Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The size of iso electronic species F-Ne and Na^+ is affected by |
|
Answer» NUCLEAR charge |
|
| 2. |
What will be the order of energy levels 3s, 3p and 3d in case of H-atom ? |
| Answer» SOLUTION :AL' have EQUAL ENERGY | |
| 3. |
The. van der Waal's equation of law of corresponding states for 1 mole of gas is : |
|
Answer» `(pi + 3/(phi^2)) (3 phi - 1) = 8 theta` `P = pi P_C = (pi .a)/(27 b^2 ) , V = phi , V_C = 3bphi` `T = theta T_C = (a theta)/(27 Rb)` `IMPLIES ((pi a)/(27 b^2) + a/(9 b^2 phi^2)) (3 b phi - b) = (R xx a theta)/(27 Rb)` `implies (pi + 3/(phi^2)) (3 phi - 1) = 8 phi`. |
|
| 4. |
Which out of the following has the largest size ? |
|
Answer» `RB^(+)` |
|
| 5. |
What is blue baby syndrome ? |
| Answer» Solution :In many agricultural areas, tube well WATER is contaminated with nitrate `(NO_(3)^(-))` ions. Excessive NITRATES are especially dangerous to INFANTS. In the infants's digestive SYSTEM, nitrates ion is reduced to nitrate ion. This results in methemoglobinenia or blue baby syndrome. The baby turns blue and can die after drinking the water if not treated. | |
| 6. |
What is covalent radius ? . How would you determine the covalent radius of chlorine atom . |
|
Answer» Solution :The DISTANCE between the nuclei of two covalently bonded atoms is known as covalent distance or inter-nuclear distance. The one-half of this inter-nuclear distance is called covalent RADIUS. The covalent distance (Cl-Cl) of Cl, MOL ecule is EXPERIMENTALLY found as 198 PM (1.98 A). Its covalent radius is 99 pm `(0.99oversetoA)` . CI-CI Inter nuclear distance =`1.98 oversetoÅ` `therefore r_(Cl)=1.98//2=0.99oversetoÅ`. |
|
| 7. |
To which orbit the electron in the hydrogen atom will jump on absorbing 12.1 eV of energy? |
|
Answer» Solution :ENERGY of ELECTRON in the nth ORBIT of H-atom is `E_(N) = (-13.6)/(n^(2)) eV :. E_(1) = -13.6 eV` After absorbing 12.1 eV, the energy will be `= -13.6 + 12.1 eV = -1.5 eV` THUS, `(-13.6)/(n^(2)) = - 1.5 eV or n^(2) = 9 or n = 3` Thus, electron will jump to 3rd orbit. |
|
| 8. |
Which of the following can not be obtained when alkenes are oxidised with KMnO_(4) and then followed by acid hydrolysis ? |
|
Answer» alkanoic acids |
|
| 9. |
Which of the following substances will have positive value of DeltaH_("formation")? |
|
Answer» `Br_(2)(l)`<BR>`O_(3)(g)` |
|
| 10. |
When 4 gm of Mg burnt with O_(2) form oxide and on dilution of metal oxide form metal hydroxide which statement is/are correct for above series of reaction? |
|
Answer» Gram equivalent of metal, metal oxide an metal hydroxide are equal `MgO+H_(2)OrarrMg(OH)_(2)` Eq. of MG = Eq. of Mgo = Eq. of `Mg(OH)_(2)` `(4)/(12)=(W)/(20)impliesW=6.66gm` `therefore` Eq of MgO = 6.66 Eq. of Mg = Eq. of `Mg(OH)_(2)` `(4)/(12)=(w)/(29)impliesW=(29)/(3)=9.66gm` |
|
| 11. |
What isthe ratioof massof protonandelectron? |
| Answer» Solution :`(1.673 xx 10^(24))/(9.109 xx 10^(28))= 1.836 xx 10^(3)` | |
| 12. |
Which of the following structures is the most preferred and hence of lowest energy for SO_(3)- |
|
Answer»
 has maximum NUMBER of COVALENT bonds and hence is of LOWEST energy. |
|
| 13. |
Which orbital has only positive value of wave function at all distances from the nucleus- |
|
Answer» 3d |
|
| 14. |
Which of the following relationship are correct? |
|
Answer» <P>`( DeltaH- DeltaE)/( DeltanxxT ) =`Constant (B) is WELL known relationship (c ) is wrong because `DeltaU = q+w` (d) follows from `- DeltaG^(@) = RT ln K`. (e ) represents Kirchhoff equation. |
|
| 15. |
What is the percentage of lead in lead pencil |
|
Answer» |
|
| 16. |
What volume of oxygen at S.T.P. is required to completely burn 65.0 g ethyl alcohol ? |
|
Answer» |
|
| 17. |
Which volatile compound burns with green edged flame ? |
|
Answer» `(C_(2)H_(5))_(3)B` |
|
| 18. |
Which of the following are isoelectronic species |
|
Answer» I,II,III |
|
| 19. |
Which one of the following pairs constitutes isotones ? |
|
Answer» `._(6)C^(13) and ._(6)C^(14)` |
|
| 20. |
What do you expect the nature of hydrides formed by elements of atomic number 15, 19 ,23 and 44 with hydrogen? |
| Answer» Solution :Atomic NUMBER 15 is a non-metal (p) and its hydride is `PH_3` which is covalent .ELEMENT with atomic number 19 is an alkali metal (K) which forms ionic hydride. | |
| 21. |
Two geometrical isomers are given by the following compound |
|
Answer» Ethylidene bromide |
|
| 22. |
What products are expected from the disproportionation reaction of hypochlorous acid ? |
|
Answer» `HClO_(3)` and `Cl_(2)O` |
|
| 23. |
Write the steps to be followed while balancing redox equation by oxidation number method. |
|
Answer» Solution :Oxidation number method: This method is based on the fact that Number of electrons lost by atoms = Number of electrons gained by atoms Steps to be followed while balancing Redox reactions by Oxidation Number method: 1. Write skeleton equation representing redox reaction 2. Write the oxidation number of atoms undergoing oxidation and reduction. 3. Calculate the increase or decrease in oxidation numbers per atom. 4. Make increase in oxidation number equal to decrease in oxidation number by multiplying the formula of oxidant and reductant by suitable numbers. 5. Balance the equation atomically on both sides except O and H atoms. 6. Balance OXYGEN atoms by adding REQUIRED number of water molecules to the side deficient in oxygen atoms. 7. ADD required number of `H^(+)` ions to the side deficient in HYDROGEN atom if the reaction is in acidic medium. 8. For reactions in BASIC medium, add `H_(2)O` molecules to the side deficient in hydrogen atoms and simultaneously add equal number of `OH^(-)` ions on the other side of the equation. 9. Finally balance the equation by cancelling common species present on both sides of the equation. |
|
| 24. |
What is the derivatives of ammonia ? Give example. |
|
Answer» SOLUTION :Ammonia is a weak base, In this one or more hydrogen is SUBSTITUTE by other GROUP is known as derivatives of ammonia. e.g. : Methyl amine, `CH_3NH_2` DIMETHYL amine, `(CH_3)_2NH` |
|
| 25. |
When a strong beam of light is passed through a colloidal solution, the light will |
|
Answer» be REFLECTED |
|
| 27. |
Two solids A and D dissociates into gaseous products as follows C(s)hArrB(g)+C(g),K_(P_(1))=300 , D(s)hArrE(g)+C(g)K_(P_(2)=600 at 27^(@)C, then find the total pressure of the solid mixture. |
|
Answer» |
|
| 28. |
What information is derived by the shape of the graph given for a fixed mass of gas? |
|
Answer» Solution :The graph PV/T versus T is a straight line parallel to the temperature axis. A jump in te curve at increased temperature denotes the CHANGE of composition. the NUMBER ofmoles of the GAS may be DOUBLED due to dissociation. For example the dimer of nitrogendioxide decomposes to its monomer. `underset("1 mole")(N_(2)O(4)(g)) to underset("2 mole")(2NO_(2)(g))` |
|
| 29. |
Which one of the following is the first in first transition series? |
|
Answer» Sc |
|
| 30. |
Which one of the following is not compatible with arenes ? |
|
Answer» Greater stability |
|
| 31. |
Water is supercooled to -4^(@)C . The enthalpy( H ) is |
|
Answer» same as ice at `- 4^(@)C` |
|
| 32. |
Under the influence of electric filed, which of the following statements is true about the movement of electrons and holes in a p-type semiconductor ? |
|
Answer» Electron will move TOWARDS the posiitively charged plate through electron holes. |
|
| 33. |
Velocity distribution is depend upon ? |
| Answer» SOLUTION :(i) TEMPERATURE (II) MOLECULAR mass of gas. | |
| 34. |
Which one is least evaporite? |
|
Answer» HF Electronegativity of F is highest so STRONG HYDROGEN bond is form. So HF is leastevaporite. ..... H - F : ...... H - F : ..... |
|
| 35. |
Which of the following alcohol is used as beverage? |
|
Answer» Propanol |
|
| 36. |
The volume of water needed to dissolve 1g of BaSO_4 (K_(sp)=1.1 xx 10^(-10)) at 25^(@) Cis { M. W of BaSO_4 is 233} |
|
Answer» 820 L ` 1 gm =(1)/(233)` moles ` therefore ` Volume of water ` = (1//233)/( 10 ^(-5))= 430 L` |
|
| 37. |
What is decomposition reaction? Give two examples |
|
Answer» SOLUTION :Chemical reactions in which a compound splits up into TWO or more simpler substances are called decomposition reaction A B `RARR` A + B Example `2KClO_(3) rarr 2KCL + O_(2)` `PCl_(5) rarr PCl_(3) + Cl_(2)` |
|
| 38. |
Which of the following are incorrect about fungicides |
|
Answer» THIRAM |
|
| 39. |
The value of DeltaG^ө for the phosphorylation of glucose in glycolysis is 13.8 kJ/mol. Find the value of K_c at 298 K. |
|
Answer» Solution :`DeltaG^ө`=13.8 kJ `mol^(-1)= (13.8xx10^3) "J mol"^(-1)` `DeltaG^ө=-RT ln K_c` `therefore DeltaG^ө`=-2.303 RT LOG `K_c` `therefore log_10 K_c=-(DeltaG^ө)/(2.303RT)` `=-(13.8xx10^3 "J mol"^(-1))/((2.303)xx(8.314 "J mol"^(-1) K^(-1))xx(298 K))` =-2.4186=`bar3.5814` `therefore K_c`=Antilog `bar3.5814=3.814xx10^(-3)` |
|
| 40. |
Which of the following is used in photoelectric cells ? |
|
Answer» Na |
|
| 41. |
Which of the following is correct matching of Column-I containing formulas and Column-II containing their names: |
|
Answer» `i rarr d, ii rarr e, iii rarr a, IV rarr F, v rarr C, vi rarr b` |
|
| 42. |
Write the resonance structures of CO_3^(2-) and HCO_3^- |
Answer» SOLUTION :
|
|
| 43. |
There is S-S bond in . |
|
Answer» `S_(2)O_(6)^(2-)` `.^(Theta)O-underset(O)underset(||)OVERSET(O)overset(||)S-underset(O)underset(||)overset(O)overset(||)S-O^(Theta)` (B) `S_(4) O_(6)^(2-)` (Tetrathionate ion) `^ThetaO-underset(O)underset(||)overset(O)overset(||)S-underset(O)underset(||)overset(O)overset(||)S-O^(Theta)` (c ) `S_(2) O_(3)^(2-)` (Thiouslphate ion) `.^(Theta)O-underset(O)underset(||)overset(O)overset(||)S-O^(Theta)` (d) `S_(2) O_(7)^(2-)` (Pyrosulphate ion) `^(Theta)O-underset(O)underset(||)overset(O)overset(||)S-underset(O)underset(||)overset(O)overset(||)S-O^(Theta)` In (d) there is no `S -S` bond . |
|
| 44. |
Total number of alkenes obtained by dehydration of 3,4-diethylhexan-2-ol in acidic medium ? |
Answer» 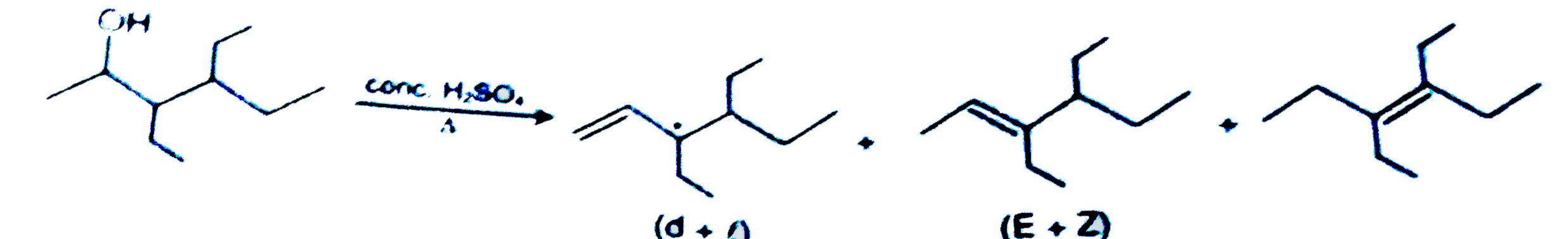
|
|
| 45. |
Which of the following ammonium salt will not give acid on heating? |
|
Answer» `(NH_(4))_(2)HPO_(4)` |
|
| 46. |
What is the hybridisation state of central I atom is I_3^(-)? |
|
Answer» `SP^(3)` |
|
| 47. |
Which of the following reactions is an example of Friedel craft alkylation. |
|
Answer»

|
|
| 48. |
The threshold frequency (v_(0)) for a metal is 5 xx 10^(14)S^(-1). Calculate the kinetic energy of emitted electron when a radiation with frequency of 2 xx 10^(15) sec^(-1) hits the metal surface. |
| Answer» SOLUTION :`4.13xx10^(-19)J` | |
| 49. |
What are the necessary conditions for an ideal solution? Give two example. |
|
Answer» Solution :For an ideal solution (i) There is no change in VOLUME of mixing two components (SOLUTE and solvent) `DV_(mixing") =0` (ii) There is no exchange of heat when the solute is dissolved in solvent `(Delta H_("mixing")=0)` (iii) Escaping tendency of the solute and the solvent present in it should be same as in pure liquids. (IV) Examples: For ideal solution: BENZENE and toluene, n-Hexane and n-Heptane, cthyl bromide and ethyl iodide, chloobenzene and bromo benzene. |
|







