Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Write the resonance structures of aniline and explain which one is the most stable ? |
Answer» SOLUTION :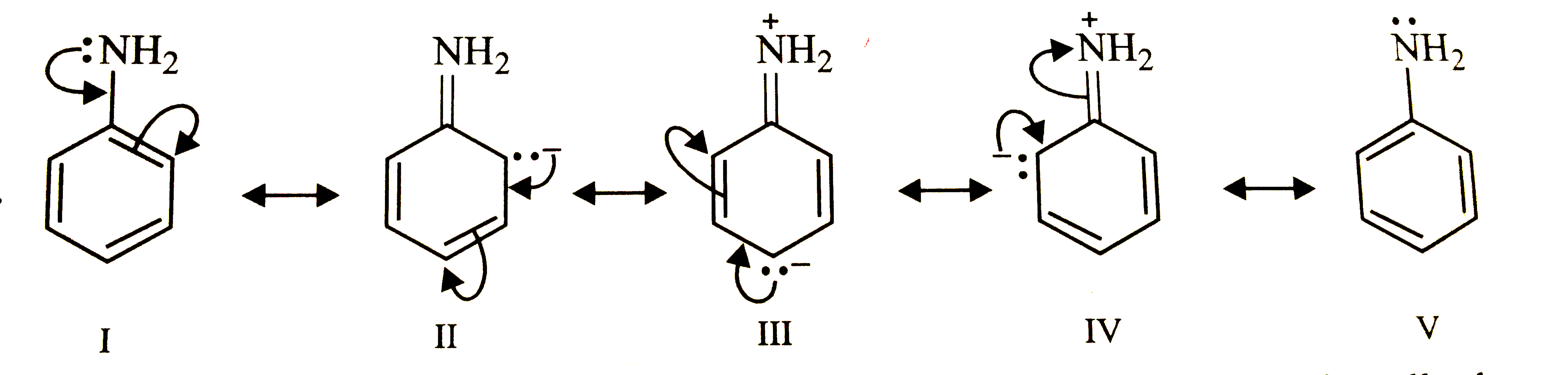 Resonance structure (I) is neutral because it does not carry and charge but all OTHERS carry separation of +ve and -ve charges. Therefore, structure (I) is the most STABLE. |
|
| 2. |
Which of the following is known as 'Wood alcohol' ? |
|
Answer» `CH_(3)OH` |
|
| 3. |
Which of the following has maximum angle strain ? |
|
Answer»
|
|
| 4. |
Which one of the following pairs does not represent example for intensive property ? |
|
Answer» Temperature and density |
|
| 5. |
Vapour density of PCl_(5) is 104.16 but when heated at 230^(@) C its vapour density is reduced to 62 . The degree of dissociation of PCl_(5) at this temperature will be |
|
Answer» `6.8%` |
|
| 7. |
The structure of IF_7is …. |
|
Answer» square pyramid 
|
|
| 9. |
The root mean square velocity of a gas molecule at 100K and 0.5atm pressure is 106.4ms^(-1). If the temperature is raised to 400K and the pressure is raised to 2 atm, the root mean square velocity becomes: |
|
Answer» `106.4m s^(-1)` |
|
| 10. |
What is zero over lap? |
| Answer» SOLUTION :when REFERRING to electric trimmers, SLASH OUTLINES, i.e., BLADES can be adjusted for zero overlap | |
| 11. |
When KMnO_(4) reacts with acidified FeSO_(4) |
|
Answer» only `FeSO_(4)` is oxidised |
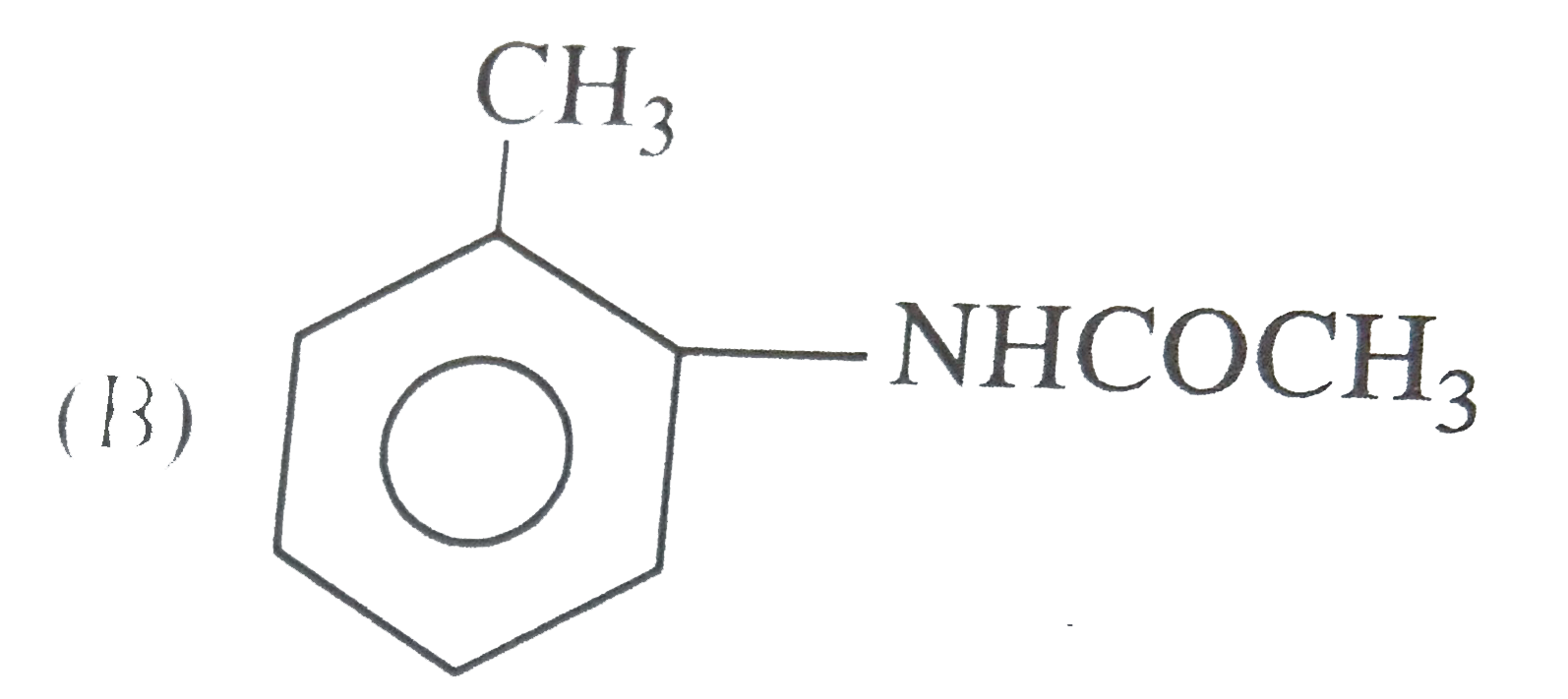
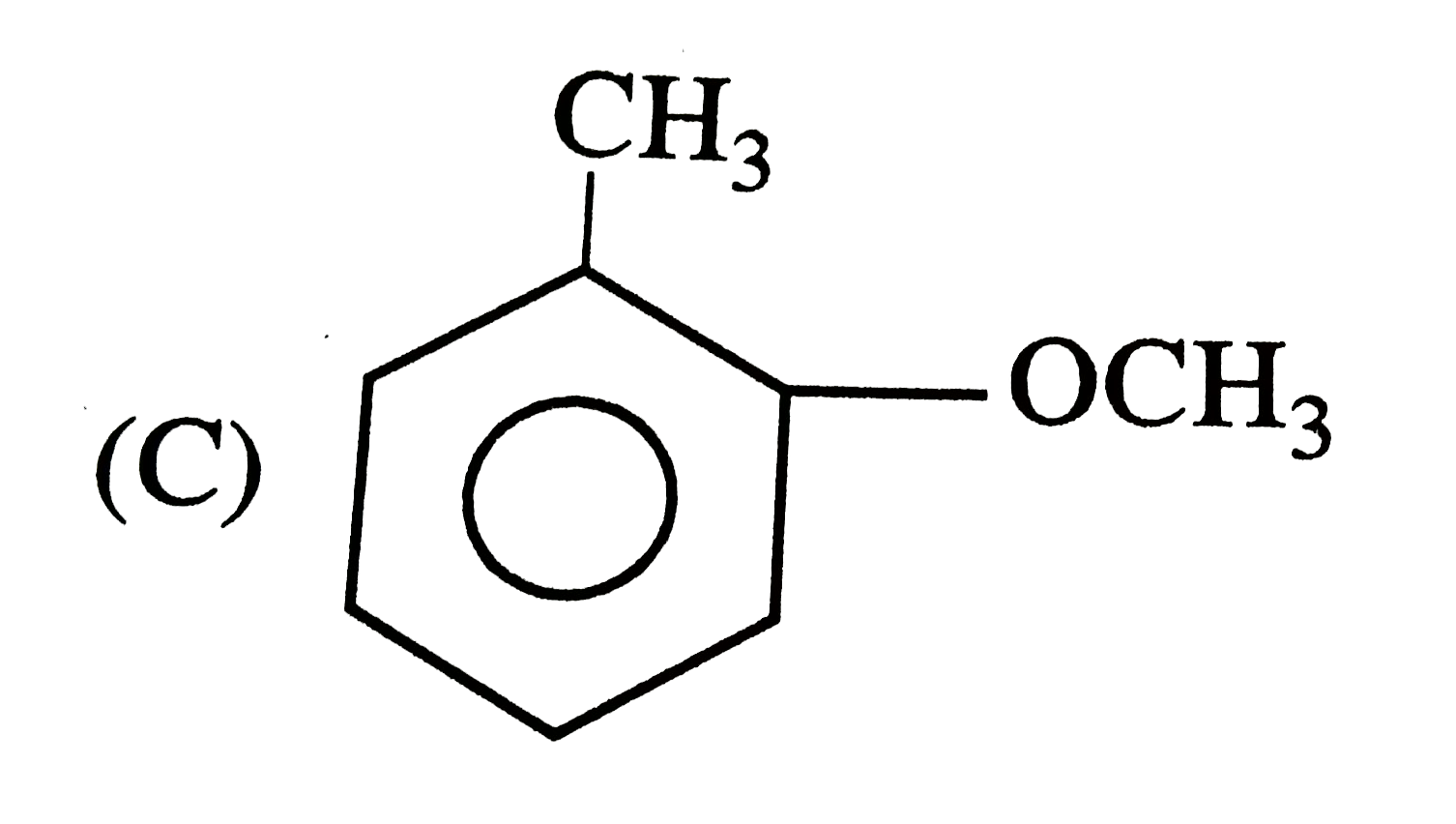
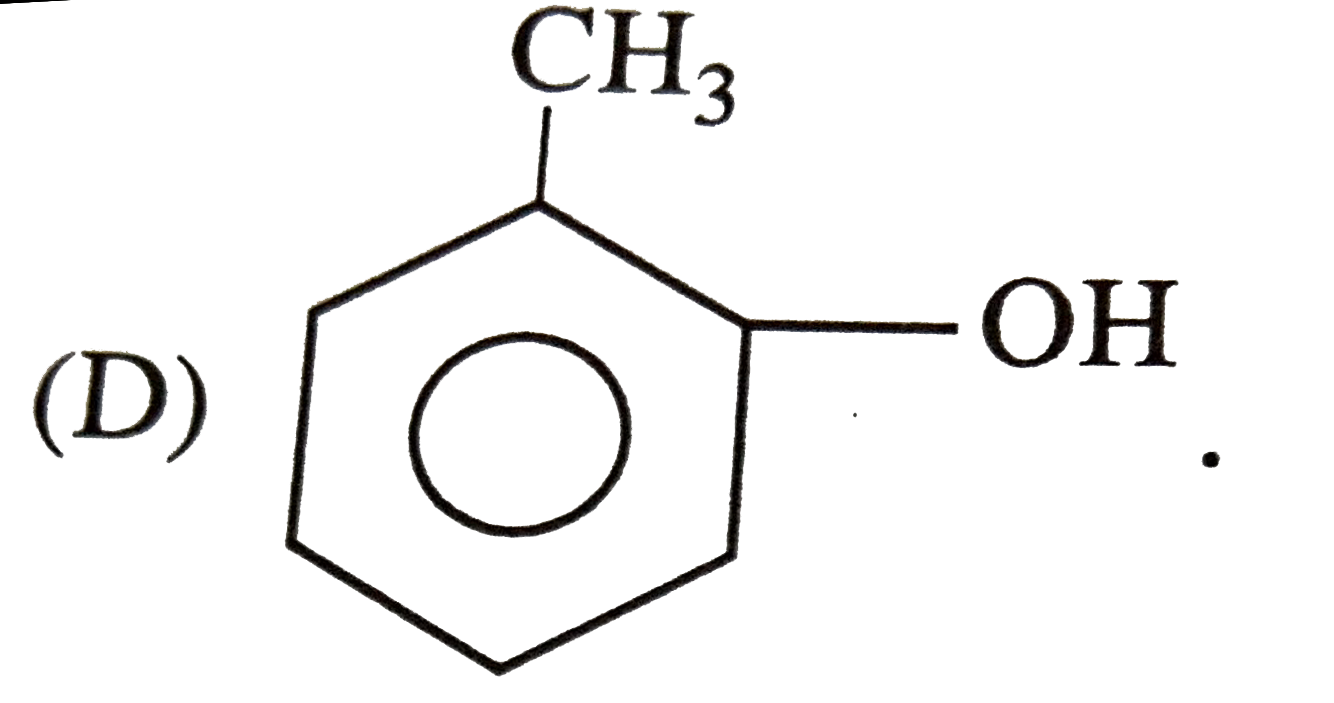
|
| 12. |
Using X-rays of wavelength 154.1 pm and starting from the glancing angle, the reflection from silver crystal was found to occur at theta=22.20^@. Calculate the spacing between the plane of Ag atoms that gave rise to the above reflection. (sin 22.20^@=0.3778) |
|
Answer» Solution :Here, `lambda`=154.1 pm , n=1, `THETA=22.20 ^@` using Bragg's EQUATION , VIZ. , `2 d sin theta= nlambda` `d=(nlambda)(2 sin theta)=(1xx154.1)/(2xx0.3778)` pm =204 pm |
|
| 13. |
Which of the following statement is/are correct: |
|
Answer» P is benzene diazonioum chloride |
|
| 14. |
X(s)hArrY(g)+2Z(g) A(s)hArrY(g)+B(g) Consider both these equilibrium to be established simultaneously in a closed container. At equilibrium, pressure of Z "and" B were found to be same and sum of pressure of Z & B is 10 atm more than that of species Y. Find ratio of standard gibb's energy of two reactions. |
|
Answer» `20` `t=eq. (a+b)2a t=eq. (a+b) b` given `P_(z)=P_(B)` `2a=b` and `P_(z)+P_(B)=P_(Y)+10` `(2a+b)=(a+b)+10` `a=10atm` `THEREFORE b=20atm` `K_(P_(1))=(a+b)(2a)^(2)=(30)(400)=12000` `k_(P_(2))=(a+b)(b)=(30)(20)=600` `DeltaG=-RTInK` `(DeltaG_(1))/(DeltaG_(2))=(lnK_(1))/(lnK_(2))=(log12000)/(log600)=(3+log12)/(2+log6)` |
|
| 15. |
Which gas is relesed during the reaction of Sodalime with Ethylene ? |
|
Answer» Nitrogen |
|
| 16. |
The salt KHC_(2)O_(4)H_(2)C_(2)O_(4).4H_(2)Omay be used as reducing agent as well as an acid. Choose thecorrect option (S): |
|
Answer» The equivalent weight of the salt is 72.5 g-`"equivalent"^(-1)`, when reacted with acidified `KMnO_4`SOLUTION. |
|
| 17. |
Which of the following compounds are aromatic accroding to Huckel's rule ? |
|
Answer» |
|
| 18. |
Write the metamers for the formula C_(5)H_(10)O with their IUPAC names. |
|
Answer» Solution :(i) `C_(2)H_(5)-UNDERSET(O)underset(||)C-C_(2)H_(5)` Pentane-3-one (II) `CH_(3)-underset(O)underset(||)C-C_(3)H_(7)` Pentane-2-one (iii) `CH_(3)-underset(O)underset(||)C-underset(CH_(3))underset(|)(CH)-CH_(3)` 3-methyl butan-2-one |
|
| 19. |
Which of the following process is/are associated with no change of hybridisation of the underlined compound? |
|
Answer» `ul(B)_(2) H_(6)` is DISSOLVED in THF |
|
| 20. |
What is the oxidation state of chromium in potassium dichromate? |
|
Answer» Solution :Let the OXIDATION state of CR in `K_(2)Cr_(2)O_(7)` be x. The sum of the oxidation STATES of all ATOMS in a neutral molecule is zero. 2x(+1) +2x(x)+7(-2)=0, x=+6 |
|
| 21. |
Which mixture of solution will have pH gt 7 ? |
|
Answer» HCl+NaOH |
|
| 23. |
Which of the following is correct about equilibrium constant ? |
|
Answer» Unpredict the direction in which the 'net reaction will take place. |
|
| 24. |
Which of the following does not liberate hydrogen one reacting with acids Boron and its compounds |
|
Answer» Al |
|
| 25. |
when changes from gaseous phase to soild phase , which one of the following startements is correct regarding H_(2)O_(2)? |
|
Answer» dral ANGLE changes from `920^(@)"to" 111^(@)` |
|
| 26. |
The twotypes of bonds present in diborane are "………….." and "…………..". |
|
Answer» |
|
| 27. |
What wil be the products of electrolysis of an aqueous solution of AgNO_(3) with silver electrodes ? |
|
Answer» AG from Ag anode dissolves while `Ag^(+)` from solution gets deposited on cathode. At cathode : `Ag_((aq))^(+)+e^(-)rarr Ag, E^(@)=+0.80 V` `2H_(2)O_((l))+2e rarr H_(2)+2OH^(-), E^(@)=-0.83 V` Since reduction potential of `Ag^(+)` is higher than `H_(2)O`, `Ag^(+)` are REDUCED to Ag. At anode : `Ag rarr Ag_((aq))^(+) + e^(-), E^(@)=-0.80 V` `2H_(2)O rarr O_(2)+4H^(-)+4e^(-), E^(@)= - 1.23 V` Since oxidation potential of Ag is higher than `H_(2)O`, Ag of SILVER anode is oxidised. Theoxidation potential of `NO_(3)^(-)` is lower than `H_(2)O`. |
|
| 28. |
The total number of tetrahedral voids in the face centred unit cell is ______ |
|
Answer» 6 |
|
| 29. |
Which one of the following acts as the best coagulating agent of ferric hydroxide sol ? |
|
Answer» magnesium chloride |
|
| 30. |
What are pesticides and herbicides? Explain giving examples |
|
Answer» SOLUTION :Pesticides. These are synthetic toxic chemicals which are used in agriculture to control the damages caused by insects, RODENTS, weeds and various crop diseases. Their repeated use gives rise to pests that are resistant to that group of pesticides. As a result, these pesticides become ineffective for these pests. Earlier DDT was used. As insect resistance towards DDT INCREASED, other organic toxins such as Aldrin and Dieldrin were introduced for use as pesticides. But these were non-biodegradable and slowly TRANSFERRED to human being through food chain causing metabolic and physiological DISORDERS Consequently, a new series of pesticides, organophosphates and carbamates have been introduced These are biodegradable but are severe nerve toxins and hence more harmful to humans and have caused even deaths, Thus, the insecticide industry is engaged in developing new insecticides Herbicides. These are the chemicals used to control weeds. Earlier, inorganic compounds such as sodium chlorate `(NaCIO_3)` and sodium arsenite `(Na_3AsO_3)` were used but arsenic compounds, being toxic to mammals, are no longer preferred. Instead, organic compounds such as triazines are now considered as better herbicides, especially for the corn-fields |
|
| 31. |
Which of the following compound contains intramolecular H-bonds? |
|
Answer» Phenol 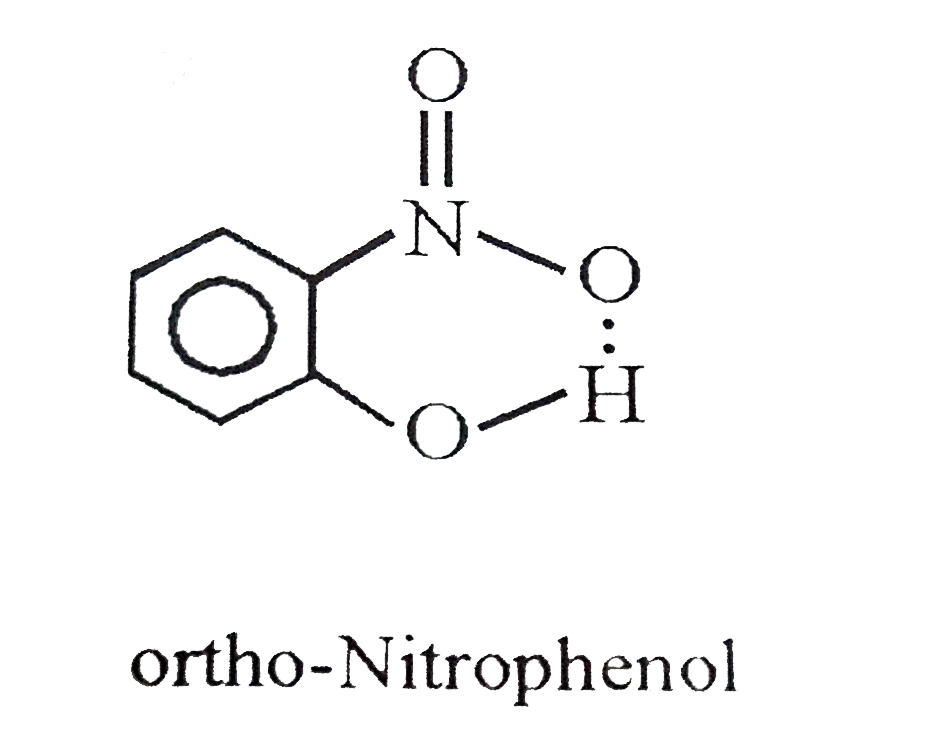
|
|
| 32. |
Which of the following alcohols will be more suspectible to acidic dehydration? |
|
Answer» `CH_(3)CH_(2)CH_(2)CH_(2)OH` Due to the FORMATION of most stable `3^(@)` carbocation. |
|
| 33. |
What are bond line structures? Give one example. |
|
Answer» Solution :Condensed STRUCTURAL formula is simplified in which only lines are USED. In this type of representation of ORGANIC compounds, carbon and hydrogen ATOMS are not shown and the lines representing carbon-carbon bond are shown in zig zag fashion. The only atoms specially written are oxygen, chlorine, nitrogen etc. Example, TERT. butyl chloride `underset(CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)C-Cl)("Condensed structure")` 
|
|
| 34. |
Under what condition , a reversible process becomes irreversible ? |
| Answer» Solution :If one of the the products (GASEOUS) is aloowed to ESCAPE out (i.e., in open vessel) or the REACTION results in the FORMATIONOF PRECIPITATE. | |
| 35. |
Which is not an allotrope of carbon ? |
|
Answer» Graphite |
|
| 36. |
What is the expression for K_(eq) for the reaction , 2 N_(2) O (g) + O_(2) (g)hArr 4 NO (g) ? |
|
Answer» `([N_(2)][O_(2)])/([NO])` |
|
| 37. |
Which period contains two elements in periodic table ? |
|
Answer» 1 |
|
| 38. |
Which of the following compounds can give E1cB reaction ? |
|
Answer» `CF_3-CHCl_2` |
|
| 39. |
Which one of the following has highest dipole moment ? |
|
Answer» `BF_(3)` |
|
| 40. |
Which statement is correct for Caro.sacid ? |
|
Answer» Oxidation NUMBER of two OXYGEN is (-1) 
|
|
| 41. |
What is the structure of H_(2)O_(2)? |
|
Answer»
|
|
| 42. |
Which information obtained from following figure ? |
|
Answer» Solution :(i) As PER figure initially only HI in SYSTEM. (ii) as the reaction take place concentration of HI DECREASES. (iii) After some TIME the concentration of HI does not change `2HI_((g)) HARR H_(2(g)) + I_(2(g))` |
|
| 43. |
What is structural isomerism ? Give two isomers of chain isomerism of alkane. |
|
Answer» Solution :* Structural isomers : Compounds having same molecular formula but different structural arrangement is known to be structural isomers. This phenomenon is known as structural isomerism. * Branched isomers (Chain isomers) : If two or more compounds having same molecular formula but the carbon chain structures are different such compounds are known as chain isomers and this is known as chain isomers and this is known as chain isomerism. Having more than three carbon alkanes havechain isomerism. EXAMPLE -1 : Butane has 2 structural isomers. (i) n-butane can be joined either in a CONTINUOUS chain or with a branched chain. (ii) Isobutane shows branched structure. (i) n-butane : `H-UNDERSET(H)underset(|)OVERSET(H)overset(|)(C)-underset(H)underset(|)overset(H)overset(|)(C)-underset(H)underset(|)overset(H)overset(|)(C)-underset(H)underset(|)overset(H)overset(|)(C)-H` (ii) 2-Methylpropane :  Example - 2: Penane `(C_(5)H_(12))` has 3 isomers. (i) Normal pentane has chain like structure. (ii) Isopentane and (III) Neopentane has branched like structure. (i) Pentane : `underset("n-pentane")(H-underset(H)underset(|)overset(H)overset(1|)(C)-underset(H)underset(|)overset(H)overset(2|)(C)-underset(H)underset(|)overset(H)overset(3|)(C)-underset(H)underset(|)overset(H)overset(4|)(C)-underset(H)underset(|)overset(H)overset(5|)(C)-H)` In short `CH_(3)CH_(2)CH_(2)CH_(2)CH_(3)` 
|
|
| 44. |
Which of the following substance will not poduce methane by its treatment with methyl magnesium iodide? |
|
Answer» Ethanol |
|
| 45. |
Which one is most reactive towards electrophilic reagent |
|
Answer»
Among the given GROUP `-OH` has the higher electron donating TENDENCY and thus, activates the ring towards electrophilic substitution 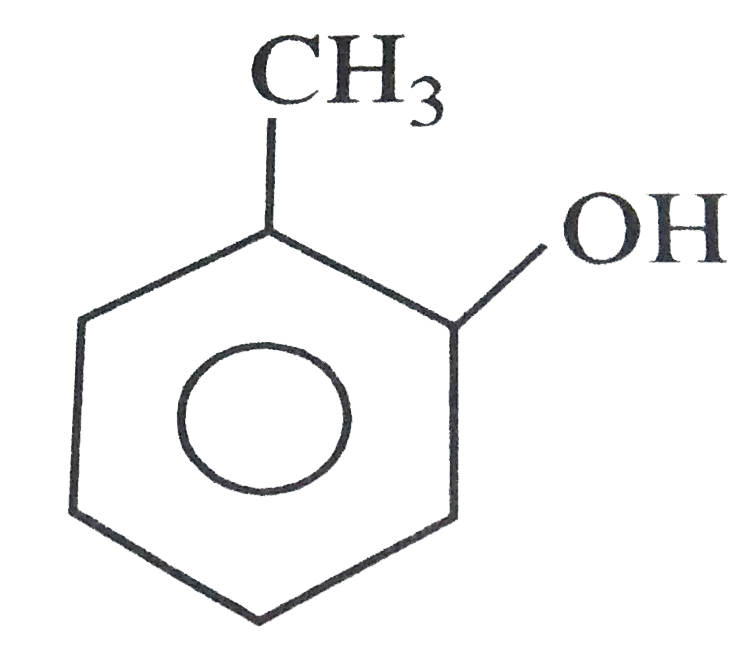 is more reactive towards electrophilic reagents. |
|
| 46. |
Which of the following solution pairs can be separated by fractional distillation ? |
|
Answer» Water-`HNO_(3)` |
|
| 47. |
Which of the following carboxylic acids undergoes decarboxylation easily ? Explain briefly |
|
Answer» `C_(6)H_(5) - CO - CH_(2) - COOH` 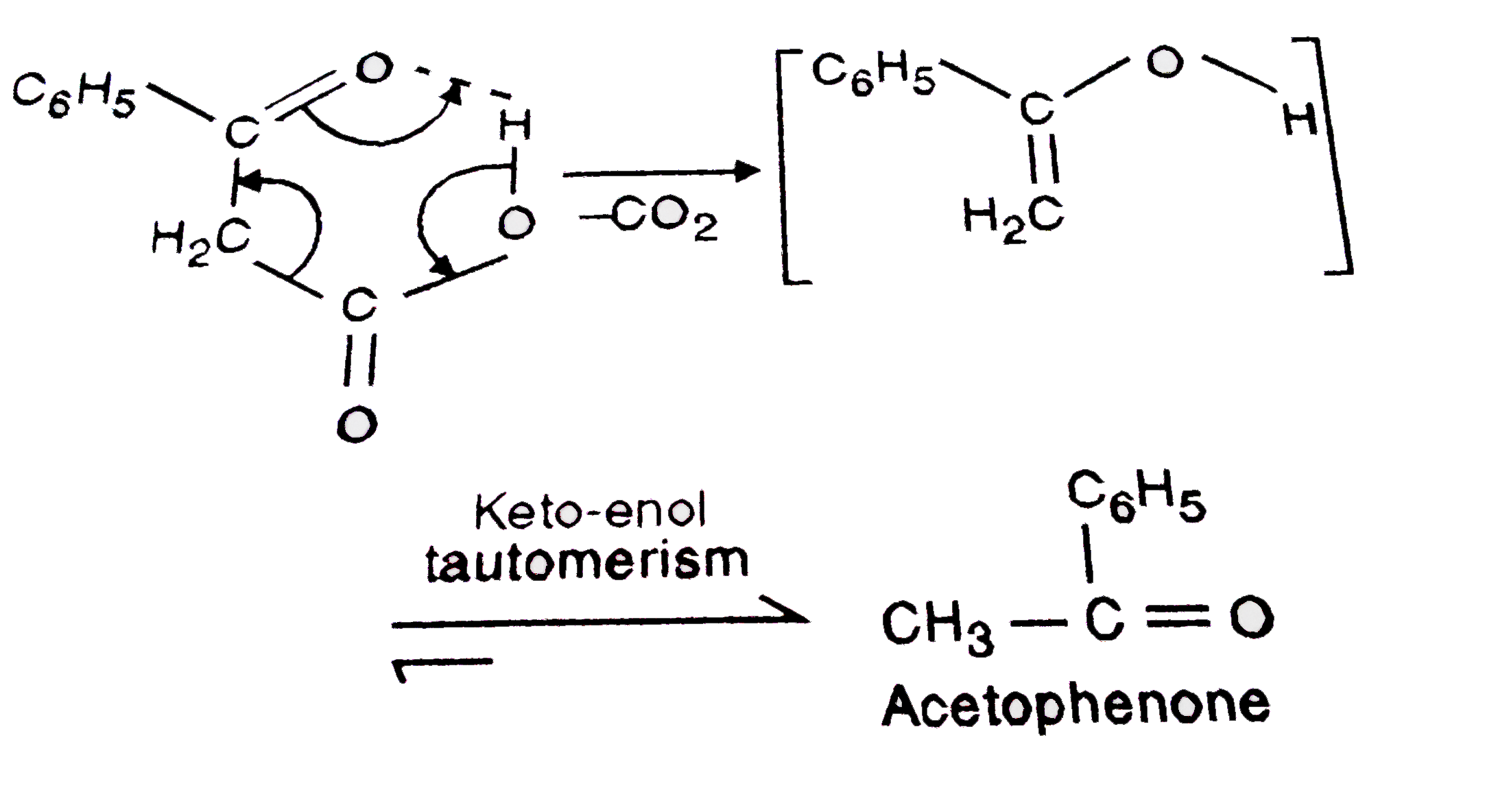
|
|
| 48. |
The standard heats of formation ofCH_(4)(g), CO_(2)(g) and H_(2)O(g) are - 76.2, - 394.8 and -241.6 kJ mol^(-1) respectively . Calculate the amount of heat evolved by burning 1m ^(3) of methanemeasured under normal conditions. |
|
Answer» |
|
| 49. |
Which are the type of chromatography? |
| Answer» SOLUTION :(a) ADSORPTION CHROMATOGRAPHY (B) PARTITION chromatography | |