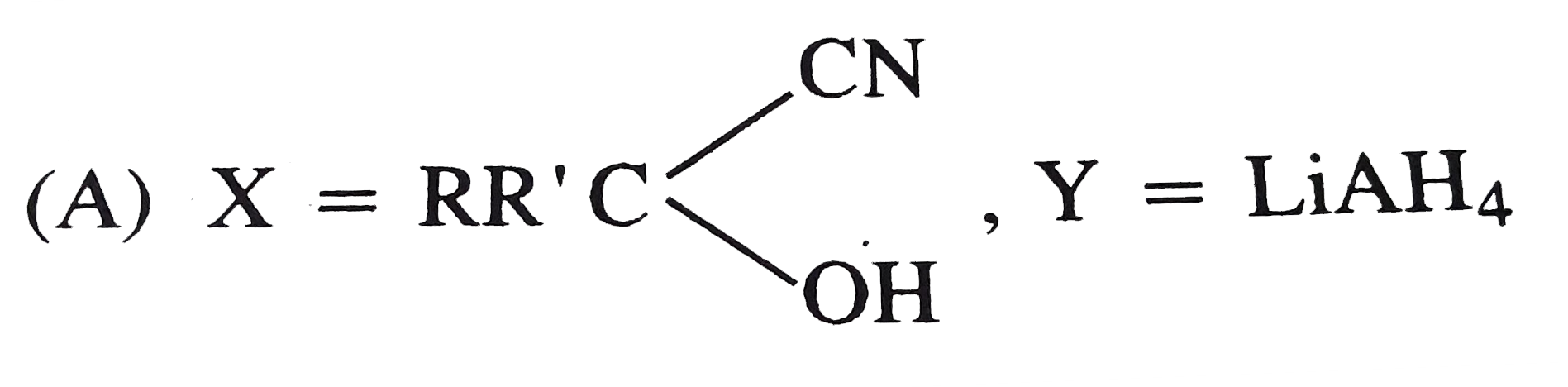
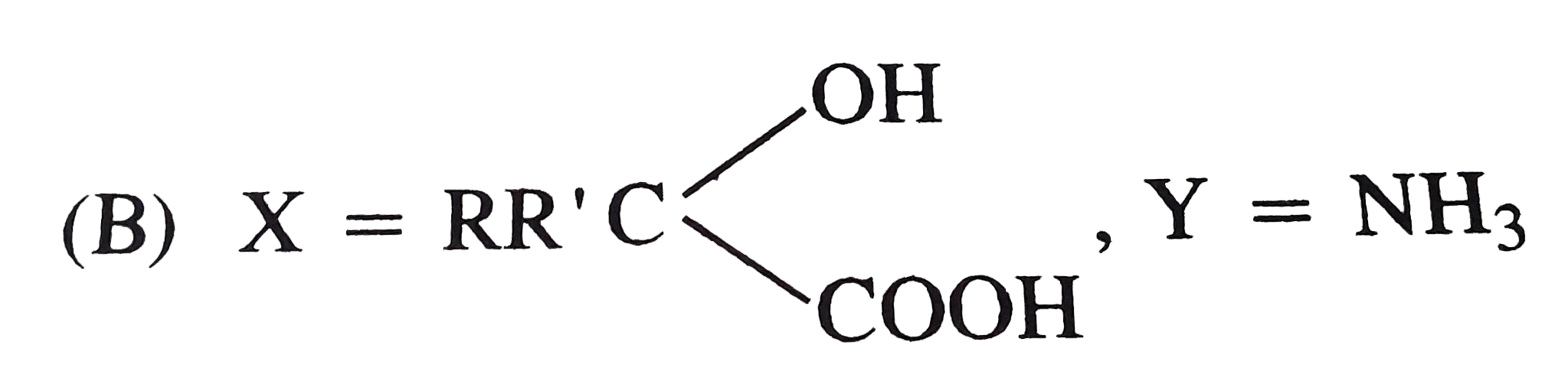
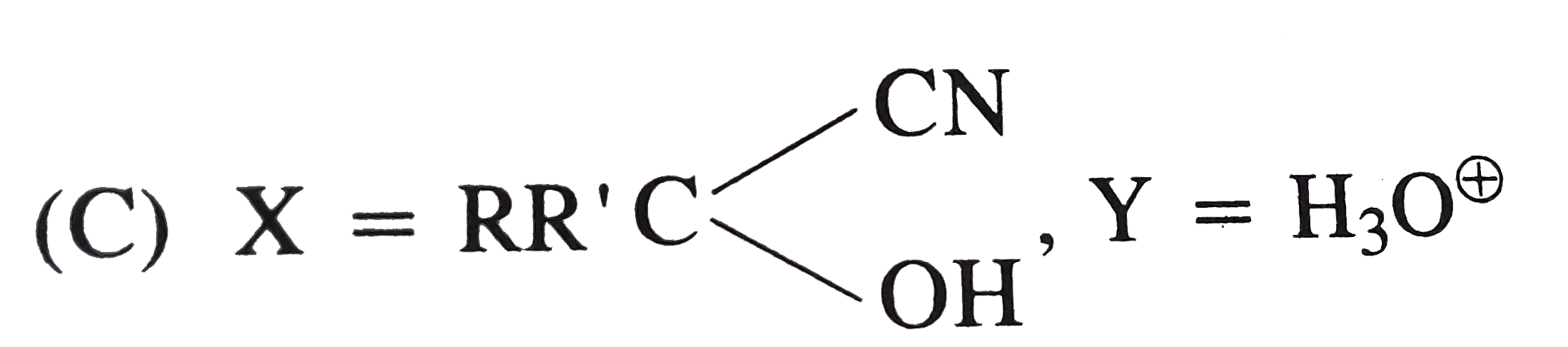
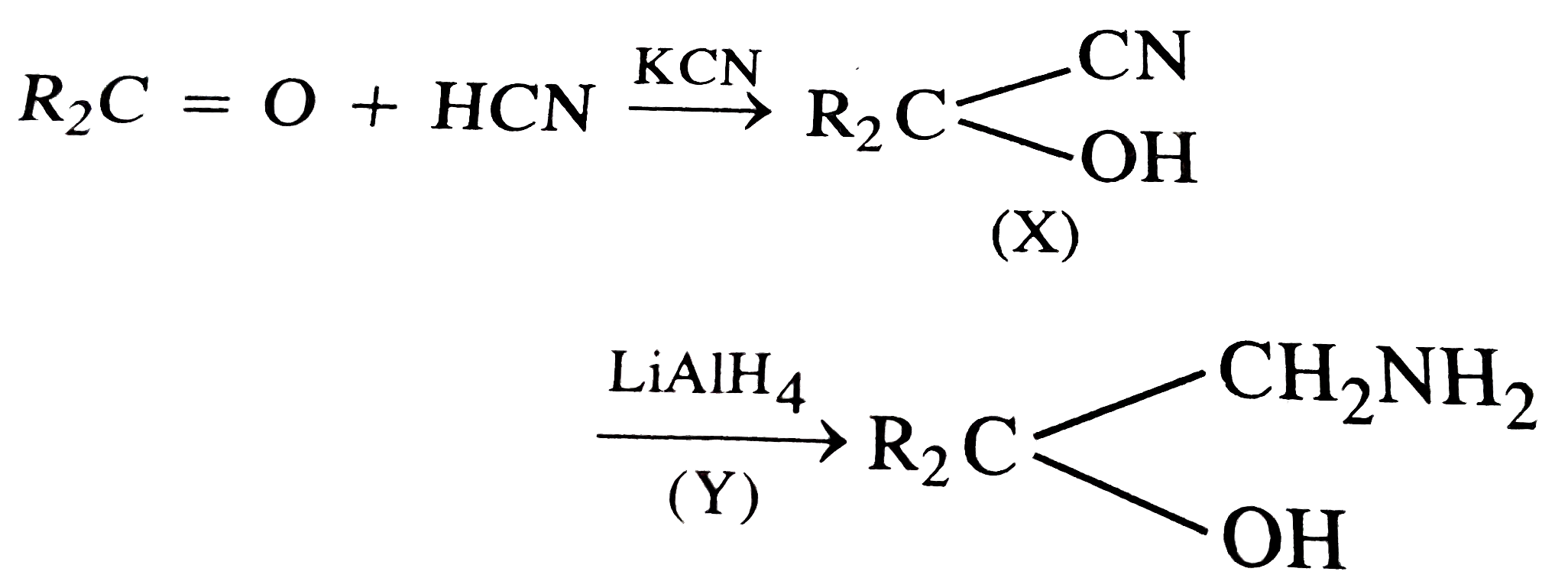Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The valence electron configuration of an element with atomic number 23 is |
|
Answer» `3D^(5)` |
|
| 2. |
Which does not show substituion in benzene ring? |
|
Answer» `CIDO_(3)H` |
|
| 3. |
What changes observed when gas is heated ? |
| Answer» Solution :When we heated GAS, SPEED of molecules and kinetic energy increases. Pressure increases with increases of AVERAGE speed of kinetic energy increases it will collide with wall of CONTAINER. | |
| 4. |
Which of the following pairs of metals is purified by Van Arkel method ? |
|
Answer» NI and Fe |
|
| 5. |
Which of the following expressions represents the electron probability function (D) ? |
|
Answer» `4pi r dr PSHI^(2)` |
|
| 6. |
Which of the following alkyl halides is not suitable for Corey-House synthesis of alkanes? |
|
Answer» `CH_3Br` |
|
| 7. |
Which of the following have been arranged in decreasing order of oxidation number of sulphur? |
|
Answer» `Na_(2)S_(4)O_(6)gtH_(2)S_(2)O_(7)gtNa_(2)S_(2)O_(3)gtS_(8)` |
|
| 8. |
Write redox reaction between fluorine and water. |
|
Answer» Solution :`F_(2)` is a STRONG oxidising AGENT. It oxidises `H_(2)O` to either `O_(2)` or `O_(3)` `2F_(2)(g) + 2H_(2)O(L) to O_(2) (g) + 4H^(+) (AQ) + 4F^(-) (aq)` `3F_(2)(g) + 3H_(2)O(l) to O_(3)(g) + 6H^(+) (aq) + 6F^(-)(aq)` |
|
| 9. |
Which cannot be oxidised by H_(2)O_(2)? |
|
Answer» Solution :`Na_(2)SO_(3)` is oxidised by `H_(2)O_(2)" to "Na_(2)SO_(4).PbS` is oxidised by `H_(2)O_(2)" to "PbSO_(4).KI` is oxidised by `H_(2)O_(2)" to "I_(2)O_(3)` cannot be oxidised by `H_(2)O_(2)` but it is reduced to `O_(2)" by "H_(2)O_(2)`. `H_(2)O_(2)+O_(3) RARR H_(2)O+2O_(2)` |
|
| 10. |
Which of the following exponential equation of DeltaG^@= - RT lnK ? |
|
Answer» `K=e^(DELTAG//RT)` |
|
| 12. |
Urine normally has a pH of 6.0 . A patient eliminates 1.3 litres of uring per day . How many moles of H^(+) ions does he eliminate in a day ? |
|
Answer» `1.3 xx 10^(-3)` |
|
| 13. |
What is meant by 'demineralised water ' and how can it be obtained ? |
| Answer» Solution :What which is free from all CATIONS and anions is CALLED demineralised WATER. It is obtained by passing hard water first through CATION exchange RESIN and then through anion exchange resin. | |
| 14. |
The velocity of light is 3 xx 10^(10) cm/s. Express it in miles/hour. |
|
Answer» SOLUTION :Since, `1 cm = 6.214 XX 10^(-6)` miles and `1 s = 1/(3600)` HOUR. Therefore, the CONVERSION factor for cm is: `1 = (6.214 xx 10^(-6))/(1 cm)` and that for second would be: `1=(1/3600) "hours")/(1 s)` Hence, `3 xx 10^(10) cm//s = (3 xx 10^(10) cm xx (6.214 xx 10^(-6) "miles")/(1 cm))/(1s xx (1/(3600 "hours"))/(1 s))` `=(3 xx 10^(10) xx 6.214 xx 10^(-6))/(1/3600)` `=6.711 xx 10^(8)` miles/hour |
|
| 15. |
What is the oxidation number ofNi in Ni(CO)_4 |
| Answer» SOLUTION :OXIDATION number of NI in `Ni(CO)_4` is x+4(0)=0,x=0 | |
| 17. |
What is oxidation number of .C. in HClO_(4)andHClO_(3) ? |
|
Answer» Solution :In `HClO_(4)toCl^(+7)` In `HClO_(3)toCl^(+5)` |
|
| 18. |
Which reaction is possible? |
|
Answer» `KMnO_(4)+Na_(2)SO_(4) rarr` |
|
| 19. |
Which of the following order is correct- |
|
Answer» `AlCl_(3) lt MgCl_(2) lt NaCl` , polarising power `overset((+1))(NaCl) lt overset((+2))(MgCl_(2)) lt overset((+3))(AlCl_(2))` Bond order`PROP(1)/("Bond length")` Bond order=`("bond order of each C-O bond")/("Total no. of resonating structures")` `CO to C-=O implies (2+1)/(1)=3.0` `CO_(2) to O =C=O implies (2+2)/(2)=2.0`  Hence, the decreasing order of bond length is, `CO_(3)^(2-) gt HCO_(2)^(-) gt CO_(2) gt CO` (c)  Correct order of dipole moment: `BeCl_(2) lt NF_(3) lt NH_(3)` (d) Correct order of bond angle: `underset(120^(@))(BF_(3)) gt underset(109^(@)28')(SiH_(4)) gt underset(107^(@))(NH_(3)) gt underset(94^(@))(H_(2)S)` |
|
| 20. |
Water molecule has bent structure even it undergoes sp^(3) hybridization. |
| Answer» SOLUTION :Due to the REPULSION between the two lone pair of ELECTRONS in water molecule, the STRUCTURE become bent. | |
| 21. |
Why can sugar (sucrose) melts on heating but common salt (sodium chloride) doesnot melt so easily ? |
| Answer» Solution :Common salt (NaCl) is an ionic compound in which `Na^(+)` ions and `Cl^(-)` ions are held together by strong electrostatic forces of attraction. Hence, its MELTING point is very high. SUCROSE is a covalent compound and no ions are present. Molecules are held together by WEAK van der Waal forces and hydrogen bonds. Hence, it has LOW melting point. | |
| 22. |
Which oneamong thefollowingelementshas thelowestfirstionisationenthalpyand which of thehashighestfirstionisationenthalpy? Li, K , Ca , S andKr. |
| Answer» Solution :Li belongs to the 2ndperiod. S TOTHE 3rdwhile K and Krbelongto the 4thperiod.BothLi and Kare ALKALI metalsbut the sizeof K isbiggerthan thatofLi . Furthersince alongany period, `Delta_(i) H_(1)` of alkalimetas is thelowestwhile of thatinertgas is THEHIGHEST. Thereforek hasthelowest`Delta_(i) H_(1)` while Krhas thehighest`Delta_(i) H_(1)` | |
| 23. |
Which pair of gases has the same average rate of diffusion at 25^(@)C? |
|
Answer» He and Ne |
|
| 24. |
underset(("Orange solid"))((A))overset(Delta)rarrunderset(("Yellow"))((B))+underset(("Green"))((C)) +underset(("Paramagnetic"))((D)) Compound (C) is also obtained on heating of: |
|
Answer» `(NH_(4))_(2)Cr_(2)O_(7)` |
|
| 25. |
Weakest acid among CH_(4)COOH, CH_(2)ClCOOH, CHCl_(2)COOH is …….. |
| Answer» SOLUTION :`CH_(3)COOH` | |
| 26. |
Why are lithium salts commonly hydrated and those of the other alkali ions usually anhydrous ? |
|
Answer» Solution : LITHIUM is the smallest in size AMONG the alkali metals. Hence, `Li^(+)`ion can polarize water molecules more easily than other alkali metals. As a RESULT, water molecules get ATTACHED to lithium salts as water of crystallization. Hence, lithium salts such as trihydrated lithium chloride are commonly hydrated. As the size of the ions increases, their polarizing power decreases. Hence, other alkali metal ions usually form anhydrous salts. |
|
| 27. |
The value of DeltaH for cooling 2 moles of an ideal monoatomic gas from 125^@ Cto 25^@ C at constant pressure will be [given C_p = 5/2 R] ………………. . |
|
Answer» `-250 R` `T_(f)=25^(@)C = 298K` `DeltaH = nC_(P)(T_(i)-T_(f))` `DeltaaH =2XX(5)/(2)R(298-398)` `DeltaH =-500R` |
|
| 28. |
Which of the following statements is trud about the science of atoms are molecules? |
|
Answer» We can see, weigh and perceive the atoms and MOLECULES through naked eyes. |
|
| 29. |
The reagent used to convert RCOOH rarr RCH_(2)OH is |
|
Answer» `NaBH_(4)` |
|
| 31. |
Which one of the following is a correct set? |
|
Answer» `H_(2)O, SP^(3),` BENT |
|
| 32. |
What is meant by Convection ? |
|
Answer» Solution :A FUNCTIONAL group is an atom or a specific combination of bonded atoms that react in a CHARACTERISTIC way, IRRESPECTIVE of ORGANIC molecule in which it is present. The reaction of an organic compound takes place at the functional group. e.g., Alcohol -OH group Ether -O-group |
|
| 33. |
What will happen when Br_2//C Cl_4 react with (a) cis But-2-ene (b) trans But-2-ene. |
|
Answer» |
|
| 34. |
The structure of 2-Iodo-2-methylpropane is ……………………….. . |
| Answer» SOLUTION :`CH_3-UNDERSET(I)underset(|)OVERSET(CH_3)overset(|)C-CH_3` | |
| 35. |
Which one of the following does not involve coagulation ? |
|
Answer» FORMATION of delta region |
|
| 36. |
The value of K_(p) for the water gas reaction, CO +H_(2)O hArr CO_(2) +H_(2)is 1.06 xx 10^(5) at 25^(@)C . Calculate the standard free energy change for the reaction at 25^(@)C. |
|
Answer» |
|
| 37. |
Total number of valence electrons in the molecule CH_(3)CHO is |
|
Answer» 18 |
|
| 38. |
The reagent used in the conversion: Hg to HgO is |
|
Answer» `H_2O_2` |
|
| 40. |
Which of the following set has atomic numbers of only representative elements ? |
|
Answer» 2,10,17,35 |
|
| 41. |
Which of the following pairs of compounds is ring-chain isomer ? |
|
Answer» `CH_(3)-undersetunderset(OH)(|)CH-undersetunderset(OH)(|)CH-CH_(3)` |
|
| 42. |
Which one of the followingis an aromatic compounds |
|
Answer»
|
|
| 43. |
What units do the following abbreviations represent ? (a) mug (b) dm (c) ps (d) kA (e) m mol ? |
|
Answer» |
|
| 44. |
The tatio of the cationic radius to anionic radius in an ionic crystal is greater than 0.732. Its co-ordination number is |
|
Answer» 1 |
|
| 46. |
What is the value of gas constant R in J mol^(-1 ) K^(-1)? |
|
Answer» 82.1 |
|
| 47. |
Which one is present in troposphere? |
|
Answer» `CO_2` |
|
| 48. |
Write the atomic number of the element in the third period and group 17 of the period table. |
| Answer» Solution :In the third period third SHELL is filled and there are 8 elements (3S 3p). These will be two Ar(18). So element 11 and 12 are s BLOCK while 13 to 18 are p block belonging to groups 13 to 18. So the elment in the `17^th` GROUP must have an atomic number 17 (CHLORINE). | |
| 49. |
The solubility product (K_(sp)) of the following compound s are given at 25^(@)C {:("Compound" ,K_(sp)),(AgCl,1.1xx10^(-10)),(Agl,1.0xx10^(-16)),(PbCrO_(4),4.0xx10^(-10)),(Ag_(2)CO_(3),8.0xx10^(-12)),(,):} The mostsoluble and least soluble compound are respectively |
|
Answer» `AGCL and PbCrO_(4)` `S=sqrt(K_(sp))`, i.e., `10^(-5)M(AgCl), 10^(-8) M (AgI), 2xx10^(-7)M (PbCrO_(4))`. Last salt isof the type `A_(2)B` for which `S=(K_(sp)//4)^(1//3)=(2xx10^(-12))^(1//3)~=10^(-4)` M. Thus, most soluble is `Ag_(2)CO_(3)` and least soluble is AgI. |
|