Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which one of the following order of acid strength is correct ? |
|
Answer» `RCOOH GT HC -= CH gt HOH gt ROH` 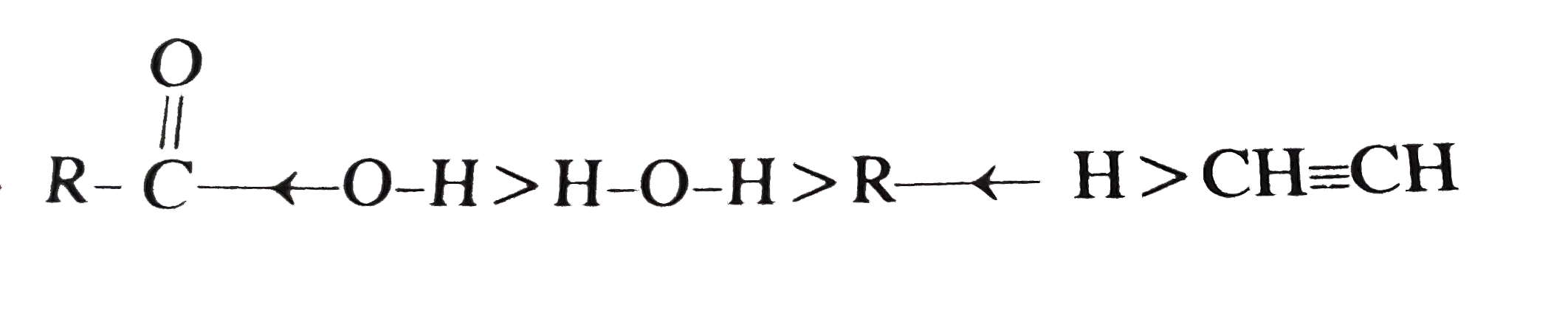
|
|
| 2. |
Volume of 100 liter gas increases to 120 liter at constant temperature and 10 bar external pressure. Calculate the work done by the system [1 liter bar = 24.21 calory ] |
| Answer» SOLUTION :`W= - 4842` CALORY | |
| 3. |
Which of the following are correct about the reaction, FeS_(2) + O_(2)rarrFe_(2)O_3 + SO_(2) |
|
Answer» E.q. wt. of `FeS_2`is M/11 Eq.wt. of `FeS_(2) = (M)/(22//2) = M/11` `2FeS_(2)+11/2O_(2) rarr 4SO_2 +Fe_(2)O_3` In this reaction 22 eletrons INVOLVED for 4 moles of `SO_2` . Eq.wt of `SO_(2)=(M)/(22//4) =M/(5.5) ` `O_(2)^(0) +4erarr2(O^(2-))` has -1 oxidation state . |
|
| 4. |
Which of the following have been arranged in decreasing of oxidation number of sulphur ? |
|
Answer» `Na_(2)S_4O_6 GT H_2S_2O_7 gt Na_2S_2O_3 gt S_8` |
|
| 5. |
The type of hybridisation of carbon atoms of the molecule of the gas involved in Bhopal gas tragedy |
|
Answer» `SP,sp^2` |
|
| 6. |
The signs of DeltaH,DeltaS and DeltaG for a non-spontaneous reaction at all temperature would e |
|
Answer» `+,+,-` |
|
| 7. |
Which one is diamagnetic ? |
|
Answer» `H_(2)^(-)` Molecule/ion`H_(2)^(-) "" H_(2)^(+) "" H_(2) "" He_(2)^(+)` TOTAL `E^(-) " no. " "3123 " ` ODD No. Odd No. Even No. Odd No. only `H_(2)` Has even ELECTRON so, `H_(2)` is diamagnetic |
|
| 8. |
Which is an acid hydrocarbon ? |
|
Answer» `CH_3CH_2CH_2CH_3` |
|
| 9. |
What is the proportion of s-character in sp^(3), sp^(2) and sp hybrid orbitals? What is the order of electronegativity in it? |
Answer» SOLUTION :
|
|
| 10. |
X gm of KHC_(2)O_(4) requires 100 ml of 0.02 M KMnO_(4) in acidic medium. In another experiment, y gm of KHC_(2)O_(4) requires 100 mkl of 0.05 M Ca(OH)_(2). The ratio of x and y is |
|
Answer» Solution :Eqts of `KHC_(2)O_(4)` = Eq of `KMnO_(4)` `(x)/((MW)/(2))=(100xx0.02xx5)/(1000)"......."(1)` Eqts of `KHC_(2)O_(4)` = Eq of `Ca(OH)_(2)` `(y)/(MW)=(100xx0.05xx2)/(1000)""......(2)` from `(1), (2)(2x)/(y)=1""x:y=1:2` |
|
| 11. |
What is the oxidising agent in chlorine water ? |
|
Answer» HCl `HOCl toHCl+[O]` HOCl can FURNISH, NASCENT oxygen. |
|
| 12. |
The _____readily undergoes addition reaction with HBr. |
|
Answer» `(CH_(3))_(2)C=CH_(2)` |
|
| 13. |
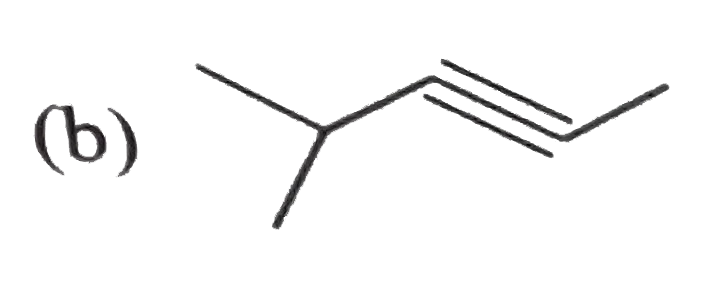
Which of the following compound does not react with NaNH_(2) ? Why ? CH_(3) -= C CH_(3), CH_(3)CH_(2)C-=CH, C_(2)H_(5)C-=C C_(2)H_(5), HC -= CH, CH_(3)CH=CH_(2), CH_(2) = CH-C-=CH |
| Answer» Solution :`CH_(3)C-=C CH_(3), C_(2)H_(5)C -= C C_(2)H_(5), CH_(3)CH -= CH_(2)` does not react with sodamide because there is ABSENCE of `-= CH` group. | |
| 14. |
What is the relation between K_(P) and K_(C), Give one example for which K_(P) is equal to K_(C), |
|
Answer» <P> SOLUTION :The relation between `K_(P)` and `K_(C)` is `K_(P)=K_(C)(RT)^((Deltan_(g)))``K_(P)` = equilibrium constant is TERMS of partial pressure `K_(c)` = equilibrium constant is terms of concentration R = gas constat T = Temperature `Deltan_(g)` = Difference between the sum of the number of MOLES of products and the sum of number of moles of products and the sum number of moles of reactants in the gas phase When `Deltan_(g)=0` `K_(P)=K_(C)(RT)^(0)=K_(C)"ie",K_(P)=K_(C)` Example : `H_(2)(g)+I_(2)(g)hArr2HI(g)` `Deltan_(g)=2-2=0` `:.K_(P)=K_(C)` for the synthesis of HI |
|
| 15. |
Which of the following is not a general characteristic of equilibrium involving physical processes ? |
|
Answer» EQUILIBRIUM is possible only in a close system at a GIVEN temperature |
|
| 16. |
Which of the following is a primary alcohol ? |
|
Answer» neo-Pentyl alcohol |
|
| 17. |
Which of the following statementabout the followingreaction are wrong? 2Cu_(2)(s)+Cu_(2)S(s)rarr 6 Cu(s)+SO_(2)(g) |
|
Answer» Both `Cu_(2)O` and `Cu_(2)S` are REDUCED |
|
| 18. |
The standard molar entropy of H_(2)O (l) " is " 70 JK^(-1) mol^(-1). Will the standard molar entropy of H_(2)O (s) be more, or less than 70 JK^(-1) mol^(-1) ? |
|
Answer» Solution :The standard MOLAR entropy of `H_(2)O(L)` is `70 JK^(-1) mol^(-1)`. The solid form of `H_(2)O` is ice. In ice, molecules of `H_(2)O` are less random than in liquid water. Thus, molar entropy of `H_(2)O (s) LT` molar entropy of `H_(2)O(l)`. The standard molar entropy of `H_(2)O (s)` is less than `70 JK^(-1) mol^(-1)`. |
|
| 19. |
What is plaster of paris? How is it parpared? Give its any two important uses. |
|
Answer» Solution :Plaster of PARIS is ` CaSO_(4) (1)/(2) H_(2) O`. It is prepared by heating gypsum at 373 K. `CaSO_(4). 2H_(2) O underset(373 K)overset("heat")(to) CaSO_(4) (1)/(2) H_(2)O + (3)/(2) H_(2)O` uses: (i) It is used for MANUFACTURE of STATUS. (ii) It is used for FILLING gaps before white washing. |
|
| 20. |
Which statement is correct ? |
|
Answer» Ethanol is more ACIDIC than phenol |
|
| 21. |
The triad of nuclei that is isotonic is |
|
Answer» `._6^14C, ._7^15N, ._9^17F` |
|
| 22. |
Writeelectron configurationof chromium and coppergivereasonof are (z=24)Cu (z=10and Cr=4s^(1) |
|
Answer» Solution :ELECTRON configurationof `Cr : [Ar]^(18) 3d 4S^(1)` Orbitaldiagram of Cr :  Electron configurationof `Cu : (Ar) ^(18)3d^(10)4s^(1) ` Orbitaldiagramof Cu :  |
|
| 23. |
Which ion is present in higher amount than its desirable limit in drinking water may cause blue baby disease ? |
|
Answer» Sulphate |
|
| 24. |
What is absolute entropy ? |
| Answer» SOLUTION :Entropy of a mole substance at STANDARD state is called standard molar entropy or absolute entropy. | |
| 25. |
What is peroxide effect? Give example . |
Answer» Solution :The addition of `HBr` to an alkene in the PRESENCE of organic PEROXIDE, gives the anti Markovniko's PRODUCT. This effect is CALLED peroxide effect. 
|
|
| 26. |
What do you understand by (i) electron deficien t(ii) electron precise and (iii) electron rich compounds of hydrogen? Provide justification with sutable examples |
| Answer» SOLUTION :For ANSWER, CONSULT SECTION 9.6 | |
| 28. |
What volume of 0.2M Ba(MnO_(4))_(2) solution is required for complete oxidation of 25 gm of 89.6% pure Fe Cr_(2)O_(4) in acidic medium according to the reaction MnO_(4)^(-) +FeCr_(2)O_(4) rarr Fe^(+3) +Cr_(2)O_(7)^(-2) +Mn^(+2) |
|
Answer» `700ml` No. of milli equivalents of `FeCr_(2)O_(4)` n-factor of `Ba(MnO_(4))_(2) = 10` n-factor of `FeCr_(2)O_(4) = 7` `:. V xx 0.2 xx 10= (25 xx 89.6 xx 10^(3))/(100xx((224)/(7))):. V = 350ml` |
|
| 29. |
Which inert element is the most reactive ? |
|
Answer» He |
|
| 30. |
Which of the following salts is the most basic in aqueous solution ? |
|
Answer» `Al(CN)_(3)` `CH_(3)CO OK` - salt of weak acid and strong base. `FeCl_(3)`-salt of weak baseand strong acid `Pb(CH_(3)CO O)_(2)`-salt of weak acid and weak base Thus, solution of `CH_(3)CO OK` is most basic. |
|
| 31. |
What is the weight in gram of available O_(2) per litre from a solution of H_(2)O_(2), 10 mL of which when titrated with N//20 KMnO_(4) solution required 25 mL for the reaction ? 2KMnO_(4)+5H_(2)O_(2)+4H_(2)SO_(4)to 5O_(2)+8H_(2)O+2KHSO_(4)+2MnSO_(4) |
|
Answer» |
|
| 32. |
The structure of isobutyl group in an organic compound is ............. . |
|
Answer» 1) `CH_(3)-CH_(2)-CH_(2)-CH_(2)-` |
|
| 33. |
Which of the following will not give white precipitate with ammoniacal silver nitrate solution ? |
| Answer» ANSWER :A::B::C | |
| 34. |
Who stated that "matter is composed of small indivisible particles" ? |
|
Answer» DALTON |
|
| 35. |
Which of the following has/have aromatic character: |
|
Answer»
|
|
| 36. |
Which of the following statement(s) is / are correct for anhydrous calcium chloride |
|
Answer» It is prepared by heating hydrated calcium chloride above 533 K |
|
| 37. |
Which of the following is responsible to rule out the existence of definite paths or trajectories of electrons ? |
|
Answer» Pauli's exculsion principle |
|
| 38. |
Which is/are correct for real gases? |
|
Answer» `underset(P to 0)(L t) (PV_m)` = constant at constant high temperature |
|
| 39. |
Which of the following reagents is used for the separation of acetaldehyde from acetophenone? |
|
Answer» `NH_(2)OH` |
|
| 40. |
What is the correct order of decreasing stability of the following cations ? underset(I)(CH_(3)-overset(o+)(C)H-CH_(3)) "" underset(II.)(CH_(3)-overset(o+)(C)H-OCH_(3)) underset(III.)(CH_(3)-overset(o+)(C)H-CH_(2)-OCH_(3)) |
|
Answer» `II GT I gt III` 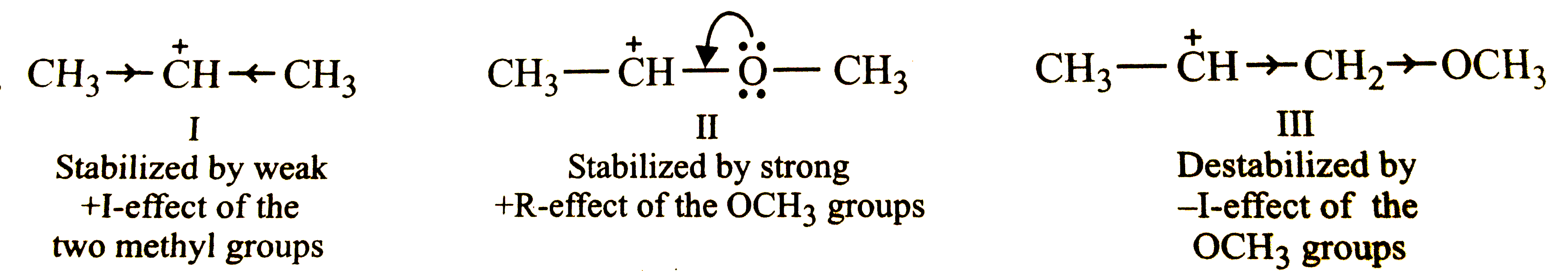 THUS, the stability of CARBOCATIONS decreases in the ORDER: `II gt I gt III`, i.e., OPTION (a) is correct. |
|
| 41. |
Which of the following act as an oxidising as well as reducing agent? |
| Answer» Answer :D | |
| 42. |
Which of the following is carcinogenic? |
|
Answer» BENZOPYRENE |
|
| 43. |
Write down the possible isomers of C_(5)H_(11)Br and give their IUPAC and common names. |
|
Answer» Solution :All possible isomers off `C_(5)H_(11)Br`. Therefore are 8 isomers that have the molecule FORMULA `C_(5)H_(11)Br`. (i) `CH_(3)-CH_(2)-CH_(2)-CH_(2)-CH_(2)Br`1-bromo pentane. (ii) `CH_(3)-CH_(2)-CH_(2)-underset(Br)underset(|)(C)H-CH_(3)` 2-bromo pentane (III) `CH_(3)-CH_(2)-underset(Br)underset(|)(C)H-CH_(2)-CH_(3)`3-bromo pentane. (iv) `CH_(3)-CH_(2)-underset(CH_(3))underset(|)(C)H-CH_(2)Br`1-bromo-2-methyl BUTANE. |
|
| 44. |
What is disproportionation reaction ? Give example. |
Answer» SOLUTION :In which REACTION one COMPONENT forms oxidation and reduction both such type of reaction is known as DISPROPORTIONATION reaction. 
|
|
| 45. |
Three moles of B_2H_6 are completely reacted with methanol. The number of moles of boron containing product formed is: |
|
Answer» |
|
| 46. |
van der Waals constant b in corrected equation for real gases represents |
|
Answer» MEASURE of effective size of gas molecules |
|
| 47. |
The visible part of electromagnetic radiations is |
|
Answer» `2000 Å to 4000 Å` |
|
| 48. |
Which of the following gases has the highest density under standard conditions? |
| Answer» Answer :D | |
| 49. |
Which one of the following is/are buffer solution(s)? |
|
Answer» `0.8 M H_2S + 0.8 M KHS ` <BR>` 2 M C_6 H_5 NH_2 +2 M C_6H_5 NH_3 Br` |
|
| 50. |
What are free radical initiators ? |
| Answer» Solution :The types of reagents that remote homelytic in substrate are called as free radical INITIATORS . They are SHORT LIVED and are HIGHLY REACTIVE . | |