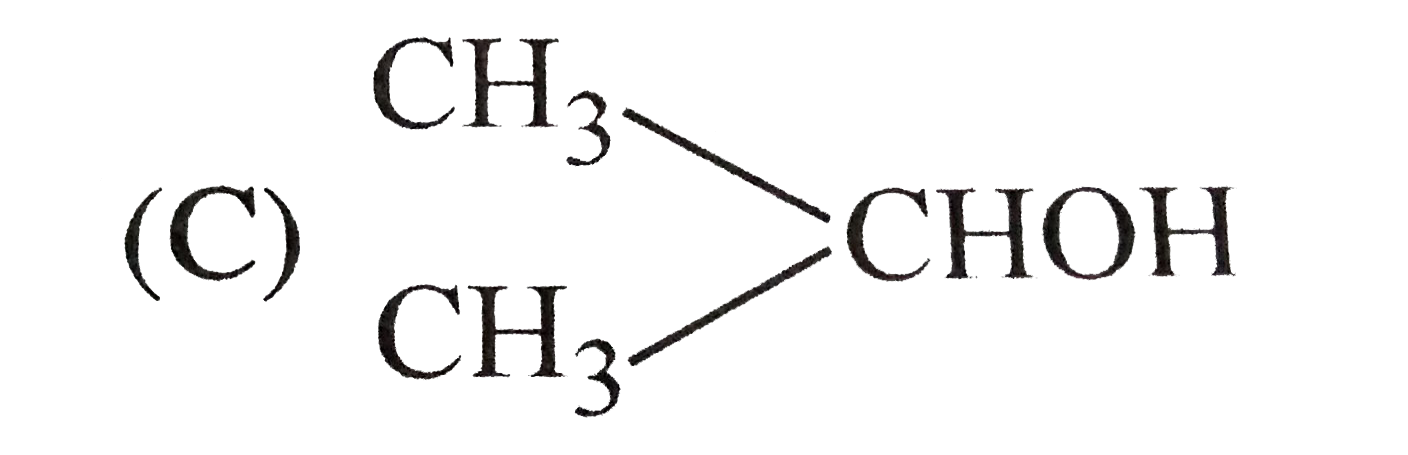Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which among the following, does not conduct electric current and why? Molten NaCl, Solid Pb,AgNO_3 solution and methanol. |
| Answer» SOLUTION :METHANOL will not CONDUCT ELECTRIC CURRENT because it does not ionise. | |
| 2. |
Which of the following is a redox decomposition reaction ? |
|
Answer» `CaCO_(3)(s)overset(Delta)toCaO(s)+CO_(2)(G)` |
|
| 3. |
What happens to an wquilibrium in a reversible reaction if a catalyst is added to it ? |
| Answer» Solution :When CATALYST is added, the state of EQUILIBRIUM is not disturbed but equilibrium is attained quickly. This is because the backward the satalyst increases the RATE of FORWARD and backward reaction to the same extent. | |
| 4. |
Tlaloalkanes undergo nucleophilic substitution reaction whereas haloarenes undergo clectrophilic substitution reaction. Comment. |
|
Answer» Solution :Haloalkanes undergo nueleophilic substitution reactions due to high clectronegativity of the halogen atom. The C-X bond in haloalkanes is SLIGHTLY polar, thereby the C atom acquires a slight positive charge in C - X bond. Hence C atom is a good targetfor attack for nucleophiles. Therefore- X atom of haloalkanes is replaced by a nucleophile easily. `NU: + overset(DELTA+)R - overset(delta-)XTO R - Nu + X` On the other hand in HALOARENES, the halogen atom releases electron to the benzene nucleus relatively electron rich with respect to the halogen atom. As a result the electrophilic attack occursat ortho and para positions. Hence haloarenes undergo electrophilic substitutionreactions. |
|
| 5. |
Write the structural formulae of the following compounds : (i) Picric acid (ii) Cumene (iii) Styrene (iv) Phthalimide (v) Hydroquinone (vi) Pyrogallol (vii) 1-Phenylpropanone (viii) 3-Bromo-1-Phenylpropene (ix) Anisole (x) Benzophenone (xi) Cinnamic acid (x) Benzophenone (xi) Cinnamic acid (xii) p-Toluidene (xiii) 2,6-Dimethylphenol (xiv) p-Methoxybenzaldehyde. |
Answer» SOLUTION :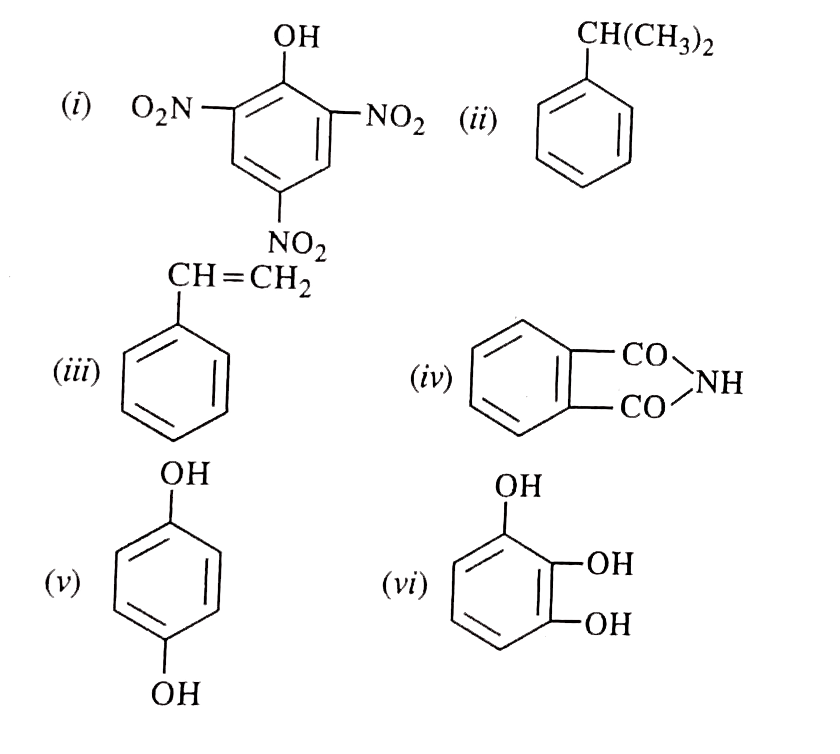 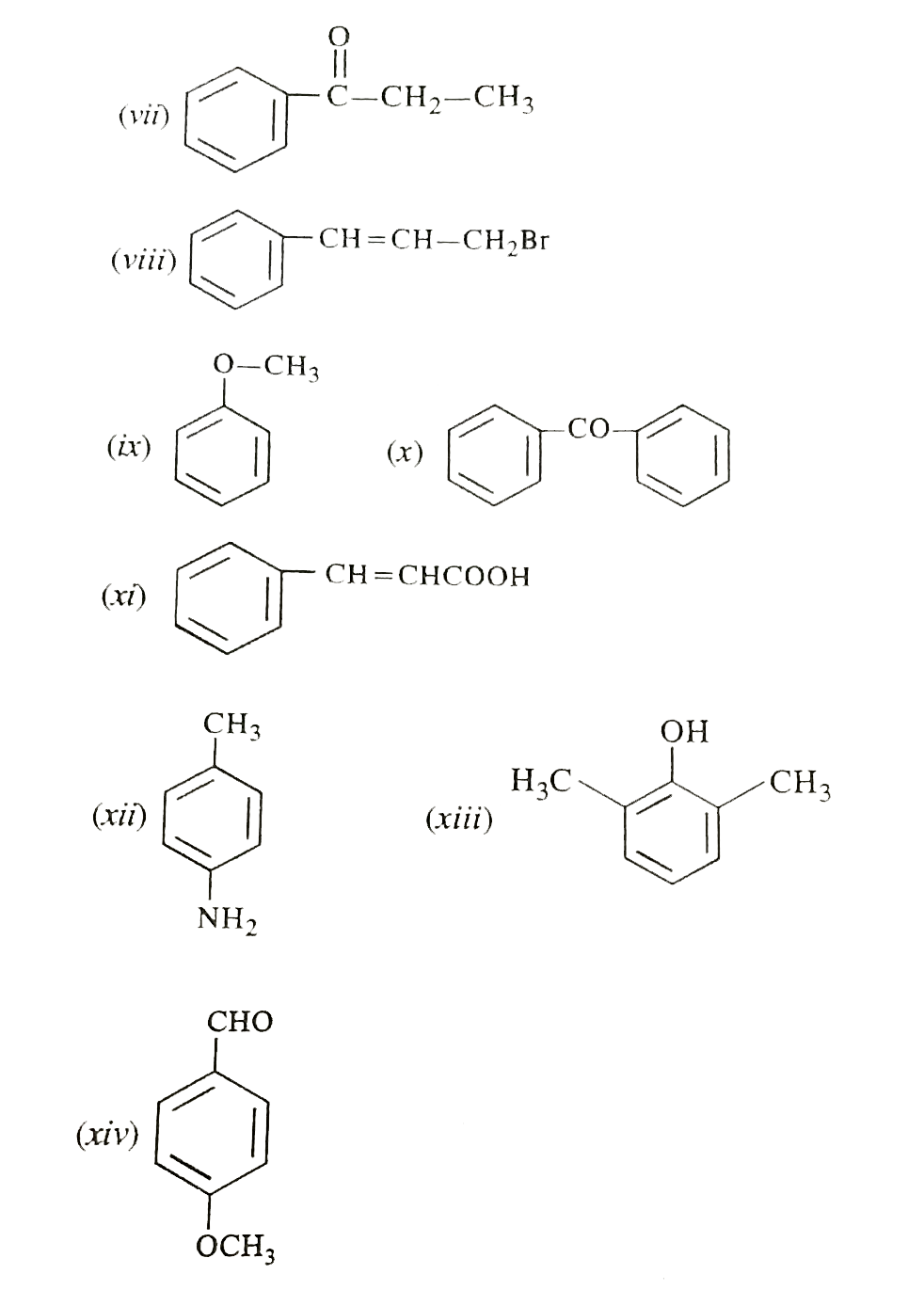
|
|
| 6. |
The standard enthalpies of formationof A(NH_(3)), B(CO_(2)), C(HI)and D(SO_(2))are respecitvely -46.19,-393.4, + 24.94 and -296.9 kJ mol^(-1) respectively. The increasing order of their stability is |
|
Answer» `Blt D lt A lt C` |
|
| 7. |
Which of the followingreaction does not stick to the cleassical idea of redox reactions ? |
|
Answer» ` 3Fe_(3)O_4(s) +8Al(s) rarr 9 Fe(s) +4Al_2O_2(s)` `Cl_(2)+3F_(2) rarr 2ClF_(3)` as both `Cl_(2)` and `F_(2)` are electronegative elements. |
|
| 8. |
Which of the following carbocations would have the greatest stability ? |
|
Answer» `CH_(3)-S-CH_(2)^(+)` 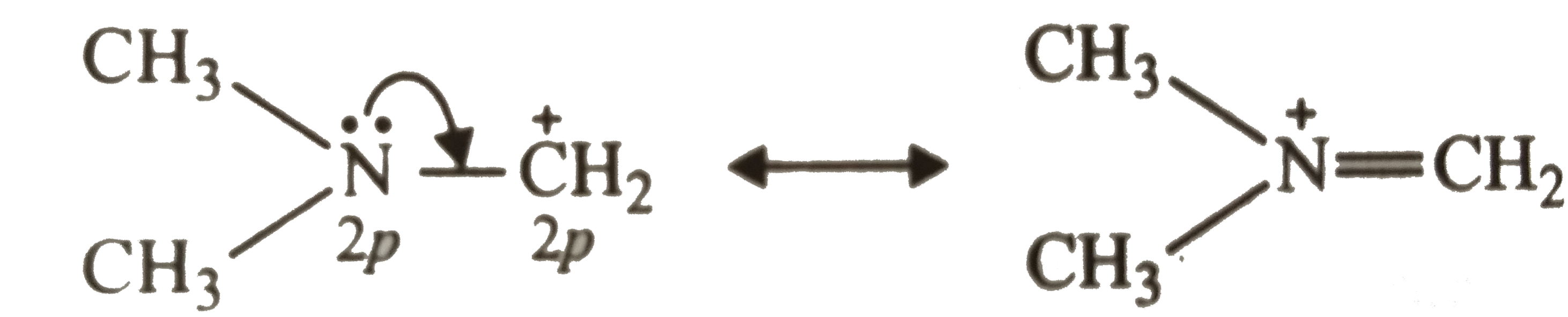
|
|
| 9. |
What is octet rule? |
| Answer» SOLUTION :The tendency of atoms of electrons to ATTAIN octet STRUCTURE of the nearset noble gas during FORMATION of a chemical bond is KNOWN as octet rule. | |
| 10. |
Write two uses of interstitial hydrides . |
| Answer» Solution :(i) Storing `H_(2)` and (II) CATALYSTS for HYDROGENATION REACTIONS. | |
| 11. |
What effect does a catalyst have on the equilibrium position of a reaction ? |
|
Answer» a catalyst favours the FORMATION of products |
|
| 12. |
The total number of orbitals associated with the principal quantum number n=3 is…………….. |
|
Answer» 9 n`=3,1=1,m_1=-1,0,1-`three p orbitals `n=3,1=2,m_1=-2,-1,0,1,2-`FIVE d oritals, overrall NINE orbitals are POSSIBLE. |
|
| 13. |
The stability of exactly half-filled and completely filled configuration is due to.........and ......... |
| Answer» SOLUTION :symmetrical DISTRIBUTION and LARGE EXCHANGE energy | |
| 14. |
Toluene undergoes nitration easilypolymerisation process |
|
Answer» Solution :(i)toluenehas amnethylgroupon thebenzeneringwhichis electronreleasinggroupandhenceactivatethe benzene ringby pushingthe electronon the benzenering. (ii) `CH_(3)` GROUPIS ORTHO -paradirectorand ringactivator . thereforein tolueneorthoandsubstitutionreaction (ii) The methylgrouphencemakesit around25 timesmorereactivethanbenzene . Thereforeit undergoesnitrationeasilythanbenzene. 
|
|
| 15. |
What is the equilibrium expression for the reaction, P_(4(s)) + 5O_(2(g)) hArr P_(4)O_(10(s)) ? . |
|
Answer» `K_c = ([P_4O_(10)])/([P_4][O_2]^5)` |
|
| 16. |
Which statement(s) about oxidation number is(are) correct? |
|
Answer» The oxidation number is the number of electrons lost (+ve) or GAINED (-ve) by an atom during the FORMATION of ionic compounds |
|
| 17. |
V and P can be |
|
Answer» `CAO,CaCO_(3)` |
|
| 18. |
What type of hybridisations are possible in the following geometeries? (a) octahedral (b) tetrahedral © square planar. |
|
Answer» Solution :(a) OCTAHEDRAL geometry is POSSIBLE by `SP^(3)d^(2)` (or) `d^(2)sp^(3)` hybridisation. (b) Tetrahedral geometry is possible by `sp^(3)` hybridisation. ( C) Square planar geometry is possible by `dsp^(2)` hybridisation. |
|
| 19. |
What is bond order ? Give example. |
| Answer» Solution :It is the NUMBER of covalent BONDS holding the atoms in the MOLECULE. Example: If the BOND is formed by the sharing of two electron pairs, then the bond oder is 2. O=O or C=C bond in alkenes. | |
| 20. |
What is effective nuclear charge ? |
|
Answer» Solution :The net nuclear charge experienced by valence electrons in the outermost SHELL is CALLED the effective nuclear charge. `Z_(eff)=Z-S` Where, Z - Atomic NUMBER S = SCREENING constant calculated by using Slater.s RULES. |
|
| 21. |
The weight ratio of elements carbon, nitrogen and hydrogen in a compound of molecular mass 108 is 18:2:7. What is its molecular formula? |
|
Answer» Solution :`C:H:N:=(18)/(12):2/1:7/(14)=3:4:1` Empirical formula is `C_(3)H_(4)N` Empirical formula weight =54 `n=(108)/(54)=2` Molecular formula `=2 XX (C_(3)H_(4)N)=C_(6)H_(8)N_(2)` |
|
| 22. |
Which colorless gas turns brown in air? |
|
Answer» NO |
|
| 23. |
To neutralize completely 20 ml of 0.1 M phosphorus acid, 40 mol of KOH was required. What volume of this KOH solution will be required to neutralize 0.66 g of H_(3)PO_(2)? |
|
Answer» SOLUTION :Eqts of `H_(3)PO_(3)` = Eqts of KOH `20xx0.1xx2=1xx40xxMimpliesM=0.1` Now Eqts of KOH = Eqts of `H_(3)PO_(2)` `0.1xx1xxV=(0.66)/(66)impliesV=0.1" lit"` `V=100ml` |
|
| 24. |
The structure of Vinyl iodide is ………………. . |
| Answer» SOLUTION :`CH_2=CHI` | |
| 25. |
Two elements P and Q form the compounds P_(2),Q_(3) , and PQ_(2). If 0.15 mole of P_(2),Q_(3), weighs 15.9 g and 0.15 mole of PQ_(2) weighs 9.3 g, find the atomic weights of P and Q. |
|
Answer» Solution :Molecular weight of `P_(2),Q_(3)` = wt. of 1 mole `(15.9xx1)/(0.15) = 106.` Molecular weight of `PQ_(2)` = wt. of 1 mole =`(9.3xx1)/(0.15) = 62.` SUPPOSE that the atomic WEIGHTS of P and Q are RESPECTIVELY x and y. Thus, 2x + 3y = 106 `(P_(2)Q_(3) =106)` x + 2y = 62. `(PQ_(2) = 62)` On solving: x = 26 y = 18. |
|
| 26. |
What is properties related to single particle of matter ? |
| Answer» Solution :Properties related to SINGLE particle of MATTER : (i) Atomic VOLUME (ii) Ionisation enthalpy (III) Electronic charge density (iv) MOLECULAR shape (v) Molecular polarity etc. | |
| 27. |
When hydrochloric acid is added to cobalt nitrate solution at room temperature, the following reaction takes place and the reaction mixture becomes blue. On cooling the mixture it becomes pink. On the basis of this information mark the correct answer. {:([Co(H_(2)O)_(6)]^(3+)(aq)+4Cl^(-)(aq)hArr[CoCl_(4)]^(2-)(aq)+6H_(2)O(l)),("(pink)" "" "(blue)"):} |
|
Answer» `Delta H gt 0` for the REACTION |
|
| 28. |
Which of the following is not an aromatic compound: |
|
Answer»
 is not an AROMATIC COMPOUND DUE to Hukele.s RULE is not an AROMATIC COMPOUND DUE to Hukele.s RULE
|
|
| 29. |
Which of the following is used for cutting and welding? |
|
Answer» ATOMIC HYDROGEN and OXY hydrogen torches |
|
| 30. |
Thespliting of spectral linein magnetic andelectricalfieldis known as . |
|
Answer» Photoelectronandstarkeffect |
|
| 31. |
Which system possess eqilibrium from the following ?(i) Saturated solution of sugar in open cup.(ii) At constant temperature mercury in thermometer and its vapour(iii) boiling water in open vessel(iv) floating ice in water at 0^@C(v) ice in water at 15^@C. |
|
Answer» Solution :(iii) and (v) is not EQUILIBRIUM. (i) Sugar solid `hArr` sugar `sol^n` equilibrium (ii)`Hg_((s)) hArr Hg_((g))`(con.temp) equilibrium (iv)`H_2O_((s)) hArr H_2O_((L))` (`0^@` C, 1 atm) equilibrium (v) It is not equilibrium as in short time ice will DISSOLVE in WATER. |
|
| 32. |
Which of the following will oxidise Hydrogen peroxide? |
|
Answer» ACIDIFIED KMnO4 |
|
| 33. |
Whybenzene undergoes electrophilic substitution reaction whereas alkenes undergoes additonreaction |
|
Answer» Solution :(i) Benzenepossesan unlybridisedp-orbitalcontainingone ELECTRON. Thelateraloverlapof theirp-orbitalsproduces 3`pi`- bond. (II) Due todelocalisation strongs `pi`- BONDIS formedwhichmakesthe moleculestable thereforebenzeneundergoes electrophilicsubstitutionreactionwheresalkenesundergoesadditionreaction . |
|
| 34. |
What is Bronsted Lowry base ? |
|
Answer» SOLUTION :Accordic to Bronsted-Lowry concept the ACID and base is defined of following: Acid : An acid is defined as a substance which has the tendency to give a PROTEN and acid is a proten donor. Base : A base is defined as a substance which has a tendency to accept a proten and base is a proten ACCEPTOR. |
|
| 35. |
What are meta directorsexplain with suitable example |
Answer» Solution :The group which increasethe electron density at meta position are CALLED as meta directorlet us consider the directive influcence of aldehydic (CHO) group BENZALDEHYDE is resonence hybrid of the following structures  In these resonance structures the postivie charge residue is presenton the ring structure it is quite evident that resonance delocalisles the positive charge on the atoms of the ringattack however resonating structures show that electron densityh is more at meta positionas compard to ORTHO and PARA positions hence CHO group is a meta director and a deactivator |
|
| 36. |
What is the name of the reaction in which bromoethane is converted to iodoethane by reacting with Nal in acetone? |
|
Answer» Hunsdicker REACTION |
|
| 37. |
Why carbon forms covalent compounds whereas lead forms ionic compounds ? |
|
Answer» Solution :Carbon cannot lose all the four valence electrons to form `C^(4+)` ions because the sum of the first four ioniatization enthalpies of carbon is very large. It alsocannot GAIN four electronto form `C^(4-)`ion as their anionwould be highlyunstablebecause it would be very difficultfor a nucleuswith only 6 protons to hold on family 10 electrons. Therefore,carbon forms only covalentcompounds by SHARING of electrons: In contrast due to inert pair effect, +2 oxidationstate of PB is more stablethanits +4 oxidationstate. Further on movingdown the group, the ionization enthalpy decreases. As a result , the sum of the first two IONIZATIONENTHALPIES of Pb is quitelow and hence Pb caneasily lose two electronsto form `Pb^(2+)` ions. Consequently, Pb form ionic compounds. |
|
| 38. |
What is oxidation number of Cr and N in (underline(N)H_(4))_(2)underline(Cr)O_(4)? |
| Answer» ANSWER :C | |
| 39. |
What is the mole fraction of the solute in a 1.00m aqueous solution ? |
|
Answer» 0.0354 `n_(H_2O)=1000/18` = 55.5 mol `H_2O` `X_"solute"=n_"solute"/(n_"solute" + n_(H_2O))` `=1/(1+55.5)`=0.0177 |
|
| 40. |
Which of the following is not a greenhouse gas ? |
|
Answer» Methane |
|
| 41. |
Use the molecular orbital energy level diagram to show that N_(2) would be expected to have atriple bond, F_(2) a single bond and Ne_(2), no bond. |
|
Answer» Solution :Formation of `N_(2)` molecule : Electronic configuration of N-atom `""_(7)N =1s^(2), 2s^(2) , 2p_(x)^(1) 2p_(y)^(1) , 2p_(z)^(1)` `N_(2)` molecule = `sigma 1s^(2), sigma^(**) 1s^(2), sigma 2s^(2) , sigma^(**) 2s^(2), PI 2sp_(x)^(2) = pi 2p_(y)^(2), sigma 2p_(z)^(2)`  Bond ORDER value of 3 means that `N_(2)` CONTAINS a triple bond Formation of `F_(2)` molecule : `""_(9)F = 1s^(2), 2s^(2, 2p_(x)^(2), 2p_(y)^(2), 2p_(z)^(1)` `F_(2) " molecule " = sigma 1s^(2), sigma^(**) 1s^(2), sigma 2s^(2) , sigma^(**) 2s^(2), sigma 2p_(z)^(2) , pi 2p_(x)^(2) = pi 2p_(y)^(2) , pi 2p_(x)^(2) = pi 2p_(y)^(2)`  Bond order = `(1)/(2) [N_(b) - N_(a) ]= (1)/(2) (10 - 8) =1 ` Bond order value 1 means that `F_(2)` contains SINGLE bond. Formation of `Ne_(2)` molecule : `""_(10)Ne = 1s^(2), 2s^(2) , 2p_(x)^(2) , 2p_(y)^(2) , 2p_(z)^(2)` `Ne_(2) "molecule" = sigma 1s^(2), sigma^(**) 2s^(2), 2 sigma 2s^(2) , sigma^(**) 2s^(2) ,sigma 2p_(z)^(2), pi 2p_(x)^(2) = pi 2p_(y)^(2), pi 2p_(x)^(2) = pi 2p_(y)^(2) = sigma 2p_(z)^(2)` Bond order = `(1)/(2) [ N_(b) - N_(a) ]= (1)/(2) (10 - 10) =0 `  Bond order value zero means that there is no formation of bond between two Ne-atoms. Hence, `Ne_(2)` molecuule does not exist, |
|
| 42. |
The unequal sharing of the bond pair of electrons between two atoms in a molecule causes |
|
Answer» NON POLAR covalen bond |
|
| 43. |
Which of the following alcohol is most acidic ? |
|
Answer» `CH_(3)OH` |
|
| 44. |
The tendency of BF_3, BCl_3 and BBr_3 to behave as Lewis acid decreases in the sequence: |
|
Answer» `BCl_3 GT BF_3 gt BBr_3` |
|
| 45. |
Which one of the following statement is correct ? |
|
Answer» Melting point of and BOILING point of HI are |
|
| 46. |
Which of the following is an amphoteric ? |
|
Answer» `SnO_2` |
|
| 47. |
Which of the following is not a monomer for ahigh molecularmass silicone polymer ? |
|
Answer» `Me_(3)SiCl` |
|
| 48. |
Which of the fallowing alkyl halides is not suitable for Corey-house synthesis of alkanes? |
|
Answer» `CH_3I` |
|
| 49. |
Thermonuclear reaction is related to |
|
Answer» FUSION of HYDROGEN |
|