Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
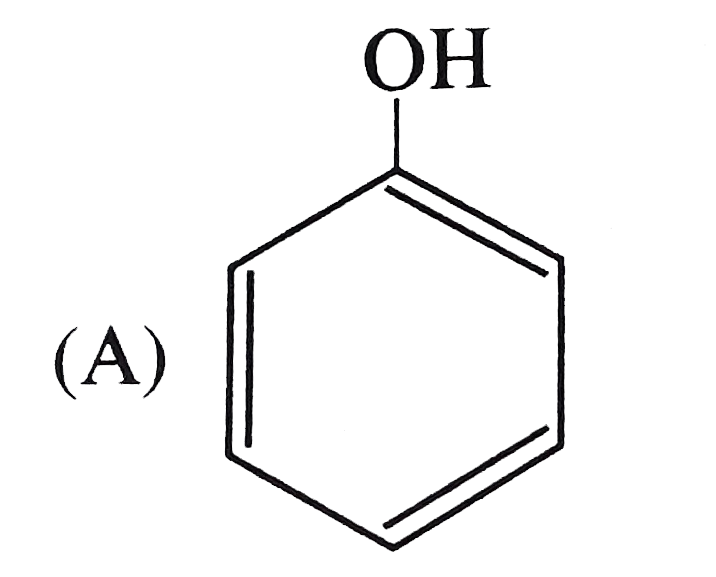
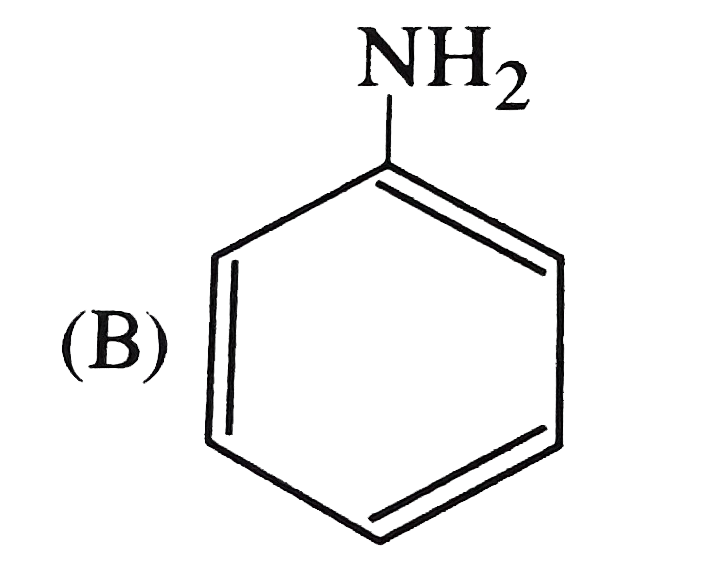
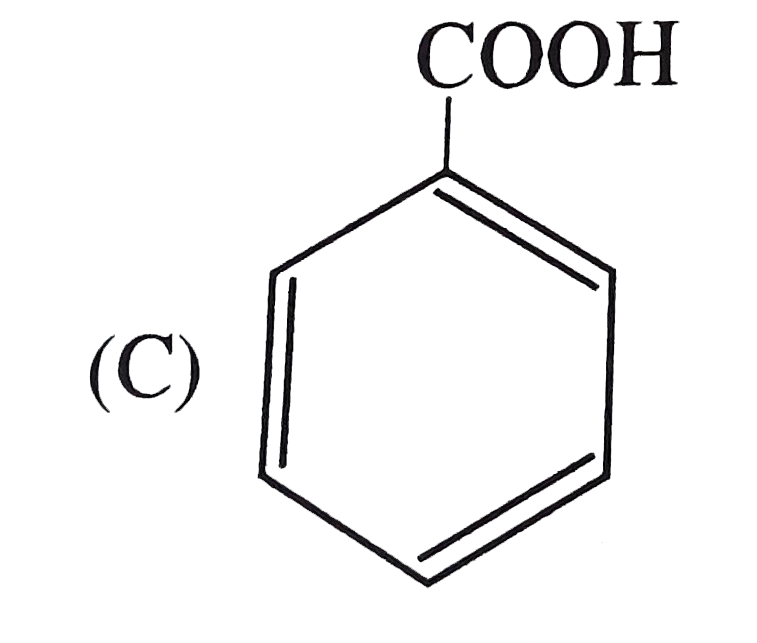
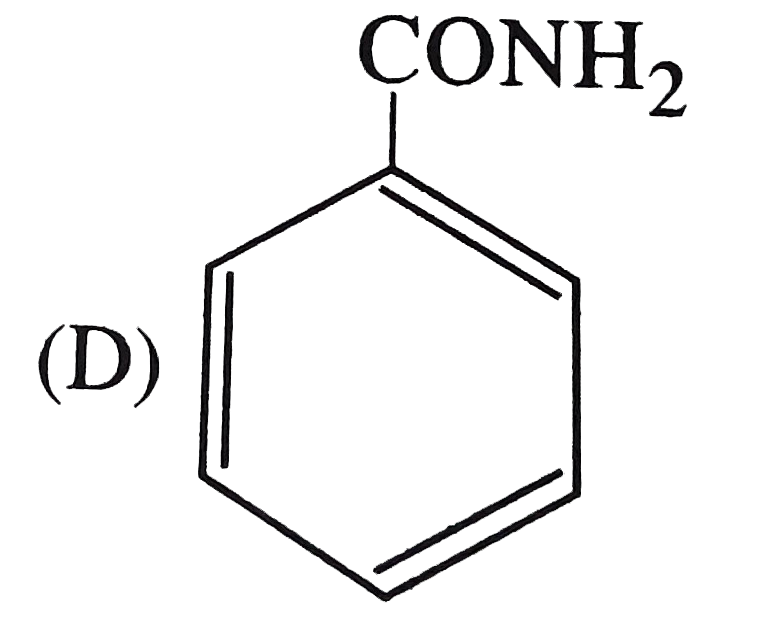
The strongest acid among the following is |
|
Answer»

|
|
| 2. |
Which of the following is indicated by the magnetic quantum number ? |
|
Answer» Size |
|
| 3. |
Which one used instead of CFC in refrigerator ? |
|
Answer» 1, 1, 2, 2 Tetrachloroethane |
|
| 4. |
What happens when diborane reacts withair ? |
|
Answer» SOLUTION :DIBORANE burns in oxygen liberating an enormous amount of ENERGY. `B_(2) H_(6(g)) + 3O_(2(g)) to B_(2)O_(3(g))+3H_(2)O_((I))` |
|
| 5. |
The size of particulates of H_2SO_4 fog lies in the range |
|
Answer» 5-100 NM |
|
| 6. |
What is the DeltaS^(@) for the reaction in cal if the graph of ln K vs 1//T is as follows. |
|
Answer» `1=(DeltaS^(@))/(R)` `:. DELTA S^(@)=2`cal |
|
| 7. |
The r.m.s. velocity of hydrogen is sqrt(7) times the r.m.s. velocity of nitrogen. If T is the temperature of the gas, |
|
Answer» `T(H_(2))=T(N_(2))` `sqrt(7)=sqrt((T(H_(2)))/(T(N_(2)))xx(28)/(2)),7=(T(H_(2)))/(T(N_(2)))xx14` or `T(N_(2))=2T (H_(2))` i.e., `T(N_(2)) GT T(H_(2))" or "T(H_(2)) lt T(N_(2))`. |
|
| 8. |
Which of the following is correct regarding aromatic nature ? A) Benzene B) Cyclopropane anion C) Cyclopentadienyl cation D) Cycloheptarienyl anion |
|
Answer» |
|
| 10. |
Which of the following are commonly used in making tooth paste. |
|
Answer» `Mg(OH)_(2)` |
|
| 11. |
The work function for Ceasium atom is 1.9 eV. Calculate the threshold frequency |
| Answer» Solution :WORK FUNCTION `(w_0)=h_v,` where V0 is the THRESHOLD frequency `w_0=1.9__eV=1.9xx1.602xx10^(-19)JthereforeV_0=w_0/h=(1.9xx1.602xx10^(-19))/(6.626xx10^(-34))=4.594xx10^14s^(-1)` | |
| 12. |
Which of the following are Lewis acids? H_(2)O,BF_(3),H^(+),CO_(4)^(+). |
| Answer» SOLUTION :`BF_(3), H^(+)and NH_(4)^(+) ` (Remember that all CATIONS are LEWIS acids). | |
| 13. |
Which of the following is a cumulative dienes |
|
Answer» Buta 1,3-diene |
|
| 14. |
Which one of the following compounds gives characteristic deep colour with FeCl_(3) solution ? |
|
Answer» acetone |
|
| 15. |
Which atom in the following reactions undergoes a change of oxidation state of -5 ? |
|
Answer» `MnO_(4)^(-)toMn^(2+)` |
|
| 16. |
Total number of orbitals associated with third shell will b e ....... |
|
Answer» 2 |
|
| 17. |
Which of the following conditions represaents an equilibrium ? |
|
Answer» Freezing of ice an open vessel, TEMPERATURE of ice is constant |
|
| 18. |
What is Eutrophication ? |
| Answer» Solution :The process in which nutrient enriched water bodies support a dense plant population. Which kills ANIMAL life by depriving it of OXYGEN and results in SUBSEQUENT LOSS of biodiversity is known as Eutrophication. | |
| 19. |
What is the solubilityproduct (K_(sp))of calcuim phosphate in pure water ? [S=molar solubility] |
|
Answer» `108 X^(2) ` `"" 3X "" 2X ` ` Ksp= (3x) ^(3)(2x)^(2) =108x^(5)` |
|
| 20. |
Write the systematic names of the following aromatic compounds: (i) (ii) (xix) para-Bromo - tert-butylbenzene Strategy: If there is only one group on the ring, no number is needed to designate its position. If there are two groups, we use the traditional designations (1,2-for ortho, 1,3-for meta, and 1,4-for para). If there are three or more goups on the ring then location numbers are assigned to give the lowest set of locants. |
|
Answer» Solution :(i) Ethylbenzene (ii) `n`-PROPHYLBENZENE (iii) (`2`-Methylpropyl) benzene (or Isobutylbenzene) (iv) `p`-Ethyltoluene (V) `1`-Bromo-`3`-nitrobenzene or `m`-Bromonitrobenzene (vi) `m`-Ethylisopropylbenzene (vii) `2`-Methyl-`3`-phenylpentane (VIII) 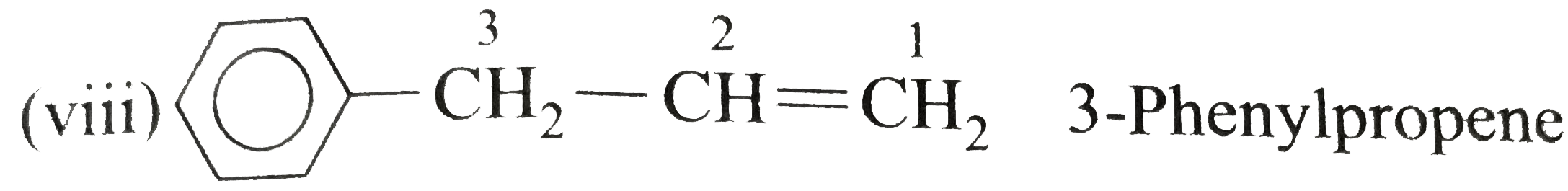 (ix) `2`-Phenylethyne (or phenylacetylene) (xi) `2,4`-Difluorobenzenesulphonic acid (xii) 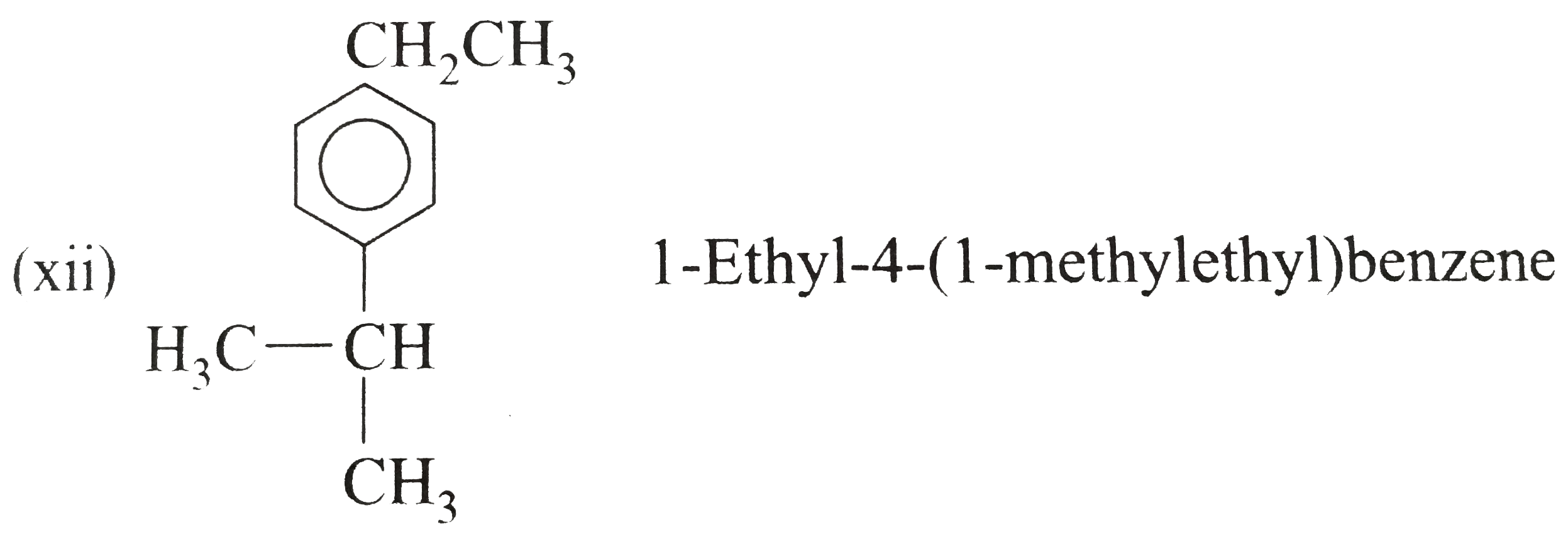 (xiii) 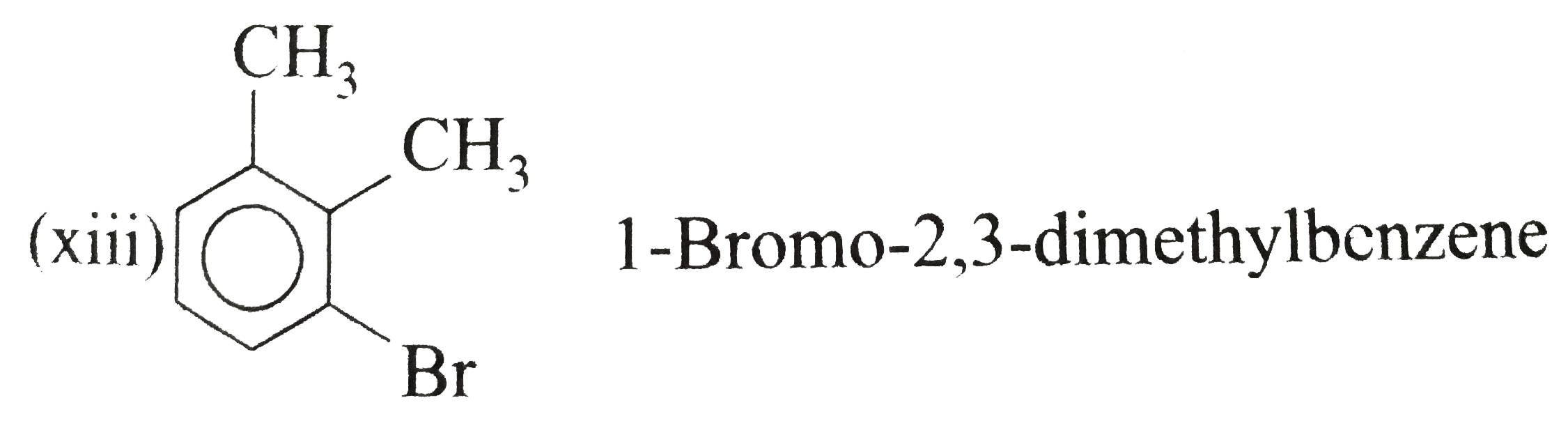 (xiv) `1,2,4`-Trinitrobenzene (xv) `1`-Ethenyl -`3`-ethyl `-5`- etynylbenzene (xvi) `1`-Iodo-`2`-metylbenzene ( or `o`-Iodotoluene) (xvii) `1`-Bromo-`3`-ethenylbenzene (or `m`-bromostyrene) (xviii) `4`-Ethyl -`1,2`-dimethylbenzene (not `1`-Ethyl-`3,4`-dimethyl benzene which has higher SET of locants) (xix) 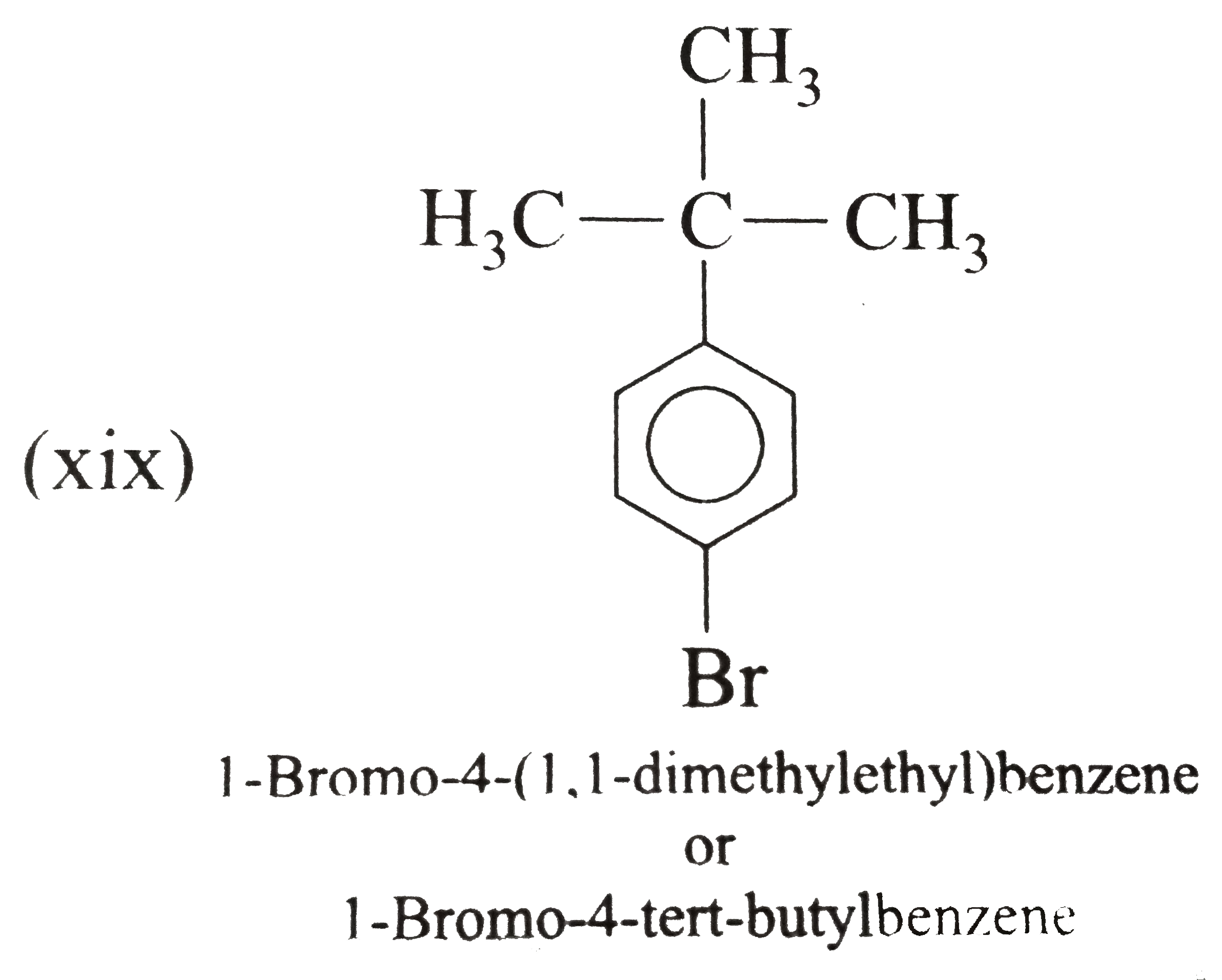
|
|
| 21. |
What is the difference between milk of lime and lime water ? |
| Answer» Solution :A suspension of slaked lime , i.e., `Ca(OH)_(2)` in water is CALLED MILK of lime but a CLEAR decanted or filtered solution of slaked lime in water is called lime water . | |
| 22. |
What can be formed on heating gypsum at 120^(@)C ? |
|
Answer» PLASTER of Paris |
|
| 23. |
Urea on hydrolysis produces ammonia and carbon dioxide. The standard entropies of urea, H_2O , CO_2 , NH_3are 173.8,70,213.5 and 192.5 "J mole"^(-1) K^(-1)respectively. Calculate the entropy change for this reaction. |
|
Answer» Solution :`S^0"(UREA)=173.8 "J mol"^(-1) K^(-1)` `S^0(H_2O)=70 "J mol"^(-1)K^(-1)` `S^0(CO_2)=213.5 "J mol"^(-1) K^(-1)` `S^0(NH_3)=192.5 "J mol"^(-1) K^(-1)` `NH_2-CO-NH_2+H_2O to 2NH_3+CO_2` `DeltaS_r^0 =sum (S^0)_"products" -sum (S^0)_"reactants"` `DeltaS_r^0=[2S^0(NH_3)+S^0(CO_2)]-[S^0"(urea)"+S^0(H_2O)]` `DeltaS_r^0`=[ 2 x 192.5 + 213.5 ]-[173.8 +70 ] `DeltaS_r^0`=[598.5]-[243.8] `DeltaS_r^0=354.7 "J mol"^(-1) K^(-1)` |
|
| 24. |
What is the total number of sigma and pi bonds in the following molecules? (a) C_(2)H_(2)""(b)C_(2)H_(4) |
|
Answer» Solution :(i) `H-C-=C-H""{:(SIGMA" BONDS = 3"),(pi" bonds = 2"):}` (II) `H-UNDERSET(H)underset(|)C=underset(H)underset(|)C-H""{:(sigma" bonds = 5"),(pi" bonds = 1"):}` |
|
| 25. |
Write the expression for Delta U under an isothermal process. |
|
Answer» <P> SOLUTION :`DU = q - p .dV` (or) `q = dU + p.dv`When work is done by system, `W = -p.dv`. |
|
| 26. |
What is the minimum volume of water required to dissolve 1 g of calcium sulphate at 298 K. For calcium sulphate, K_(sp)=9.1xx10^(-6). |
|
Answer» Solution :`CaSO_(4)(s)hArrCa^(2+) (aq) + SO_(4)^(2-) (aq)` If s is the SOLUBILITY of `CaSO_(4)` inmoles `L^(-1)`, then `K_(sp)=[Ca^(2+)]xx[SO_(4)^(2-)]=s^(2)` or `s=sqrt(K_(sp))=sqrt(9.1xx10^(-6))=3.02xx10^(-3) "mol" L^(-1) = 3.02xx10^(-3) xx 136 G L^(-1) = 0.411 g L^(-1)`[Molar mass of `CaSO_(4) = 136 g "mol"^(-1)`) THUS, for dissolving 0.411 g, water required = 1 L `:. ` For dissolving 1 g, water required `=(1)/(0.411) L = 2.43 L`. |
|
| 27. |
Which of the following is an alloy of non-transition elements? |
|
Answer» Elektron |
|
| 28. |
What is the effect of increae in temperature for the reaction? 2NO_(2(g))hArrN_(2)O_(4(g))""DeltaH=-572.5kJ |
| Answer» SOLUTION :Increase in temperature INCREASES the RATE of backward REACTION because forward reaction is exothermic. | |
| 30. |
Which of the following in viable particulates |
| Answer» Solution :Moulds | |
| 31. |
X and Y both are m oving particles. The uncertainty in measure of momentum of Y is half than that of X. If uncertainity of position of X is pm 0.05A than that of Y is .... |
|
Answer» `2.5XX10^(-10)` CM `triangle` p of y=`1/2` of x =`1/2 triangle p_(x)` For x `triangle x triangle p =(H)/(4pi)` `0.05 A xx triangle p_(x)=(h)/(4pi)` `thereforetrianglex =0.05 xx2` `=0.1 xx10^(-10)` cm `=1xx10^(-9)` cm |
|
| 32. |
Which among the following represents an amide |
|
Answer»
|
|
| 33. |
Which one of the following is reduced with Zn, Hg and HCl acid to give the corresponding hydrocarbon ? |
|
Answer» ETHYL acetate |
|
| 34. |
Which of the following is not a state function? |
|
Answer» S |
|
| 35. |
Which of the following are obtained as the only product after hydration of suitable alkyne with H_2SO_4, and HgSO_4 ? |
|
Answer»

|
|
| 36. |
Which type of intermediate (A) is formed during the reaction? CH_(3)CH_(2)-N=N-CH_(2)CH_(3) overset("heat")to (A)+N_(2) |
|
Answer» Carbocation |
|
| 37. |
Those elements impart colour to the flame on heating in it, the atoms of which require low energy for the ionisation (i.e., absorb energy in the visible region of spectrum). The elements of which of the following groups will impart colour to the flame ? |
|
Answer» 2 
|
|
| 38. |
Which of the following statement is not correct about Lother Meyer's plot of atomic volume against atomic weight ? |
|
Answer» Alkali metals are at the peak |
|
| 39. |
Which has a higher energy : a photon of red light with a wavelegth of 7500 Å or a photon of green light with a wavelength of 5250 Å? |
|
Answer» |
|
| 40. |
The sizes of A, A^+ and A^- follows the order |
|
Answer» `A^(+) GT A^(-) gt A` |
|
| 41. |
What are the effects of smoking habits for pregnant women ? |
| Answer» Solution :If the pregnant women has a HABIT of SMOKING, the CO RATIO increases in order to increase the chances of MISCARRIAGE, spontaneous ABNORMALITIES and disorders in the baby. | |
| 43. |
What are the sources of the human body that produces free radicals? |
|
Answer» SOLUTION :Sources for HUMAN body produces free radicals. (i) Human body is exposed to X-rays. (ii) Cigarette smoke (iii)INDUSTRIAL chemicals. (iv) AIR pollutants. |
|
| 44. |
Which one of the following statement is false : |
|
Answer» WORK is a state function |
|
| 45. |
What are extensive properties? |
|
Answer» Solution :(i)The PROPERTY that is depend on the mass or size of the system is called an extensive property (II) EX. VOLUME, NUMBER of moles, mass and internal energy. |
|
| 46. |
Which of the following compounds will exhibits trans (geometrical) isomerism ? |
|
Answer» Butanol |
|
| 47. |
which of the following options temperature-dependent concentration term? |
|
Answer» PPM |
|
| 48. |
Which of the following are Lewis acids ? |
|
Answer» `PH_(3) and BCl_(3)` `AlCl_(3)` -electron deficient, incomplete octet. Note. `SiCl_(4)` can accept a lone pair of ELECTRONS in d-orbital of SILICON and HENCE acts as a LEWIS acid. Hence, option 2 can also be considered as correct 
|
|
| 49. |
What is the effect of added inert gas on the reaction of equilibrium? |
| Answer» Solution :When an inert gas (i.e., a gas which does not react with any other species INVOLVED in equilibrium) is added to an equilibrium SYSTEM at CONSTANT volume, the total number of moles of GASES present in the container increases, that is, the total pressure of gases increases, the partial pressure of the reactants and the products are unchanged. Hence at constant volume,addition of inert gas has no EFFECT on equilibrium. | |
| 50. |
Which of the following can act as an oxidising and reducing agent in neutral and alkaline solution ? |
| Answer» Solution :`KMnO_(4)` | |