Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following atatements are correct for above rreaction ? |
|
Answer» number of possible enolates are four |
|
| 2. |
Which is used as an antifreeze ? |
|
Answer» Glycol |
|
| 3. |
Which of the following can NOT change the colour of litmus paper |
|
Answer» DIL `H_2SO_4` solution |
|
| 4. |
What is the mass of the precipitate formed when the preparation of alkyl halides 50 ml of 16.9% solution of AgNO_(3) is mixed with 50 ml of 5.8 % NaCl solution? |
|
Answer» 7g `0.0497 molxxunderset(AgCl)(143.32)=7.11 g` |
|
| 5. |
Write notes on Interstitial Hydrides. |
| Answer» Solution :Interstitial Hydrides : Metallic hydrides usually obtained by HYDROGENATION of metals and alloys in which hydrogen occupies the interstitial sites (VOIDS). Hence, they are called interstitial hydrides. These hydrides show properties similar to PARENT metals and hence they are also known as metallic hydrides . Most of the hydrides are non-stoichiometric with variable composition `(TiH_(1.5-1.8)and PdH_(0.6-0.8))`, some are relatively light, inexpensive and thermally unstable which makes them useful for hydrogen storage APPLICATIONS. | |
| 6. |
Which of the following does not participate in the Solvay's process for the manufacture of Na_(2)CO_(3)? |
|
Answer» `NH_(3)` |
|
| 7. |
Write structures of different isomers corresponding to the 5th member of alkyne series. Also write IUPAC names of all the isomers. What type of isomerism is exhibited by different pairs of isomers ? |
Answer» Solution :The molecules FORMULA of the 5th MEMBER of the ALKYNE series is `C_6H_10`. The possible isomers and their IUPAC names are : 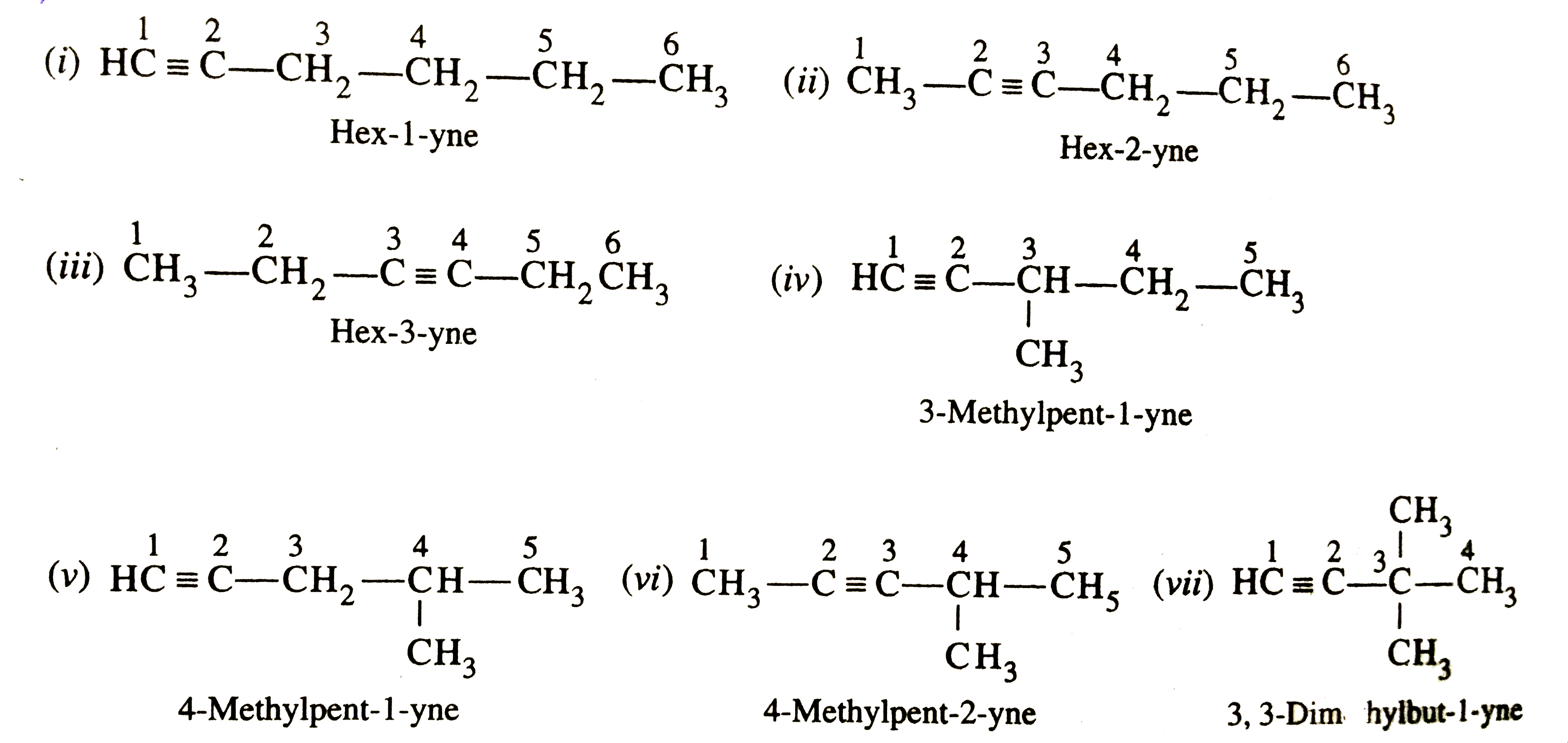 POSITION isomers . (i) and (ii) , (i) and (III) , (ii) and (iii), (v) and (vi) Chain isomers . (i) and (iv) , (i) and (v) , (i) and (vi), (i) and (vii) , (ii) and (iv) , (ii) and (v), (ii) and (vi), (ii) and (vii) , (iii) and (iv) , (iii) and (v) , (iii) and (vi) , (iii) and (vii) , (iv) and (v) , (v) and (vii) |
|
| 8. |
Which of the following will dissolve in aqueous Na_(2)CO_(3) ? |
|
Answer» PHENOL |
|
| 9. |
What are major differences between metals and non-metals? |
Answer» SOLUTION :
|
|
| 10. |
Two litres of an ideal gas at a pressure of 10 atm expands isothermally into a vacuum until its total volume is 10 litres. How much heat is absorbed and how much work is done in the expansion? |
| Answer» Solution :We have `q=-W=p_(ex)(10-2)=0(8)=0 (W_("FREE EXPANSION"=0))` | |
| 11. |
Which of the following is an example of aryl alkyl halide? |
|
Answer» BENZYL CHLORIDE |
|
| 12. |
Which one of the following does not undergo sublimation ? |
| Answer» Solution :Water | |
| 13. |
What is weed killers ? Give an example . |
| Answer» Solution :HERBICIDES are the chemical compounds used to control unwanted plants. They are otherwise known as weed killers. Example sodium CHLORATE `(NaClO_3)` and sodium arsenite `(na_2 AsO_3)` . Most of the herbicides are TOXIC to MAMMALS. | |
| 14. |
Which of the following reaction is an example of elimination reaction : |
|
Answer» `CH_3-CHOunderset(H_2O)OVERSET(CH_3MgBr)to CH_3-undersetunderset(OH)(|)CH-CH_3` |
|
| 15. |
Which of the following has large size |
|
Answer» Al |
|
| 16. |
Which of the following properties of alkali elements shows their weak bond in solid state ? |
|
Answer» LOW BOILING POINT and high ELECTRIC conductivity. |
|
| 17. |
The source of CO_2 in the atmosphere is |
|
Answer» COMBUSTION of FUEL |
|
| 18. |
Using the standard electrode potentials given in the Table-8.1, predict if the reaction between the following is feasible : (a) Fe_((aq))^(3+)andI_((aq))^(-) (b) Ag_((aq))^(+)andCu_((s)) ( c) Fe_((aq))^(3+)andCu_((s)) (d) Ag_((s))andFe_((aq))^(3+) (e) Br_(2(aq))andFe_((aq))^(2+) |
|
Answer» Solution :(a) `Fe_((aq))^(3+)andI_((aq))^(-)` probable REACTION … `2Fe_((aq))^(3+)+2I_((aq))^(-)to2Fe_((aq))^(2+)+I_(2(s))`  Value of `E^(@)` is POSITIVE, therefore reaction is possible. (b) `Ag_((aq))^(+)+Cu_((s))` Probable reaction is `2Ag_((aq))^(+3)+Cu_((s))to2Ag_((s))+Cu_((aq))^(+2)`  Value of `E^(@)` positive, therefore reaction is possible. ( c) `Fe_((aq))^(+3)+Cu_((s))` posiible reaction between them.  Value of `E^(@)` positive therefore reaction is possible. (d) `Ag_((s))+Fe_((aq))^(+3)` possible reaction between them.  `E^(@)` is negative so reaction is not possible. (e) `Br_(2(aq))+Fe_((aq))^(+2)` possible reaction between them.  `E^(@)` is positive, therefore reaction is possible. |
|
| 19. |
Which of the following reaction involves homogeneous reduction ? |
|
Answer» `CH_(2)=CH_(2) OVERSET(H_(2)//NI)rarr CH_(3)-CH_(3)` |
|
| 20. |
Which of the following given sets of quantum numbers are consistent with the theory ? |
|
Answer» `n = 2, l = 1, m = 0, s = +(1)/(2)` |
|
| 21. |
The standard molar entropy of H_(2) O_((l)) is 70 "JK"^(-1) "mol"^(-1). Will the standard molar entropy of H_(2) O_((s)) be more, or less than 70 "JK"^(-1) "mol"^(-1) ? |
|
Answer» Solution : The standard MOLAR entropy of `H_(2) O_((l))` is 70 `JK^(-1) "mol"^(-1)`. The SOLID form of `H_(2) O` is ice. In ice, molecules of `H_(2)O` are LESS random than in LIQUID water. Thus, molar entropy of `H_(2) O_((s)) lt` molar entropy of `H_(2) O_((l))`. The standard molar entropy of `H_(2) O_((s))` is less than `70 "JK"^(-1) "mol"^(-1)`. |
|
| 22. |
Three-centre two-electron bonds are present in |
|
Answer» `NH_(3)` |
|
| 23. |
When 6.3 g of sodium bicarbonate is added to 30 g of acetic acid solution, the residual solution is round to weigh 33 g. The number of moles of carbon dioxide released in the reaction is |
|
Answer» 3 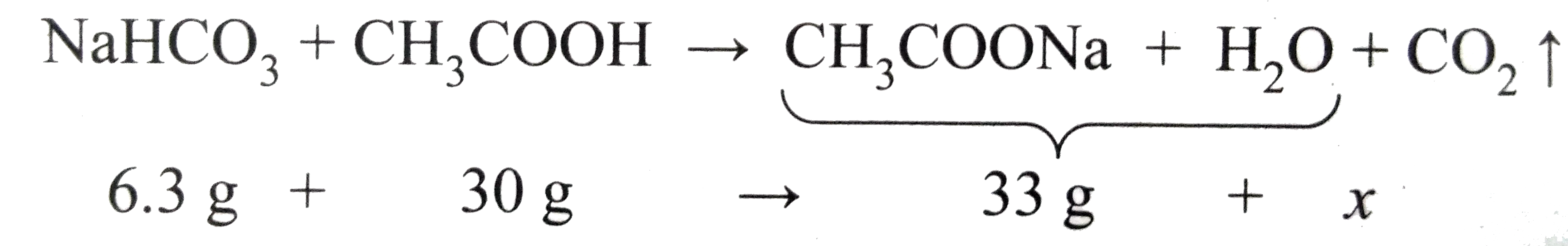 The AMMOUNT of `CO_(2)` RELEASED, x=3.3g No. of MOLES of `CO_(2)` released =3.3/44=0.75 MOL |
|
| 24. |
Using s, p, d notations, describe the orbital with the following quantum numbers : (a) n=1, l=0 (b) n=3, l=1 (c) n=4, l=2 (d) n=4, l=3. |
| Answer» Solution :(a) 1s (B) 3P (C) 4D (d) 4f. | |
| 25. |
The substance which can be used as adsorbent in column chromatography is |
|
Answer» `Na_(2)O` |
|
| 26. |
Which of the following are formed in the process of estimation of phosphorus |
|
Answer» `H_(3)PO_(4)` 
|
|
| 27. |
Which of the following expressions is correct? |
|
Answer» `Delta G^(@) = - nFE^(@)` |
|
| 28. |
What is the pressure at Kolkata, Chennai and Mumbai at 0^(@)C temperature ? |
| Answer» Solution :These THREE cities near SEA AREA of its NORMAL pressure will be 1 bar at 273.15 K its standard pressure. | |
| 29. |
What are colligative properties? |
|
Answer» Solution :The properties which do not depend on the chemical nature of the solute but DEPENDS only on the number of solute particles present in the solution are called colligative properties e.g., (i) Relative LOWERING of vapour PRESSURE `- (P ^(@) P)/(P^(@))` (ii) Osmotic pressure `-pi` (iii) Elvation of boiling point `-DELTA T_(b)` (iv) Depression in FREEZING point `- Delta T _(r).` |
|
| 30. |
What is the oxidation number of Mn is MnO_(4)^(-)? |
|
Answer» Solution :Mn in `MnO_4^(-)` , Let the oxidation number of Mn in `MnO_4^(-)` be X. WRITING oxidation number of each atom above its symbol, we get, `OVERSET(x)(Mn)overset(-2)O_4i.e., x+4(-2)=-1i.e., x=+7` Thus, the oxidation number of Mn in `MnO_4^(-)` is +7 |
|
| 31. |
What is the oxidation number of iron in the brown ring complex compound? |
|
Answer» Solution :The BROWN RING COMPLEX compound is given as : `[Fe(H_(2)O)_(5)NO] SO_(4)` The complex ion is `[Fe(H_(2)O)_(5)NO]^(2+)` Oxidation NUMBERS of `H_(2)O` is zero and `NO` is `+1`. Oxidation STATE of Fe is `+1` Experimentally, the number of unpaired electrons also support `+1` state of Fe (but not `+2` state). |
|
| 32. |
Which one is the most toxic ? |
| Answer» SOLUTION :CO is more TOXIC than carbon `CO_(2) and SO_(2)` | |
| 33. |
Which has maximum entropy change of vaporisation? |
|
Answer» WATER (L) |
|
| 34. |
Tropolone is an example of |
|
Answer» benzenoid AROMATIC COMPOUND |
|
| 35. |
What kind of compounds can be dissolved and hydrolysed in water? |
|
Answer» Solution :Water can easily dissolve all IONIC COMPOUNDS. Due to H-b onding with polar molecules, even COVALENT molecules like alco hols and carbohydrate dissolve in water. Water can hydrolyse MANY metallic and non-metallic oxides, hydrides, phosphides and other salts. `P_4O_(10(s))+6H_2Oto4H_3PO_(4(AQ))` |
|
| 36. |
Which of the following does not exist infree state |
|
Answer» `BF_(3)` |
|
| 37. |
The volume of methane at N.T.P formed from 8.2g of sodium acetate by fusion with soda lime is |
|
Answer» 10 l |
|
| 38. |
Why are rare gases are mono atomic? |
| Answer» Solution :RARE GASES process OCTET structure i.e., 8-electrons in the outer most orbit. They have no TENDENCY to react. | |
| 39. |
Which one of the following sets of quantum numbers is not possible for an electron in the ground state of an atom with atomic number 19 ? |
|
Answer» Quantum number were proposed out of NECESSITY in Bohr model of the atom. In multi `e^-` system , `l , m_l` also required E describes SHAPE. |
|
| 40. |
What will be the molar volume of nitrogen and argon at 273.15 K and 1 atm ? |
| Answer» Solution :When temperature and PRESSURE of a gas is 273.15 K and 1 atm, they are known as standard temp. and pressure conditions (STP). Due to this volume occupied by 1 mole of each and every gas is 22.4 L. HENCE, the molar volume of `N_(2)` and Ar at 273/15 K and 1 atm is 22.4 L. | |
| 41. |
The reagent used for softening the temporary harness of water is |
|
Answer» Only a & B |
|
| 42. |
Which of the following compounds exhibits Tautomerism |
|
Answer»

|
|
| 43. |
When vapour of an alcohols are passed over hot reduced copper, alcohol is converted into alkene the alcohol is |
|
Answer» Primary 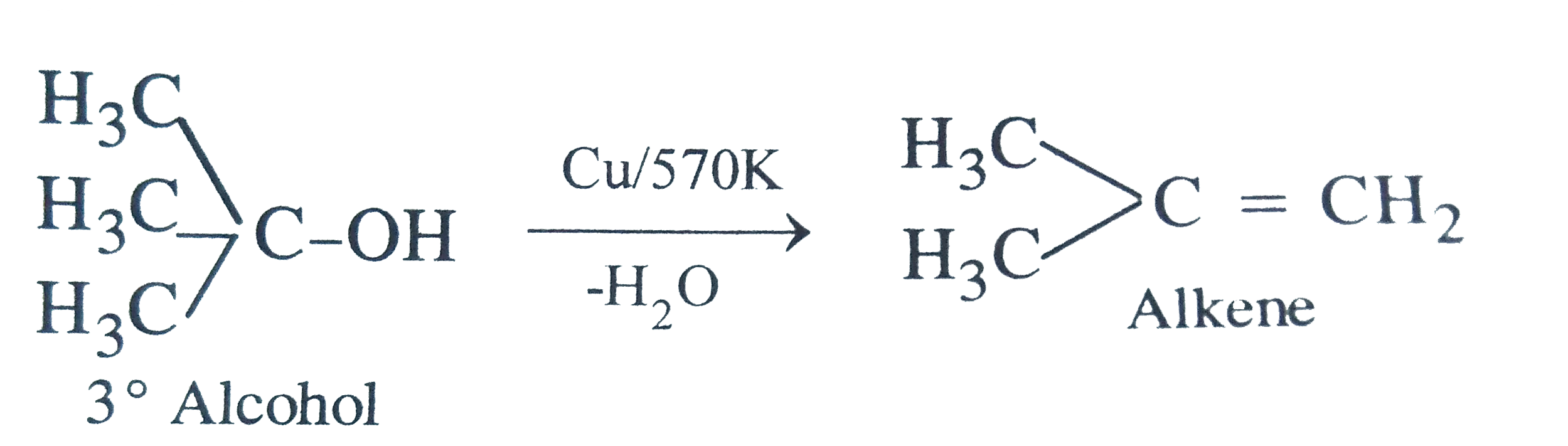
|
|
| 45. |
Which of the following oxids is acidic in nature ? |
|
Answer» `B_(2)O_(3)` |
|
| 46. |
Which of the following carboxylic acids undergoes decarboxylation on heating ? |
|
Answer» `C_(2)H_(5) - CO - CH_(2)COOH` |
|
| 47. |
Which of the following holds good to the laws of thermodynamics for the reaction ? C_2H_(4(g)) + 3O_(2(g)) to 2CO_(2(g)) + 2H_2O_((l)) |
|
Answer» `DELTA H = Delta E + RT` |
|
| 48. |
Which of the following options consist of substances that will illustrate law of reciprocal proportions? |
|
Answer» Water, carbon-di-oxdide and ethanol |
|
| 49. |
Why are falling drops spherical ? |
| Answer» SOLUTION :This is because force of SURFACE tension tends of decrease the surface AREA of the liquid to MINIMUM and for a given volume, sphere has minimum surface area. | |





