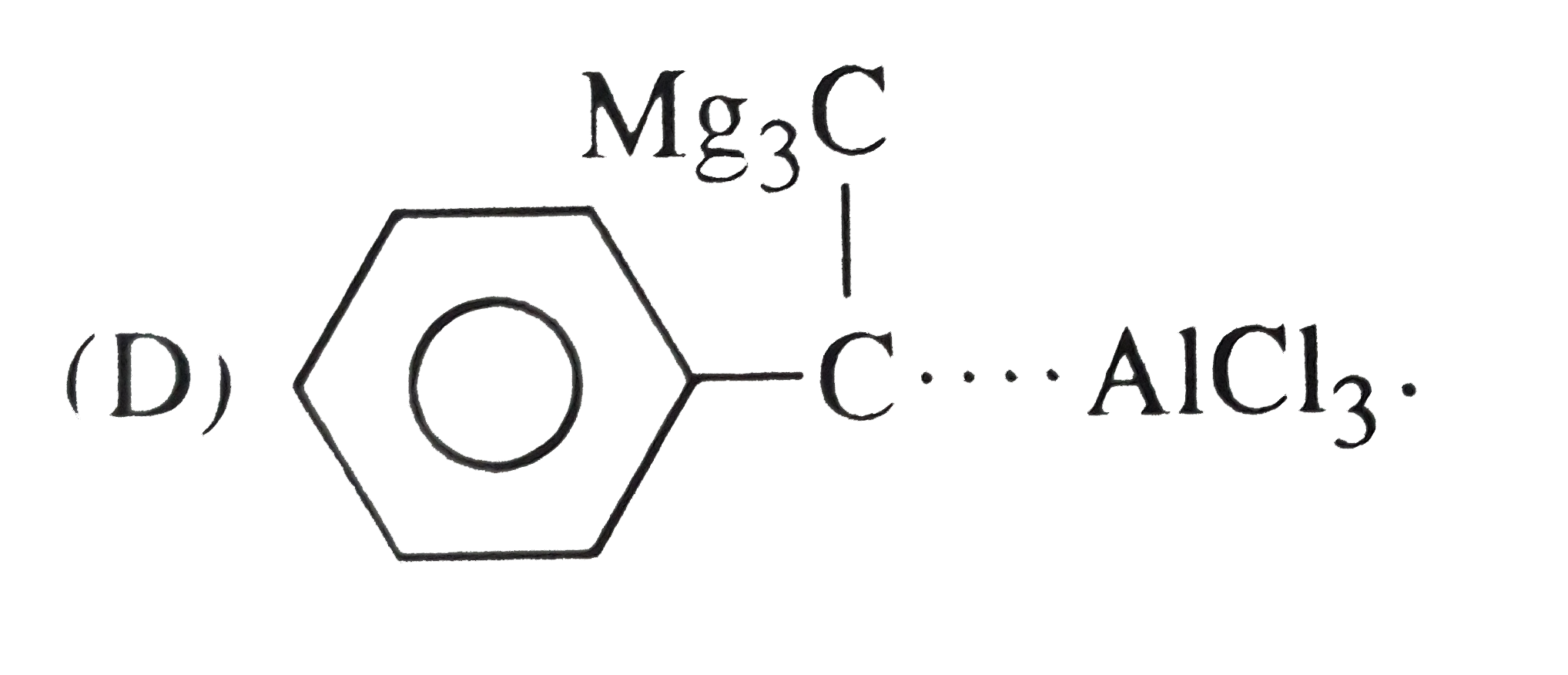Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Reagent (A) and (B) in above reaction are: |
|
Answer» `A = RCO_(3)H, B=H_(2)O_(2)` |
|
| 2. |
Read the following statements i) Crystal of NaOH are deliquescent ii) The by product formed in Solvay process is CaCl_(2) iii)NaHCO_(2) is a mild antiseptic for skin infection |
|
Answer» i and III are CORRECT |
|
| 3. |
Read the following passage and answer the questions that follow.Electron-precise compounds have the required number of electrons to write their conventional Lewis structures. All elements of group 14 form such compounds which are tetrahedral in geometry. Electron - rich hydrides have excess of electrons which are present as lone pairs. Elements of groups 15-17 form such compounds. Metallic hydrides are formed by many d-block and f-block elements. These hydrides conduct heat and electricity though not as efficiently as their parent metals do. They are non-stoichiometric. Incorrect statement among the following is |
|
Answer» The electrical conductivity of CRH is less than that of Chromium. |
|
| 4. |
Read the following passage and answer the questions that follow.Electron-precise compounds have the required number of electrons to write their conventional Lewis structures. All elements of group 14 form such compounds which are tetrahedral in geometry. Electron - rich hydrides have excess of electrons which are present as lone pairs. Elements of groups 15-17 form such compounds. Metallic hydrides are formed by many d-block and f-block elements. These hydrides conduct heat and electricity though not as efficiently as their parent metals do. They are non-stoichiometric. Electron precise compound among the following is |
|
Answer» `NH_3` |
|
| 5. |
Read the following passage and answer the questions that follow.Electron-precise compounds have the required number of electrons to write their conventional Lewis structures. All elements of group 14 form such compounds which are tetrahedral in geometry. Electron - rich hydrides have excess of electrons which are present as lone pairs. Elements of groups 15-17 form such compounds. Metallic hydrides are formed by many d-block and f-block elements. These hydrides conduct heat and electricity though not as efficiently as their parent metals do. They are non-stoichiometric. A non-stoichiometric hydride is formed by |
|
Answer» Na |
|
| 6. |
Read the following passage and answer the questions followed by is silicates are a group of minerals which have the general formula SiO_(2), the most common of which is quartz. Quartz is a frame work silicate with SiO_(4), tetrahydra arranged in spirals. The spirals can turn in clockwise or anticlock wise direction a feature that results in there being two mirror images opticlaly active varities of quartz. The silicate anion in the mineral kinoite is a chain of three SiO_(4), tetrahedra that share coners with adjacent tetrahedra. The mineral also contain Ca^(2+),Cu^(2+) ions and water molecules in 1 : 1 : 1 ratio. The mineral is represented as |
|
Answer» `CaCuSi_(3)O_(10). H_(2)O` |
|
| 7. |
Read the following message passage and answer the followed by is silicates are a group of minerals which havegeneral formula SiO_(2) the most common of which is quartz. Quartz is a frame work silicate with SiO_(4) tetrahydra arranged in spirals. The spirals can turn in clockwise or anticlork wise direction a feature that result in there being two mirror images optically active varities of quartz The following represents various silicate anions . Their formulae are respectively |
|
Answer» `SiO_(3)^(2-), SiO_(7)^(2-)` |
|
| 8. |
Read the following message passage and answer the followed by is silicates are a group of minerals which havegeneral formula SiO_(2) the most common of which is quartz. Quartz is a frame work silicate with SiO_(4) tetrahydra arranged in spirals. The spirals can turn in clockwise or anticlork wise direction a feature that result in there being two mirror images optically active varities of quartz Si_(3)O_(10)^(8-)(having three tetrahedral units ) is representedas |
|
Answer»
|
|
| 9. |
Read of the following passage giving the role of CO_(2) buffer in controlling pH of blood. The importance of pH maintance in Blood. Maintenance of the pH in blood and intracellur fluids is absolutely crucial to the processes the occur in living ogranisms. This is primarily because the functioning of enzymes-catalysts for these processe- is sharply pH dependent. The normal pH value of blood plasma i 7.4. Severe illness or dear can result from subtained variations fo a few tenths of pH unit. Among the factors that lead to a condition of acidosis, in which there is decreas in the pH of blood are heart failure, kidney failure, diabetis mellitus, persistent diarroheoa or a long term high protein diet,temporary condition acidosis may result from proglonged, intensive in ph of blood, may several factors are involved in the control of the pH of blood. A particularly important one is the ratio of dissolved,OH_(3)^(-) to H_(2)CO_(3).CO_(2)(g) is moderarately soluble in water and in aqeous solution reactns only a limilated extent to product H_(2)CrO_(3) CO_(2) + H_(2)O hArr H_(2)CO_(3(aq)) H_(2)CO_(3) + H_(2)O hArr HCO_(3)^(-) + H_(3)O^(+) , pK_(al) = 6.11 HCO_(3)^(-) + H_(2)O hArr CO_(3)^(2-) + H_(3)O^(+), pK_(a2) = 10.25 In the H_(2)CO_(3), HCO_(3)^(-) buffer system we deal only with the first ionisation step (K_(al)) H_(2)CO_(3) is a wear acid and HCO_(3)^(-) is the conjugate base (salt). CO_(2) enters the blood from issues as the by product metabolic reaction. In lungs, CO_(2)(g) is exchanged for O_(2)(g), which is transported throughout the body the blood. Following reaction occurs in the body If CO_(2) escape from the system |
|
Answer» pH will decreases |
|
| 10. |
Read of the following passage giving the role of CO_(2) buffer in controlling pH of blood. The importance of pH maintance in Blood. Maintenance of the pH in blood and intracellur fluids is absolutely crucial to the processes the occur in living ogranisms. This is primarily because the functioning of enzymes-catalysts for these processe- is sharply pH dependent. The normal pH value of blood plasma i 7.4. Severe illness or dear can result from subtained variations fo a few tenths of pH unit. Among the factors that lead to a condition of acidosis, in which there is decreas in the pH of blood are heart failure, kidney failure, diabetis mellitus, persistent diarroheoa or a long term high protein diet,temporary condition acidosis may result from proglonged, intensive in ph of blood, may several factors are involved in the control of the pH of blood. A particularly important one is the ratio of dissolved,OH_(3)^(-) to H_(2)CO_(3).CO_(2)(g) is moderarately soluble in water and in aqeous solution reactns only a limilated extent to product H_(2)CrO_(3) CO_(2) + H_(2)O hArr H_(2)CO_(3(aq)) H_(2)CO_(3) + H_(2)O hArr HCO_(3)^(-) + H_(3)O^(+) , pK_(al) = 6.11 HCO_(3)^(-) + H_(2)O hArr CO_(3)^(2-) + H_(3)O^(+), pK_(a2) = 10.25 In the H_(2)CO_(3), HCO_(3)^(-) buffer system we deal only with the first ionisation step (K_(al)) H_(2)CO_(3) is a wear acid and HCO_(3)^(-) is the conjugate base (salt). CO_(2) enters the blood from issues as the by product metabolic reaction. In lungs, CO_(2)(g) is exchanged for O_(2)(g), which is transported throughout the body the blood. Important diagnostic analysis in the blood is |
|
Answer»
|
|
| 11. |
Read of the following passage giving the role of CO_(2) buffer in controlling pH of blood. The importance of pH maintance in Blood. Maintenance of the pH in blood and intracellur fluids is absolutely crucial to the processes the occur in living ogranisms. This is primarily because the functioning of enzymes-catalysts for these processe- is sharply pH dependent. The normal pH value of blood plasma i 7.4. Severe illness or dear can result from subtained variations fo a few tenths of pH unit. Among the factors that lead to a condition of acidosis, in which there is decreas in the pH of blood are heart failure, kidney failure, diabetis mellitus, persistent diarroheoa or a long term high protein diet,temporary condition acidosis may result from proglonged, intensive in ph of blood, may several factors are involved in the control of the pH of blood. A particularly important one is the ratio of dissolved,OH_(3)^(-) to H_(2)CO_(3).CO_(2)(g) is moderarately soluble in water and in aqeous solution reactns only a limilated extent to product H_(2)CrO_(3) CO_(2) + H_(2)O hArr H_(2)CO_(3(aq)) H_(2)CO_(3) + H_(2)O hArr HCO_(3)^(-) + H_(3)O^(+) , pK_(al) = 6.11 HCO_(3)^(-) + H_(2)O hArr CO_(3)^(2-) + H_(3)O^(+), pK_(a2) = 10.25 In the H_(2)CO_(3), HCO_(3)^(-) buffer system we deal only with the first ionisation step (K_(al)) H_(2)CO_(3) is a wear acid and HCO_(3)^(-) is the conjugate base (salt). CO_(2) enters the blood from issues as the by product metabolic reaction. In lungs, CO_(2)(g) is exchanged for O_(2)(g), which is transported throughout the body the blood. The pH of blood system is maintained by a proper balance of H_(2)CO_(3) and NaHCO_(2) concentration. The volume of 5M NaHCO_(3) solution should be mixed with a 10 m sample of blood which is 2M in H_(2)CO_(3) order to maitain its pH |
| Answer» Solution :N//A | |
| 12. |
Reactivity towards chemical reactions is very low for elements of group-18 because |
|
Answer» they have stable electronic configuration in outermost SHELL. |
|
| 13. |
Reactivity of H-atoms bonded to different types of C-atoms in alkanes in the order |
|
Answer» `3^(@) gt 1^(@) gt 2^(@)` |
|
| 14. |
Reactivity of borazole is greater than that of benzene because |
|
Answer» BORAZOLE is non-polar COMPOUND `:.`Borazole is reactive substance |
|
| 15. |
Reactivity of alkene |
Answer» Solution :In alkene, carbon-carbon has weak `pi`-bond.  Electrons of `pi`-bonds shows weak dynamic electrons. This type of `pi`-electrons source is present in alkene. So such type of REACTANT and COMPOUNDS attack on `pi`-bond and alkene and break `pi`-bond easily and FORMS compounds having single bond. So alkane compounds easily gives addition reaction. `pi`-bond because alkene is more reactive and less stable than alkanes. So, STRENGTH of C-C in alkane (bond ethalpy `384 kJ mol^(-1)`), is less then strength of C-C in alkene (bond enthalpy 681 kJ/mol). |
|
| 16. |
Reactions that have standardfree energy change less than zero always have equilibrium constant to be |
|
Answer» unity |
|
| 17. |
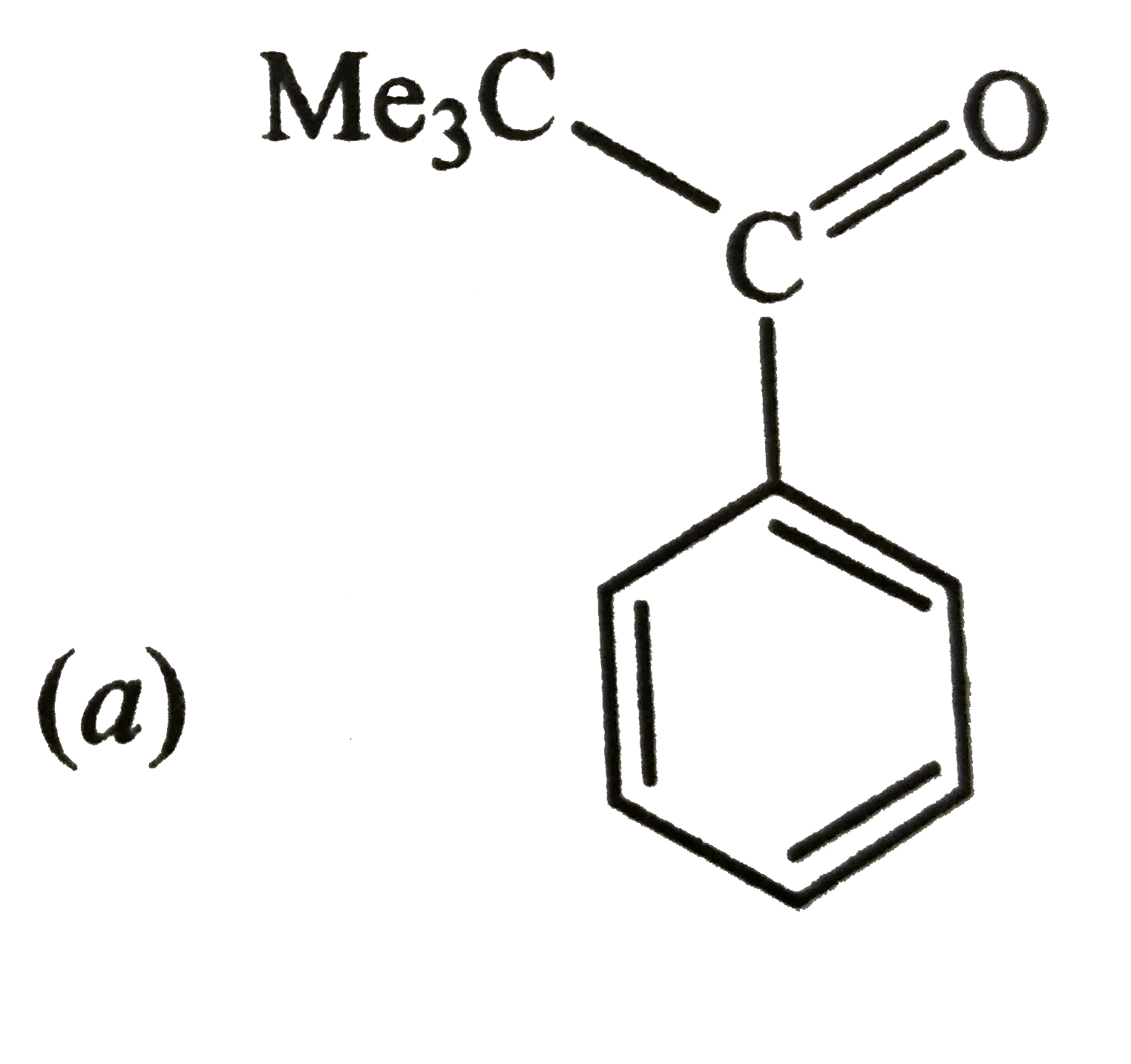
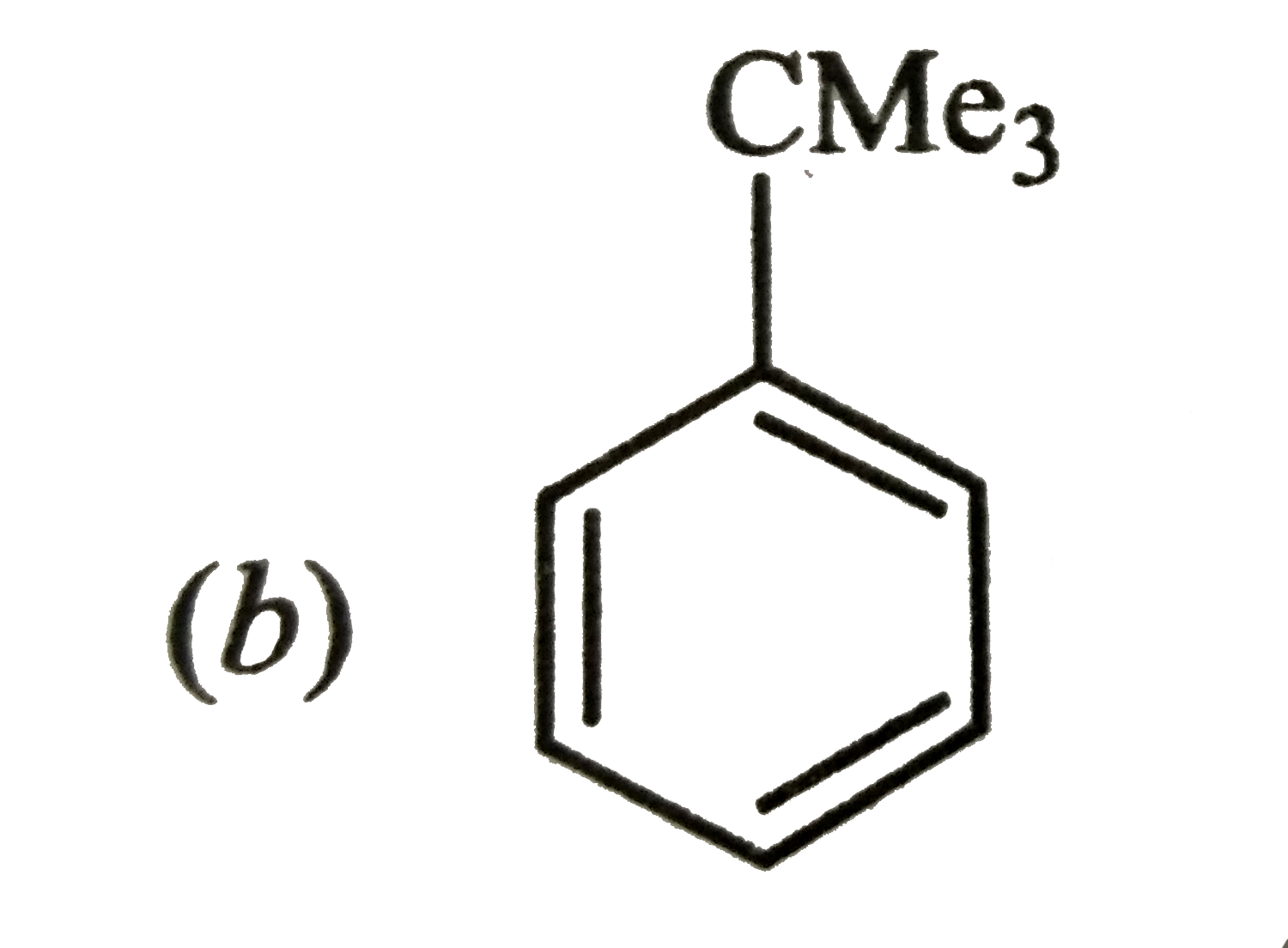
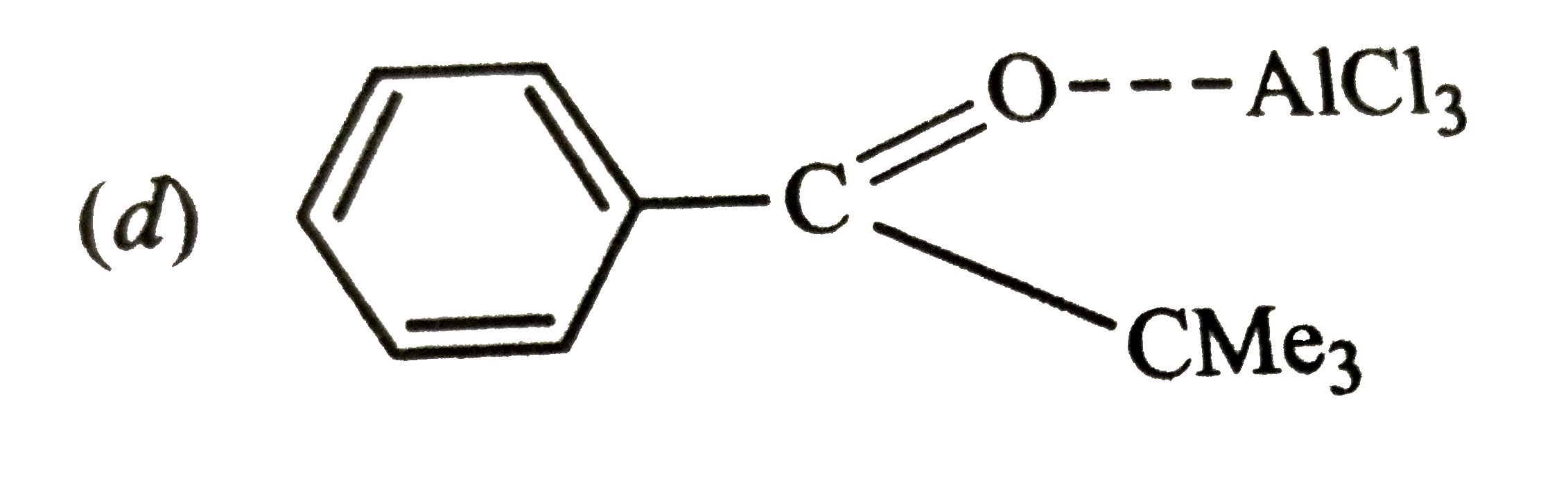
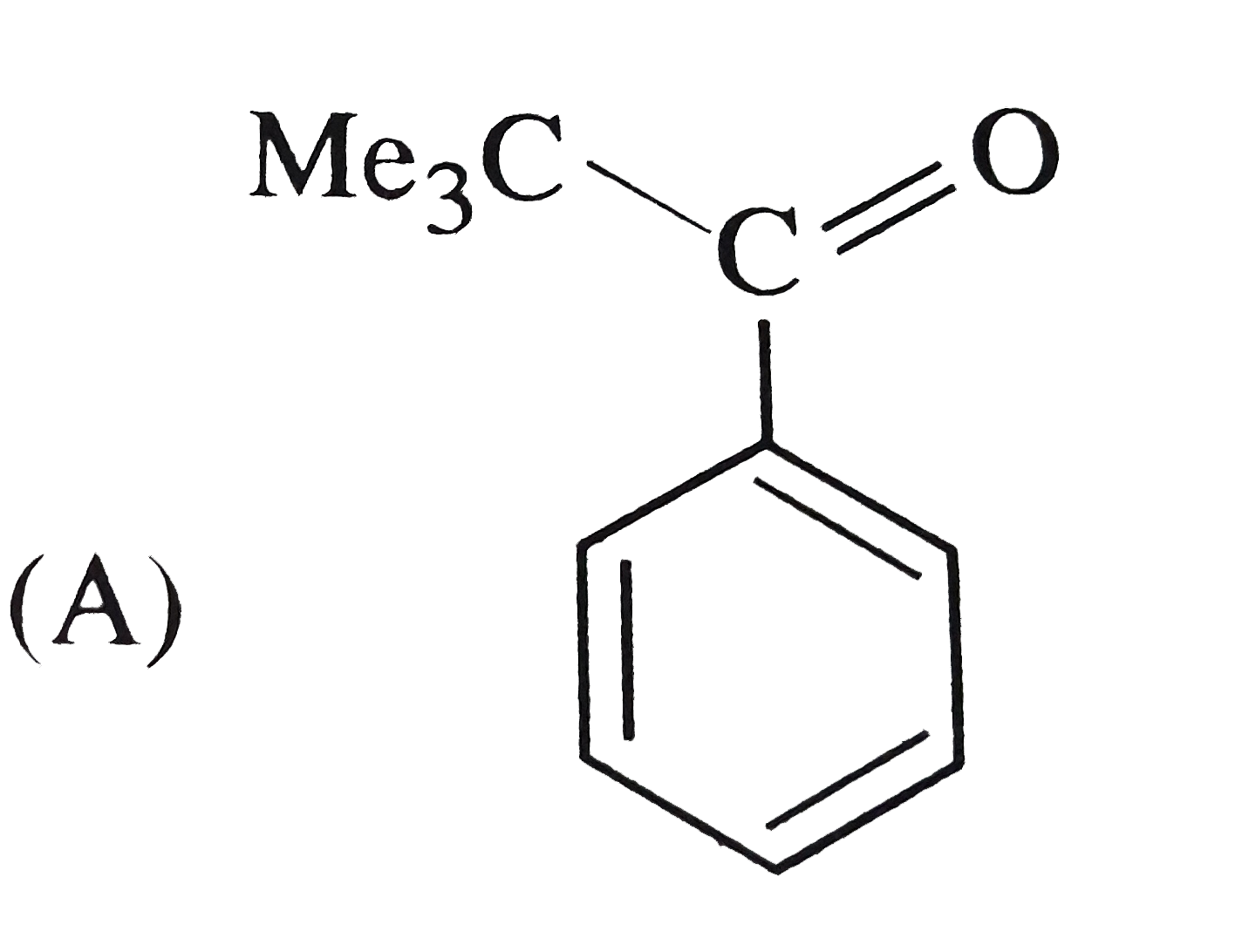
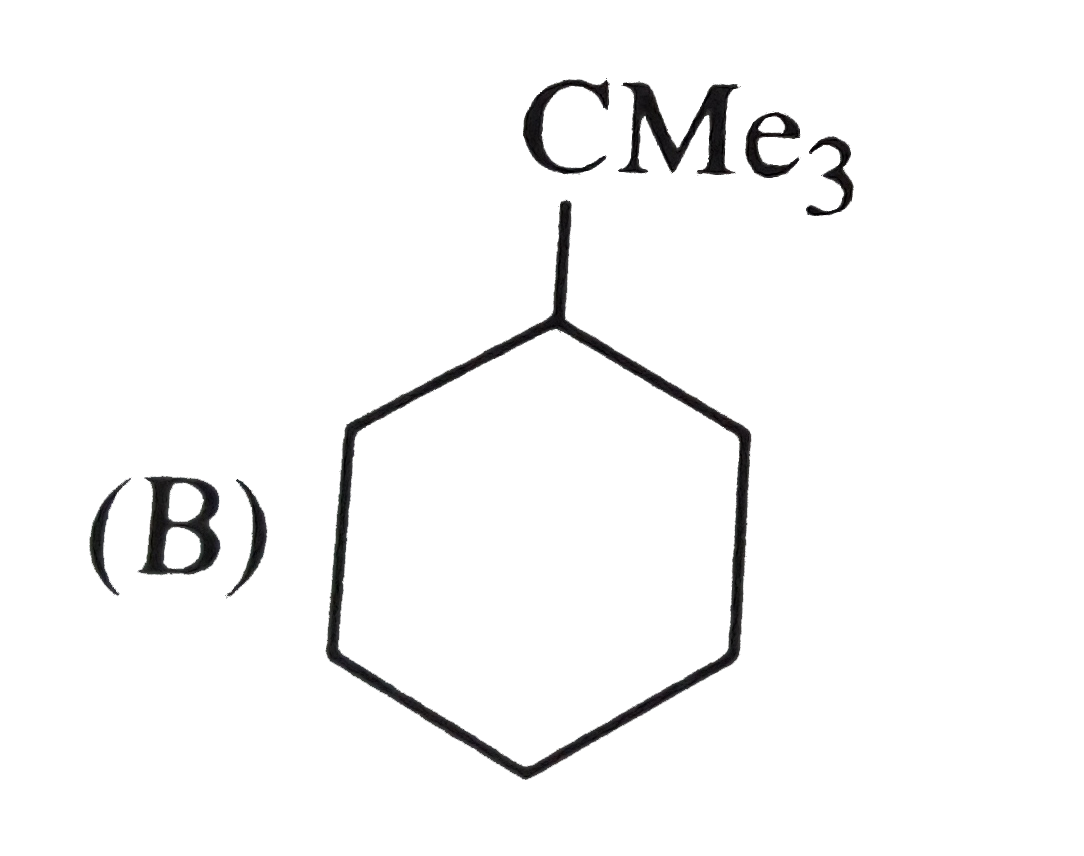
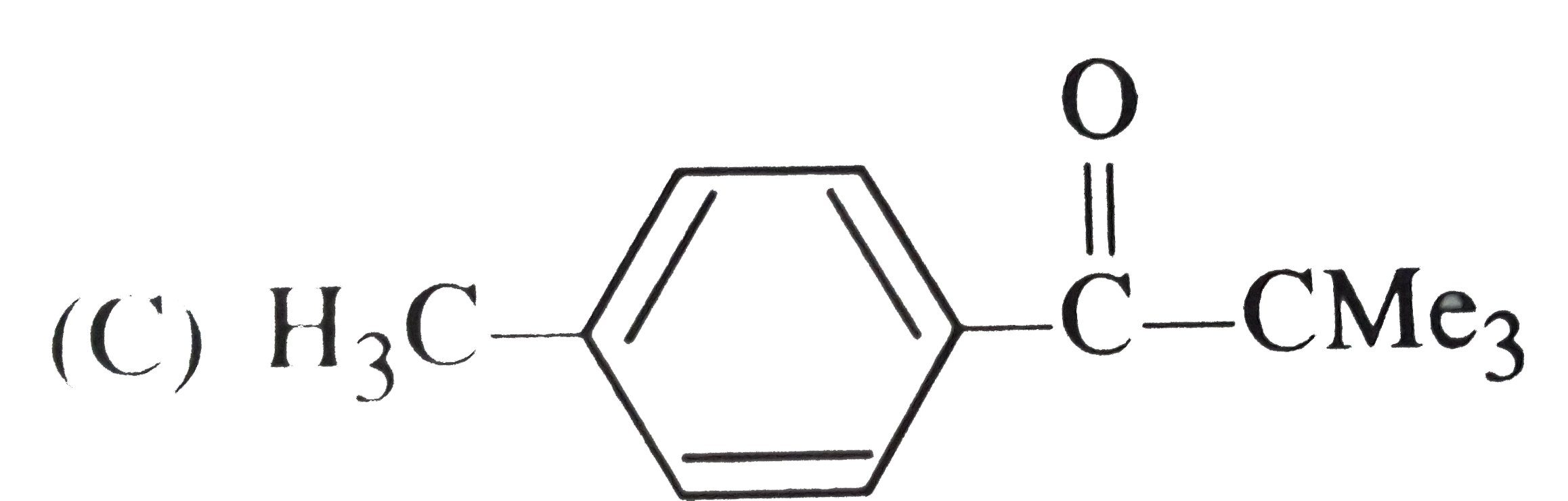
Reactions of benzene with Me_3C COCl in the presence of anhydrous AlCl_3 gives |
|
Answer»
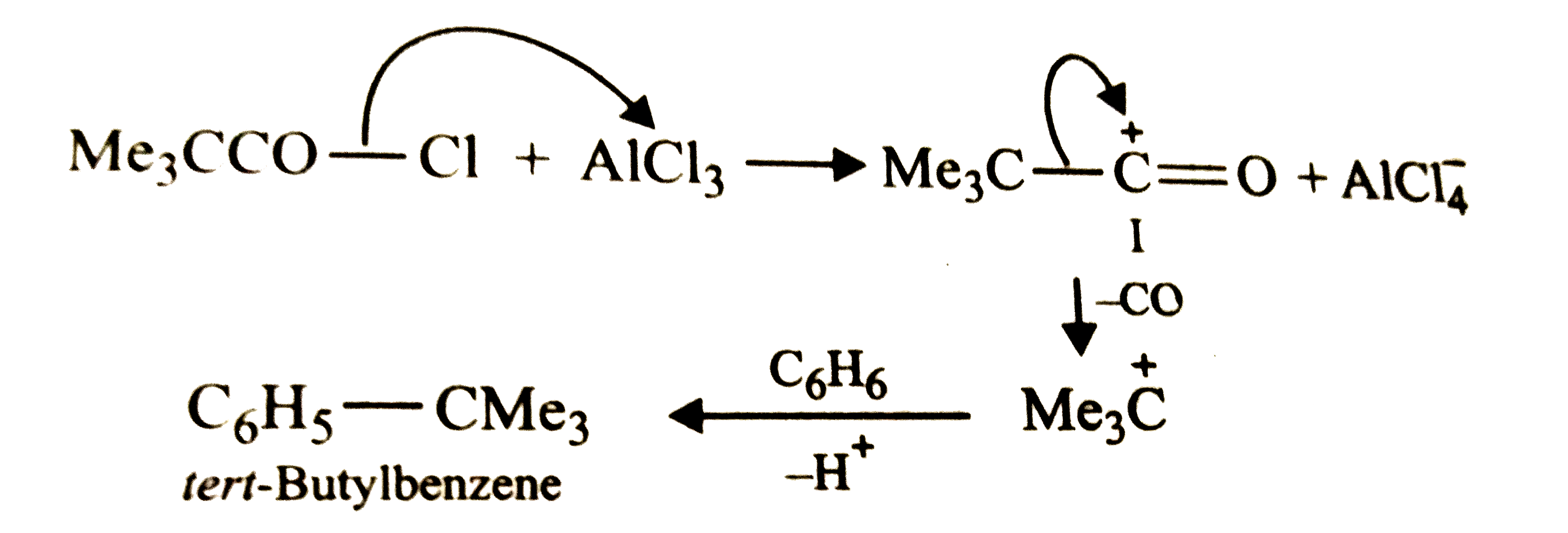 The initially formed acylium ion (I) readily ELIMINATES a stable molecule of CO to FORM tert-butyl carbocation which then attacks the BENZENE RING to form tert-butylbenzene. |
|
| 18. |
Reactions involving gold have been of particular interest to alchemists. Consider the following reactions, Au(OH)_(3) + 4 HCl ""^(3)//""_(4)®HAuCl_(4) + 3 H_(2)O, DH = - 28 kcal Au(OH)_(3) + 4 HBr""^(3)//""_(4)®HAuBr_(4) + 3 H_(2)O, DH = -36.8 kcal In an experiment there was an absorption of 0.44 kcal when one mole of HAuBr_(4) was mixed with 4 moles of HCl. Then the fraction of HAuBr_(4) converted into HAuCl_(4) (percentage conversion) |
|
Answer» 0.05 Reaction is `= -28 - (-36.8) = 8.8` K.cal. So one MOLE of `HAuBr_(4)` can be converted in to `HAuCl_(4)` by absorption of 8.8K. Cal So Absorption of 0.44 K.Cal can give `(1)/(8.8) xx 0.44` moles of `HAuCl_(4)`. So no. of mole of `HAuCl_(4)` obtained = 0.05 So % `=(0.05)/(1) xx 100= 5%` |
|
| 19. |
Reactions involving heterolytic fission are said to be produced via |
|
Answer» IONIC mechanism |
|
| 20. |
Reaction that is not related to the preparation of sodium carbonate by Solvay process is |
|
Answer» `2NH_(3)+H_(2)O+_CO_(2)rarr(NH_(4))_(2)CO_(3)` |
|
| 21. |
Reaction stoichiometry, kinetics and thermodynamics ltbgt Nitrosyl chloride (NOCl), is a yellow gas that is most commonly encountered as a decomposition product of aqua regia. It is toxic and irritating to the lungs. On heating NOCl decomposes as 2NOClrarr2NO+Cl_(2). The enthalpy change (DeltaH) for the formation of 1 mole of Cl_(2) by the decomposition of NOCl is 75.3KJ between 100.K "to" 600K. The standard entropies (S^(@)_(298K)) of different species are as given below: {:("Substance",NOCl,NO,Cl_(2)),(S_(298K)^(@),264,211,223):} A gaseous mixture of NO,Cl_(2) and NOCl with partial pressures (in bar) 1.5,0.88 and 0.065 respectively was taken at 475K. Deduce whether the net reaction will lead to increase in NOCl concentration. |
|
Answer» |
|
| 22. |
Reaction of with RMgX followed by hydrolysis will lead to produce |
|
Answer» `RCHOHR` 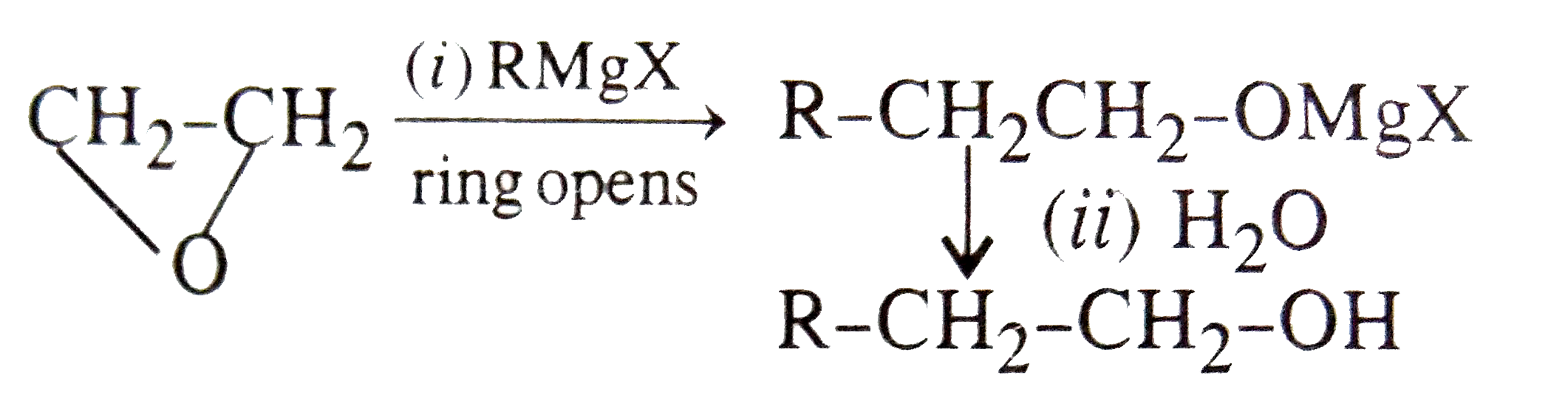
|
|
| 23. |
Reaction of R-CONH_(2) with Br_(2)//KOH gives R-NH_(2) as the main product. The intermediate involved in this reaction are |
|
Answer» `R - OVERSET(O)overset(||)(C) - NHBR`<BR>`R - overset(I)overset(uarr)(NH) - Br` |
|
| 24. |
Reaction of PCl_(3) and PhMgBr would give |
|
Answer» bromobenzene |
|
| 25. |
Reaction of hydrogen bromide with propene in the absence of peroxide is a/an |
|
Answer» FREE RADICAL reaction |
|
| 26. |
Reaction of ethyl chloride with sodium metal leads to the formation of …………………. . |
| Answer» SOLUTION :n-Butane | |
| 27. |
Reaction of ethyl formate with excess of CH_(3)MgI followed by hydrolysis gives |
|
Answer» `N-`PROPYL alcohol 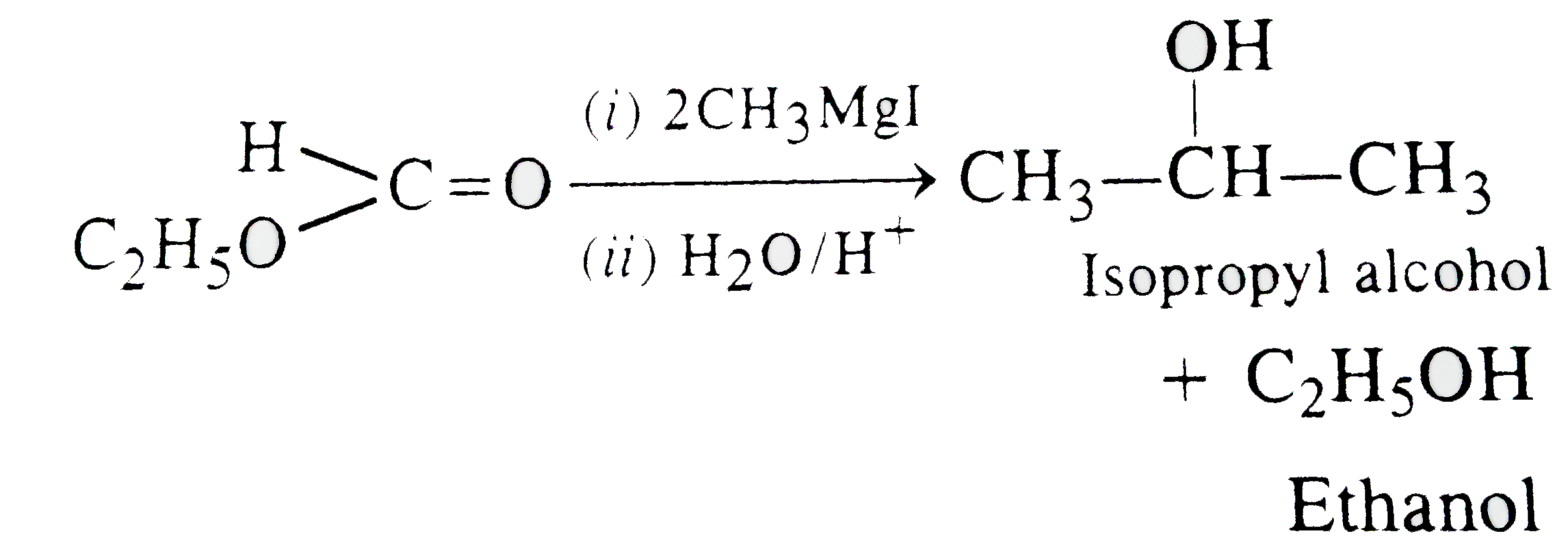
|
|
| 28. |
Reaction of ethyl acetate with excess of CH_(3)MgI followed by hydrolysis gives : |
|
Answer» `N-`Propyl alcohol |
|
| 29. |
Reaction of diborane with ammonia gives initially |
|
Answer» `B_(2)H_(6).NH_(3)` |
|
| 30. |
Reaction of CH_(3)CHO and aluminium ethoxide is called |
|
Answer» Tischenko REACTION |
|
| 31. |
Reaction of Br_(2) with Na_(2)CO_(3) in aqueous solution gives sodiu bromide and sodium bromate with evolution of CO_(2) gas The number of sodium bromide molecules involved in the balanced chemical equation is |
|
Answer» Thus 5 moles of NaBr are formed |
|
| 32. |
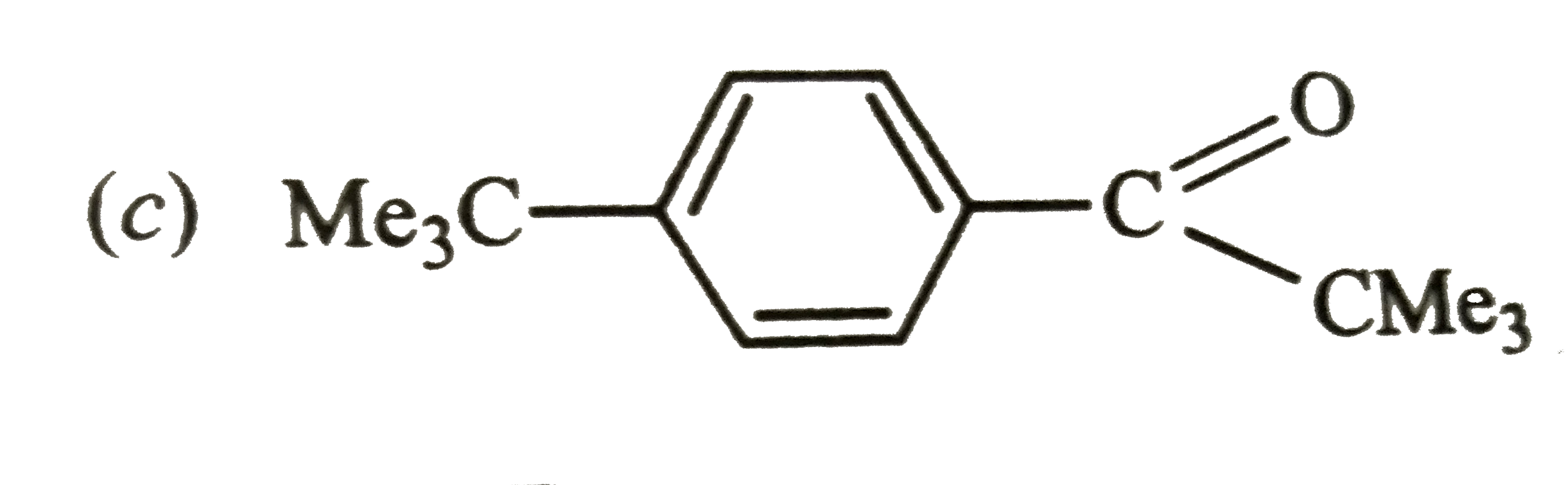
Reaction of Benzene with Me_(3)C COCl in the presence of anhydrous AlCl_(3) gives |
|
Answer»
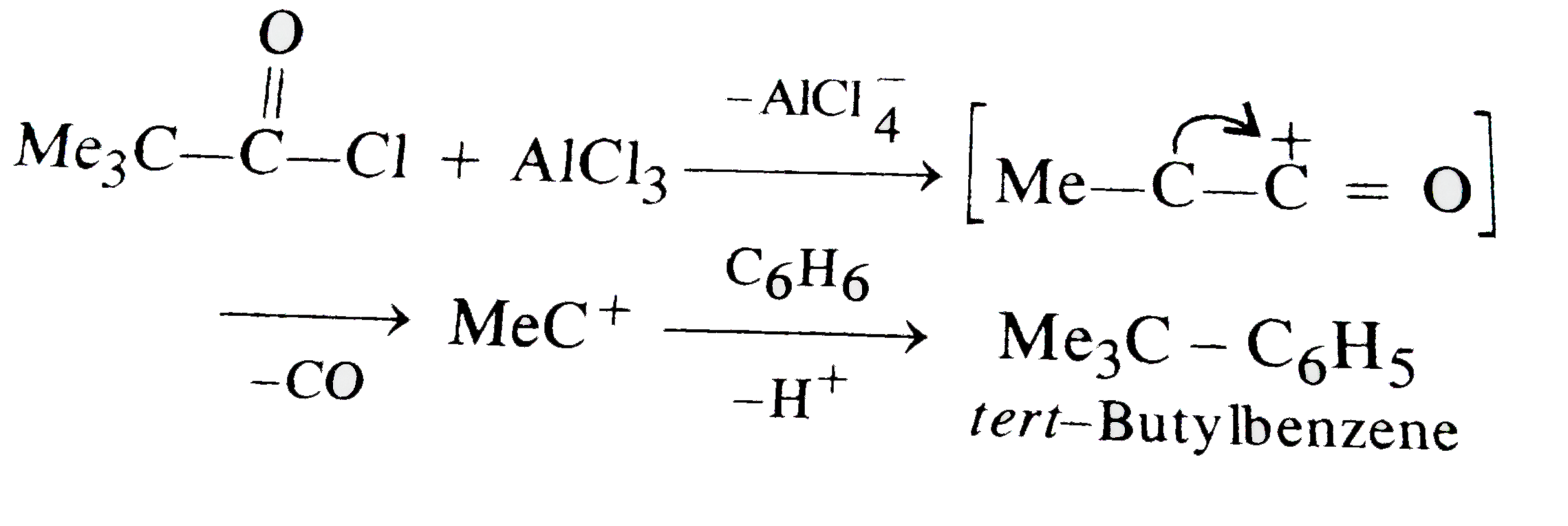
|
|
| 33. |
Reaction of benzene with isobutylchloride (CH_3CH(CH_3)CH_2Cl) in the presence of anhydrous AlCl_3 yields. |
|
Answer» tert-butylbenzene |
|
| 34. |
Reaction of alkanes with halogen is explosive in case of |
|
Answer» `F_2` |
|
| 35. |
Reaction of alkene with ___________ are explosive in nature |
|
Answer» `Cl_2 ` |
|
| 36. |
Reaction N_(2(g))+ 2O_(2(g)) hArr 2NO_(2(g))equilibrium constant is 100. Find equilibrium constant for following reaction. (1) 2NO_(2(g)) hArr N_(2(g)) + 2O_(2(g)) (2) NO_(2(g)) hArr 1/2N_(2(g)) + O_(2(g)) |
|
Answer» SOLUTION :(1)`K_1=([N_2][O_2]^2)/[NO_2]^2=1/100=1xx10^(-2)` (2)`K_2=([N_2]^(1/2)[O_2])/([NO_2])=(K_1)^(1/2)`=0.1 |
|
| 37. |
Reaction N_(2(g))+ 3H_(2(g)) hArr 2NH_(3(g)) , At 400 K is K_p=41 , So, calculate K_p of following reactions at 400 K. (a)2N_(2(g)) + 6H_(2(g)) hArr 4NH_(3(g)) (b)2NH_(3(g)) hArr N_(2(g))+ 3H_(2(g)) (c)1/2N_(2(g)) + 3/2H_(2(g)) hArr NH_(3(g)) |
| Answer» SOLUTION :(a)`1.67xx10^(-3)`, (B) 0.024 , (C) 6.4 | |
| 38. |
Reaction intermdiates are short lived species and are highly reactive. They are formed by heterolytic and homolytic bond fission. There are varioustypes of reaction intermediates in which the most important are carbocation , carbanion and free radical. Carbocation is an organic species in which carbon have positive charge and six electrons in its outermost shell. The stability of carbocation can be increased by positive inductive effect, hyperconjugation and delocalisation. If alpha-atom with respect to carbocation has one or more lone pair of electron then lone pair of electron strongly stabilises the carbocation due to octet completion. Species in which carbon have negative charge is called carbanion. Carbanion carries three bond pairs and one lone pair. The stability of carbanioncan be increased by negative inductive effect, negative mesomeric effect and delocalisation. Free radical is a species which have seven electrons in its outermost shell. The stability of free radical can be increased by hyperconjugation and delocalisation. The stability order of following carbocations is |
|
Answer» `I GT II gt III` |
|
| 39. |
Reaction intermdiates are short lived species and are highly reactive. They are formed by heterolytic and homolytic bond fission. There are varioustypes of reaction intermediates in which the most important are carbocation , carbanion and free radical. Carbocation is an organic species in which carbon have positive charge and six electrons in its outermost shell. The stability of carbocation can be increased by positive inductive effect, hyperconjugation and delocalisation. If alpha-atom with respect to carbocation has one or more lone pair of electron then lone pair of electron strongly stabilises the carbocation due to octet completion. Species in which carbon have negative charge is called carbanion. Carbanion carries three bond pairs and one lone pair. The stability of carbanioncan be increased by negative inductive effect, negative mesomeric effect and delocalisation. Free radical is a species which have seven electrons in its outermost shell. The stability of free radical can be increased by hyperconjugation and delocalisation. Which of the following is the most stable carbanion intermediate ? |
|
Answer»
|
|
| 40. |
Reaction intermdiates are short lived species and are highly reactive. They are formed by heterolytic and homolytic bond fission. There are varioustypes of reaction intermediates in which the most important are carbocation , carbanion and free radical. Carbocation is an organic species in which carbon have positive charge and six electrons in its outermost shell. The stability of carbocation can be increased by positive inductive effect, hyperconjugation and delocalisation. If alpha-atom with respect to carbocation has one or more lone pair of electron then lone pair of electron strongly stabilises the carbocation due to octet completion. Species in which carbon have negative charge is called carbanion. Carbanion carries three bond pairs and one lone pair. The stability of carbanioncan be increased by negative inductive effect, negative mesomeric effect and delocalisation. Free radical is a species which have seven electrons in its outermost shell. The stability of free radical can be increased by hyperconjugation and delocalisation. The stability order of following free radicals is: C_(6)H_(5)underset(I)CH_(2)overset(*)CH_(2)""CH_(3)underset(II)CH_(2)overset(*)CH_(2)""underset(""III)(C_(6)H_(5))overset(*)CH_(2)""underset(IV)(CH_(3)^(*) |
|
Answer» `I GT II gt III gt IV` |
|
| 41. |
Reaction (I) and raection (II) are respectively, |
|
Answer» `S_(N)1, S_(N)1` |
|
| 42. |
Reaction HO_3 SOOSO_3 H_((aq)) underset(H_2O)overset"Hydrolysis"to 2H_2SO_4 + x. What is product x ? |
| Answer» SOLUTION :The PRODUCT X will be `H_2O_2` . | |
| 43. |
Reaction H_(2(g))+ I_(2(g)) hArr 2HI_((g))0.4 mol H_2 and I_2 taken in 2L vessel if 0.5 mol HI form at equilibrium than calculate K_p |
| Answer» SOLUTION :`K_p`=11.11 | |
| 44. |
Reaction: CH_(3)-underset(underset(Cl)(|))(CH)-CH_(3) overset("Alcohol, KOH")rarr x underset(y)overset(Cl_(2))rarr Identify x and y respectively |
|
Answer» PROPENE and 2, 2-dichloropropane |
|
| 45. |
Reaction between red phosphorus and liquidbromineis an exothermic reaction represented as follows: 2P (s) +3Br_(2)(l) rarr 2P Br_(3)(g),DeltaH^(@) = - 243kJ mol^(-1) What will be the enthalpy change when 2.63g of phosphorusreacts accordingto the above reaction. Take atomic mass of phosphorus as 31.0 . |
|
Answer» |
|
| 46. |
Reaction between N_2 and O_(2-) takes place as follows : 2N_(2(g)) + O_(2(g)) hArr 2N_2O_((g))If a mixture of 0.482 mol N_2 and 0.933 mol of O_2 is placed in a 10 L reaction vessel and allowed to form N2O at a temperature for which K_c = 2.0 xx 10^(-37), determine the composition of equilibrium mixture. |
|
Answer» SOLUTION :`{:("Reaction:",2N_(2(g)) + , O_(2(g)) hArr , 2N_2O_((g))),("Initial MOL:",0.482,0.933,0),("Change in reaction:",-2x,-X,+2x),("Mol at EQUILIBRIUM:",(0.482-2x),0.933-x,2x),("Equilibrium mol L"^(-1), "0.482-2x"/10,"0.933-x"/10,(2x)/10):}` `[N_2]=(0.482-2x)/10 = 0.482/10 "mol L"^(-1) = 0.0482 "mol L"^(-1)` `[O_2]= (0.933-x)/10 = 0.933/10 "mol L"^(-1) = 0.0933 "mol L"^(-1)` `[N_2O]=(2x)/10 = 0.2x "mol L"^(-1)` `K_c=[N_2O]^2/([N_2]^2[O_2])=(0.2x)^2/((0.0482)^2(0.0933))` `therefore 2.0xx10^(-37)=(0.04x^2)/((0.0482)^2(0.0933))` `therefore 0.04x^2=2.0xx10^(-38)xx(0.0482)^2(0.0933)` `therefore x=sqrt((20xx10^(-38)xx(0.0482)^2(0.0933))/(0.04))` `therefore x=sqrt(0.1084xx10^(-38))= 0.3292xx10^(-19)` Now, `[N_2O]=2x=2(0.3292xx10^(-19))` `=0.6584xx10^(-19)=6.584xx10^(-20) "mol L"^(-1)` `[N_2]=0.482-2x=0.482-0.6584xx10^(-19) approx 0.482` `[O_2]= 0.0933-x=0.0933-0.6584xx10^(-19) approx` 0.0933 |
|
| 47. |
Reaction between nitrogen and oxygen takes place as follows : 2 N_(2) (g) + O_(2) (g) hArr 2 N_(2) O (g) If a mixture of 0*482 mol of N_(2) and 0*933 mol of O_(2) is placed in a reaction vessel of volume 10 L and allowed to form N_(2)Oat a temperature for which K_(c) = 2*0 xx 10^(-37) . Determine the composition of the equilibrium mixture. |
|
Answer» Solution :` {:(,2 N_(2) (g) ,+,O_(2)(g),hArr,2 N_(2)O(g)),("Intial",0*482 "mol",,0*933"mol",,),("At eqm.",0*482 "mol",,0.933-x//2,,x),("Molar conc." ,(0*482-x)/10,,(0*933-x//2)/10,,x/10):}`, As K ` = 2*0xx 10 ^(-37)` is very small , this means that the amount of `N_(2) and O_(2) ` reacted (x)is very very small . Hence , at equilibrium , we have `[N_(2)] = 0* 0482 "mol"L^(-1), [O_(2)] = 0* 0933 "mol" L^(-1) , [N_(2)O]= 0*1x ` ` :. K_(c) = (0*1 x)^(2) /((0*0482)^(2) (0*0933)) = 2* 0 xx 10^(-37) ` (GIVEN) On solving, this GIVES `x = 6*6XX 10^(20)` `:. [N_(2)O ] = 0*1x 6*6xx 10^(-21)"mol"L^(-1)` |
|
| 48. |
Reaction between H_(2)SO_(4) and NaOH is given below- H_(2)SO_(4)+2NaOH to Na_(2)SO_(4)+2H_(2)O The amount and molarity of he Na_(2)SO_(4) produced when 0.1(M) 1L H_(2)SO_(4) reacts with 0.1 (M) 1L NaOH will be- |
|
Answer» 7.10g |
|
| 49. |
Reaction between hydrogen sulphide and sulphurdioxide gives sulphur. Which type of redox reaction is this? |
|
Answer» SOLUTION :The reaction between HYDROGEN sulphide and sulphur dioxide is given as, `2H_(2)S+SO_(2)RARR 3S+H_(2)O` OXIDATION state of S in `H_(2)S` is `-2` and is oxidised to .O.. Oxidation state of S in `SO_(2)` is `+4` and is reduced to .O.. THEREFORE this reaction is an example of comproportionation. |
|