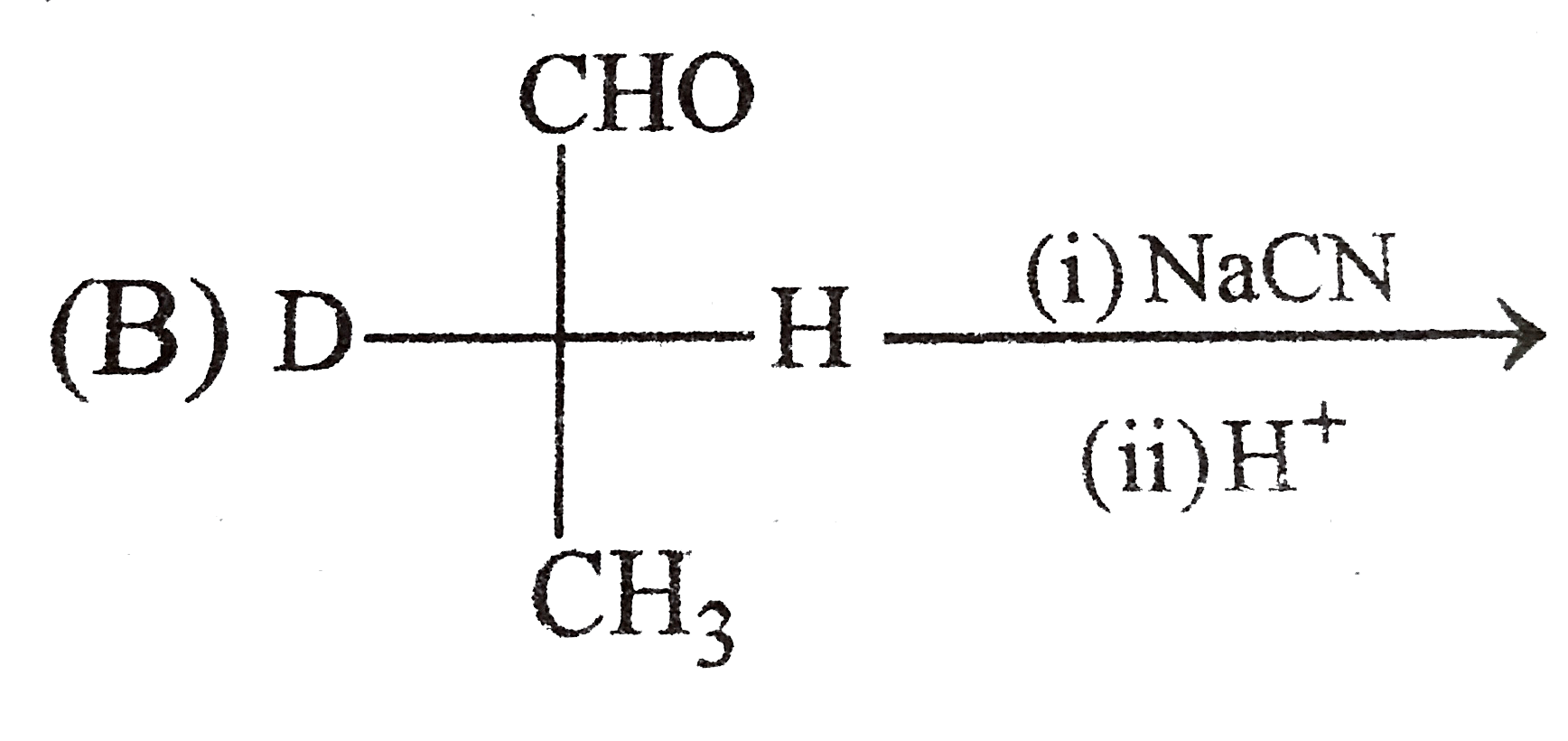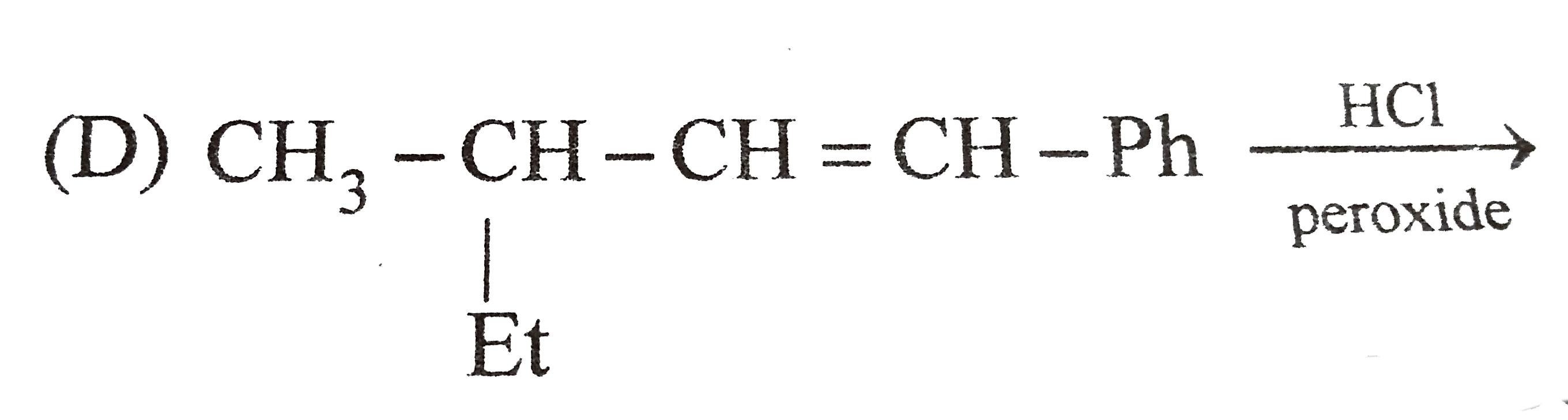Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 2. |
When the real gas is behave like an ideal gas ? |
| Answer» Solution :Real gases follow these laws only under CERTAIN conditions when FORCES of interaction are paractically negligible. In all other situations these deviate from IDEAL BEHAVIOUR. | |
| 3. |
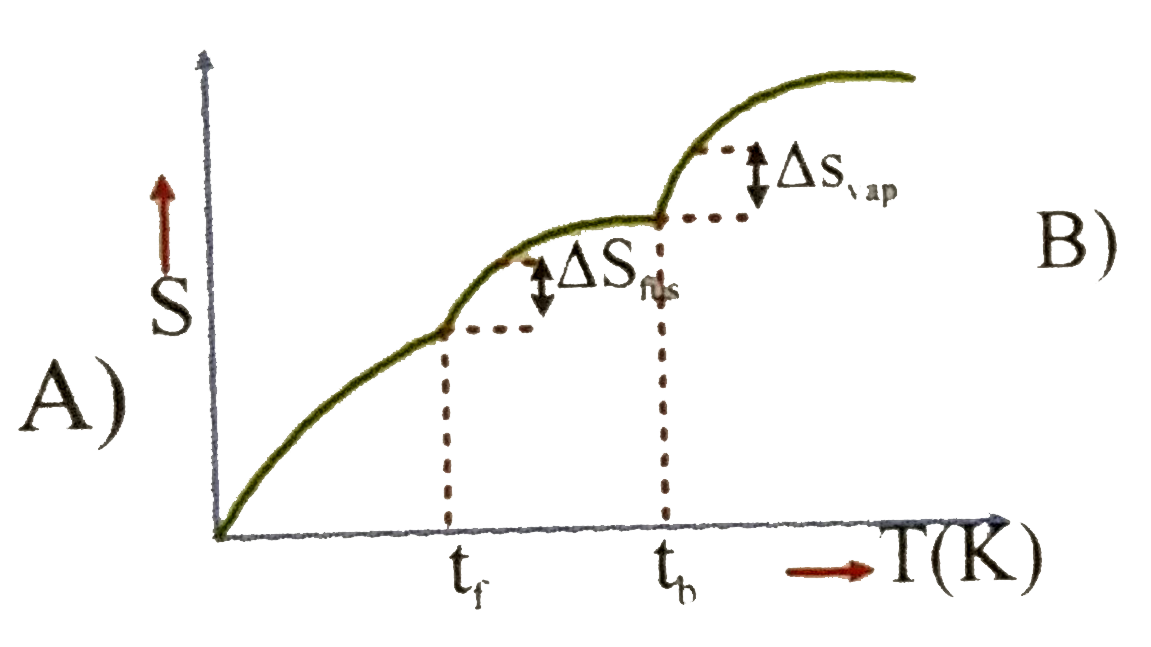
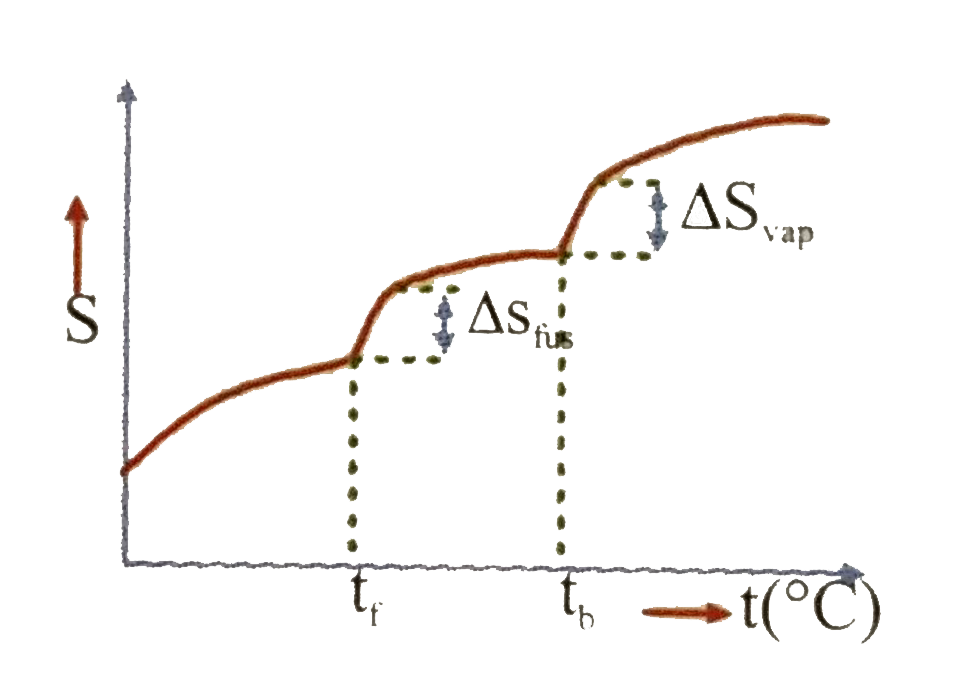
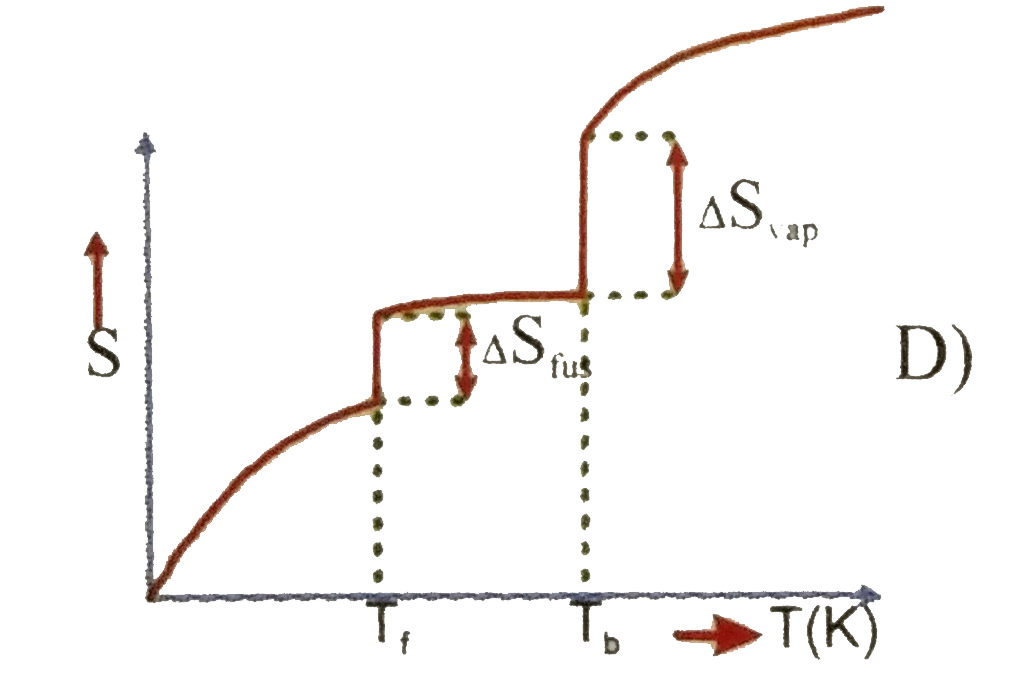
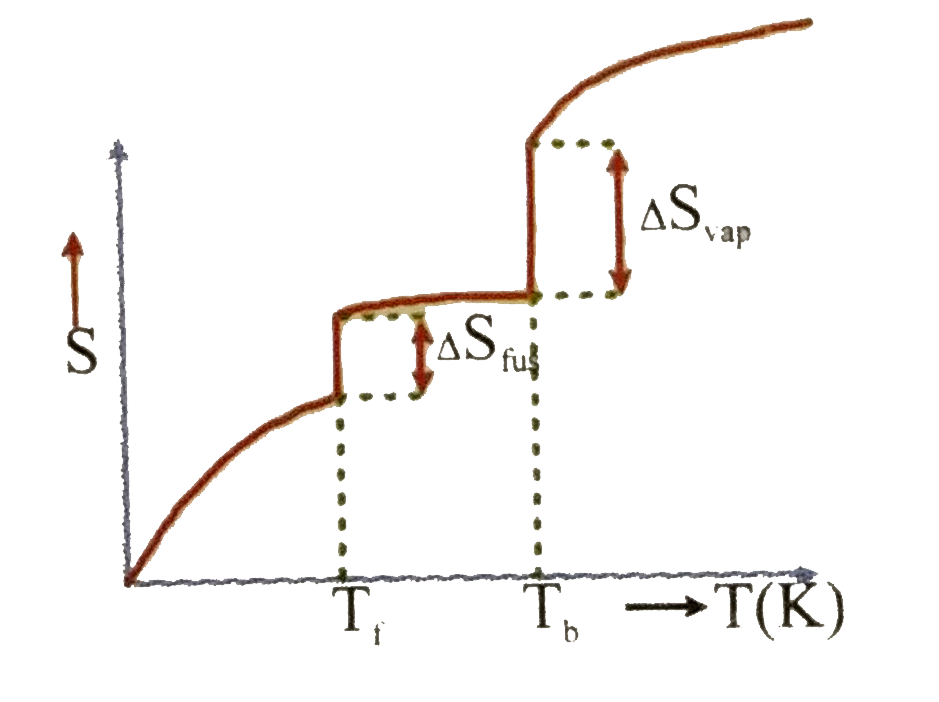
Which of the following graphs best illustrates the variation of entropy of a substance with temp. |
|
Answer»
|
|
| 4. |
When BCl_(3) is treated with water, it hydrolyses and forms [B(OH)_(4)]^(-) only whereas AlCl_(3) in acidified aqueous solution forms [Al(H_(2)O)_(6)]^(3+)ion, Explain what is the hybridisation of boron and aluminium in these species? |
|
Answer» SOLUTION :In trivalent state, most of the compounds being covalent are HYDROLYSED in water, e.g., `BCl_(3)` on HYDROLYSIS in water form `[B(OH)_(4)]^(-)` species, the hybridisation state of B is `sp^(3)` `{:(BCl+3H_(2)OrarrB(OH)_(3)+3HCl),(B(OH)_(3)+H_(2)Orarr[B(OH)_(4)]^(-)+H^(+)):}` `AlCl_(3)` in acidified aqueous solution form OCTAHEDRAL `[Al(H_(2)O)_(6)]^(3+)` ion. In this complex, the 3d orbital of Al are INVOLVED and the hybridisation state of Al is `sp^(3)d^(2)` 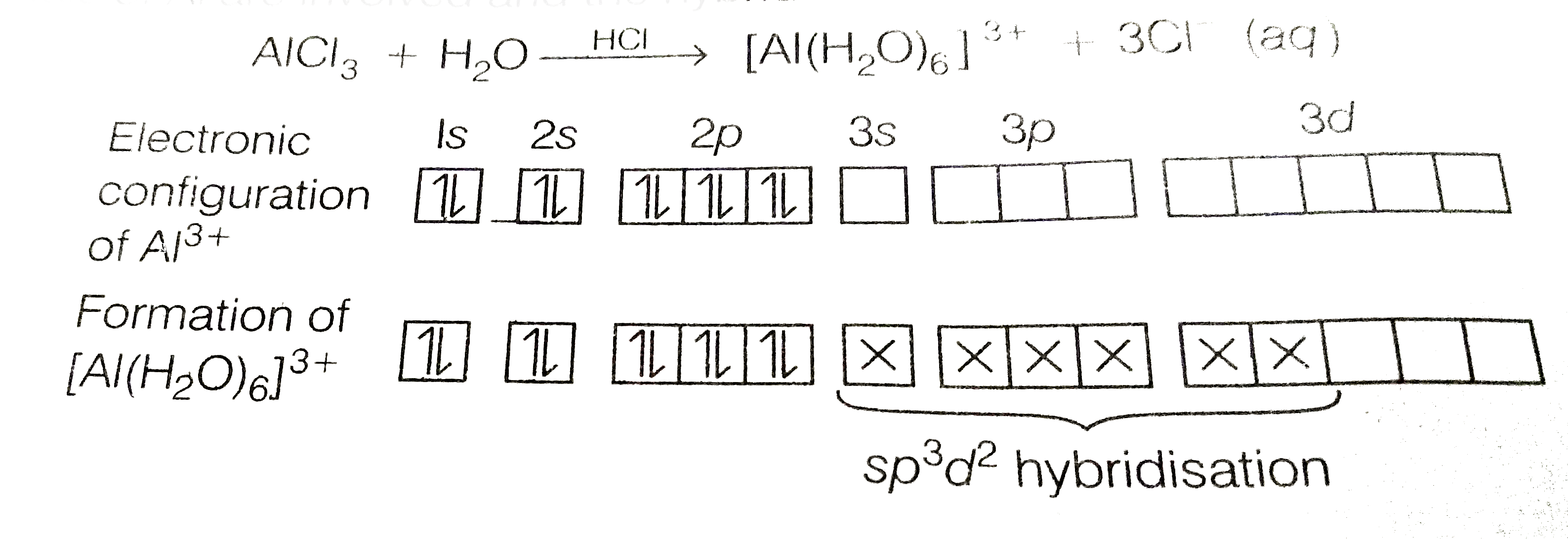
|
|
| 5. |
Which of the following property does not change regularly down the group in the case of IA group metals ? |
|
Answer» Density |
|
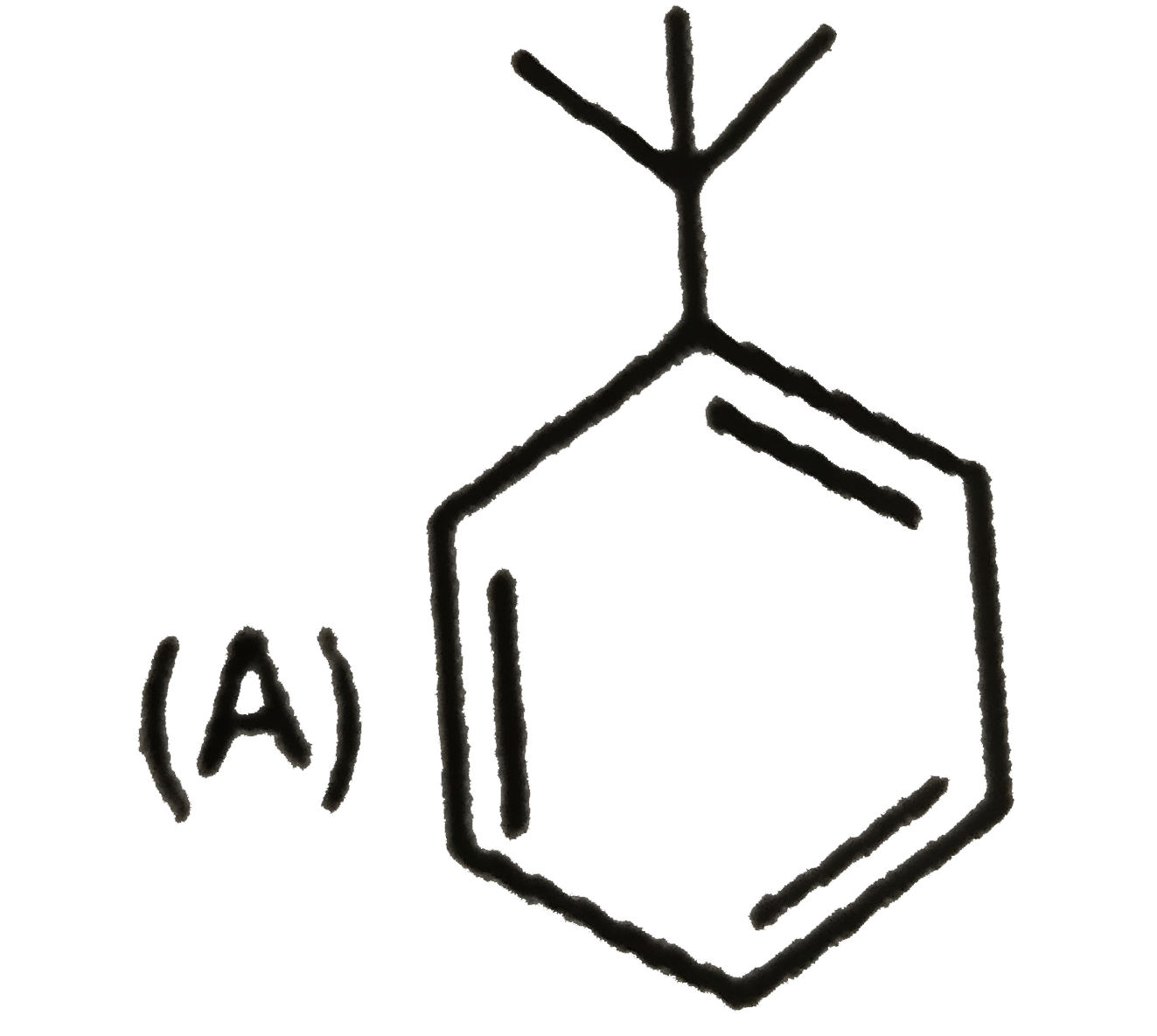
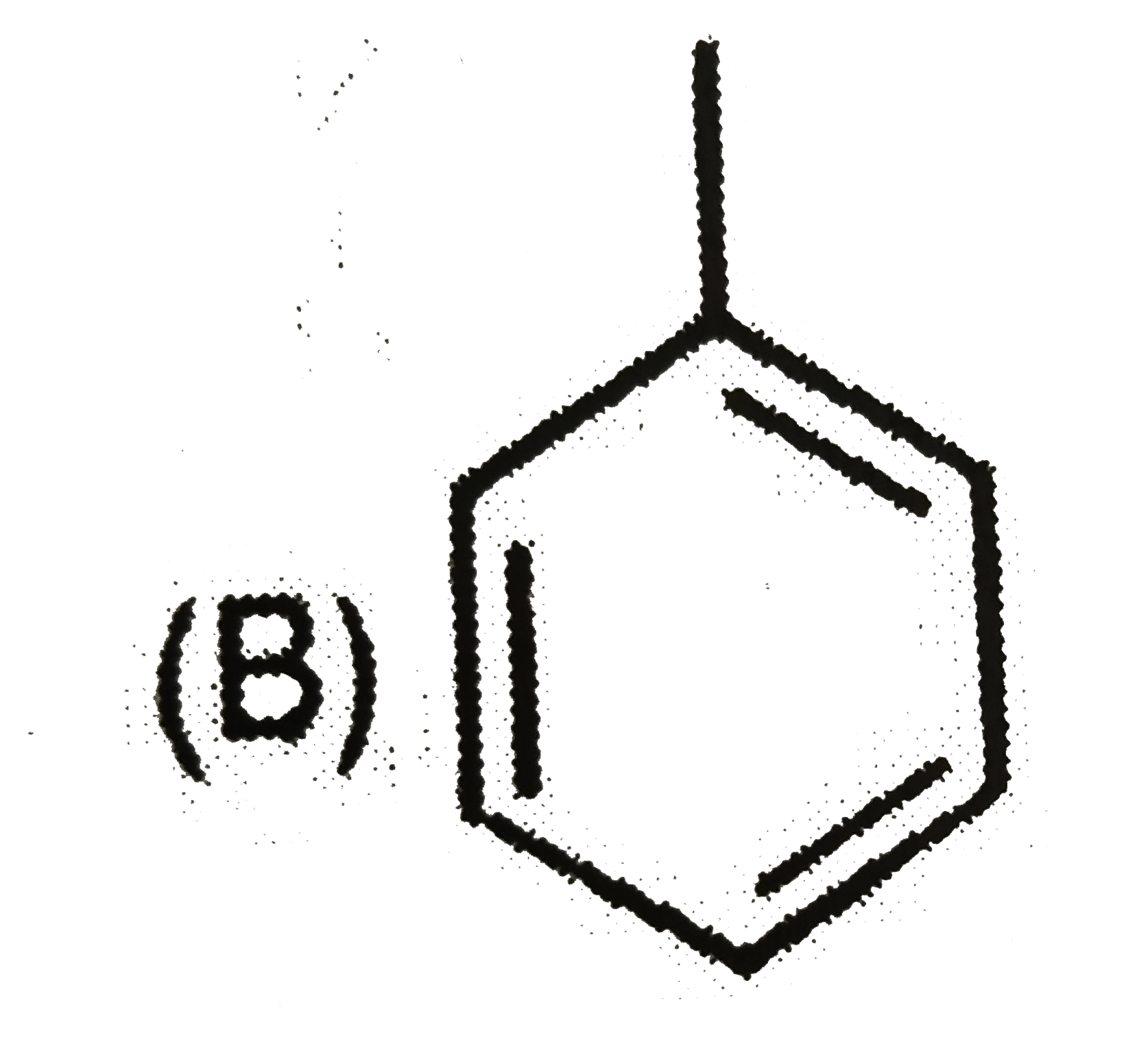
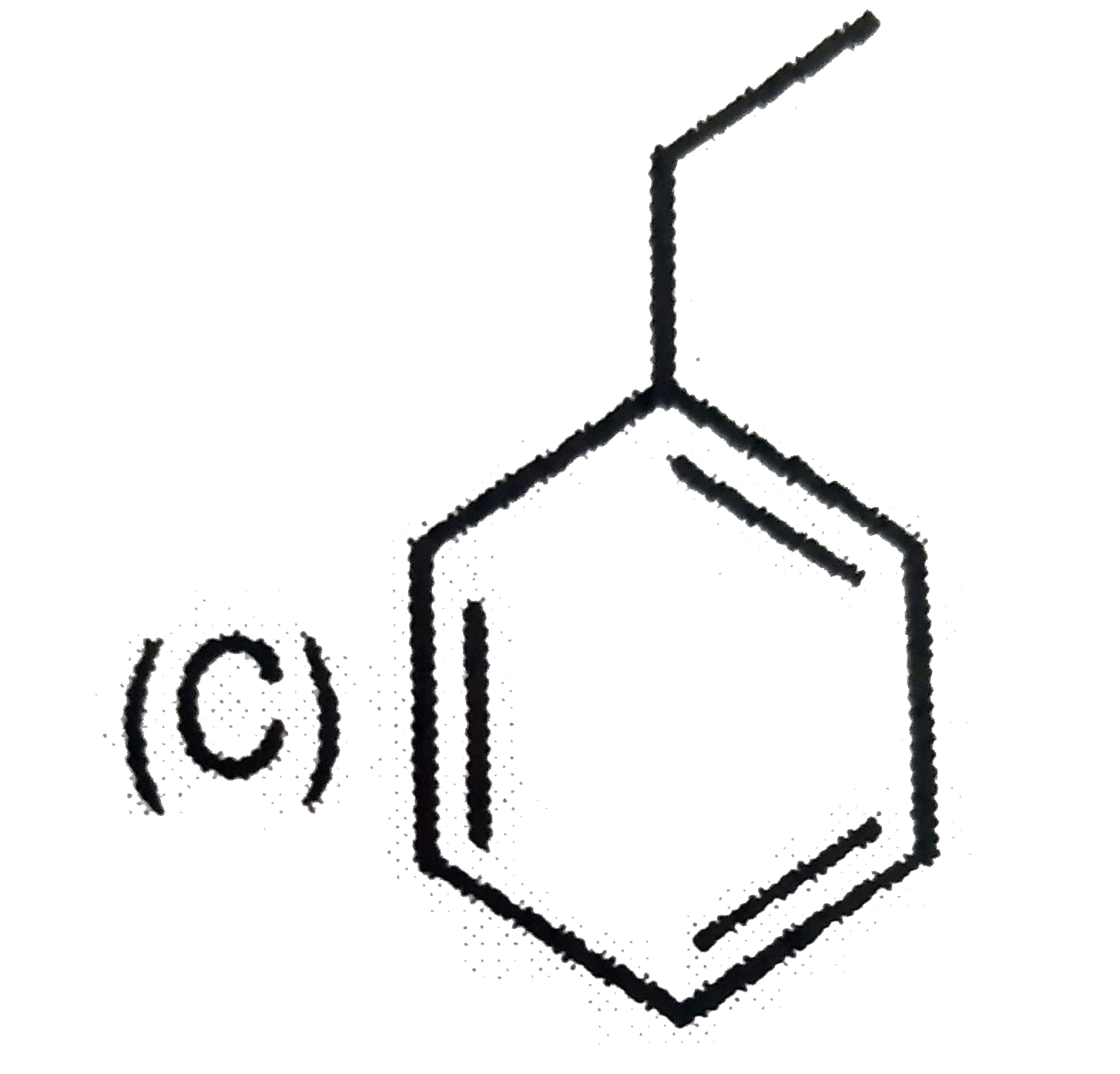
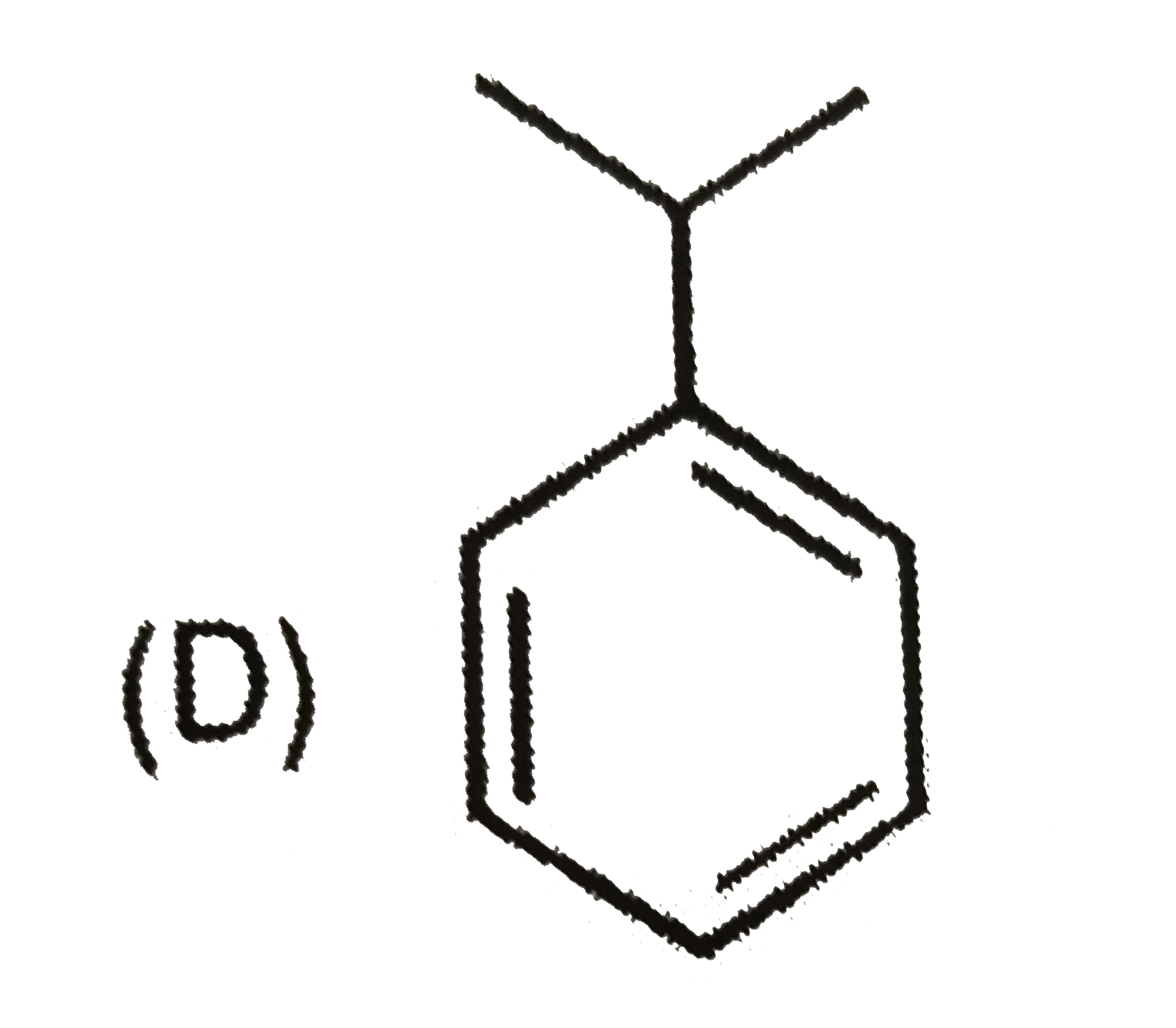
| 6. |
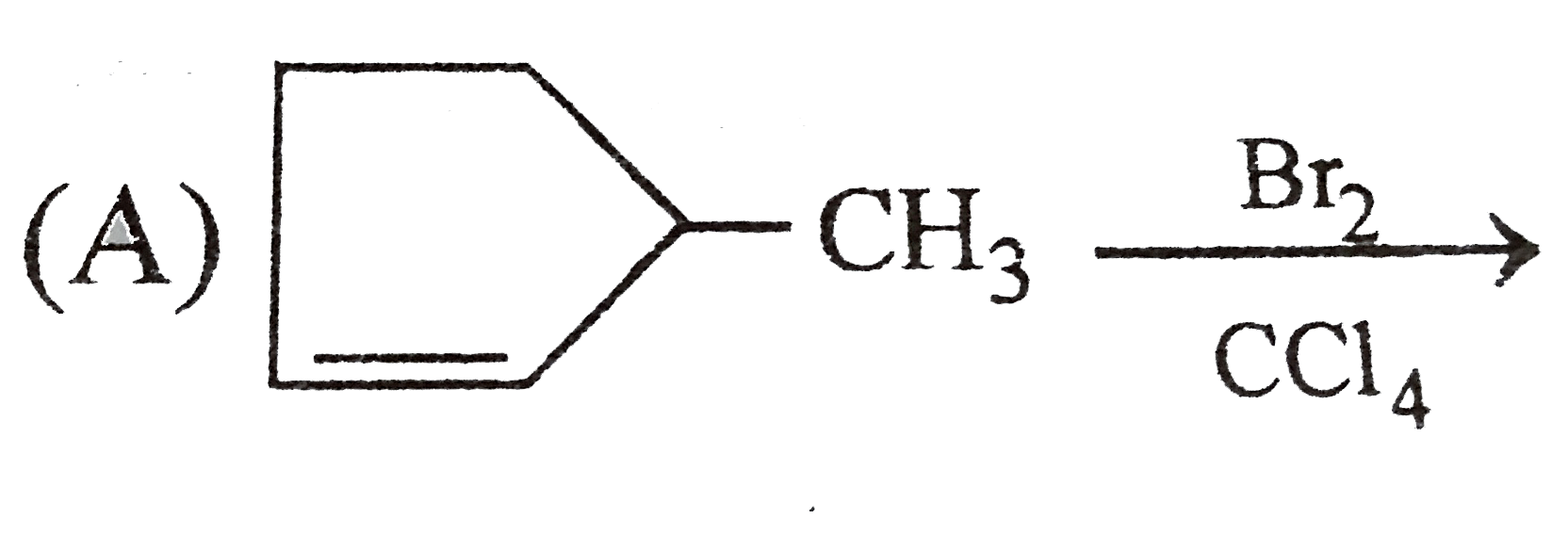
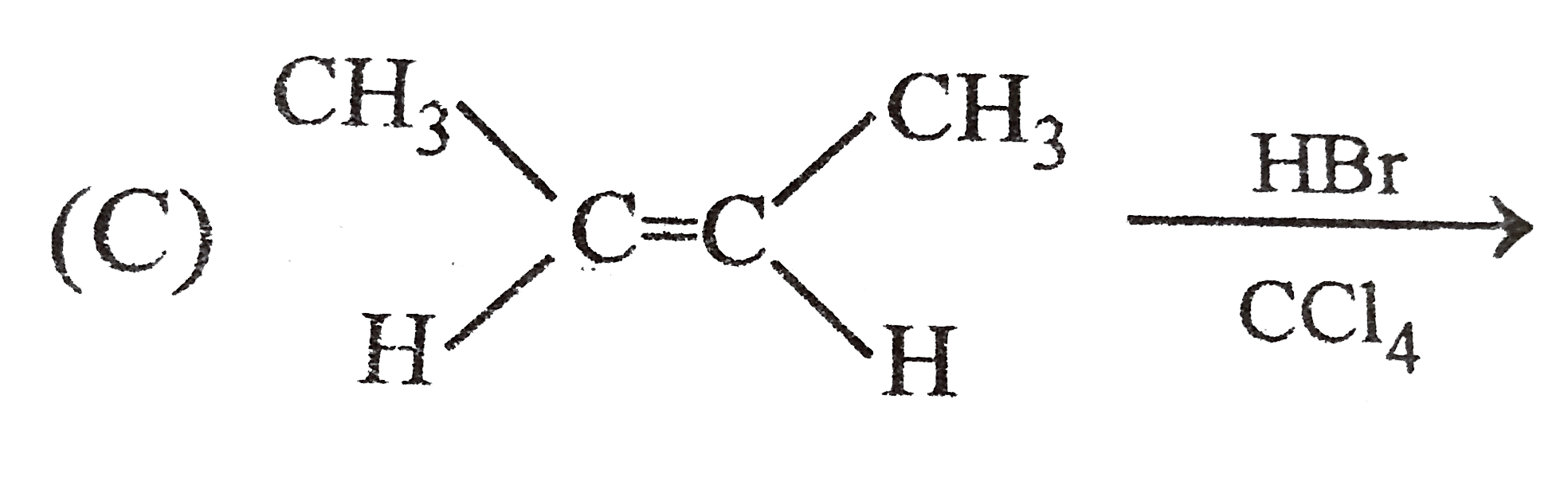
Which of the following reaction product is Diastereomer: |
|
Answer»
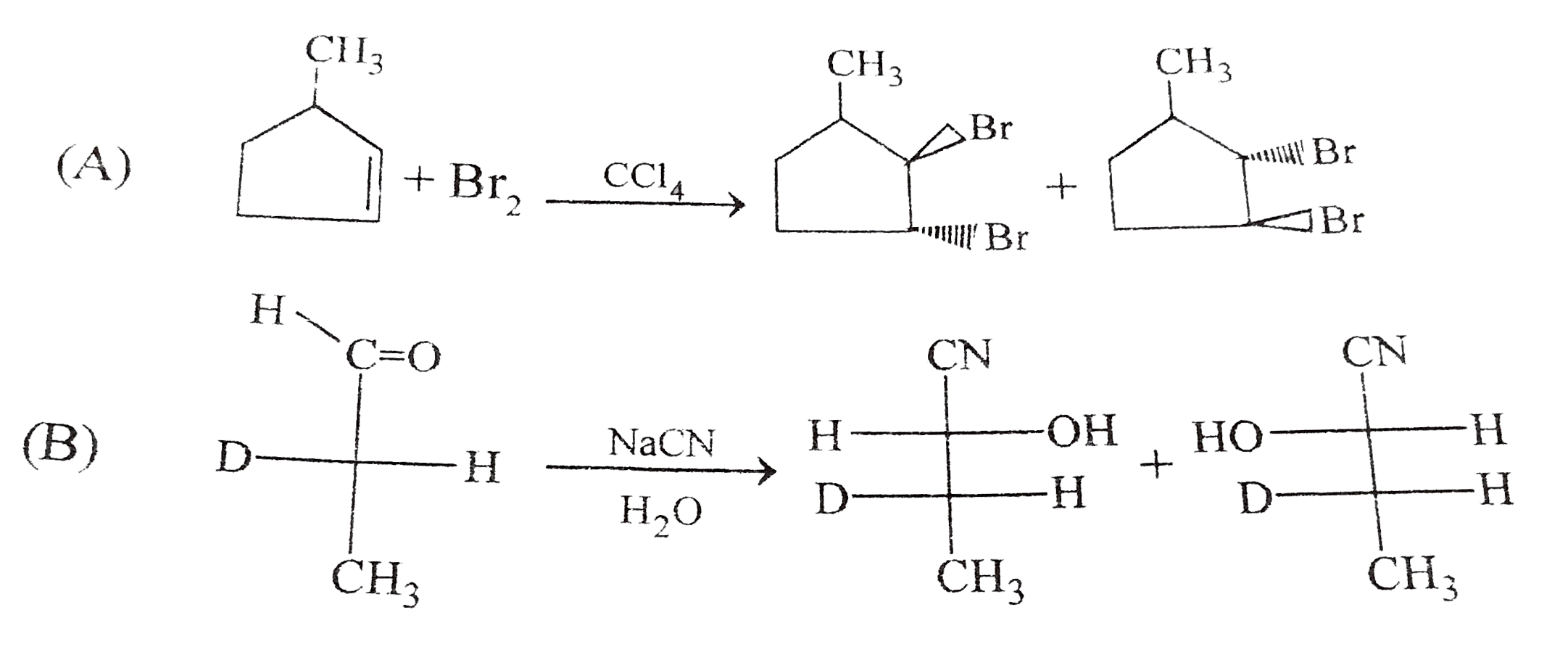 (D).HCL does not show peroxide effect `CH_(3)-UNDERSET(ET)underset(|)(C)H-CH=CH-PH+HCltoCH_(3)-underset(Et)underset(|)overset(H)overset(|)(C)-CH_(2)-underset(Cl)underset(|)overset(H)overset(|)(C)-Ph+CH_(3)-underset(Et)underset(|)overset(H)overset(|)(C)-CH_(2)-underset(H)underset(|)overset(Cl)overset(|)(C)-Ph` |
|
| 7. |
The separation of lanthanides by the ion exhange method is based on |
|
Answer» The SOLUBILITY of thenitrates |
|
| 8. |
What is the oxdation number of phosphorus in H_(3)PO_(2) molecule ? |
| Answer» Solution :If x is the O.N. of P in `H_(3)PO_(2)`, then `3(+1)+x+2(-2)=0orx=+1`. | |
| 9. |
Which of the following solution will have no effect on pH dilution? |
|
Answer» `0.3M CH_3 COOK` (b)pH = 7+ ` ((pKa- pKb)/( 2 )) = 7 ` does not DEPEND C ( c )`pH =pKb + log ""(S)/(B)= pKb`doesnot depend on C (d)`pH=pKa_1 + log "" (S)/(A)= pKa_1 `, doesnot depend on C |
|
| 10. |
What is quick lime ? What happens when we add water to it ? |
| Answer» SOLUTION :CaO , SLAKED lime , `CA(OH)_(2)` is formed , i.e, `CaO + H_(2)O to Ca(OH)_(2)`. | |
| 12. |
Which are the methods for measurement of pH ? |
|
Answer» Solution :(1) Though ACCURATE measurement of PH of a solution is done with help of an istrument called pH meter. (pH change LIKE 0.0001) (2) Approximate pH (within the range of 0.5) can be determined with the help of pH PAPER which show different colours when DIPPED in solution of different pH. (3) By use of litmus paper to know about acid base. (4) Acid-base indicator give information about pH range. |
|
| 13. |
Which will be hydrolyse of the following in aqueous solution ? Cl^(-) , CH_3COO^(-) , NO_3^(-), Cu^(2+) , CN^(-) , CH_3NH_3^(+) , SO_4^(2-) , Na^(+) , K^(+) , Mg^(2+) , PO_4^(3-) |
| Answer» SOLUTION :`CH_3COO^(-) , Cu^(2+) , CN^(-) , CH_3NH_3^(+) , MG^(2+) , PO_4^(3-)` | |
| 14. |
Which of the following compounds possesses 3^(@) carbon atom ? |
|
Answer» 1-Chloro butane `H_(3)C-underset(3^(@))underset(UARR)overset(CH_(3))overset(|)(CH)-CH_(3)` |
|
| 15. |
Which of the following does not involve coagulation ? |
|
Answer» Clotting of BLOOD by the use of all FERRIC chloride |
|
| 16. |
Which of the following option(s)is correctly matched for atomic /molecular weight determination method? |
|
Answer» Ag-Dulong and PETT's method |
|
| 17. |
Which of the following diseases is caused by ozone layer depletion ? |
| Answer» Solution :Skin Cancer | |
| 18. |
Which of the carbonates given below is unstable in air and is kep in CO_(2) atmosphere to avoid decomposition . |
|
Answer» `BeCO_(3)` `BeCO_(3) hArr BeO + CO_(2)` |
|
| 19. |
Which one of the following pairs of solution is not an acidic buffer ? |
|
Answer» `H_2CO_3` and `Na_2CO_3` |
|
| 20. |
Which of the following electronic configuration corresponds to an inert gas ? |
|
Answer» `1s^(2)2s^(2)2p^(5)` |
|
| 21. |
Which of the following orbitals will not form sigma bond afteroverlapping ? |
|
Answer» <P>s - ORBITAL and s- orbital |
|
| 22. |
Which of the following compound decolourises bromine water also reacts with PCl_(5) to give white fumes of HCl? |
|
Answer» `CH_(3)CH_(2)OH` `H_(3)C-CH=CH-CH_(2)OH overset(PCl_(5))rarr HCl+POCl_(3)+H_(3)C-CH=CH-CH_(2)-CL` |
|
| 24. |
What is the way to measure estimated volume of on individual atom? |
|
Answer» Solution :There is no practical WAY by which the size of an individual atom can be measured. One practical approach to estimate the size of an atom of a non-metallic ELEMENT is to measure the distance between two ATOMS when they are bound together by a single bond in a covalent molecule. There are three methods : (1) Metalic radius for metals. (2) Covalent radius for non-metals. (3) Vander Vaal.s radius for NOBLE gases. |
|
| 25. |
Which is act as an oxidising agent in the following reaction ? 2H_2O_((l)) + 2Na_((s)) to 2NaOH_((aq)) + H_(2(g)) |
| Answer» Solution :`H_2`is obtained by REDUCTION of `H^+` in `H_2O` . THEREFORE it act as an OXIDIZING agent. | |
| 26. |
Which statement about the given reaction is incorrect? N_(2)+3H_(2)hArr2NH_(3), DeltaH=-ve |
|
Answer» At `200^(@)C`, the YIELD of `NH_(3)` is `15%` |
|
| 27. |
Which of the following reactions involves carbon-carbon bond formation |
|
Answer» Reimer- Teimann's reaction<BR>Hydroboration-oxidation |
|
| 28. |
When magnesium ribbon is burnt in air two products are formed magnesium oxid and magnesium nitride point out the oxidising and reducing agents |
|
Answer» Solution :REDUCING agent OXIDISING agent `2MG (s)+O_(2)(G)rarr 2 overset(+2)Mg overset(-2)O(s)` `3overset(0)Mg(s)+N_(2)(g)rarr overset(+2)Mg_(3) overset(-3)N _(2)(s)` |
|
| 29. |
Write the IUPASC name of the element with atomic number 104. |
|
Answer» |
|
| 31. |
When a mole of magnesium bromide is prepared from 1 mole of magnesium and 1 moleof liquid bromine, 524 kJ of energy is released. The heat of sublimation of Mg metal is 148 "kJ mol"^(-1). The heat of dissociation of bromine gas into atoms is 193 "kJ mol"^(-1). The heat of vaporization of liquid bromine is 31 kJ "mol"^(-1). The ionisation energy of magnesium is 2187 kJ mol^(-1) and the electron affinity of bromine is - 662 "kJ mol"^(-1). Calculate the lattice energy of magnesium bromide. |
|
Answer» Solution :Given : `MG(s) + Br_2(l) to MgBr_(2)(s)"" DeltaH_f^0=-524 "KJ MOL"^(-1)` Sublimation : `Mg(s) to Mg(g) "" DeltaH_1^0=+148 "KJ mol"^(-1)` Ionisation : `Mg(g) to Mg^(2+)(g) +2e^(-) "" DeltaH_2^0 =2187 "KJ mol"^(-1)` Vapourisaton : `Br_2(l) to Br_(2)(g)"" DeltaH_3^0=+31 "KJ mol"^(-1)` Dissociation : `Br_2(g) to 2Br(g) "" DeltaH_4^0=+193 "KJ mol"^(-1)` Electron AFFINITY : `Br(g) +E^(-) to Br^(-) (g) "" DeltaH_5^0=-331 "KJ mol"^(-1)` 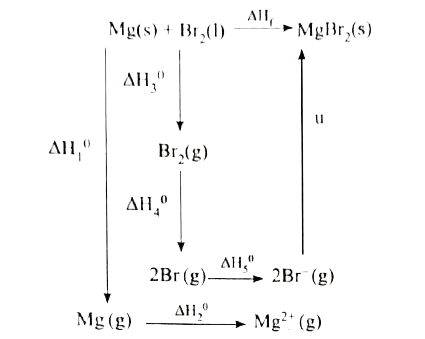 `DeltaH_f=DeltaH_1 + DeltaH_2+DeltaH_3+DeltaH_4+2DeltaH_5+u` -524=148+2187+31+193+(2 x -331) + u -524 =1897 + u u=-524 - 1897 `u=-2421 "kJ mol"^(-1)` |
|
| 32. |
Two systems, PCI_(5(g)) harr PCI_(3(g)) + Cl_(2(g)) and COCl_(2(g)) harr CO_((g)) + Cl_(2(g)) are simultaneouslyinequilibrium in a vessel at constant volume. If some CO is introduced into the vessel, then at the new equilibrium, the concentration of: |
|
Answer» `PCL_5` is greater than that before |
|
| 33. |
What will be the value of [OH^(-)]^(2) in the 0.1 M solution of ammonium hydroxide having K_(b)=1.8xx10^(-5)? |
|
Answer» `1.8xx10^(-7)` `K_(b) =([NH_(4)^(+)][OH^(-)])/([NH_(4)OH])=([OH^(-)]^(2))/([NH_(4)OH])` `[:' [NH_(4)^(+)]=[OH^(-)]]` `:. [OH^(-)]^(2) = K_(b) xx [ NH_(4)OH]` `=1.8xx10^(-5)xx0.1=1.8xx10^(-6)` |
|
| 34. |
What are the products in the following sequence of reaction Compound (E ) is |
|
Answer» `OHC(CH_(2))_(4)COOH` |
|
| 35. |
What are organic oxidation reactions? Give an example. |
|
Answer» Solution :Most of the OXIDATION reaction of organic compounds involves GAIN of oxygen or loss of HYDROGEN. EXAMPLE:`underset("acctaldehyde")(CH_(3)CHO) underset(H^(+)|K_(2)Cr_(2)O_(7)) overset((O))tounderset("Acetic acid")(CH_(3)COOH)` |
|
| 36. |
What angles are associated with the following orbitals ?sp,sp^(2) and sp^(3) |
| Answer» Solution :` SP = 180^(@), sp^(2) = 120^(@) , sp^(3) = 109^(@) 28`' | |
| 37. |
Which of the following processes are accompanied by increase or entropy: (a) Dissolution of iodine in a solvent. (b) HCI is added to AgNO_(3) and a precipitate of AgCI is obtained. (c) A partition is removed to allow two gases to mix. |
| Answer» SOLUTION :INCREASE of ENTROPY: (a) and (C) | |
| 38. |
Which of the following reactions do not yield H_(2)O_(2) as a product ? |
|
Answer» `(BaO_(2)+H_(3)PO_(4))` |
|
| 39. |
Which one ofthe following is correct for a spontaneous reaction at any temperature ? |
|
Answer» `DELTAH = -ve , DELTAS = +ve, DELTA G = -ve` |
|
| 40. |
Which of the following exhibits both +4 and -4 oxidation states ? |
|
Answer» Na |
|
| 41. |
When phenol is treated with CHCl_(3) and NaOH the product formed is |
|
Answer» benzaldehyde 
|
|
| 42. |
Which is more basic Na_(2)CO_(3) or NaHCO_(3)? |
|
Answer» `NaHCO_(3)` |
|
| 43. |
The species formed in the depletion of ozone layer by chlorotlurocarbons in free radial mechanism is |
|
Answer» `CLO^(**)` |
|
| 44. |
The weight of a gaseous mixture containing 12.044xx10^(23) atoms of He and 3.011xx10^(23) molecules of hydrogen is _____________g. |
|
Answer» Hydrogen weight = `(3.011xx10^(23))/(6.023xx10^(23))xx2=1g` Total = `8+1=9g` |
|
| 45. |
Which of the following reaction() is (are) not oxidation, reduction ? |
|
Answer» `H^(+) +OH^(-) rarrH_(2)O` 
|
|
| 46. |
Which one serve as starting materials for many organic synthesis? |
|
Answer» HALOALKANES |
|
| 47. |
Two liquids having vapour pressure P_1 ^(@) and P_2^(@)in pure state in the ratio of 2 :1 are mixed in the molar ratio of 1 : 2 The ratio of their moles in the vapour state would be |
| Answer» ANSWER :A | |
| 48. |
What happens when PCl_(5) is heated ? |
|
Answer» Solution :`PCl_(5)` has three equatorial (202 pm) and two AXIAL (240 pm) bonds (Fig. 11.35, page 11/134). Since axial bonds are weaker (REPELLED by three bond pairs) than equatorial bonds (repelled only by two bond pairs), therefore, when `PCl_(5)` is heated, the less stable axial bonds break to form `PCl_(3)`. `PCl_(5)OVERSET("Heat")toPCl_(3)+Cl_(2)`. |
|
| 49. |
Which of the following layering pattern will have a void fraction of 0.260? |
|
Answer» ABCCBAABC |
|
| 50. |
What are the uses of Calcium ? |
|
Answer» Solution :Calcium hydroxide is USED (i) in the preparation of mortar, a building material. (ii) in white wash DUE to its disinfectant NATURE. (iii) in glass MAKING and tanning industry (iv) for the preparation of bleaching powder and for the purification of SUGAR. |
|