Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When is the law of constant proportions not obeyed ? |
| Answer» Solution :Law of CONSTANT PROPORTIONS is not OBEYED when an element exists in the form of TWO or more isotopes. | |
| 2. |
Usingthe PeriodicTable , predict theformulas of thecompoundswhichmightbe formedby thefollowingpairsof elements: (a)silliconand bromine (b)aluminiumand sulphur. |
|
Answer» Solution :(a) SILICON belongto group14.It has fourvalenceelectronsand henceits valence=4 . Brominebelongsto GROUP 17. It has sevenvalence electronsand henceits valence =8-7 =1 thereforethe FORMULAOF thecompoundformedwould be 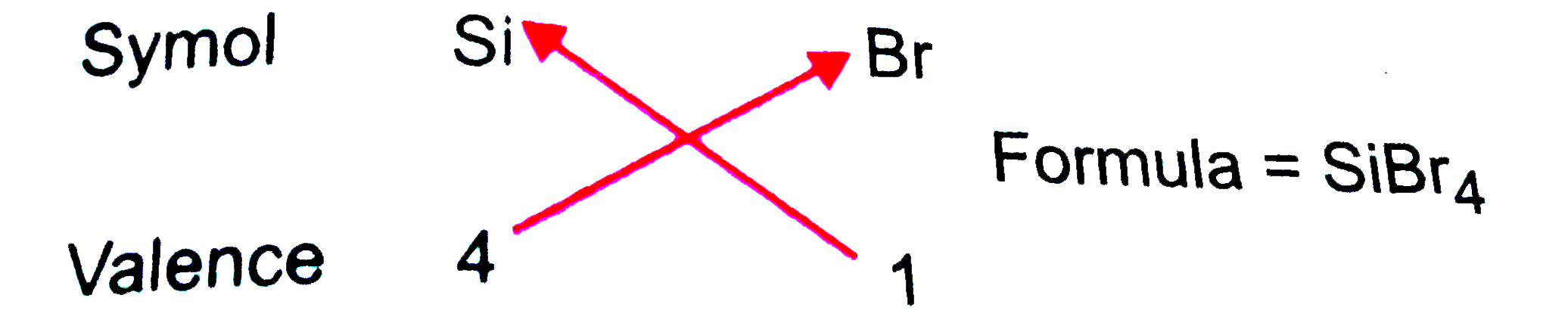 (b) Aluminiumbelongs group 13. Ithas three element in thevalenceshelland henceits valence =3Sulphur belongsto group 16. Ithas 6 valence electronsand henceitsvalence =8 -6 =2 Thereforethe formulaof the compoundformedwould be 
|
|
| 3. |
Which of the following molecules acts as a Lewis acid ? |
|
Answer» `(CH_(3))_(2)O` |
|
| 4. |
Which of the following reactions is an example of autoreduction ? |
|
Answer» `Fe_(3)O_(4)+4COrarr3Fe+4CO_(2)` It is not intermolecular redox reaction . |
|
| 5. |
What is Zeeman effect? |
| Answer» SOLUTION :If a SUBTANCE which GIVES a line emission SPECTRUM, is placed in a magnetic field, the lines of the spectrum get split up into ANUMBER of closely spaced lines. This phenomenon is known as Zeeman effect. | |
| 6. |
Which bond is short C = O, or N = O ? Why ? |
| Answer» SOLUTION :CO, because its ELECTRONEGATIVITY DIFFERENCE is more than No. | |
| 7. |
Which of the following molecules is both planar and linear |
|
Answer» `CH_(2)=CH-CH=CH_(2)` |
|
| 8. |
What is wrong with the names givenfor these compounds ? Provide the correct name for each. |
|
Answer» Solution :(i) The parent CHAIN has six carbon atoms and not five Correct IUPAC name : 3-Methylhex-3-ene (ii) The LOWEST set of LOCANT rule has not been followed Correct IUPAC name : 2,7,8-Trimethyldecane (III) Lowest number has not been given to the locant for the secondary suffix. Correct IUPAC name : Pent-4-yn-1-ol (iv) The functional group is a N-methylamine Correct IUPAC name : N-Methylbut-3-enamine (v) RING has more carbon atoms than the alkyl group Correct IUPAC name : Propylcyclohexane (vi) Lowest sum rule has not been followed Correct IUPAC name : 1,2,4-Trimethylcyclopentane (vi) Lowest number has been given to the lacant for the prefix and not to en. Correct IUPAC name : 3-Chlorocyclopentene. |
|
| 9. |
Which of the following has maximum freezing point? |
|
Answer» PURE `H_2O ` |
|
| 10. |
What is mole fraction of methyl alochol in a 5.2 molal aqueous solution of methyl alcohol, CH_(3)OH. |
|
Answer» 0.1 `:.` 5.2 MOLE of methanol in 1000 G WATER. Mole of water `= (1000)/(18) = 55.56` TOTAL mole `= 5.2 + 55.56 = 60.76` Mole fraction of methanol `=("mole of methanol")/("total mole")` `= (5.2)/(60.76) = 0.08558 ~~0.086` |
|
| 11. |
Which of the following elements shows diagonal relationship ? |
|
Answer» LI and Mg |
|
| 12. |
What is meant by water pollution? |
| Answer» SOLUTION :Water pollution is DEFINED as the ADDITION of foreign substances or factors like HEAT which degrades the quality of water so that it becomes an health HAZARD or untit for use. | |
| 13. |
What is the K_(c) value for formation of HI at 700 K ? Predict the extent of the reaction ? |
|
Answer» Solution :`H_(2)(g)+I_(2)(g) hArr 2HI (g) at 700 K` `K_(C)=57.0` `10^(-3) lt K_(c) lt 10^(3)`. So both forward and backward REACTION make SIGNIFICANT progess. Neither forward nor reverse reaction predominates. |
|
| 14. |
Write the structure of the following compounds, (a) 1-Brumo-4-ethyl cyclobexane (b) 1,4-Dichlorobut-2-ene (c) 2-Chloro-3-metyl pentane |
Answer» SOLUTION :(a) 1-bruno-4 ethylcyclo HEXANE (B) 1,4-dichloro but-2-ene (C) 2-Chloro-3-methyl PENTANE 
|
|
| 15. |
What are combuistion reactions |
|
Answer» Solution :A combustionreactionis achemicalreactionbetweensubstaneand oxygenwithevol of heatand lightin thepresenceof sufficientoxygenalkane undergoncombustionignited and producesscarbondioxideand WATER `CH_(4) + 2O_(2) toCO_(2)2H_(2) ODelta H^(@) = 890.4 KJ` |
|
| 16. |
What is volume of different gas of n mole different gases at constant temperature and pressure ? |
|
Answer» <P> SOLUTION :Volume of different GASES are same at constant T and constant p because in given formula n, R, T, p are same.`V=(NRT)/(p)` |
|
| 17. |
What are the conjugate bases of the following ? CH_(3)OH, HN_(3), [Al(H_(2)O)_(6)]^(3+). |
| Answer» Solution :`{:(CH_(3)O^(-)",",N_(3)^(-)",",[AL(H_(2)O)_(5)OH]^(2+)(H^(+)"ion has been removed from one "H_(2)O "MOLECULE")),("methoxide ion","AZIDE ion",),(,,):}` | |
| 18. |
The values of K_(P_(1)) and K_(P_(2)) for the reactions: X hArr Y'Z and A hArr 2B are in the ratio 9:1 if degree of dissociation and initial concentration of X and A be equal then total pressure at equilibrium P_(1), and P_(2) are in the ratio. |
|
Answer» `36:1`  `(K_(P_(1)))/(K_(P_(2)))=(P_(y)P_(Z))/(P_(x))XX(P_(A))/(P_(B)^(2))` `(K_(P_(1)))/(K_(P_(2)))=(P_(y)P_(Z))/(P_(x))XX(P_(A))/(P_(B)^(2))` 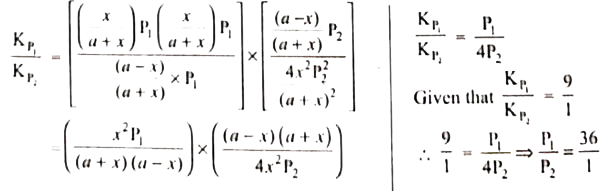
|
|
| 19. |
Which state of Matter having definite volume but not definite shape ? |
| Answer» SOLUTION :Only liquid have definite VOLUME but it volume is as per the SHAPE of the VESSEL. | |
| 20. |
Which one of the following is present as an active ingredient in bleaching powder for bleaching action ? |
|
Answer» `CaOCl_(2)` |
|
| 21. |
The total number of diproticacids among the following is H_(3)PO_(4)"" H_(2)SO_(4) " " H_(3)PO_(3) " " H_(2)CO_(3) " " H_(2)S_(2)O_(7)" " H_(3)BO_(3) " " H_(3)PO_(2) " " H_(2)CrO_(4) " " H_(2)SO_(3) |
Answer» 
|
|
| 22. |
Which of the following does not occur as sulphide ore. ? |
|
Answer» Zn |
|
| 23. |
What is the standard pressure of atmosphere ? |
| Answer» SOLUTION :1 ATMOSPHERE = 1.013 BAR | |
| 24. |
What is the reason behind the cause of earh pain while climbin a mountain? How it can be rectified? |
|
Answer» Solution :(i) When one ascends a mountain in a plain, the EXTERNAL PRESSURE drops while the pressure WITHIN the air cavities remains the same. This creates an imbalance. (ii) The greater internal pressure forces the eardrum to bulge outward CAUSING pain. (iii) With TIME and with the help of a yawn or two, the excess air within your ear.s cavities escapes thereby equalizing the internal and external pressure and relieving the pain. |
|
| 25. |
What is a spontaneous process ? Give an example. |
|
Answer» Solution :A PROCESS which takes place on its own, WITHOUT any external aid under the given condition is called a spontaneous process. EXAMPLE : FLOW of heat from higher to lower temperature. |
|
| 26. |
Wurtz reaction using bromoethane yields |
|
Answer» (i) 2-bromobutane |
|
| 27. |
The temperature at which the reaction Ag_(2)O(s) rarr 2Ag(s) +1/2 O_(2) (g) at 1 atmospheric pressure will be in equilibrium is ____ K. The value of DeltaH and DeltaS for the reaction are 30.58 kJ and 66.11 JK^(-1) respectively and these value do not change much with temperature. |
|
Answer» 462.6 K |
|
| 28. |
Which of the following molecular formula belongs to alkyne series ? |
|
Answer» `C_(7)H_(14)` |
|
| 29. |
Two moles of an ideal gas expand isothermally and reversibly from 1 litre to 10 litre at 300K, The enthalpy change (in kJ) for the processis |
|
Answer» 11.4 kJ Forisothermal expansion of ideal gas, `DeltaU = 0` `( :' DeltaU =C_(v) DeltaT ` and `DeltaT =0)` `:. DeltaH =0 +0, i.e., DeltaH =0` or putting `DeltaU = C_(v) DeltaT , DeltaH = C_(v) DeltaT + R DeltaT` `= (C_9v) +R) DeltaT = C_(p) DeltaT` for 1 mole. For N moles, `DeltaH = n C_(p) DletaT =0` |
|
| 31. |
Which of the following compounds gives blood red colouration when its Lassaigne's extract is treated with alkali and ferric chloride ? |
|
Answer» Phenyl hydrazine |
|
| 32. |
When we apply brakes of our car, which property of the brake fluid is applicable |
| Answer» Solution :The PROPERTY of incompressibility of the liquids (brake FUID) to transmit the PRESSURE which we apply with out foot to the brake shoes on the wheels. | |
| 33. |
The wrong statement in the case of an ideal gas is |
|
Answer» It cannot be CONVERTED into a liquid |
|
| 34. |
Which of the following elements differs from the rest in several physical and chemical properties?(a)Li(b)Na(c)Rb(d)Cs |
|
Answer» LI |
|
| 35. |
Which of the following compound on reductive ozonolysis does not give glyoxal as one of the product |
|
Answer»
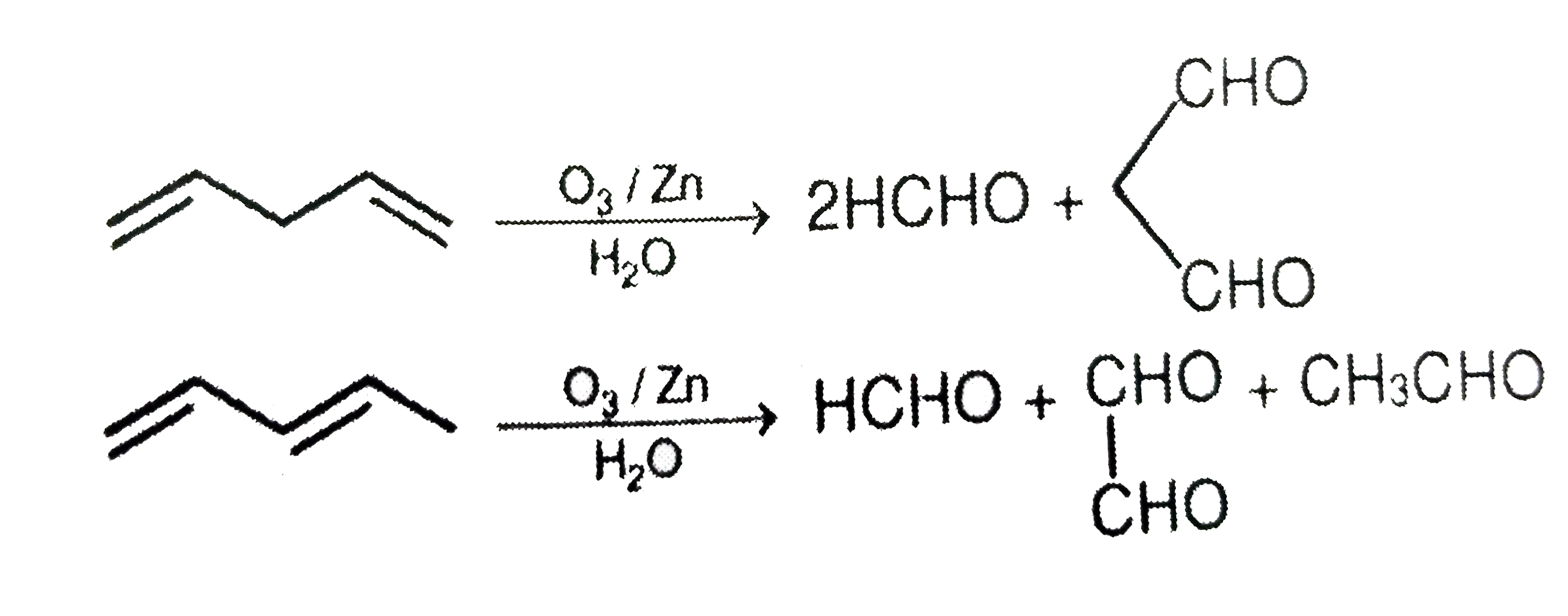 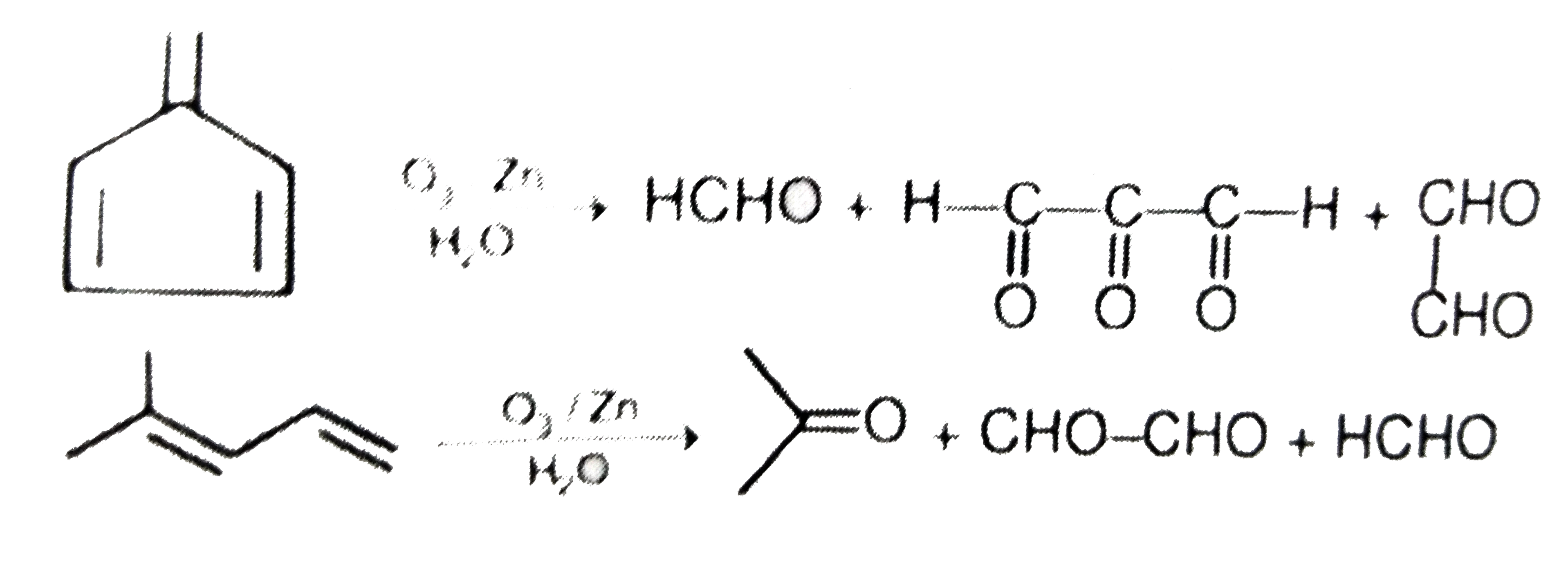
|
|
| 36. |
The three main components of photochemical smog are ...... |
| Answer» SOLUTION :HYDROGEN SULPHIDE, DUST and PAN. | |
| 37. |
Value of universal gas constant (R) is same for all gases. What is its physical significance ? |
| Answer» Solution :From the gas equation, PV=nRT, `R=(pV)/(nT)`. If p is measured in PASCAL, V/n i.e.,molar VOLUME per mole in `m^(3)` and T in kelvin, units of R are Pa `m^(3)K^(-1)mol^(-1)" or J "mol^(-1)K^(-1)`. As joule is the unit of work, R is the work done per mole per kelvin. | |
| 38. |
Using the data ( al values are in kilocalories per moleat 25^(@)C ) given below, calculate the bond energy of C-C and C-H bonds. DeltaH^(@)combustion( ethane) = - 372.0 DeltaH^(@)combustion ( propane) = - 530.0DeltaH^(@)forC ( graphite rarr C (g) = 172.0 Bond energy of H-H= 104.0 Delta_(f)H^(@) ofH_(2)O(l)= - 68.0,Delta_(f)H^(@) forCO_(2) = - 94.0 |
|
Answer» Solution : We are GIVING (i)`C_(2)H_(6)(g)+(7)/(2) O_(2)(g) rarr 2CO_(2)(g)+H_(2)O(l),DeltaH^(@) =- 372.0 kcal` (ii) `C_(3)H_(8) +5O_(2)(g) rarr 3CO_(2)(g)+4H_(2)O(l),DeltaH^(@) = - 530.0 kcal` (III) `C(s) rarr C(g), DeltaH^(@) =172.0kcal` (iv) `H_(2)(g) rarr 2H(g) , DeltaH^(@) = 104.0kcal` (vi)` C(g)+ O_(2)(g) rarrCO_(2)(g) , DeltaH^(@) = - 94.0 kcal` Supposethe bond energyof`C-C` bond `=X kcal ` mol`e^(-1)` and thatof`C-H` bond `=y` kcal` mol e^(-1)`. Then for `C_(2)H_(6)(g), i.e.,H- underset(H) underset(|) overset(H) overset(|) (C) - underset( H ) underset(|) overset(H) overset(|) (C)-H rarr C(g) +H(g),DeltaH = x+6y`....(vii) andfor `C_(3)H_(8) (g)`, i.e., `H-underset(H) underset(|) overset(H) overset(|)(C) - underset(H) underset(|) overset(H) overset(|) (C) - underset(H) underset(|) overset(H) overset(|) (C) - H rarr 3C(g) +8 H(g), DeltaH = 2x + 8 y `.....(viii) To get Eqn. (vii) , operateEqn. (i) `+ 2 xx `Eqn. (iii) ` + 3 xx` Eqn. (iv) `- 3 xx` Eqn. (v) `- 2 xx` Eqn. (vi). This gives`DetlaH = 676 kcal` To get Eqn. (viii) , operateEqn. `+` Eqn. (iii) `+`Eqn. (iii) `+ 4 xx` Eqn. (vi)` - 4 xx` Eqn. (v) `- 3 xx `Eqn. (vi). This gives `DeltaH = 956kcal` Thus,`x + 6y= 676 ` `2x + 8y=956` On solving these equations, we get `x= 82,y = 99` Hence, `C-C` bond energy `= 83 kcal mol^(-1)` and `C-H` bond energy `= 99` kcal `mol^(-1)` |
|
| 39. |
What is oxidation number? |
|
Answer» SOLUTION :The gain of electron or the loss of electron from the atom, molecules or ions, the number of electron gain or LOOSED termed as oxidation number. Oxidation number INDICATES the electric charge of any atom at its oxidation stage. In simple ionic COMPOUNDS oxidation number of elements is similar to electric charge of the element. |
|
| 40. |
The vapour density of a mixture containing NO_(2) " and "N_(2)O_(4) is 38.3 " at " 33_(@)C calculate the no. of moles of NO_(2) if 100kg "of" N_(2)O_(4) were taken initially. |
|
Answer» Solution :`N_(2)O_(4)(G)iff2NO_(2)(g)` `M_(mix)=2xx38.3=76.6` `N_(2)O_(4)iff2NO_(2)` `{:(t=0,a,0),(t=t,a-aalpha,2aalpha):}` no. of MOLES of `NO_(2)=`"no. of moles of" N`O_(2)=2_(aalpha)=(2xx100xx0.2)/(92)=0.435` |
|
| 41. |
The series limit for Balmer series of H-spectra is |
|
Answer» 3800 |
|
| 42. |
When aqueous solution of borax is acidified with hydrochloric acid, a white crystalline solid is formed which is soapy to touch. Is this solid acidic or basic in nature ? Explain. |
|
Answer» Solution :An aqueous solution of borax is acidified with HCl to give boric acid. `Na_2B_4O_7 + 2HCL + 5H_2O to 2NaCl + underset"Boric acid"(4H_3BO_3)` Boric acid is a weak monobasic acid. It is not a protonic acid but acts as a Lewis acid by ACCEPTING ELECTRONS from a hydroxyl ion : `B(OH)_3 + 2HOH to [B(OH)_4]^(-)+ H_3O^+` |
|
| 43. |
Which of the following combinations shall give rod azo dye with HNO_(2)? (II) (KMnO_(4)+dil. H_(2)SO_(4)) (IV) (FeSO_(4)+dil. CH_(3)COOH) |
|
Answer» REACTION with (I) FOLLOWED by reaction with (II) |
|
| 44. |
Which one of the following is most reactive towards nucleophilic substitution reaction? |
|
Answer»
|
|
| 45. |
The total number of following ions, which interfere with the brown ring test for NO_(3)^(-) is/are: Br^(-),I^(-),CO_(3)^(2-),NO_(2)^(-),CIO_(3)^(-),CrO_(4)^(-2) |
|
Answer» |
|
| 46. |
Which among the following are auto - redox reactions ? |
|
Answer» `P_(4)+OH^(-) rarrH_(2)PO_(2)^(-) +PH_(3)` `*S_(2)O_3^(2-)rarrSO_4^(2-)+S` `* H_2O_2 rarrH_2O+1/2O_2` |
|
| 47. |
When water freezed to ice, entropy .......... . |
|
Answer» Increases |
|
| 48. |
Which of the following reactions will get affected by increasing the pressure ? Also, mention whether change will cause the reaction to go into forward or backward direction. (i)COCl_(2(g)) hArr CO_((g)) +Cl_((2)(g)) (ii)CH_(4(g)) + 2S_(2(g)) hArr CS_(2(g)) + 2H_2S_((g)) (iii)CO_(2(g)) + C_((s)) hArr 2CO_((g)) (iv)2H_(2(g)) + CO_((g)) hArr CH_3OH_((g)) (v) CaCO_(3(s)) hArr CaO_((s)) + CO_(2(g)) (vi)4NH_(3(g)) + 5O_(2(g)) hArr 4NO_((g)) + 6H_2O_((g)) |
|
Answer» Solution :In CHEMICAL equilibrium the components in gases phase and `Deltan_((g)) ne 0`so, the effect of pressure on reaction. Only in reaction (ii) `Deltan_((g)) ne 0` and remaining reaction `Deltan ne 0`. So, effect of pressure does not occurs on reaction (ii) and remaining reaction (i), (iii), (iv), (v), (vi) the effect of pressure is occurs. According to increases in pressure, the forward reaction is (iv), REVERSE reactions are (i), (ii), (v) an (iv). Note: There is no effect of reaction (ii). Explain : On increasing pressure reaction moves in such direction moles of gases decreases and NEW equilibrium is established as per Le Chatelier.s PRINCIPLE. i.e. `Deltan_((g)) lt 0`in that direction reaction is forward. 
|
|
| 49. |
Which one of the following has maximum entropy ? |
| Answer» Answer :D | |