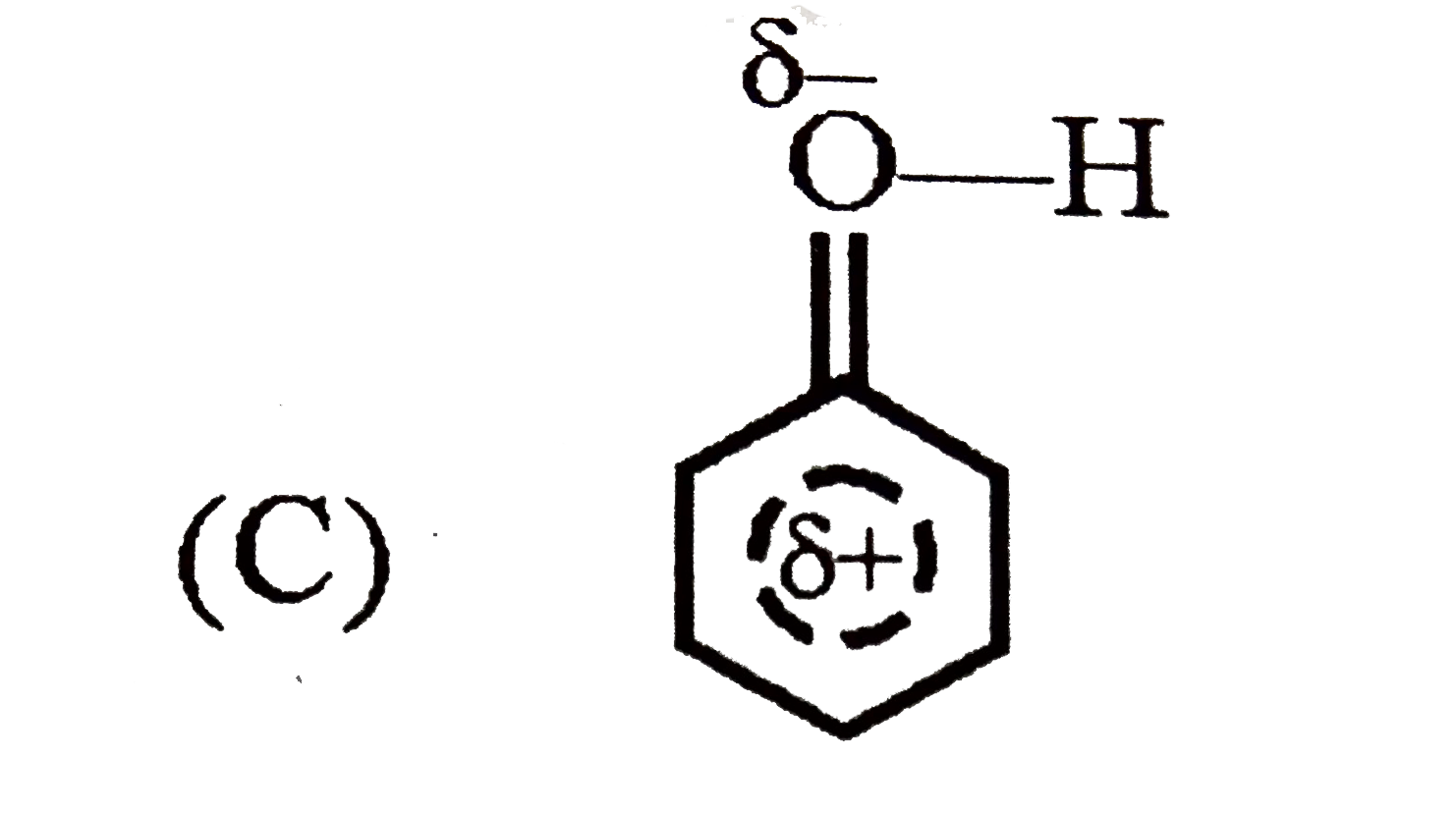
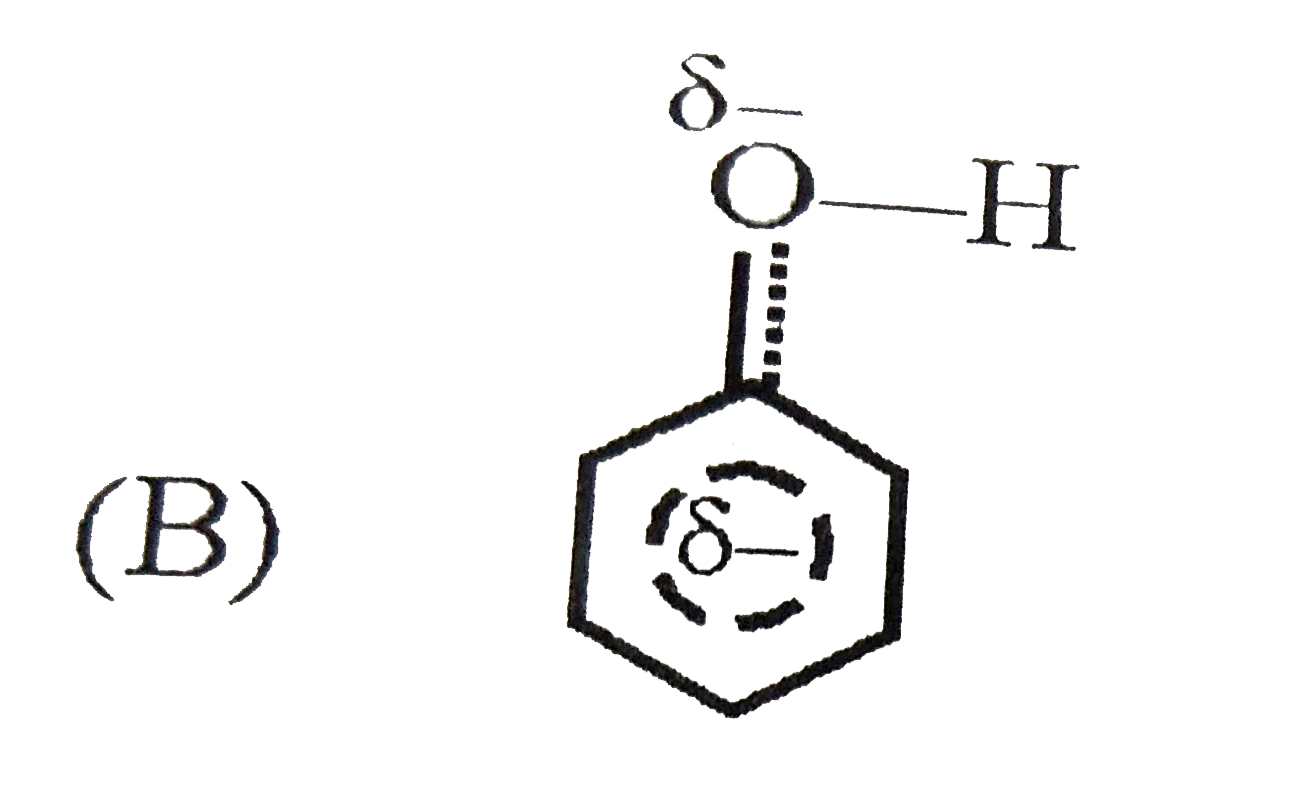
| 3. |
Which of the following best represents the phenol molecule ? |
|
Answer»
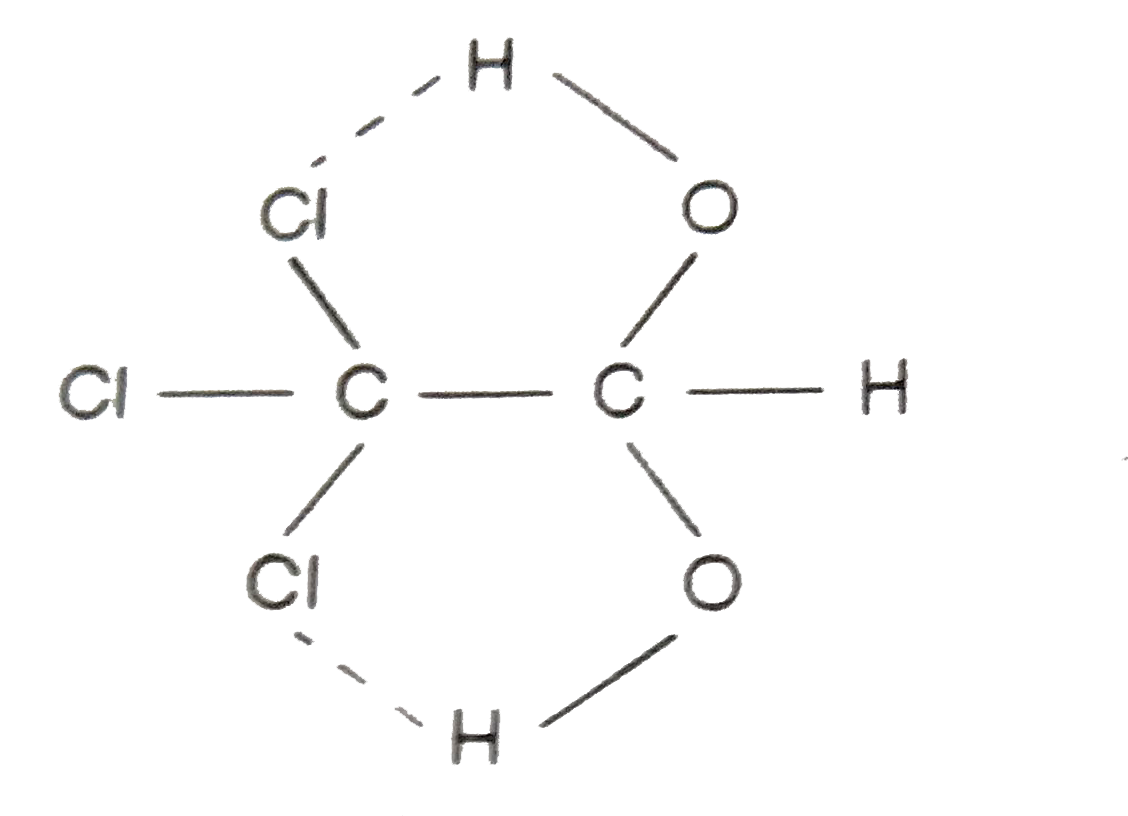
|
|
| 4. |
What is the difference between the terms hydrolysis and hydration ? |
|
Answer» Solution :INTERACTION of `H^(+) and OH^(-)` with the anion and the cation of a salt respectively original ACID and the original base is called hydrolysis. For example, `underset("Salt")(Na_(2)CO_(3)) + 2H_(2)O to underset("Base")(2NaOH)+ underset("Acid")(H_(2)CO_(3))` Hydration, on the other hand, means addition of `H_(2)O` to ions or moleculesto form hydrated ions on hydrated salts. For example, `underset("Salt")(Na^(+)Cl^(-)(s)) + H_(2)O(l) to Na^(+)(aq) + Cl^(-)(aq)` `underset("Colourless")(CuSO_(4)(s)) + 5H_(2)O(l) to underset("(Blue)")(CuSO_(4).5H_(2)O(s))` |
|
| 5. |
which of the following compounds on reaction with NaOh and H_(2)O_(2) gives yellow colour ? |
|
Answer» `ZN(OH)_(2)` |
|
| 6. |
What is the hybrid state ofBeCl_(2) ? What will be the change in the hybrid state ofBeCl_(2)in the solidstate ? |
|
Answer» <P> Solution :In the vopour stateat hight temperature , `BeCl_(2)`existsas linear molecule , `Cl - Be - Cl`. The hybridizationof the central atom sp . In the solid state, it has a polymericwith chlorine bridges as follows : 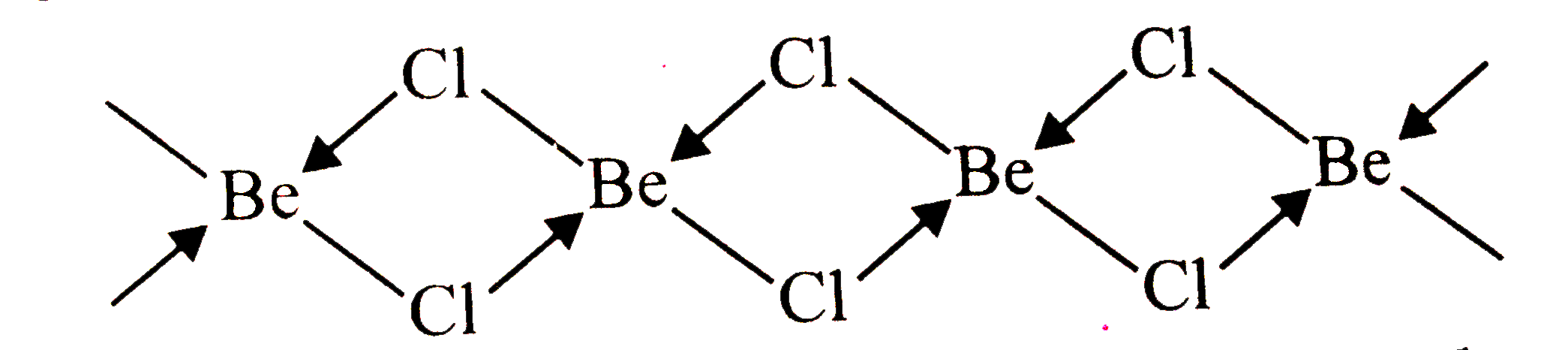 Two Clatoms are linked to Be atom by two coordinate bonds and two by covalent bonds. Forthese bonds to be formed , Be in the excited state with the configuration` 1s^(2)2s^(1) 2 p_(X)^(1) 2p_(y) ^(0) 2p_(Z)^(0)`undergoes ` sp^(3)`hydridisation . Two half-filled HYBRID orbitals will form normal covalent bonds with two Cl atom. The other two Cl atoms are COORDINATED to Be atom by donating ELECTRON pairs into the empty hybrid orbitals |
|
| 7. |
Two gases A and B present separately in two vessels X and Y at the same temperature and pressure with molecular weights M and 2M respectively are effused out. The orifice in vessel X is circular while that in Y is a square. If the radius of the circular orifice is equal to that the length of the square orifice, the ratio of rates of effusion of gas A to that of gas B is |
|
Answer» `PI sqrt(2)` |
|
| 8. |
What amount of CaO is produced by 1 gm of calcium with oxygen. |
|
Answer» |
|
| 9. |
Write the following numbers in exponential notation (i) 4250000 (ii) 0.0008432 |
|
Answer» |
|
| 10. |
Which of the folllowing species contains three bond pairs one lone pair around the central atom ? |
|
Answer» `H_(2)O` 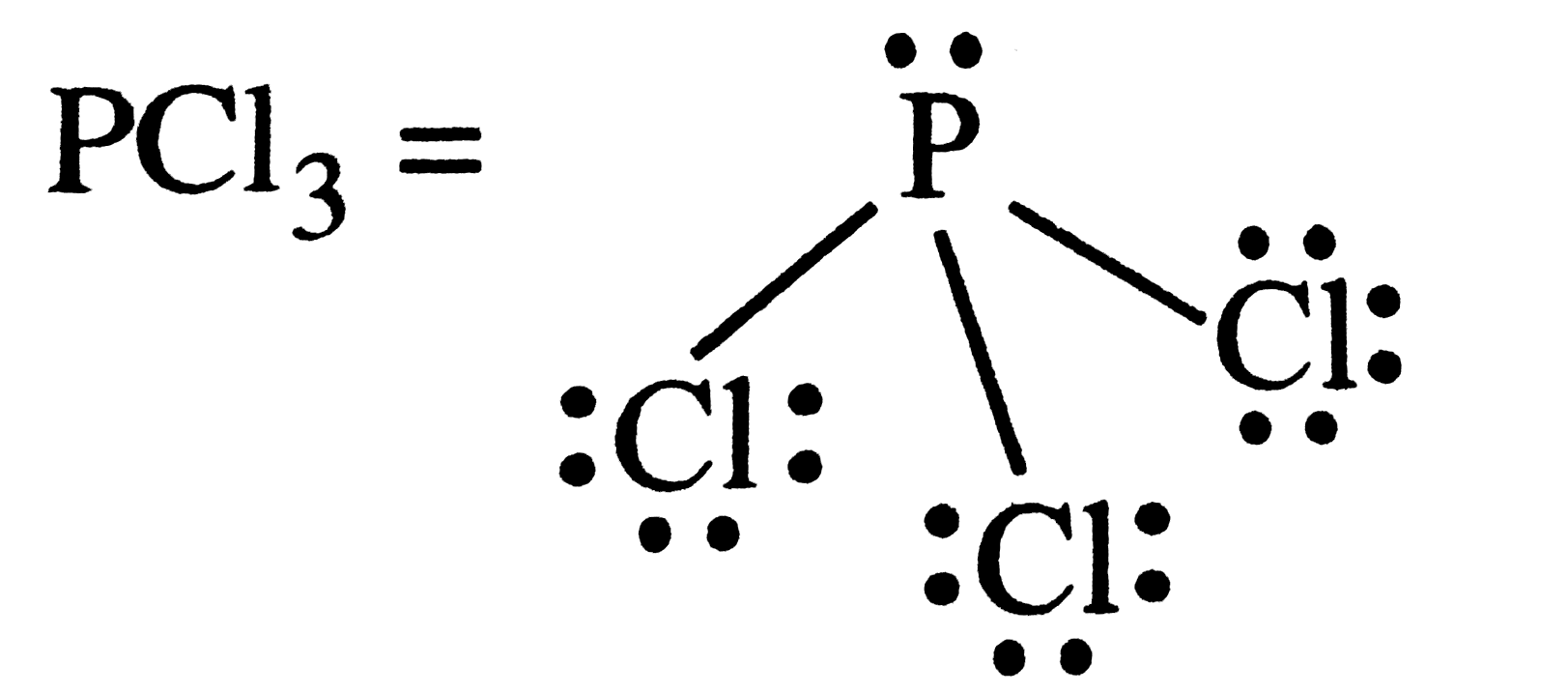 Each bond represents one bond pair . |
|
| 11. |
What is Epsom salt ? What is the action of heat on it ? |
| Answer» SOLUTION :`MgSO_(4) . 7 H_(2)O , MgSO_(4) . 7H_(2)O overset(423 K) underset(-6H_(2)O)(to) MG SO_(4) . H_(2)O overset(573K)underset(-H_(2)O)(to) MgSO_(4) overset("Heat")(to) MgO + SO_(3)`. | |
| 12. |
The rms speed of N_2 molecules in a gas is u. If the temperature is doubled and the nitrogen molecules dissociate into nitrogen atoms, the rms speed becomes : |
|
Answer» Solution :`(C_1)/(C_2) = sqrt((T_1)/(M_1) xx (M_2)/(T_2)) implies U/(C_2) = sqrt((T_1)/(28) xx 14/(2T_1))` `(U_2)/(C_2^2) = 1/4 implies C_2 = 2U`. |
|
| 13. |
Which one decrease the amount of CO_2 in atmosphere? |
|
Answer» PLANTS |
|
| 14. |
Which gas is produced when calcium nitride (Ca_(3)N_(2)) is hydrolysed by water ? |
|
Answer» `N_(2)` |
|
| 15. |
Which of the following on reduction with LiAlH_(4) will give ethyl alcohol ? |
|
Answer» `(CH_(3)CO)_(2)O` (B) `CH_(3)COCl + 4[H] overset(LiAlH_(4))rarr CH_(3)CH_(2)OH + HCl` (C) `CH_(3)COOC_(2)H_(5) + 4[H] overset(LiAlH_(4))rarr 2C_(2)H_(5)OH` Amides on reduction with `LiAlH_(4)` give corresponding AMINES |
|
| 16. |
What is the grade of H_2O_2 whose concentration is 3.125m for selling in open market ? |
|
Answer» 30 |
|
| 17. |
What are the uses of calcium hydroxide? |
|
Answer» Solution :Calcium hydroxide is used (i) in the preparation of mortar, a building MATERIAL. (II) in white wash due to its DISINFECTION nature. (iii) in GLASS making and tanning industry. (iv) for the preparation of bleaching POWDER and for the purification of sugar. |
|
| 18. |
Which bond is stronger sigma or pi? Why ? |
|
Answer» Solution :Sigma bonds `(sigma)` are stronger than Pi bonds `(pi).` Because, sigma bonds are formed from bonding orbitals directly between the nuclei of the bonding atoms, resulting in greater OVERLAP and a strong sigma BOND (AXIAL OVERLAPPING). (ii) Pi bonds results from overlap of atomic orbitals that are in contact through two areas of Overlap (LATERAL overlapping). Pi bonds are more diffused bonds than sigma bonds. |
|
| 19. |
What do you mean by ozone hole ? What are its consequences ? |
|
Answer» Solution :With the depletion of ozone layer, more UV RADIATION filters into troposphere. UV radiations lead to ageing of skin, cataract, sunburn, skin cancer, killing of many phytoplanktons, damage to fish productivity ETC. Plant proteins get easily affected by UV radiations which leads to the harmful mutation of cells. It also increases EVAPORATION of SURFACE WATER through the stomata of the leaves and decreases the moisture content of the soil. Increase in UV radiations damage paints and fibres, causing them to fade faster. |
|
| 20. |
What is meant by the term average bond enthalpy ? Why is there difference in bond enthalpy of O - H bond in ethanol (C_(2) H_(5) OH) and water ? |
|
Answer» Solution :The similar bonds in a molecule do not possess the same bond ENTHALPIES. e.g., in `H_(2)` O (H-O-H) molecule after the breaking of first O-H bond , the secon O-H bond undergoes some change because of changed CHEMICAL environment . Therefore, in polyatomic molecules the TERM mean or average bond enthalpy is used It is obtained by dividing total bond dissociation enthalpy by the number of bonds BROKEN. e.g. `H_(2)O_((g)) rarr H(g) + OH_((g)) ` `Delta_(a) H_(1)^(0) = 502 " kJ mol"^(-1) OH_((g)) rarr H + O_((g))`, `Delta_(a) H_(2)^(0) = 427 KJ mol^(-1)` Average 0-H bond enthalpy =`(502 + 427)/(2)` = 464.5 kJ `mol^(-1)` The bond enthalpies of O - H bond in `C_(2)H_(5)` OH and `H_(2)O` are different because of the different chemical (electronic) environment around OXYGEN atom. H - C - `underset(H)underset(|)overset(H)overset(|)(C) - underset(H)underset(|) overset(H)overset(|)(O) - H , H - O - H ` ` ""(C_(2) H_(5) OH) "" (H_(2) O)` |
|
| 21. |
Which is incorrect statement in respect of Mendeleev's periodic table ? |
|
Answer» It has made the study of elements easier and systematic |
|
| 22. |
Which of the following products is obtained when methyl magnesium bromide reacts with ethyl alcohol ? |
|
Answer» Acetone |
|
| 23. |
Which of the following statements above hydrogen is incorrect ? |
|
Answer» HYDROGEN has three isotopes of which tritium is the most common. |
|
| 24. |
The vapoour density of a mixture containing NO_(2) and N_(2)O_(4) is 38.3 at 300 K. the number of moles of NO_(2) in 100 g of the mixture is approximately |
|
Answer» 0.44 |
|
| 28. |
Which type of isomerism is shown by 2,3-dichlorobutane ? |
|
Answer» diastereomerism 
|
|
| 29. |
Which of the following statements about inductive effect is correct? |
|
Answer» It involves electrons in `sigma` bond |
|
| 30. |
When ethylene glycol is heated with acidified potassium permaganate, the main organic compound obtained is : |
|
Answer» OXALIC ACID |
|
| 31. |
Writethe products A & B for the followingreaction CI-CH_(2)-CH_(2)-Ci underset(HCI)overset(KOH)rarr(A)underset(HCI)overset(KOH)rarr(B) |
Answer» SOLUTION :
|
|
| 32. |
When acetylene is passed through dil. H_(2)SO_(4) in the presence of the HgO_(4), the compound formed is |
|
Answer» Ether |
|
| 33. |
Which is the composition of fusion mixture ? |
|
Answer» `K_(2)CO_(3) +NH_(4)Cl` |
|
| 34. |
Which of the following reaction will shift in forward direction. When the respective change is made at equilibrium |
|
Answer» `N_(2)(g)+3H_(2)(g)hArr2NH_(3)(g)` increase in pressure at eq. |
|
| 35. |
Which of the following sets of quantuin numbers represents the highest energy of an atom ? |
|
Answer» n = 3 , L = 0 , m = 0, s = + `(1)/(2)` n = 3, l = I represents 3p orbital n = 3, l = 2 represents 3d orbital n = 4, l = O represents 4s orbital The order of increasing energy of the orbitals is 3s `LT 3p lt 4s lt 3d`. |
|
| 36. |
What fraction (n/N) of the lattices sites are vacant at 298 K for a crystal for which the energy required to make a defect is 1 eV (1 eV=1.602xx10^(-19) J ) ? |
|
Answer» Solution :If `E_upsilon` is the energy REQUIRED to create one VACANT SITE, then by Boltzmann formula `n/N=e^(-E_upsilon //kT)` (k=Boltzmann CONSTANT =`R/N_0`) orIn `n/N=-E_upsilon / "kT"` or log `n/N=- E_upsilon / "2.303 kT"=(1.602xx10^(-19))/(2.303(8.314//6.023xx10^23)xx298)` `=-0.00169185xx10^4 =-16.9185=bar(17).0815` `n/N`=Antilog `bar(17).0815=1.206xx10^(-17)` |
|
| 37. |
The RMS velocities of two gases at the same temperature are u_1 and u_2 their masses are m_1 and m_2 respectively. Which of the following expression is correct? |
|
Answer» `(m_2)/(u_1) = (m_2)/(u_2)` |
|
| 38. |
What happens whensodium metal is dropped in water ? |
|
Answer» SOLUTION : When Na metal is DROPPED in water, it reacts violently to form sodium hydroxide and hydrogen GAS. The chemical equation involved in the reaction is: `2Na_((s))+2H_(2)O_((l)) to 2NaOH_((aq))+2H_(2(g))` |
|
| 39. |
Write short note on the following bond parameter . Resonance |
| Answer» SOLUTION :Resonance : Whenever we write Lewis structure for a moleculemore than one VALID structures are POSSIBLE in which the relative position of the attoms are same. They only differ in position of bonding and LONE paid of electrons. Such structures are called resonance structures and this phenenomenon is called resonance. | |
| 40. |
Write the complete chemical equation for the following . Oxidation of 4-hydroxy phenol |
Answer» SOLUTION :
|
|
| 41. |
Water is said to be polluted if the D.O value of water is less than |
|
Answer» 152 ppm |
|
| 42. |
X and Y has R_(f) 0.75 and 0.25 respectively. In column chromatrogaphy. Which is obtained first? |
| Answer» Solution :Y is OBTAINED first as it `R_(F)` is LESS. So it REMAIN at lower in column comes out first. | |
| 43. |
The total number of atomic orbitals in fourth energy level of an atom is |
|
Answer» 4 `= N^(2) = 4^(2) = 16` |
|
| 44. |
Vapour pressure of pure water at 298 K is 23.8 mm Hg. 50 g of urea (NH_(2) CONH_(2)) is dissolved is 850 g of water. Calculate the vapour pressure of water for this solution and its relative lowering. |
|
Answer» Solution :Here `P _(1) ^(0) =23.8mm` `W _(2)= 50 g` `M_(2) `(urea) `=60g mol^(-1)` `W_(1)= 850G` `M _(1) `(Water) `= 18g mol ^(-1)` Here we have to CALCULATE `P _(S)` Applying Raoult.s law, `(P ^(0) - P_(S))/(P^(0))= (n_(2))/( n _(1) + n _(2)) = ((W _(2))/(M _(2)))/((W _(1))/(M _(1)) + (W_(2))/(M _(2)))` ` = ((50)/(60))/((850)/(18) + (50)/(60))= ( 0.83)/(48.05) =0.017` Thus, relative lowering of VAPOUR pressure `=0.017` Substituting `P^(0) =23.8` mm Hg `(23.8 -P_(s))/(P _(S)) = 0.017` We get, `23.8 - P_(S) = 0.017 P _(S)` `P _(S) =23.4 mm Hg` Thus, vapour pressure of water in the solution `=23.4` mm Hg |
|
| 45. |
Write Huckel rule and explain with suitable example. |
|
Answer» Solution :Benzene was considered as parent .aromatic. compound. Now the name is applied to all the ring system whether or not having benzene ring, possessing following CHARACTERISTICS. (a) Requirements for aromatic character in compound : (i) Compound must havr cyclic (ring) structure. (ii) Cyclic compound must have all atoms in ONE planer structure. (iii) Complete delocalisation of the `pi` electronsin the ring. (iv) PRESENCE of `(4n+2)pi` electrons in the rinf where .n. is an inetger (n=0, 1, 2,...). This is often referred to as Huckel Rule. (b) Huckel.s rule for aromaticity : If the number of electrons in planar cyclic compound equals to (4n+2), then these compounds possess aromatic properties, (aromaticity) where n = number of cyclic structures, (4n+2) = total delocalized electron of atoms present in aromatic ring). (c) Aromatic hydrocarbon compounds : (i) Benzene is aromatic in nature. Number of rings in benzene = n =1 NUmber of double bonds in benzene = 3 so, number of `pi2(p)` electrons = 6 `therefore` According to Huckel rule, if (4n+2) `pi` electrons are present in structure then it is considered as aromatic. So for benzene , n=1 and (4n+2) = 4(1) + 2 = 6, so benzen obeys Huckel rules and benzene is aromatic in nature and it possess aromatic characteristics.  (ii) Cyclopentadienyl anion aromatic in nature. According to Huckel rule, n = 1 and hence (4n+2) = 6 In cyclopentadienail anion two double bond have `2 xx 2 = 4PI` + two lone `pi` electrons, so it is aromatic in nature.  (iii) Cycloheptatrienyl cation is aromatic in nature. Number of ring = n = 1 `therefore (4n+2) = 6`  In this compound three double bonds are present, so no. of `pi` electrons `= (3 xx 2) = 6`. So it follows Huckel rule, so cycloheptatriene, n = 1(4n+2)=6, cation is aromatic in nature. It possess positive charge `sp^(2)` carbon so it also possess planarity. (iv) Nepthalene is aromatic in nature.  Number of ring = n= 2 It having five double bonds so no. of `pi` electrons `= (5 xx 2) = 10` electrons Napthalene follows Huckel rules and so it is aromatic INNATURE. (v) Anthrecene and Phenanathrene are aromatic compounds. 
|
|
| 46. |
What is compressibility factor (Z) ?(i) deviation factor (Z = 1 )(ii) Z gt 1(iii) Z lt 1(iv) deviation graph and (v) relation between molar volume and Z. |
|
Answer» Solution :Compressibility factor (Z) : Deviation between real gas and idea gas is known as compressibility factor (Z). `Z=(pV)/(nRT) ""`…..(Eq. -i) where, Z = 1 Z = Compressibility factor of ideal gas deviation BEHAVIOUR, `Z =(V_("real"))/(V_("ideal"))=("real molar volume")/("ideal molar volume")` .......(constant T) For `Z = 1, Z gt 1, Z lt 1` is give in points(i), (ii), (iii) (i) Z = 1 OR 1 deviation factor : For ideal gas Z = 1 at all TEMPERATURE and pressure , because pV = nRT. If Z = 1 then gas is ideal gas and `Z to p` graph is parallel line. At very low pressures all gases shown have Z = 1 and behave as an ideal gas. Ideal gas law, follow at Z = 1 at Boyle temperature of Boyle point at real pressure. (ii) `Z gt 1` OR Positive deviation : At high pressure all real gases shows `Z gt 1` they are less compressible than ideal gas. If `Z gt 1` than permanently gas. `N_(2), H_(2)O_(2)` shows positive deviation, `Z to p` graph SHOW positive deviation. According to diagram for `H_(2)` gas at high pressure `Z gt 1`. When pressure increases value of Z becomes more positive. Ideal gas shows positive deviation of higher than Boyle.s point and value of a are higher than are then intermolecular attraction force are weak. (iii) `Z lt 1` OR negativedeviation : At intermediate pressure, most gasses have `Z lt 1`. For `O_(2), CH_(4), CO_(2)` gas shows negative deviation, `Z to p` graph shows negative deviation. According to diagram, `Z lt 1` at lower pressure. Initially pressure increases than Z decreases, after that z cross ideal gas line. At lower temperature then Boyle.s point value of Z decreases at increases pressure, which REACTED at minimum value and pressure is continuously increases. (iv) Graph between `Z to p` for some gases :  (v) Relation between Z real gas and ideal molar volume : `Z = (pV_("real"))/(nRT) ""`.....(Eq. -i) If gas is an ideal, `V_("real")=(nRT)/(p) ""`......(Eq. -iii) Value of `(nRT)/(p)` is put in (iv) then we get equation (iii). `Z=(V_("real"))/(V_("ideal")) ""`....(Eq. -v) `THEREFORE Z =("Real molar volume")/("Ideal molar volume")`......(constant T) ..Compressibility factor is ratio of real molar volume and ideal molar volume... |
|
| 47. |
Which property of colloidal solution is independent of charge on the colloidal particles |
|
Answer» Electrophoresis |
|
| 48. |
Which force present in Hydrogen bond ? |
| Answer» Solution :It is special type of VAN DER WAALS DIPOLE - Dipole interaction FORCE. | |
| 49. |
Write general chemical reaction, which show the dissolution of alkali metal in liquid ammonia. |
| Answer» SOLUTION :`M+(X+y)NH_(3) to [M(NH_(3))_(x)]^(+)+[E(NH_(3))_(y)]^(-)` | |