Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which are the correct IUPAC names of the following compound? |
|
Answer» 5 - Butyl -4- isopropyldecane |
|
| 2. |
Which of the following is a Bronsted neutralisation reactions? |
|
Answer» `H^(+)+OH^(-)to H_2O ` |
|
| 3. |
What is the inductive effect? Which one of the following shows maximum hyper cojugation effect? CH_(3)CH=CH_(2)"" (CH3)_(2)=C=CH_(2)"" CH_(2)=CH_(2) |
|
Answer» Solution :The percentage displacement of bonded sigma ELECTRONS along the CHAIN the carbon atoms due to the presence of a substitutent group of different electronegativitgy is called inductive effect. `(CH_(3))_(2)C=CH_(2)` |
|
| 4. |
WhichimportantpropertydidMendeleev use toclasify the elements in hisperiodictableand didhe stick tothat ? |
| Answer» SOLUTION :Mendeleevusedatomicweightas thebasisof classification ofelementsin theperiodictable. He arrangedthe thenknownelementsin ORDEROF increasingatomicweigths groupingtogetherelements notknownat THATTIME. Forexamplethe ELEMENT galliumafteraluminiumandgermanium aftersilicon werenot known at thetimeMendeleevpreparedhisperiodictable. He calledtheseelementsas EKA- aluminium and ekasilicon. Latertheseelementswerediscoveredand theirpropertieswerefound to besimilarto thosepredictedby Mendeleev. | |
| 5. |
The species having bond angle of 120^(@) is |
|
Answer» `PH_(3)` STRUCTURE having a bond angle of `120^(@)` . All other molecules have bond angle LESS than `120^(@)` . |
|
| 6. |
Which of the given elements will gain one electron more readily in comparison to other elements of their group- |
| Answer» ANSWER :A::D | |
| 7. |
When 1 mole of C_(2)H_(6)(g)at 27^(@)C is burnt completely in some oxygen gas at 27^(@)C At constant pressure, CO_(2)(g),CO(g)and H_(2)O(g) are produce at 327^(@)C. the surropundings. The standard enthaplies of formation of C_(2)H_(6)(g).CO(g) and H_(2)O(g) are -40, -90, -30 and -60kcal//mol respectively. The molor heat capacities at constant pressure ofCO_(2)(g),CO(g) and H_(2)O (g) are 7.0,6.0 and 9.0cal//Kmol, respectively. The molar hear capacities at constnt pressure of CO_(2)(g),CO(g) and H_(2)O (g) are 7.0,6.0 and 9.0 cal//K-mol respectively.On the basis of these informations, math teh valuesof column-I with the quantities represented in column-II Assume Delta_(r)H independent of temperature. {:(,"Column-I",,"Column-II"),((a),1.0,(p),Delta_(r)H "of reaction" : 2C_(2)H_(6)(g) + 7O_(2)(g)rarr 4CO_(2)(g)+6H_(2)O(g),"is"),((b),-200"kcal",(q),Delta_(r)H"of reactIon":C_(2)H_(6)(g)+(5)/(2)O_(2)(g)rarr2CO(g)+3H_(2)O(g)"is"),((C),-3600"cal",(r),"moles of CO formedis"),((d),-640kcal,(s),"Moles of CO_(2)formed is"),(,,(t),"Work done by the system is") :} |
|
Answer» |
|
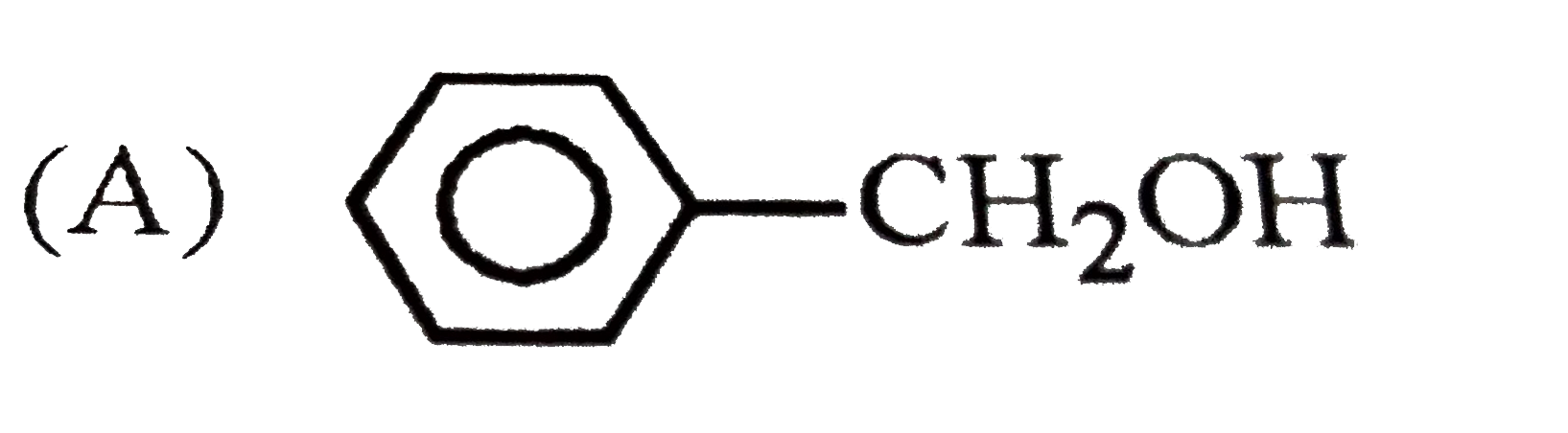
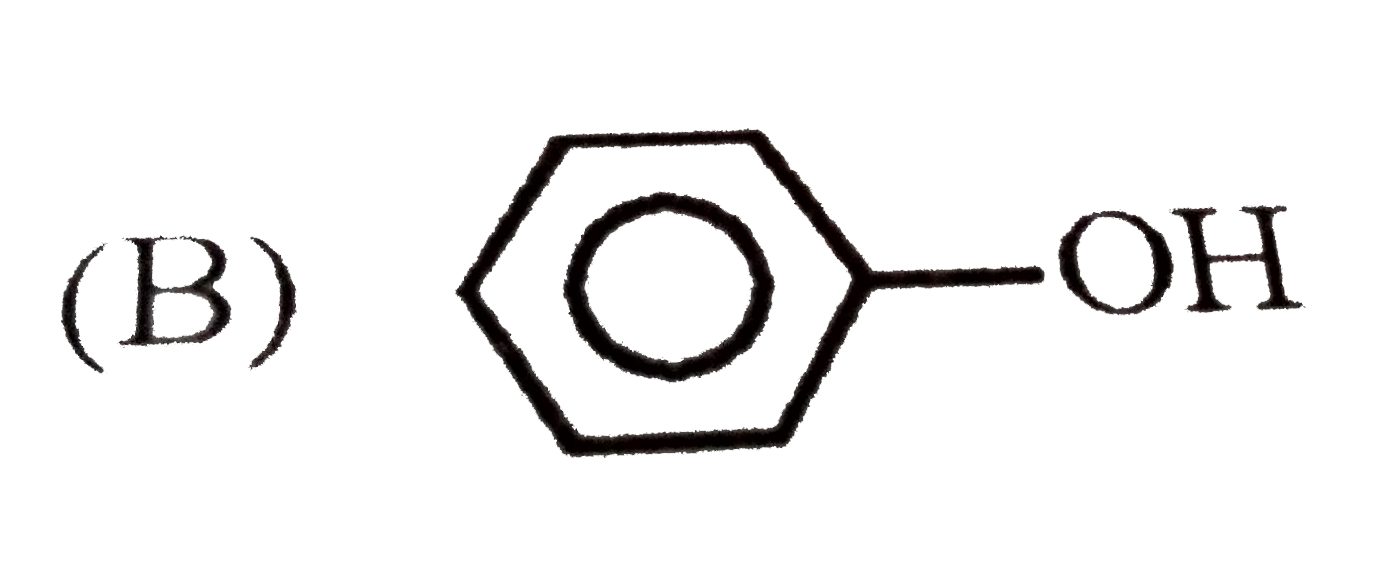
| 8. |
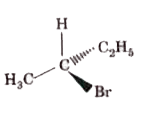
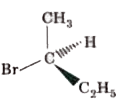
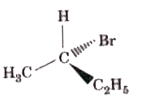
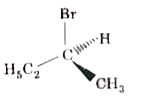
Which of the following structures is enantiomeric with the molecule (A) given below? |
|
Answer»
|
|
| 9. |
The uncertainty in the momentum of a particle is 3.31 xx 10^(-2) kgms^(-1). The uncertainity in its position is (in metres) |
|
Answer» `1.59xx10^(-33)` |
|
| 10. |
Which of thefollowing species do not show disporoportionationreaction and why ? CIO^(-),CIO_(2)^(-),CIO_(3)^(-) and CIO_(4)^(-) Also write reaction for each of the species that disproportinates |
|
Answer» Solution :The oxidation STATE of CI in all the given species are `overset(+1)CIO, overset(+3)CIO, overset(+5)CIO_(3), overset(+7)CIO_(4)` out of above species `CIO_(4)` does not undergo disproportionation since in this oxanion chloringis ALREADY present in the higherst oxidation state +7 and hence cannot be further oxidation all the remaing oxanotion have oxdation states LOWER undergo disproportion reaction as shown below `3overset(+1)CIOrarr2overset(-1)CI+overset(+5)CIO_(3),overset(+3)CIO_(2)rarr2CIO_(3),4CIO_(3)^(-)rarr3 CIO_(4)^(-)` |
|
| 11. |
Which of the following pair of elements have same atomic radius |
| Answer» Answer :B | |
| 12. |
What are bio-degradable and non-biodegradable pollutants? |
|
Answer» SOLUTION :(i) The pollutants which can be easily DECOMPOSED by the natural biological processes are called biodegradable pollutants. For example plant wastes, ANIMAL wastes. (ii) The pollutants which cannot be decomposed by the natural biological processes are called non-biodegradable pollutants. For example, METAL wastes such as HG and Pb, D.D.T. plastics. nuclear wastes |
|
| 13. |
Which of the following concentration terms is/are affected by a change in temperature ? |
|
Answer» MOLARITY |
|
| 14. |
What type of hybridisations are possible in the following geometeries ? (a) octahedral (b) tetrahedral (c ) square planar |
|
Answer» Solution :(a) OCTAHEDRAL GEOMETRY is possible by `sp^(3)" "d^(2) (or) d^(2)" "sp^(3)` hybridisation. (B) Tetrahedral geometry is possible by `sp^(3)` hybridisation. (c) Square planar geometry is possible by `dsp^(2)` hybridisation. |
|
| 15. |
Which has maximum number of molecules among the following? |
|
Answer» 44 g of `CO_(2)` No. of moles of `O_(3)=((48G))/(("48 g mol"^(-1)))=1`mol No. of moles of `H_(2)=(8G)/(("2g mol"^(-1)))=4` mol No. of moles of `SO_(2)=((64G))/(("64g mol"^(-1)))=1` mol `:.` 4 moles or 8g of `H_(2)` have max. no of molecules. |
|
| 16. |
What is the difference between ground state and excited state ? |
| Answer» SOLUTION :Ground state means the LOWEST energy state. When the electrons absorb energy and jump to outer orbits. This state is CALLED excited state. | |
| 17. |
Which are the correct IUPAC names of the following compound ? {:(""HC(CH_(3))_(2)),("|"),(H_(3)C-CH_(2)-CH_(2)-CH-CH-CH_(2)-CH_(2)-CH_(2)-CH_(2)CH_(3)),("|"),(""H_(3)C-CH-CH_(2)-CH_(3)):} |
|
Answer» 5-Butyl-4-isopropyldecane (C) 5-sec-Butyl-4-iso-propyldecane (D) 4-(1-methylethyl)-5-(1-methylpropyl)-decane |
|
| 19. |
What are the mojor differece between metals and non metals ? |
| Answer» SOLUTION :Metals are electropositive (lower IE) element with LESS negative electron gain enthalpies. They are good reducing AGENTS which from BASIC oxide and inoic COMPOUNDS. Non metals are electronegative (high IE) elements with more nagative electron gain enthalpies. | |
| 20. |
What is Sabatier - Sendersens reaction? |
|
Answer» Solution :The process of additionof `H_2 `to unsaturatedcompounds( alkenesoralkynes) in knownas hydrogenation . Theaboveprocesscan becataysedbynikelat 298K .This reactionis knownassabatier -sendersensreactionfor EXAMPLE : ` overset("( Ethene)") (CH_2 = CH_2 +H_2 )underset(298 K ) overset( NI)tounderset("(ETHANE )") (CH_3 -CH_3)` |
|
| 21. |
Using van der Waal's equation calculate the constant 'a' when 2 mole of a gas confined in a 4 L flask exerts a pressure of 11.0 atm at a temperature of 300 K. The value of 'b' is 0.05 L mol^(-1). |
| Answer» SOLUTION :`6.49 ATM L^(2) MOL^(-2)` | |
| 22. |
Which one of the following causes cancer? |
|
Answer» 1, 2-Benzpyrene |
|
| 23. |
Which of the following compounds ae correctly related |
|
Answer» `{:("Compound","use"),(MgSO_(4)*7H_(2)O,"MORDANT in dying fabrics"):}` |
|
| 24. |
Whodiscoveredprotonand neutron ? |
|
Answer» SOLUTION :PROTON DISCOVEREDBY Goldstinin 1886 NEUTRON discoveredby Chadwickin 1932. |
|
| 25. |
The speed of an electron in the inner most orbit of the hydrogen (Bohr radius = 52.9 pm, me = 9.11 xx 10^(-31) kg) is |
|
Answer» `2.19x10^(14)m.s^(-1)` |
|
| 26. |
The solubility product constants of Ag_(2)CrO_(4) and AgBr are 1.1xx10^(-12) and 5.0xx10^(-13) respectively. Calculate the ratio of molarities of theirsaturated solutions. |
|
Answer» |
|
| 27. |
Which one of the following has both ionic and covalent bond ? |
| Answer» Answer :B | |
| 28. |
Which of the following is insoluble in water? |
|
Answer» LiF |
|
| 30. |
How much minimum volume of CO at STP is needed to react completely with 0.112 L of O_(2) at 1.5 atm. Pressure and 127^(@)C to give CO_(2). |
|
Answer» Solution :Carbonmonoxide isoxidised in oxygen to give CARBON dioxide `2CO+O_(2)rarr2CO_(2)` 1 mole of `O_(2)` = 2 moles of CO 1 volume of ` O_(2)` = 2 volumes of CO 0.112L `O_(2)` at STP = 0.224L CO at STP STP conditionsGiven conditions `P_(1)=1atm""P_(2)=1.5atm` `T_(1)=273K""T_(2)=400K` `V_(1)=0.224L""V_(2)=?` Volume of carbonmonoxide needed in the given conditions `=V_(2)= (P_(1)V_(1))/(T_(1))XX(T_(2))/(P_(2)) = (1xx0.224xx400)/(273xx1.5) =229.3 L` |
|
| 31. |
Whichof the following compounds is most acidic ? |
|
Answer»
|
|
| 32. |
Various oxidation states of few elements are mentioned. Which of the options is not correctly matched ? |
|
Answer» a. Phosphorus: +3 to +5 |
|
| 33. |
Which reaction yields Bakelite ? |
|
Answer» Urea with `HCHO` 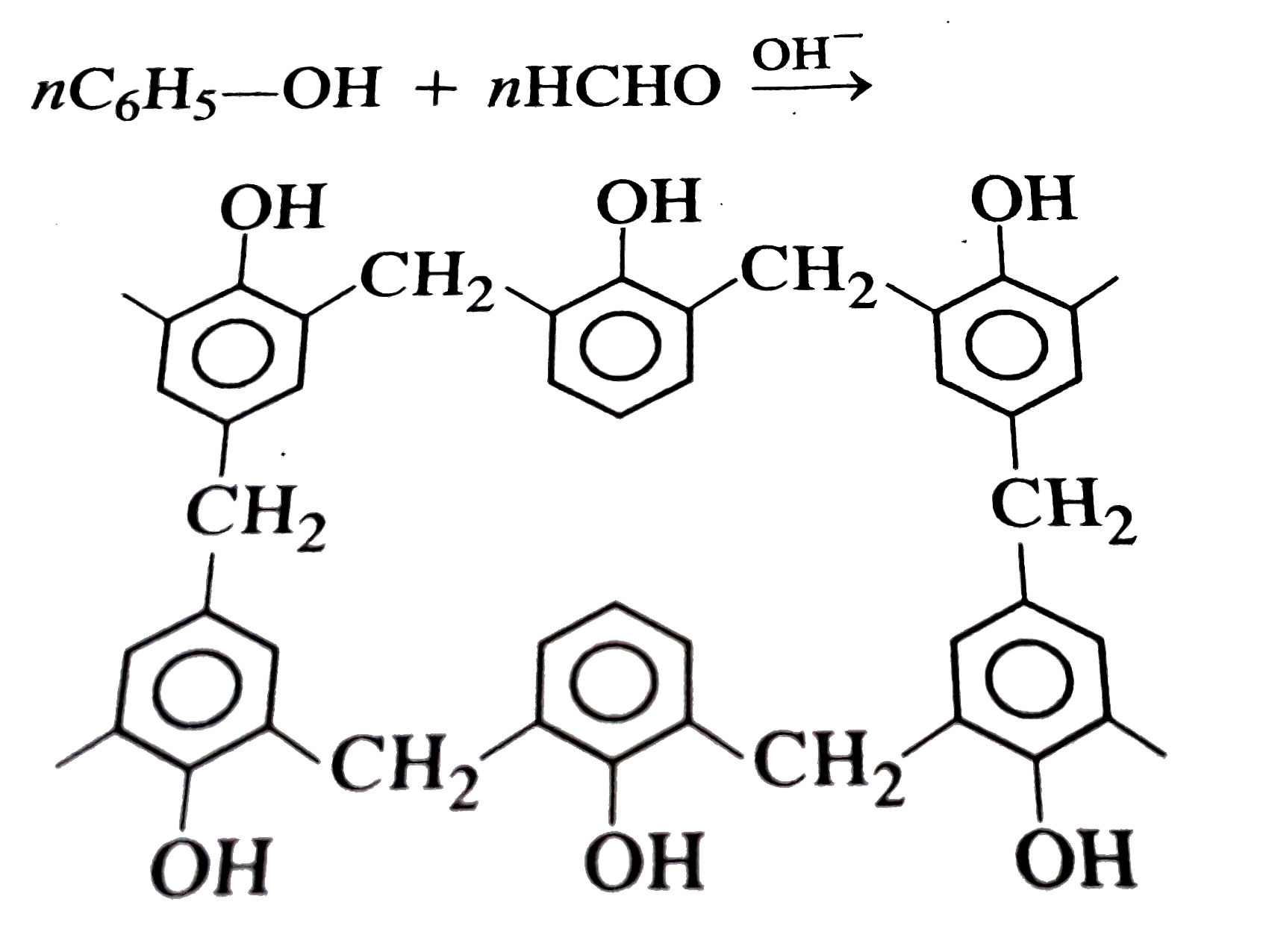
|
|
| 35. |
What isthe angularmomentumof electron in 5^(th) orbitof Bohr.smodel |
|
Answer» `1.0 (h)/(pi)` |
|
| 36. |
The shape of colloidal particles is:- |
|
Answer» SPHERE like |
|
| 37. |
which of the following is /are correct? |
|
Answer» In (HCP) unit cell, the total number of octahedral voids is 4. |
|
| 38. |
When treated with conc. HNO_(3) aluminium is rendered "………….." due to the formation ofprotective layer of its "………….." on the surfaceof the metal. |
|
Answer» |
|
| 39. |
Which law shows density and pressure ? |
|
Answer» Boyle.s |
|
| 40. |
Write the lewis dot structure of CO_(3)^(2-) molecule. |
Answer» SOLUTION :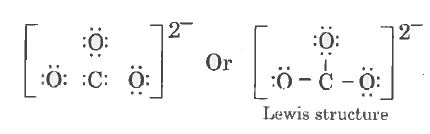
|
|
| 41. |
The system would required ……………… heat to effect a given temperature rise than at constant volume . |
|
Answer» less |
|
| 42. |
Which one of the following relations is correct with respect to first (I) and second (II) ionisation potentials of Na and Mg? |
|
Answer» `I_(NA) GT I_(MG)` |
|
| 44. |
Write the resonance structures of CH_(2)=CH-CHO. Indicate relative stability of the contributing structures order of decreasing stability. |
Answer» Solution :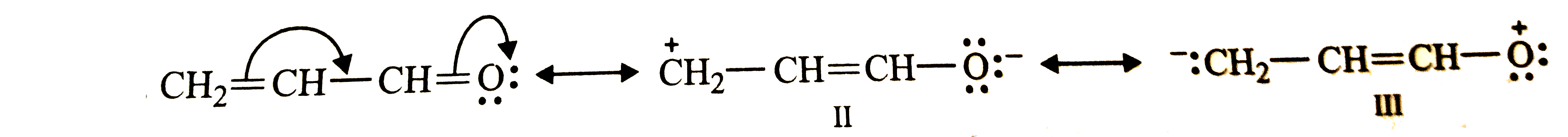 STRUCTURE (I) is most stable since both C and O atom has an octet of ELECTRONS and none of these atoms carries any charge. Structures (II and III) both involve SEPARATION of charge and HENCE both are less stable than structure (I). However, structure (II) is more stable than structure (III) since it carries a -ve charge on the more electronegative O atom and +ve charge on the less electronegative (C) atom while is structure (III), the more electronegative O atom carries the +ve charge while the less electronegative C atom carries the -ve charge. THUS, the decreasing order of stability is : `I gt II gt III` |
|
| 45. |
What is the difference in pH when adding of common ion (conjugate base) in weak acid ? |
| Answer» SOLUTION :INCREASE in PH. | |
| 46. |
Which alcohol is prepared from the following ketones via MPV reduction ? |
|
Answer» |
|
| 47. |
What is a spontaneous process? Write the criteria for spontaneity of a process in terms of DeltaG. |
|
Answer» SOLUTION :A PROCESS which take splace on its own WITHOUT any EXTERNAL AID. `DeltaG=-Ve` |
|
| 48. |
Which compound amongst the following is nitrated with most difficulty ? |
|
Answer» nitrobenzene |
|
| 49. |
Two acids A and B titrated separately each time with 25 mL of N Na_(2)CO_(3) solution and require 10 mL and 40 mL respectively for complete neutralisation. What volume of A and B would you mix to produce one litre of normal acid solution ? |
|
Answer» |
|