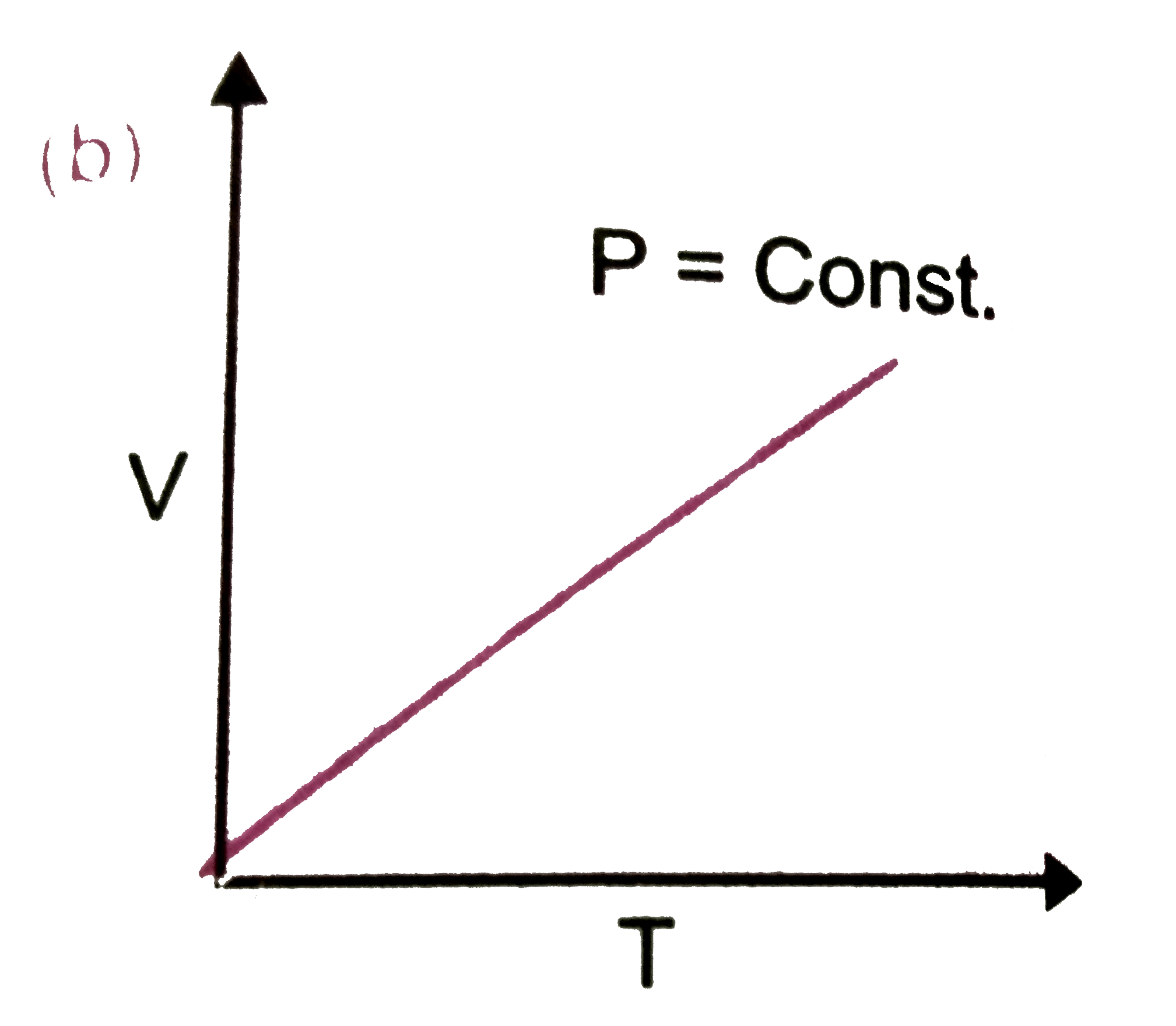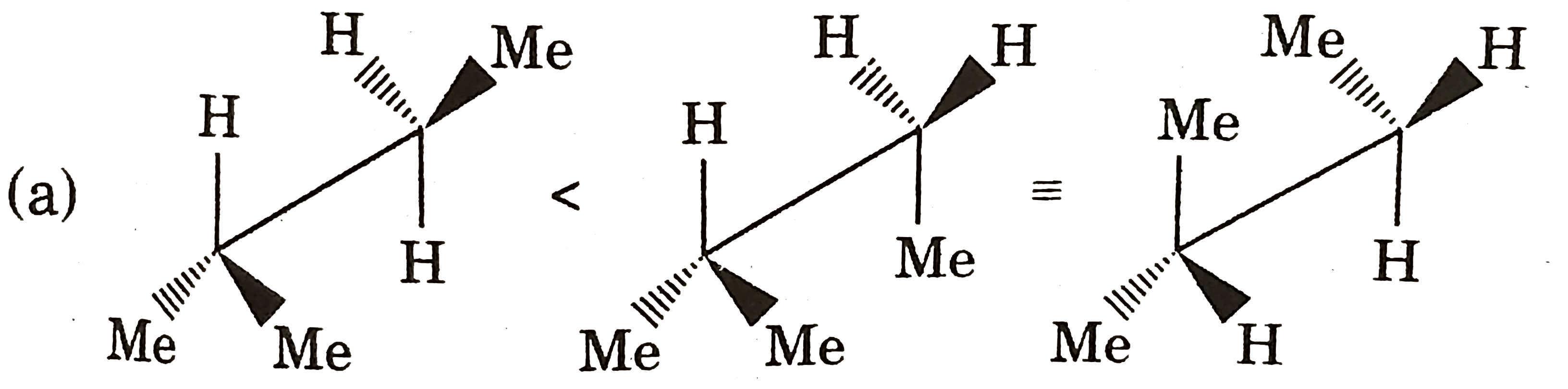Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
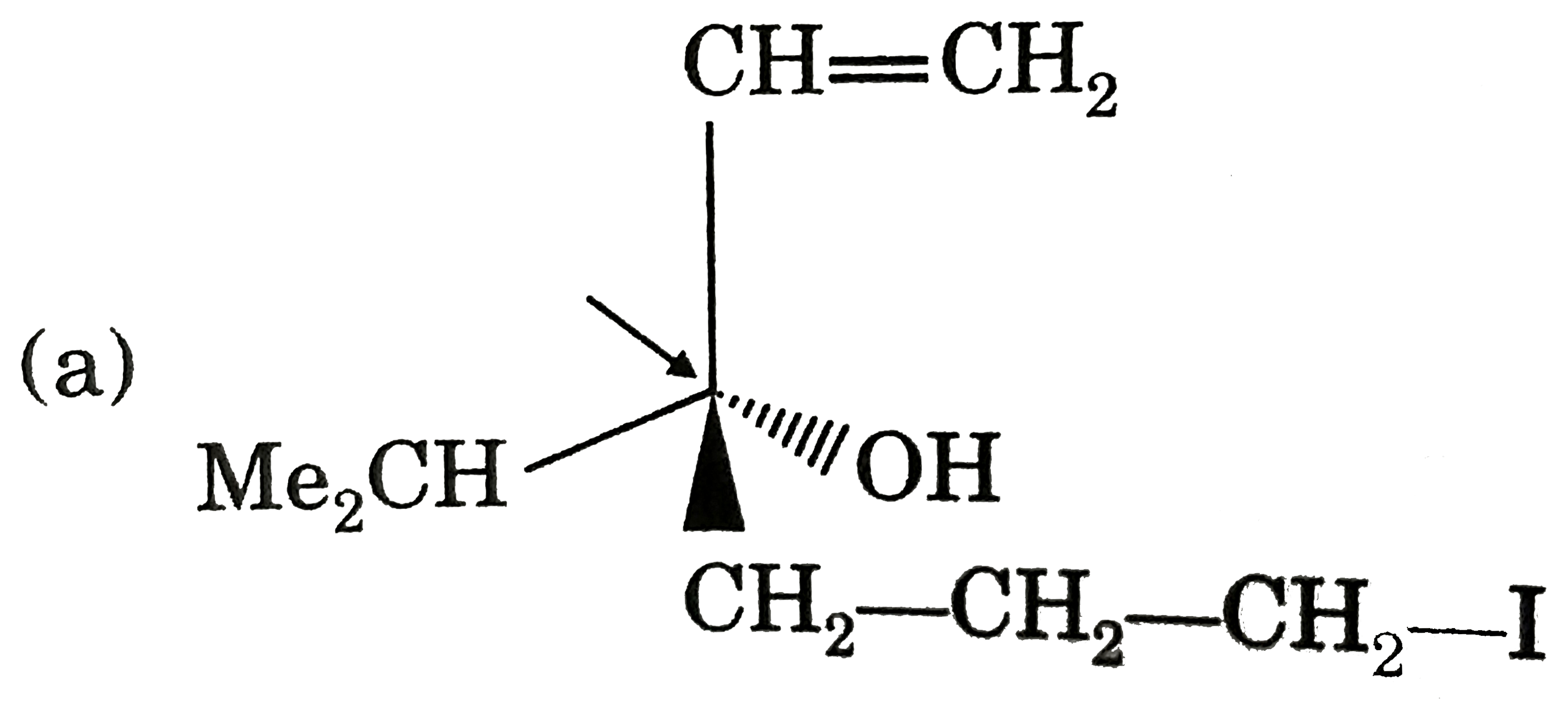
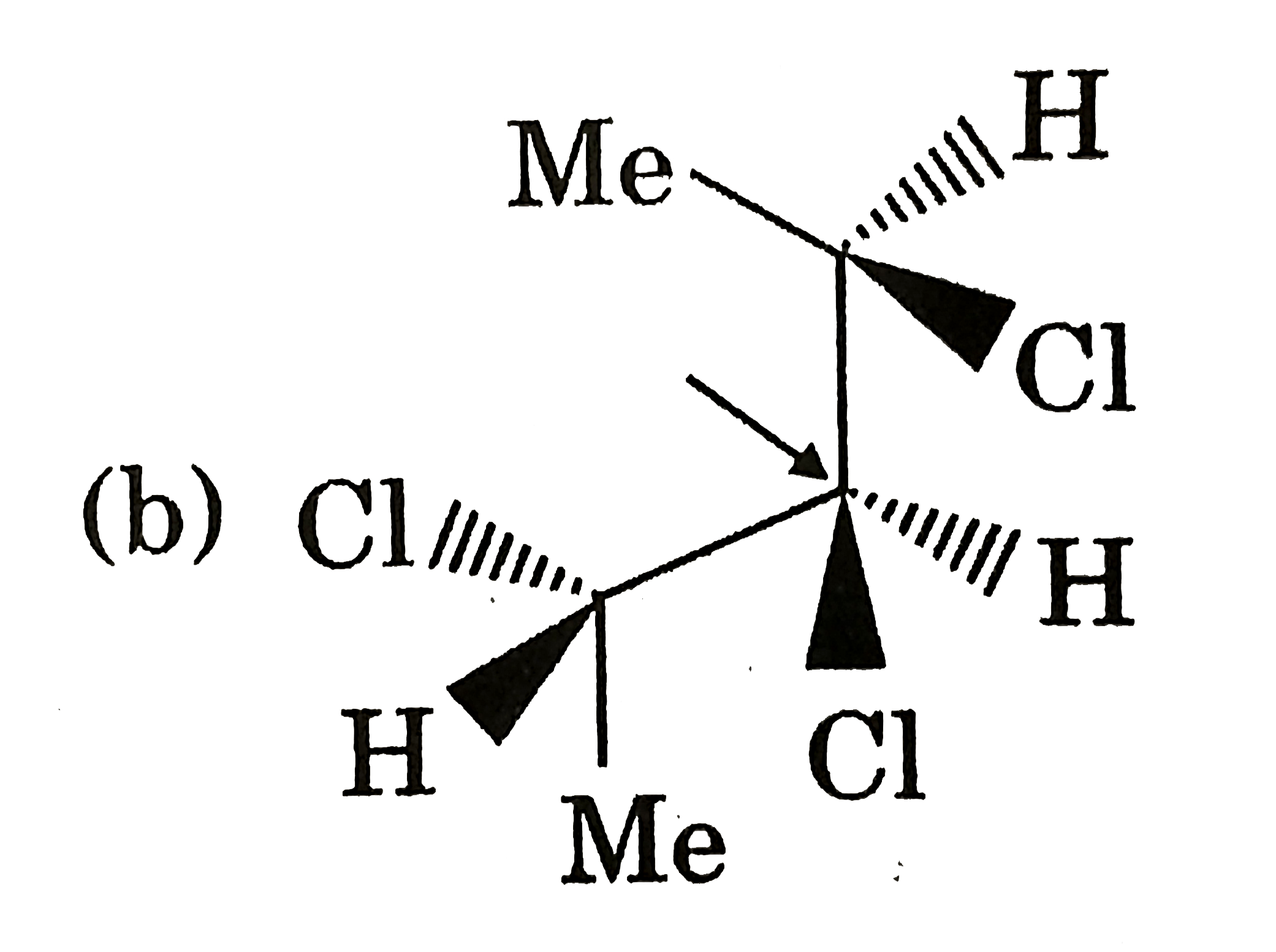
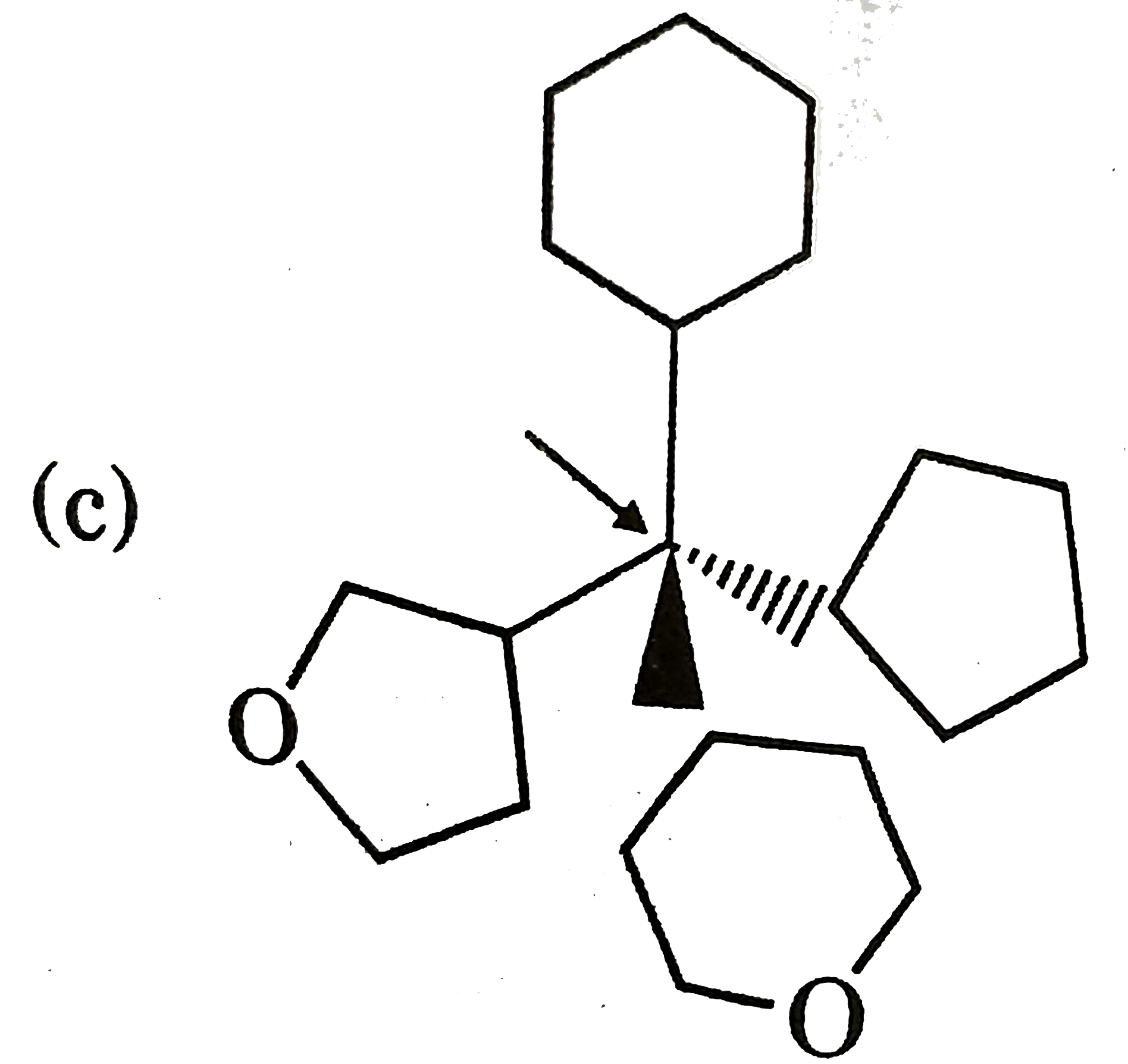
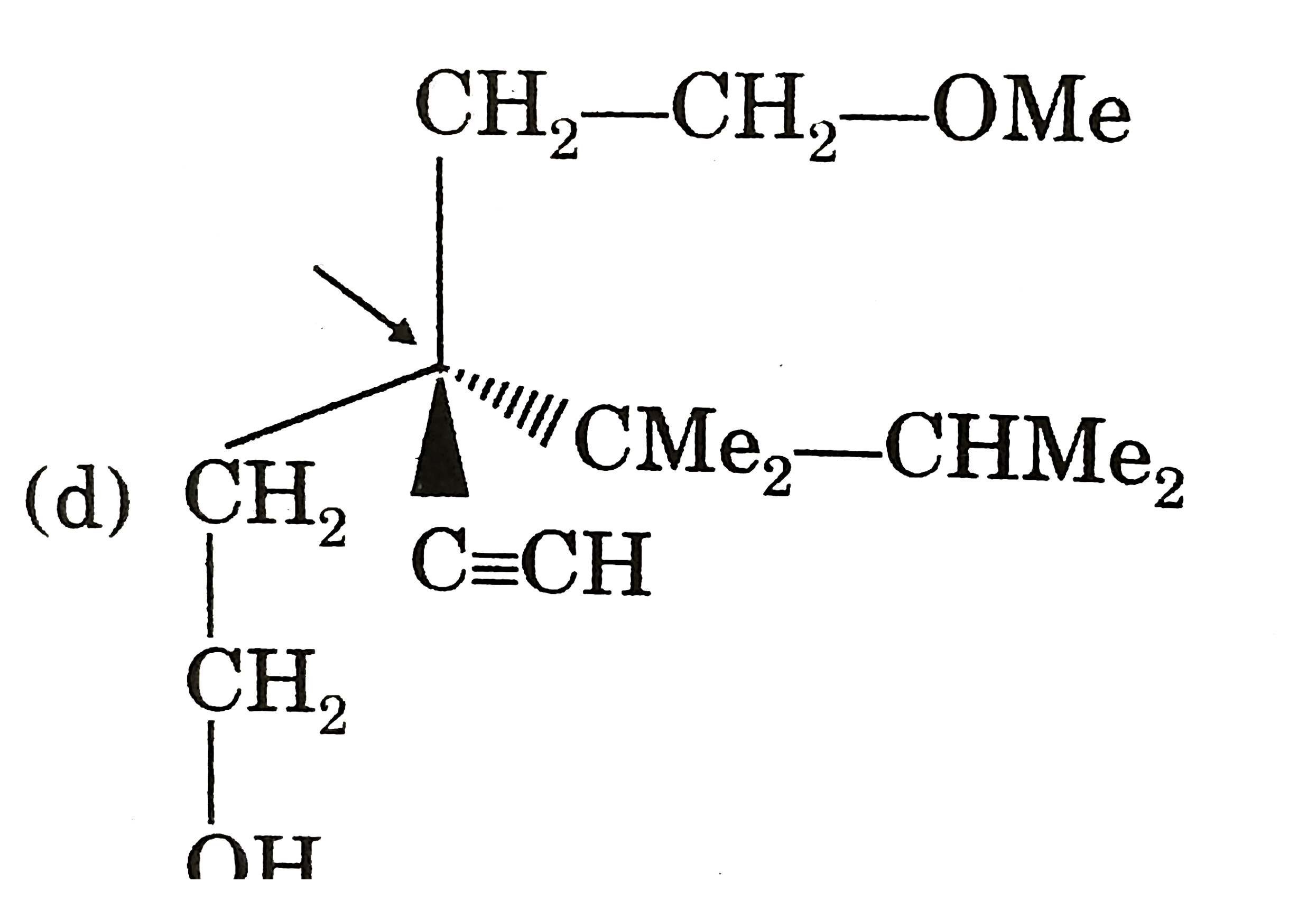
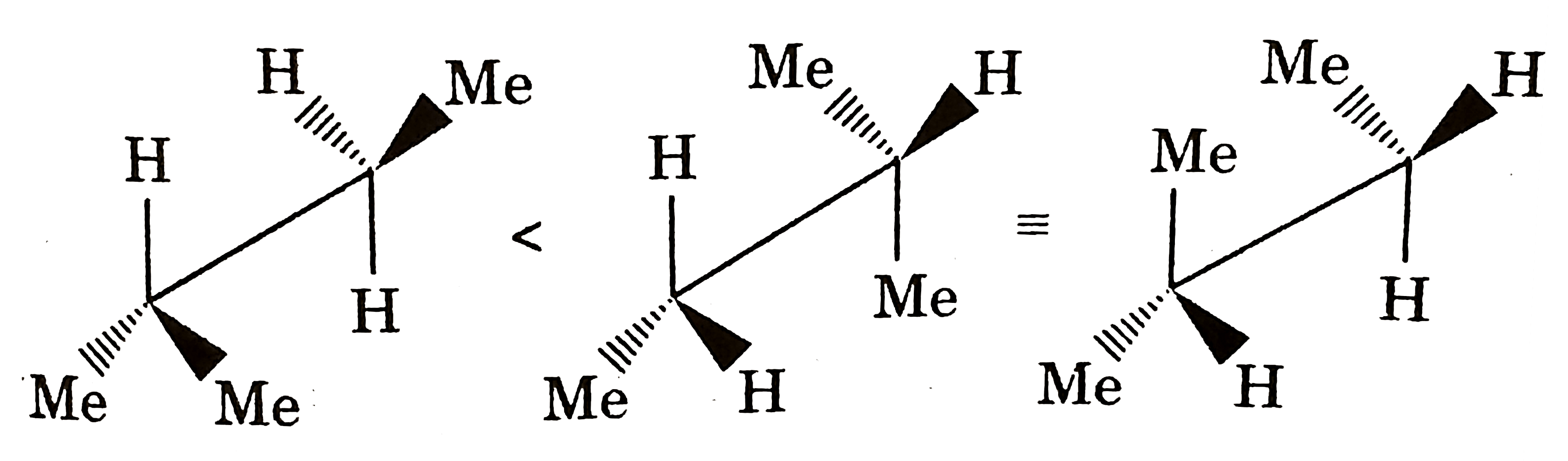
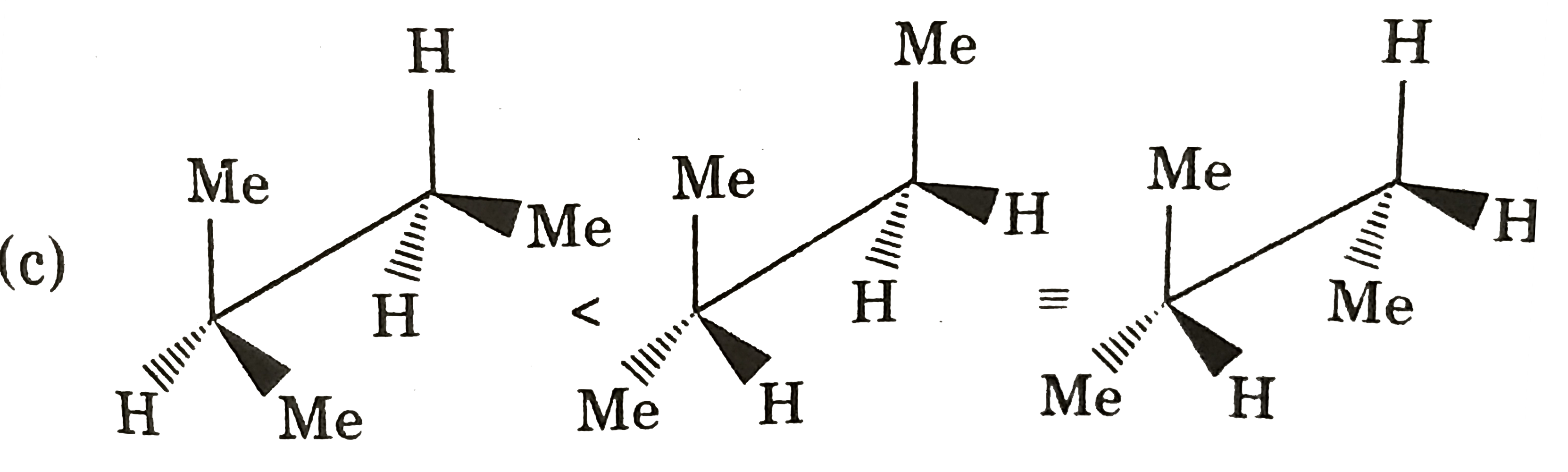
Which of the following compounds has indicate carbon-R-configured? |
|
Answer»
|
|
| 2. |
Which of the following group can participate in resonance with other suitable group : |
|
Answer» `CH_(2)=OVERSET(OPLUS)(O-)` |
|
| 3. |
What will be the molality of the solution made by dissolving 10 g of NaOH in 100g of water ? |
|
Answer» 2.5 m `m=10/40xx1000/100=2.5m` |
|
| 4. |
The velocity of an electron in excited state of H-atom is 1.093 xx 10^6 m/s, what is the circumference of this orbit? |
|
Answer» `3.32 xx10^(-10)` m `implies1.093 xx 10^6 = 2.186 xx 10^6 xx 1/n = 2 ` CIRCUMFERENCE of the ORBIT = 3.33 `xx n^2 A^0` |
|
| 5. |
What is BHC how will you prepare BHC mention its uses |
|
Answer» Solution :(i) BHCis Benzenehexachloride. (II) Benzenereacts withthreemoleculeof `CI_(2)`in the presenceof sunlightor UV lighttoBHC. THISIS aloscalledas gammaxaneor LINDANE  (iii) BHCis a powerful insecticile |
|
| 6. |
Which of the following exists as covalent crystals in the solid state : |
|
Answer» Iodine |
|
| 7. |
The thermal conductivity of graphite along an axis in the plane of hexagonal rings is |
|
Answer» exactly the same as ALONG an sxis perpendicular to this plane |
|
| 8. |
Under what conditions will CuSO_4.5H_2O be efflorescent at 25^(@)C? For the reaction CuSO_4.5H_2O_((s)) harr CuSO_4.3H_2O_((s))+2H_2O_((g)) K_p at 298K is 1086 xx 10^(-4) atm^2 and vapour pressure of water is 23.8 Torr. |
|
Answer» If relative HUMIDITY `LT` 33.3%, efflorescence takes place `K_(P)=P_(H_(2)O)^(2) implies P_(H_(2)O) = sqrt(K_(P))=7.92` mm Relative humidity = `(P_(H_(2)O))/("vapour pressure of" H_(2)O)` `=(7.92)/(23.8)=0.333` % R.H=33.3 `Q lt K`, forward direction The reaction will PROCEED only when vapour pressure of moisture in atmosphere is lesser than 7.92mm to show efflorescent nature. |
|
| 9. |
Which are the multiple bond ? Why ? |
| Answer» SOLUTION :DOUBLE bond and triple BONDS are multiple bond. As they certain more them are ELECTRON pair. | |
| 10. |
Which types of bond exist between two carbon of alkene ? |
| Answer» SOLUTION :DOUBLE bond in which ONE is `sigma` and another is `PI`-bond. | |
| 11. |
Which one of the following indicates the value of the gas constant R? |
|
Answer» `1.987 " cal " deg^(-1) MOL^(-1)` |
|
| 12. |
Which of the following can turn blue litmus to red colour |
|
Answer» Dil `H_2O_2` SOLUTION |
|
| 13. |
The study of discharge of electricity through gases led to the discovery of |
|
Answer» STRUCTURE of the atom |
|
| 15. |
Which out of the following can be used to store an alkali metal?i)H_(2)O (ii) C_(2)H_(5)OH and(iii) benzene |
|
Answer» SOLUTION :iii) BENZENE can be used to store an alkali metal, because other substances rea as below: `Na + H_(2)O to NaOH + 1//2H_(2)` `Na + C_(2)H_(5)OH to C_(2)H_(5)ONA+ .1//2H_(2)` |
|
| 16. |
Which of the following has zerooxidation number fro every atom ? |
|
Answer» Polyatomic ion |
|
| 17. |
What are organic reactions? |
| Answer» Solution :Substrate is an organic molecule reacts with reagents, which may be an organic, inorganic orany agent like heat, PHOTON ETC, that brings about the chemical CHANGE to form a PRODUCT, this is known as organic reactions. | |
| 18. |
Two vessels of capacities 1.5 litres and 2.0 litres containing hydrogen at 750 mm pressure and oxygen at 100 mm pressure respectively are connected to each other through a value. What will be the final pressure of the gaseous mixture assuming that temperature remains constant ? |
|
Answer» For partial pressure of `H_(2)`,`P_(1)V_(1)=P_(2)V_(2), i.e., 750xx1.5=P_(2)xx3.5` `p_(H_(2))=(750xx1.5)//3.5=321.43 mm` For partial pressure of `O_(2)``P_(1)'V_(1)'=P_(2)'V_(2)', i.e., 100xx2=P_(2)'xx3.5` or`p_(O_(2))=(100xx2)//3.5=57.14 mm` `P_("MIXTURE")=321.43+57.14 mm=378.57 mm` |
|
| 19. |
Which of the following gas is expected to have highest value of Van der Waal's constant 'a' |
|
Answer» `NH_3` |
|
| 20. |
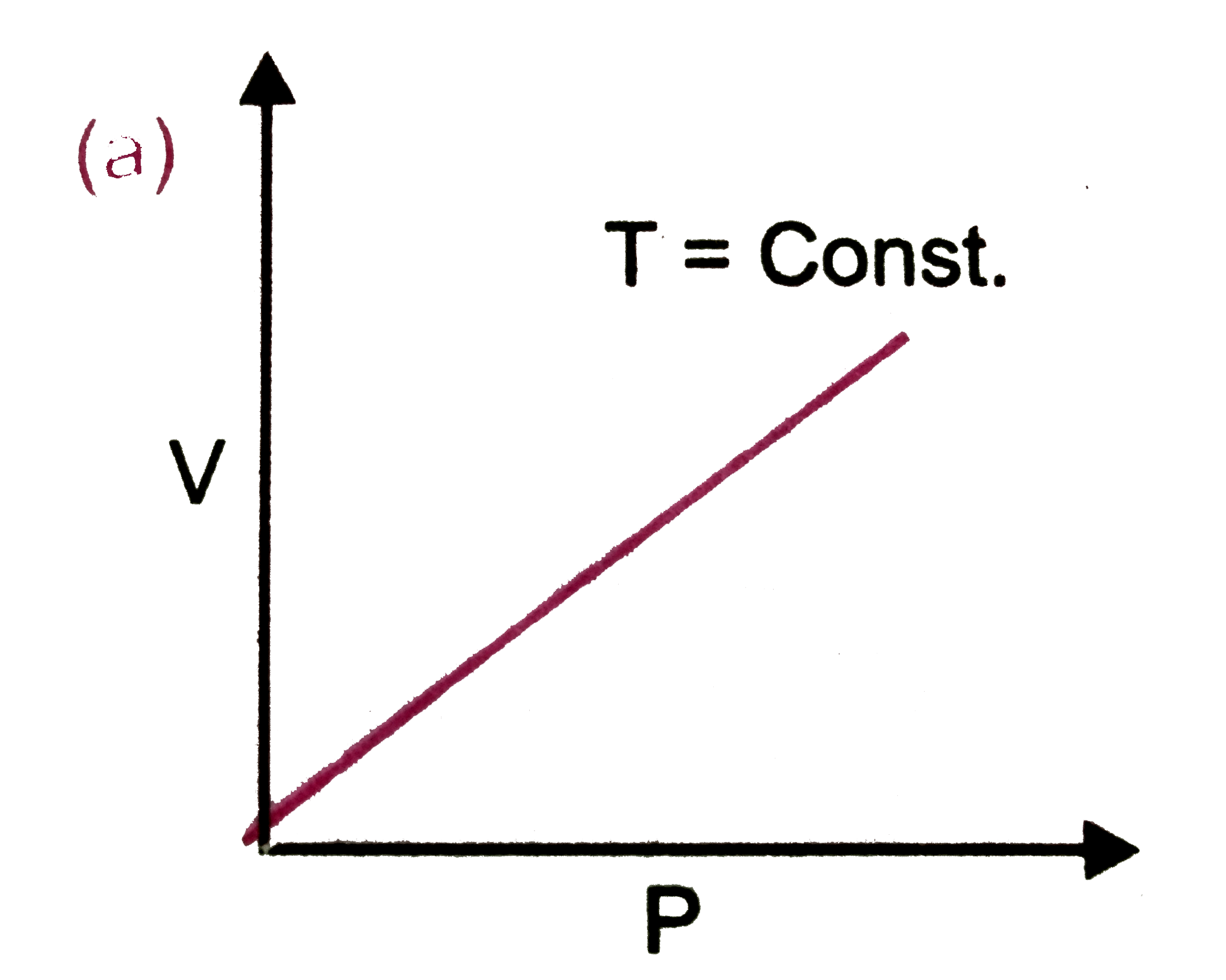
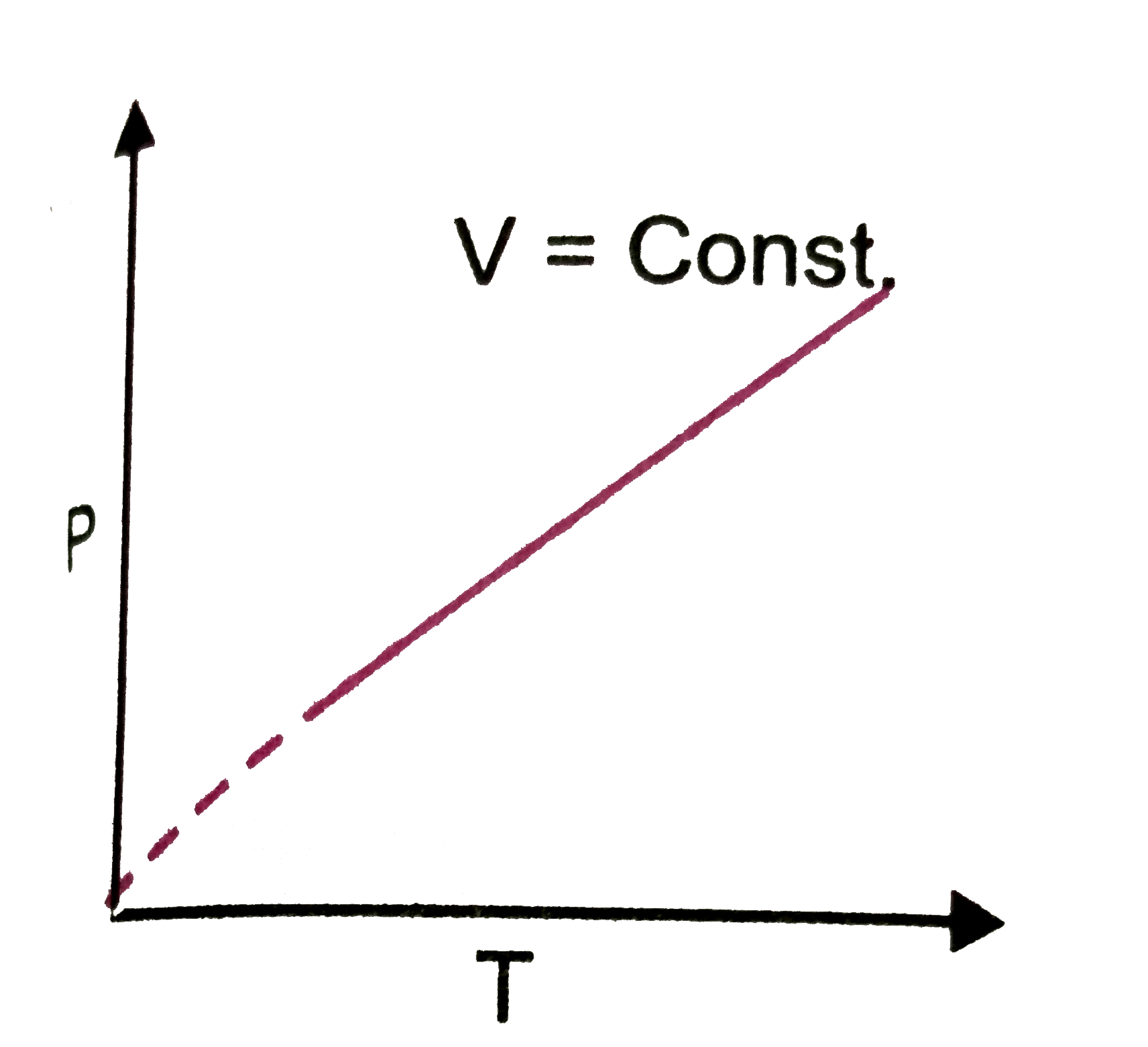
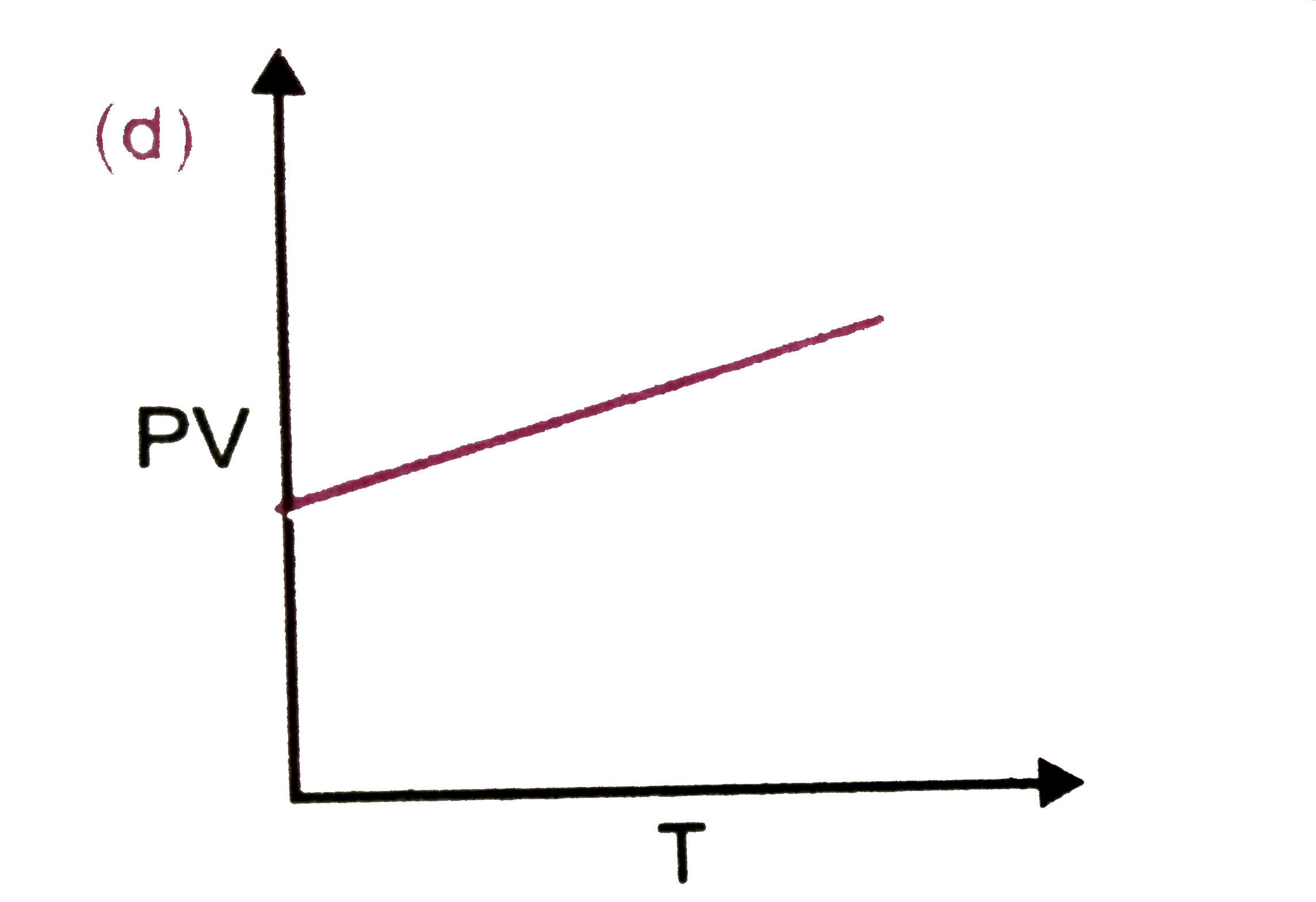
Which of the following plots are correct ? |
|
Answer»
|
|
| 21. |
Which of the following pairs of compounds is isoelectronic and isostructural ? |
|
Answer» `BeCl_(2) , XeF_(2)` `XeF_(2) = 8 + 7 xx2 = 22 ` electrons Therefore, both the species are isoelectronic. In both the species, the central atoms UNDERGO `sp^(3)` d-hybriduzation giving trigonal bipyramidal geometry where three EQUATORIAL position are occupied by lone pairs of electrons giving a linear shape to `IB_(2)^(-) and XeF_(2)` as shown in the structures below :  Thus , these two species are ALSO isostructural. |
|
| 22. |
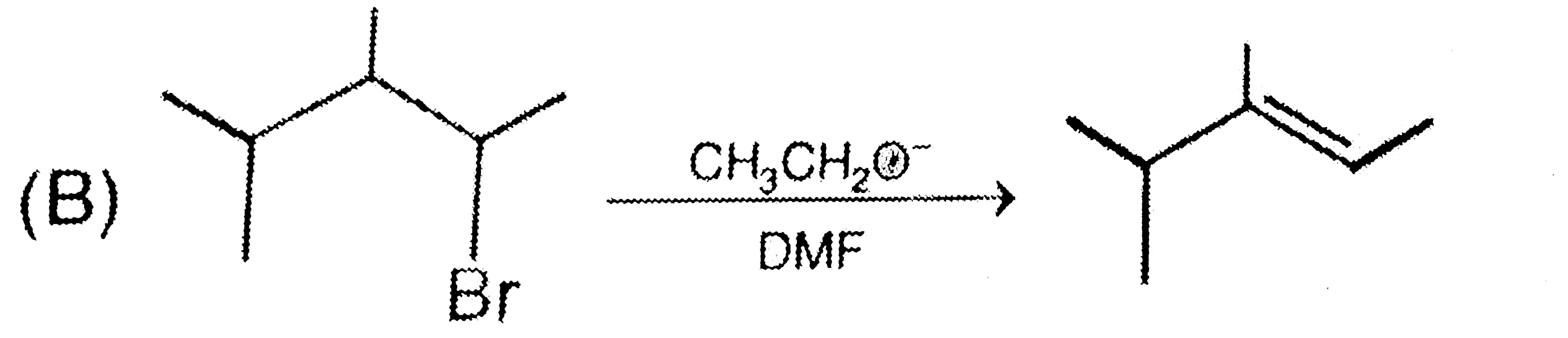
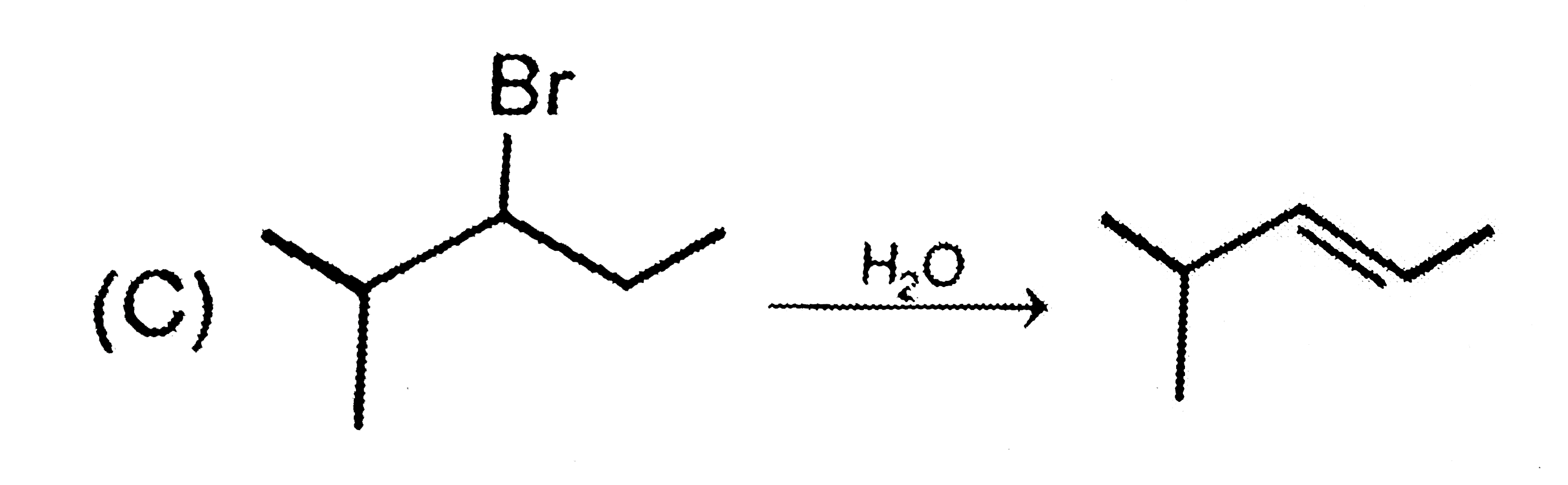
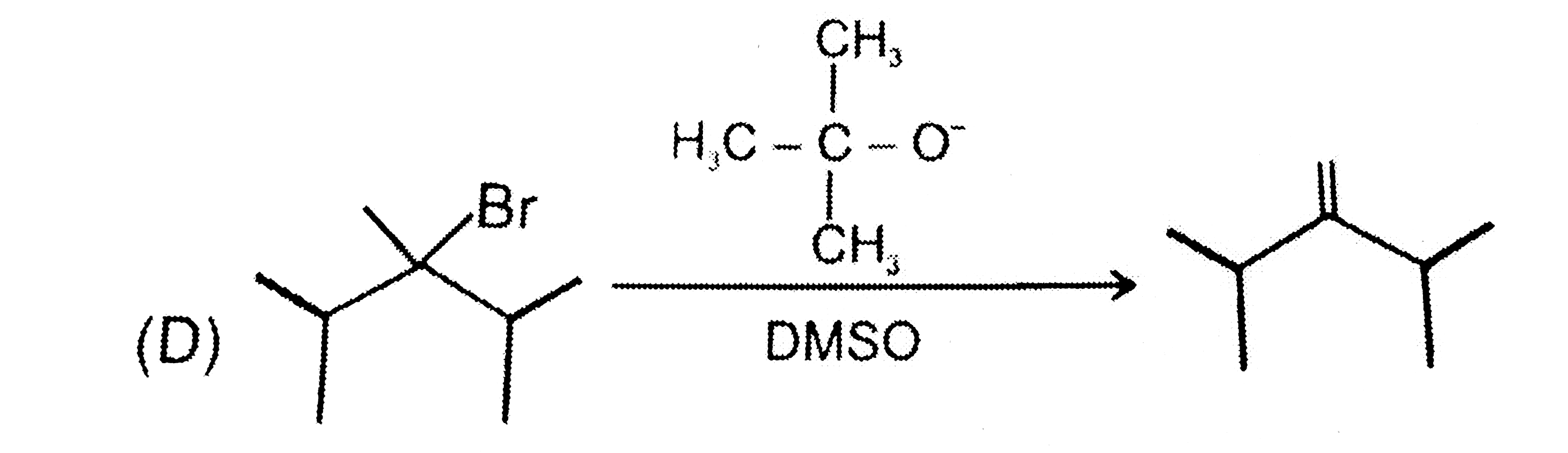
Which of the following reaction the major product . |
|
Answer»
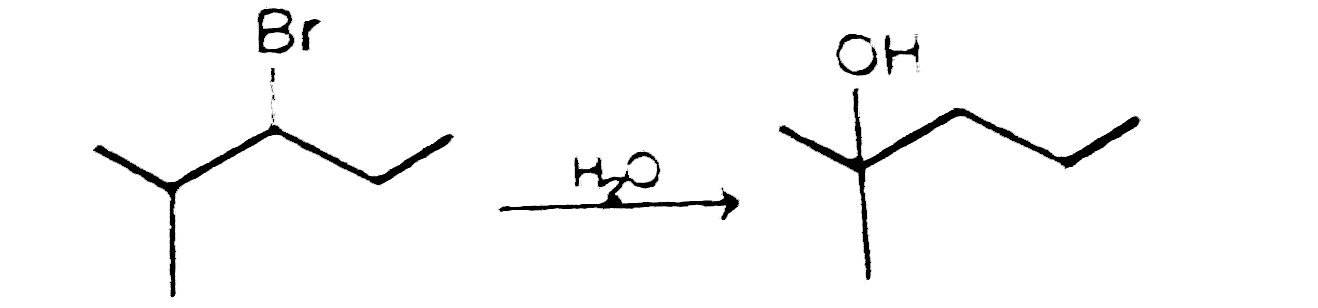
|
|
| 23. |
Which type of silicate is Beryl (Panna Ratna) ? |
|
Answer» LINEAR SILICATE |
|
| 24. |
What is free radicals, carbanions and carbocations? How they form? |
| Answer» Solution :These three are very unstable and reactive intermediate obtained during organic REACTION. The CARBOCATION and carbonions is formed by heterolytic cleavage and the electron PAIR of bond is transfer on only one atom. In formation of free radical, By hemolytic cleavage, one of the electrons of the shared pair in a covalent bond GOES with each of the bonded atom | |
| 25. |
Which of the following compound is produced when CH_(2)=CH-(CH_(2))_(2)COOH reacts with HBr in presence of peroxides |
|
Answer» `CH_(3)CH(CH_(2))_(5)COOH` |
|
| 26. |
Which of the following statements are correct ? A) Sink for CO is ocean B) Green house effect Causes lowering of temperature of eart's s suface C) To control CO emission by automobiles, usually catalytic converters fitted into exhaust pipes D) H_(2)SO_(4) herbicides and insecticides form mists |
|
Answer» A,B |
|
| 27. |
The Van't Hoff factor (1) for a dilute aqueous solution of the strong electrolyte barium hydroxide is..... |
|
Answer» 0 `ALPHA=((i-1))/((n-1))` i=a(n-1)+1 `THEREFORE`n=i=3(forBa`(OH)_(2)`,a=1) |
|
| 28. |
What is the work done in a open vessel at 27^(@)C, when 92 grams of sodium metal reacts with water. |
|
Answer» |
|
| 29. |
What is calorimerty ? |
| Answer» Solution :An EXPERIMENTAL technique from which we can MEASURE ENERGY change associate with chemical and PHYSICAL process. this technique is called calorimetry. | |
| 30. |
The solubility product of Ca(OH)_(2) at 250^(0)C is 4.42 xx 10^(-5). A 500 mL of saturated solution of Ca(OH)_(2) is mixed with equal volume of 0.4 M NaOH. How much Ca(OH)_(2) on milligram is precipitated? |
|
Answer» `758.2mg` For `Ca(OH)_(2) hArr Ca^(2+) + 2OH^(-)` `K_(sp) = s xx (2s)^(2)= 4s^(3)` Then, `4s^(3) = 4.42 xx 10^(-5)` `s=3sqrt(((4.42 xx 10^(-5))/(4))) = 0.0233M` Now `Ca(OH)_(2) + NaOH` are mixed `:.` Solution has `Ca^(2+)` and `OH^(-)` out of which some `Ca^(2+)` and `OH^(-)` out of which some `Ca^(2+)` are precipitates on mixing. `[Ca^(2+)] = (0.0223 xx 500)/(1000) = 0.01115 = 111.5 xx 10^(-4) M` `[OH^(-)] = (0.0223 xx 2 xx500)/(1000) + (500 xx 0.4)/(1000) = 0.2223 M` (from `Ca(OH)_(2)`), (from `NaOH`) `[Ca^(2+)] [OH^(-)]^(2) = KSP` `[Ca^(2+)]_("left") [0.2223]^(2)= 4.42 xx 10^(-5)` `[Ca^(2+)]_("left") = (4.42 xx 10^(-5))/([0.2223]^(2)) = 8.94 xx 10^(-4) "MOL litre"^(-1)` `:'` MOLE of `Ca(OH)_(2)` precipitated `=` Mole of `[Ca^(2+)] "INITIAL" - [Ca^(2+)]` final `= 111.5 xx 10^(-4) -8.94 xx 10^(-4)` `= 102.46 xx 10^(-4)` `:.` WEIGHT of `Ca(OH)_(2)` precipitated from `Ca(OH)_(2)` solution `= 102.46 xx 10^(-4) xx 74` `= 7582.04 xx 10^(-4) g = 758.2 mg` |
|
| 31. |
Write the net ionic equation for the rection of potassium dichromate (VI) K_(2)Cr_(2)O_(7) with sodium suphite Na_(2)SO_(3) in acid solution to give chromium (III) ion and suphate ion |
|
Answer» Solution :Step 1 WRITE the skeleton equation for the GIVEN reaction `Cr_(2)O_(7)^(2-)(aq)+SO_(3)^(2-)(aq)rarrCr^(3+)(aq)+SO_(4)^(2-)(aq)` Step 2 find out the element which undergo a change in oxidiatoin number (O.N)  here O.N of Cr decrease from +6 `Cr_(2)O_(7)^(2-)` to +3 in `Cr^(3+)` while that of s increases from +4 in `SO_(3)^(2+) to +6 in SO_(4)^(2-)` step 3 find out the total increase and decrease in O.N SINCE thereare two Cr atoms on L.H.S and only one on R.H.S therefore multiply `Cr^(3+)` onR.H.S of eq (i) by 2 and thus the total decreasein O.HN of Cr is `2xx3=6` step 4 balance increase / decrease in O.N since the toal increasein O.N is 2 and decrease is 6 therefore multiply `SO_(3)^(2-)` on L.H.S and `SO_(4)^(2-)` on R.H.S of Eq (ii) 3 combining steps 2 and 3 we have step 5 balance all atoms than H and O not needed since both cr and s atoms are already balanced step 6 balance o atoms by adding `H_(2)O` moleculed since there are seven O atoms in `Cr_(2)O_(7)^(2-)` and nine in `3So_(3)^(2-)` on L.H.S and only 12 on the R.H.S of eq (iii) therefore add 4 molecules of `H_(2)O` to R.H.S of eq (iii) we have `Cr_(2)O_(7)^(2-) (aq)+3SO_(3)^(2-)(aq)rarr2cr^(3+)(aq)+3SO_(4)^(2-)(aq)+4H_(2)O(l)` step 7 balance H toms by adding `H^(+)` IONS since the reaction occurs in the acidic medium since there are 8H on R.H.s and none o the L.H.S therefore add `8H^(+)` to the L.H.S of Eq (iv) we have `Cr_(2)O_(7)^(2-)+3SO_(3)^(2-)(aq)+8H^(+)(aq)rarr2Cr^(3+)r(aq)+3SO_(4)^(2-)(aq)+4H_(2)O(l)` thus equ (v) REPRESENT the correct balanced equation |
|
| 32. |
What is the oxidation state of oxygen of H_2O_2 in the final products when it reacts with CIO_3? |
|
Answer» `2x=0` |
|
| 33. |
When Schrodinger wave equation in polar coordinates is solved the solution for Phi is of the form Psi (r, theta , phi)= R(r) , Y(theta , phi). Here R(r) is radial part of wave function and Y(theta, phi) is angular part of the wave function. The region or space where probability of finding electron is zero is called nodal surface. If the probability of finding electron is zero then Psi^2 (r, theta, phi)=0 impliesPsi (r, theta, phi)=0 If the radial wave function is equal to zero we get radial node and if angular part is equal to zero we get angular nodes. Total no. of nodes for any orbital = n - 1. Where ‘n’ is principal quantum number.Number of radial nodes for 4f orbital |
|
Answer» 3 |
|
| 34. |
When Schrodinger wave equation in polar coordinates is solved the solution for Phi is of the form Psi (r, theta , phi)= R(r) , Y(theta , phi). Here R(r) is radial part of wave function and Y(theta, phi) is angular part of the wave function. The region or space where probability of finding electron is zero is called nodal surface. If the probability of finding electron is zero then Psi^2 (r, theta, phi)=0 impliesPsi (r, theta, phi)=0 If the radial wave function is equal to zero we get radial node and if angular part is equal to zero we get angular nodes. Total no. of nodes for any orbital = n - 1. Where ‘n’ is principal quantum number. The d-orbital with two nodal surfaces formed cones |
|
Answer» `3d_(x^2-y^2)` C) `3dx^2 - y^2 ` has no radial node. |
|
| 35. |
When Schrodinger wave equation in polar coordinates is solved the solution for Phi is of the form Psi (r, theta , phi)= R(r) , Y(theta , phi). Here R(r) is radial part of wave function and Y(theta, phi) is angular part of the wave function. The region or space where probability of finding electron is zero is called nodal surface. If the probability of finding electron is zero then Psi^2 (r, theta, phi)=0 impliesPsi (r, theta, phi)=0 If the radial wave function is equal to zero we get radial node and if angular part is equal to zero we get angular nodes. Total no. of nodes for any orbital = n - 1. Where ‘n’ is principal quantum number. represents |
|
Answer» 4s |
|
| 36. |
Which is not the correct statement for ionic solids in which positive and negative ions are held by strong electrostatic attractive forces ? |
|
Answer» The radius ratio `r_+//r_-`increases as coordination number increases |
|
| 37. |
The relative abundance of ""^(12)C and ""^(13)C are respectively 98.892 and 1.108. If the atomic masses of ""^(12)C and ""^(13)C are 12 u and 13.0035 u, respectively, calculate the ave rage atomic mass of carbon. |
| Answer» SOLUTION :Average atomic MASS of CARBON `=((98.892xx124)XX(1.108xx13.00335))/(98.892+1.108)=12.011u` | |
| 38. |
What are the following diseases and what are they due to ? (i) Asphyxiation (i) Pneumoconiosis (iii) Black foot (iv) Methemoglobinemia |
|
Answer» Solution :(i) Asphyxiation. Acute oxygen-starvation in the blood is called asphyxiation or anoxia. It happens when a person stays for long in the air having a concentration of CO more than 750 ppm. (ii) Pneumoconiosis. It is a DISEASE of lungs commonly found in industrial workers. It is due to accumulation of small particulates in the lungs which act as sites for absorption of carcinogenic compounds such as polynuclear hydrocarbons, asbestos etc. (iii) Black foot. It is a peripheral vascular disease in which the blood vessels in the lower limbs are severely damaged resulting eventually in gangrene. It is due to high concentration of ARSENIC in ground water. (iv) Methemoglobinemia. It is a blue BABY syndrome which results in a blue colour skin and a congenital (from BIRTH) HEART defect in infants which laymen often call hole in the heart'. It occurs when water used for drinking contains nitrates gt 50 ppm. |
|
| 39. |
What is the effect of temperature on degree of dissociation? |
| Answer» SOLUTION :The degree of DISSOCIATION INCREASES with INCREASE in temperature. | |
| 40. |
What is inert pair effect? |
| Answer» Solution :The pair of electrons in valence s-orbital is reluctant to take part in BOND formation due to poor shielding effect of -d and f-electron in heavier elements . It is called inert pair effect due to which lower OXIDATION STATE BECOMES more stable than higher oxidation state in case of P-block elements . | |
| 41. |
Which of the following is/are not arranged in correct order of increasing energy? |
|
Answer»
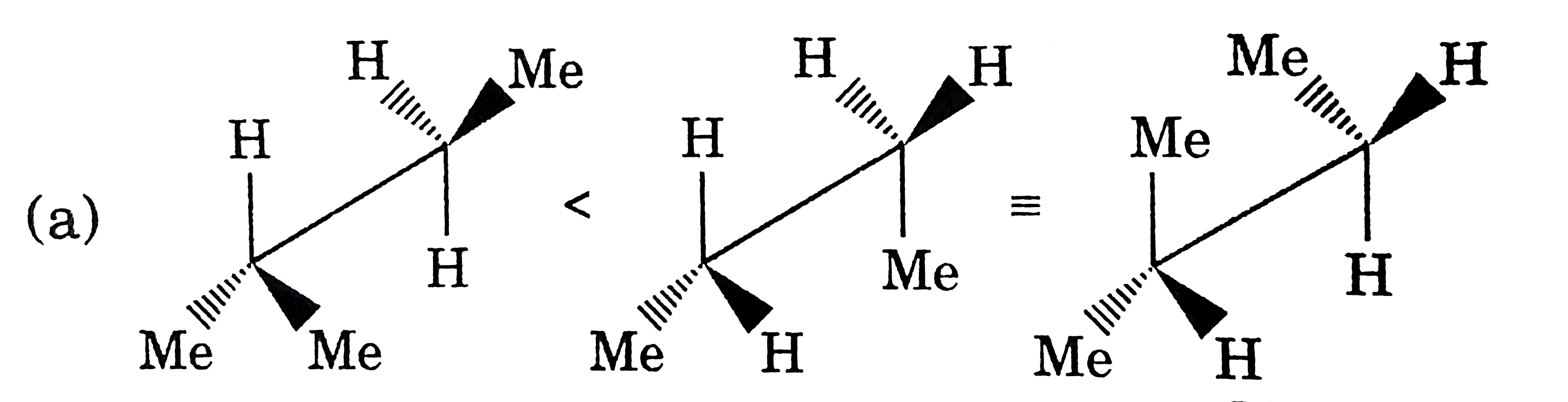   
|
|
| 42. |
Which arrangement of the orbitals of H-atom of their energy is correct? |
|
Answer» `2s lt 3s = 2p_x = 3p_y lt 4S = 4p_z = 4d_(xy)` |
|
| 43. |
The value of K_(c) for the reaction, 2 HI (g) hArr H_(2)g + I_(2) (g)is 1xx10^(-4) At a given time, the composition of reaction mixture is [HI] = 2xx10^(-5) "mol"and [I_(2)]=1xx10^(-5)mol In which direction will the reaction proceed ? |
|
Answer» Solution :REACTION quotient (Q) `= ([H_(2)][I_(2)])/([HI]^(2))=((10^(-5))(10^(-5)))/((2xx10^(-5))^(2))=(1)/(4) = 0.25 =2.5xx10^(-1)` As `Q gt K_(c)`, the reaction will proceed in the reverse direction. |
|
| 44. |
Which element of group 14 exhibitsmaximum tendency for catenation ? |
| Answer» Solution :Among the element of group 14, C-C bond has the MAXIMUM bond STRENGTH and hence carbon showsmaximum tendency for CATENATION. | |
| 45. |
When the graph of p alpha (1)/(V) is not obtained straight line ? |
| Answer» Solution :When PRESSURE of gas is very LOW then DEVIATION OBSERVED in Boyle.s Law. At that there straight LINE is not observed. | |
| 46. |
Which of the following correctly represents Boyle's Law? |
|
Answer»
|
|
| 47. |
When 4f level is completely filled with electrons, the next electron will enter into a subshell whose (n+1) is equal to - |
|
Answer» |
|
| 48. |
What are polyhalogen compounds? Give its types with example. |
|
Answer» Solution :(i) Carbon COMPOUNDS containing more than one halogen atom are called POLYHALOGEN compounds. (ii) They are classified as (i) GEM dihalides (ii) Vicinal dihalides. (a) Gem dihalide: In this compound, two halogen atoms are ATTACHED to one carbon atom. e.g., `CH_3CHCl_2`ethylidene chloride. (b) Vicinal dihalide: In this compound, two halogen atoms are attached to two adjacent carbon atoms. e.g., `{:(CH_2-CH_2),(|""|),(Cl""Cl):}`1,2-dichloro ETHANE |
|
| 49. |
Which cylinder is used to define standard mass in kilogram ? |
|
Answer» Pt-Pd |
|