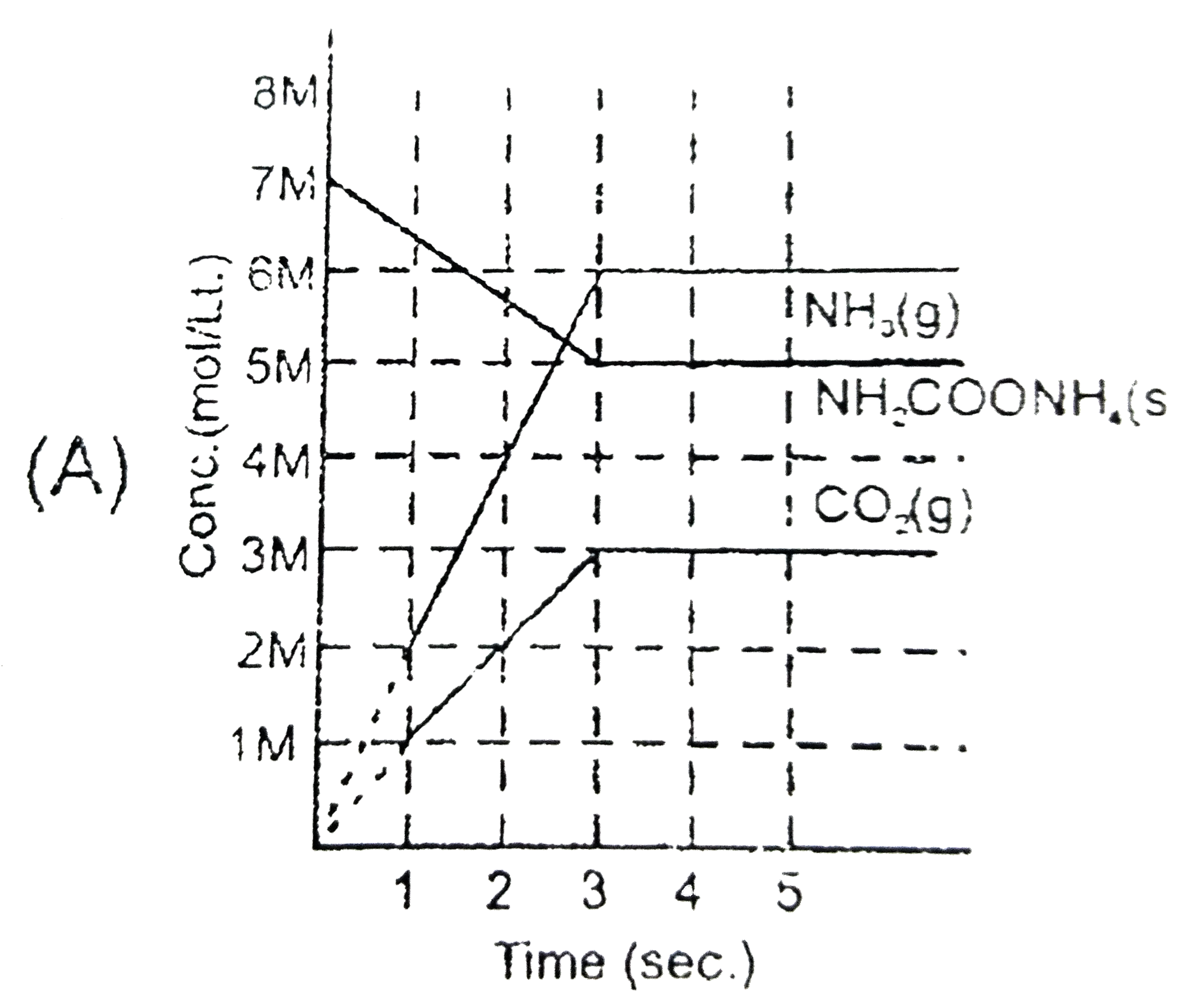Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Solubility of earbon dioxide gas in cold water can be increased by |
|
Answer» increase in pressure |
|
| 2. |
Solubility of carbon dioxide gas in cold water can be increased by............. |
|
Answer» increase in pressure |
|
| 3. |
Solubility of carbon dioxide gas in cold water can be increased by ……………… . |
|
Answer» increase in pressure increase in pressure, favours the forward REACTION. |
|
| 4. |
Solubility of an ionic compound in water is mainly dependent on A) Lattice enthalpy "" B) Hydration enathalpy Both these factors oppose each otherand the resultant of these determines thhe solubility of an ionic compound in water. If lattice enthalpy has greater value, the compound has greater value, the compound is less soluble. In case hydration enthaply has greater value, the compound is highly soluble is water BeF_(2) is soluble in water while fluorides of other alkalie earth metals are isoluble because of |
|
Answer» Covalent NATURE of `BeF_(2)` |
|
| 5. |
Solubility of an ionic compound in water is mainly dependent on A) Lattice enthalpy "" B) Hydration enathalpy Both these factors oppose each otherand the resultant of these determines thhe solubility of an ionic compound in water. If lattice enthalpy has greater value, the compound has greater value, the compound is less soluble. In case hydration enthaply has greater value, the compound is highly soluble is water Compounds of alkalie earth metals are less soluble than alkali metals, due to |
|
Answer» Theirhigh hydration enthalpy |
|
| 6. |
Solubility of an ionic compound in water is mainly dependent on A) Lattice enthalpy "" B) Hydration enathalpy Both these factors oppose each otherand the resultant of these determines thhe solubility of an ionic compound in water. If lattice enthalpy has greater value, the compound has greater value, the compound is less soluble. In case hydration enthaply has greater value, the compound is highly soluble is water Which of the following is more soluble in water |
|
Answer» `MgSO_(4)` |
|
| 7. |
Solubility of AgCl in 0.2 M NaCl is x and that in 0.1 M AgNO_3 is y then which of the following is correct ? |
|
Answer» ` x= y` ` (i)x (0.2 ) = Ksp (II)(0.1 )y= Ksp ` |
|
| 8. |
Solubility of Ag CN is maximum in: |
|
Answer» ACIDIC buffer solution |
|
| 9. |
Solubilityof solid solutein a liquidsolventincreases withincreasein ______. |
| Answer» Solution :When the temperature is increased, the AVERAGE kinetic energy of the MOLECULES of the solute and the solvent increases. The INCREASE in the kinetic energy facilitates the solvent molecules to break the intermolecular attractive forces that KEEP the solute molecules together and hence the solubility increases. | |
| 10. |
Solubility depends on the lattice energy of the solid and the hydration energy of the ions. Some lattice values for Group - II compounds are much lighter than the values for Group - I compounds, because of the effect of the increased charge on the ions in the Boron - Lande equation. Taking any one particular negative ion the lattice energy decreases as the size of the metal increases. The hydration energy also decreases as the metal ions become larger. For a substance to dissolve, the hydration energy must exceed thelattice energy The correct order of hydration energies is |
|
Answer» `Be^(2+)ltCa^(2+)ltCr^(2+)ltBa^(2+)` The hydration energy depends on size of the ion, lesser the size higher will be the hydration energy |
|
| 11. |
Solubility depends on the lattice energy of the solid and the hydration energy of the ions. Some lattice values for Group - II compounds are much lighter than the values for Group - I compounds, because of the effect of the increased charge on the ions in the Boron - Lande equation. Taking any one particular negative ion the lattice energy decreases as the size of the metal increases. The hydration energy also decreases as the metal ions become larger. For a substance to dissolve, the hydration energy must exceed thelattice energy The correct order of solubility is |
|
Answer» `BeF_(2)ltCaF_(2)gtSrF_(2)gtBaF_(2)` The high VALUE of solubility of `BeF_(2)` is due to high hydration energy of `Be^(+2)` ION |
|
| 12. |
Solubilirty product constants (K_(sp)) of salts of types MX, MX_(2), and M_(3)X at temperature T are 4.0xx10^(-8), 3.2xx10^(-14), and salts of temperature T is in the order |
|
Answer» `MX_(2)gtM_(3)XgtMX` `MX(s)hArr M^(+)underset(S)((aq.))+X^(-)underset(S)((aq.))` `K_(sp)=C_(M^(+))C_(X^(-))=(S)(S)` or `S=sqrt(K_(sp))=sqrt(4.0xx10^(-8))` `=2xx10^(-4)M` `MX_(2)(s)hArrM^(2+)underset(S)((aq.))+2X^(-)underset(2S)((aq.))` `K_(sp)=C_(M^(2+))C_(X^(-))^(2)=(S)(2S)^(2)=4S^(3)` or `S=((K_(sp))/(4))^(1//3)= ((3.2xx10^(-4))/(4))^(1//3)` `=2xx10^(-5)M` `M_(3)X(s)hArr underset(3S)(3M^(+)(aq.))+X^(3-)underset(S)((aq.))` `K_(sp)=C_(M^(+))^(3)C_(X^(3-))` `=(3S)^(3)(S)=27S^(4)` or`S=((K_(sp))/(27))^(1//4) = ((2.7xx10^(-15))/(27))^(1//4)` `=10^(-4)M` Thus, solubility order is `MXgt M_(3)XgtMX_(2)` |
|
| 13. |
Solids possess a ........... range order while liquids possess a ........... range order. |
| Answer» SOLUTION :LONG, SHORT | |
| 14. |
Solid NH_(4)I on rapid heating in a closed vessel at 357^(@)C dvelops a constant pressure of 275 mm Hg owing to partial decomposition of NH_(4)I into NH_(3) and HI but the pressure gradually increases further (when the excess solid residue remains in the vessel) owing to the dissociation of HI. Calculate the final pressure developed at equilibrium. K_(p) for HI dissociation is 0.015 at 357^(@)C. |
| Answer» SOLUTION :`307.46 MM`, | |
| 15. |
Solid NaOH dissolve in water and if solution is stirred than temperature of solution increases. Now the heating of this solution is suitable ? |
| Answer» SOLUTION :NaOH when dissolve in water the process is exothermic. `NaOH_((s)) + (AQ) toNaOH_((aq))`+ heatso HEATING is not SUITABLE. | |
| 16. |
Solid H_2O_2has non planar and non linear structure based on |
| Answer» Answer :C | |
| 17. |
Solid component of the carth consisting of soil, rocks and mountains is called |
|
Answer» Hydrosphere |
|
| 20. |
Solid barium oxide has ions, but it does not conduct electricity. Why ? |
| Answer» Solution :Electrical conductivity isdue to the movement of free electrons or freeions. Barium OXIDE is an ionic SOLID. It has no free electrons. Although solid barium oxide has ions, `Ba^(2+) and O^(2-`), the ions are not free to move in solid STATE, due to a definite crystal lattice. | |
| 21. |
Solid ammonium carbamate it taken in an empty closed container and allowed to attain equilibrium as, NH_2COONH_4(s)hArr2NH_3(g)+CO_2(g),K_(P)=500 choose the incorrect statements : |
|
Answer» EQUILIBRIUM total PRESSURE is 15 atm |
|
| 22. |
Solid ammonium carbamate dissociates to give ammonia and carbon dioxide as follows: NH_(3)COONH_(4)(s)hArr2NH_(2)(g)+CO_(2)(g) At equilibrium, ammonia is added such that partial pressures of NH_(3) at new equilibrium equals the original total pressure (at previous equilibrium). If the ratio of the total pressures now to the original total pressure is a//b then report a+b. |
|
Answer» `2P_(@) P_(@) K_(P)=(2P_(@))^(2)xxP_(@)=4P_(@)^(2)` `3P_(@) P' K_(P)=(3P_(@))^(2)xxP=9P_(@)^(2).P` `P=(4)/(9)P_(@)` `(P^(t))/(P^(t))=(3P_(@)+(4)/(9)P_(@))/(3P_(@))=(31)/(27)` |
|
| 23. |
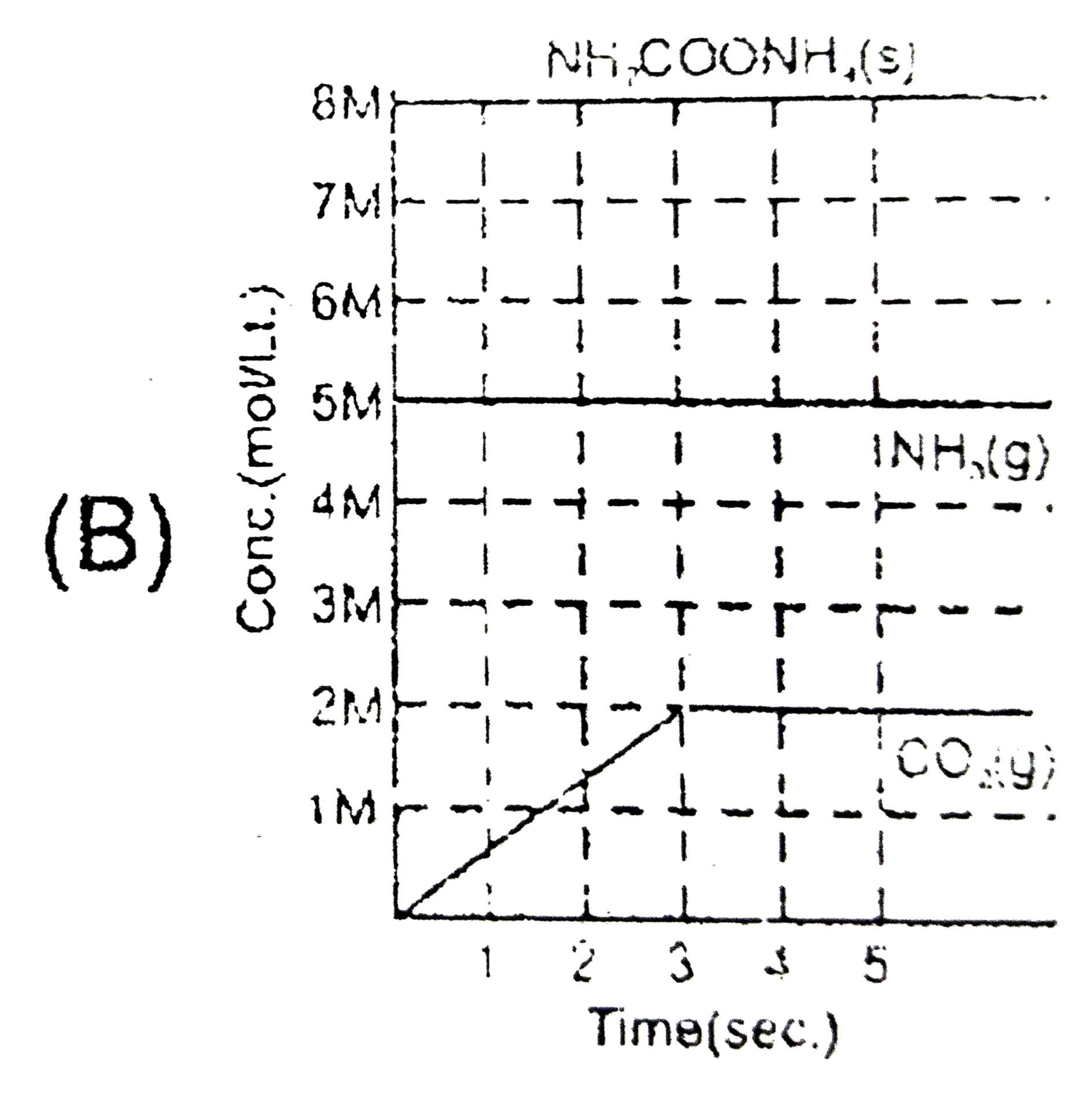
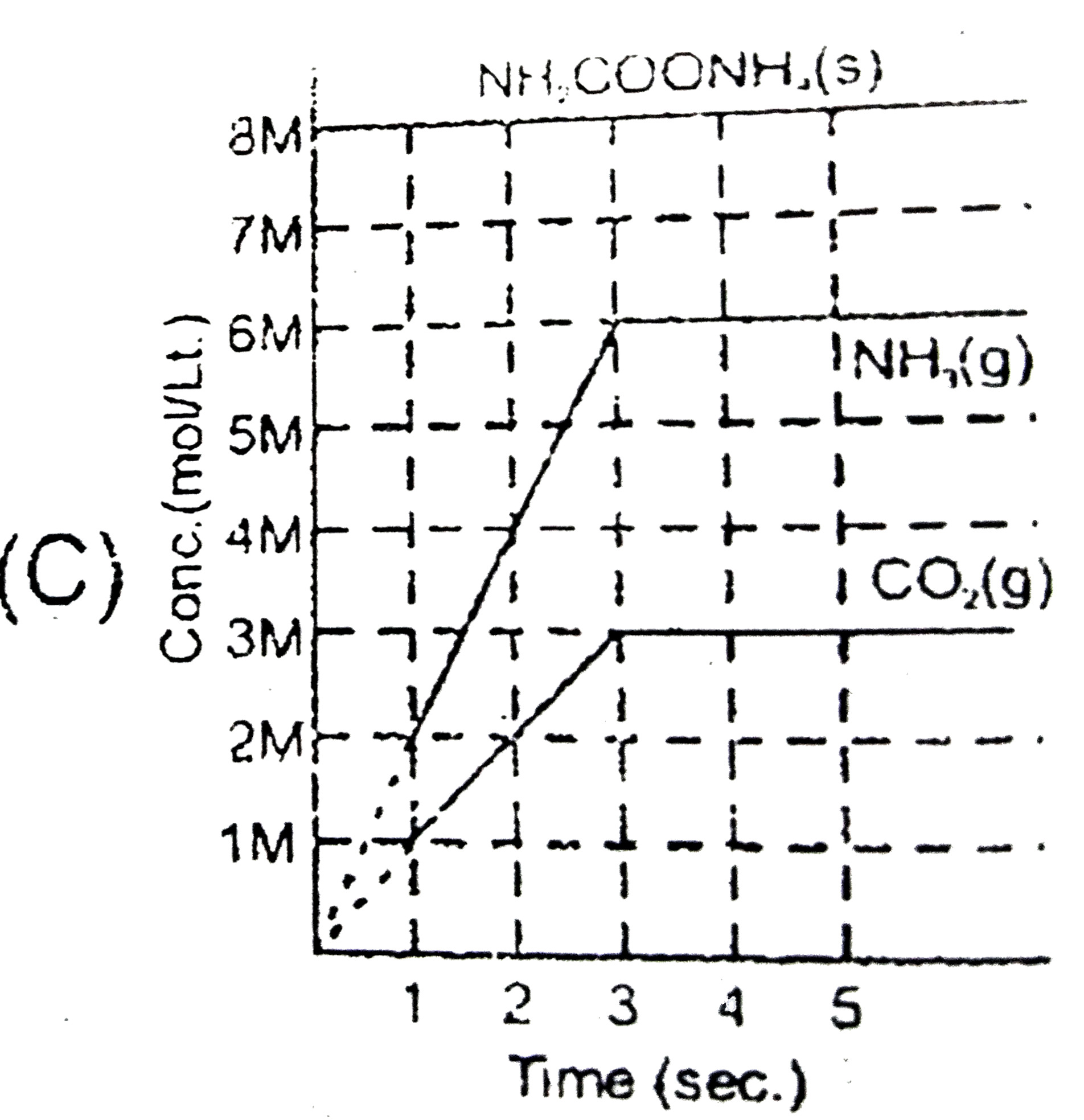
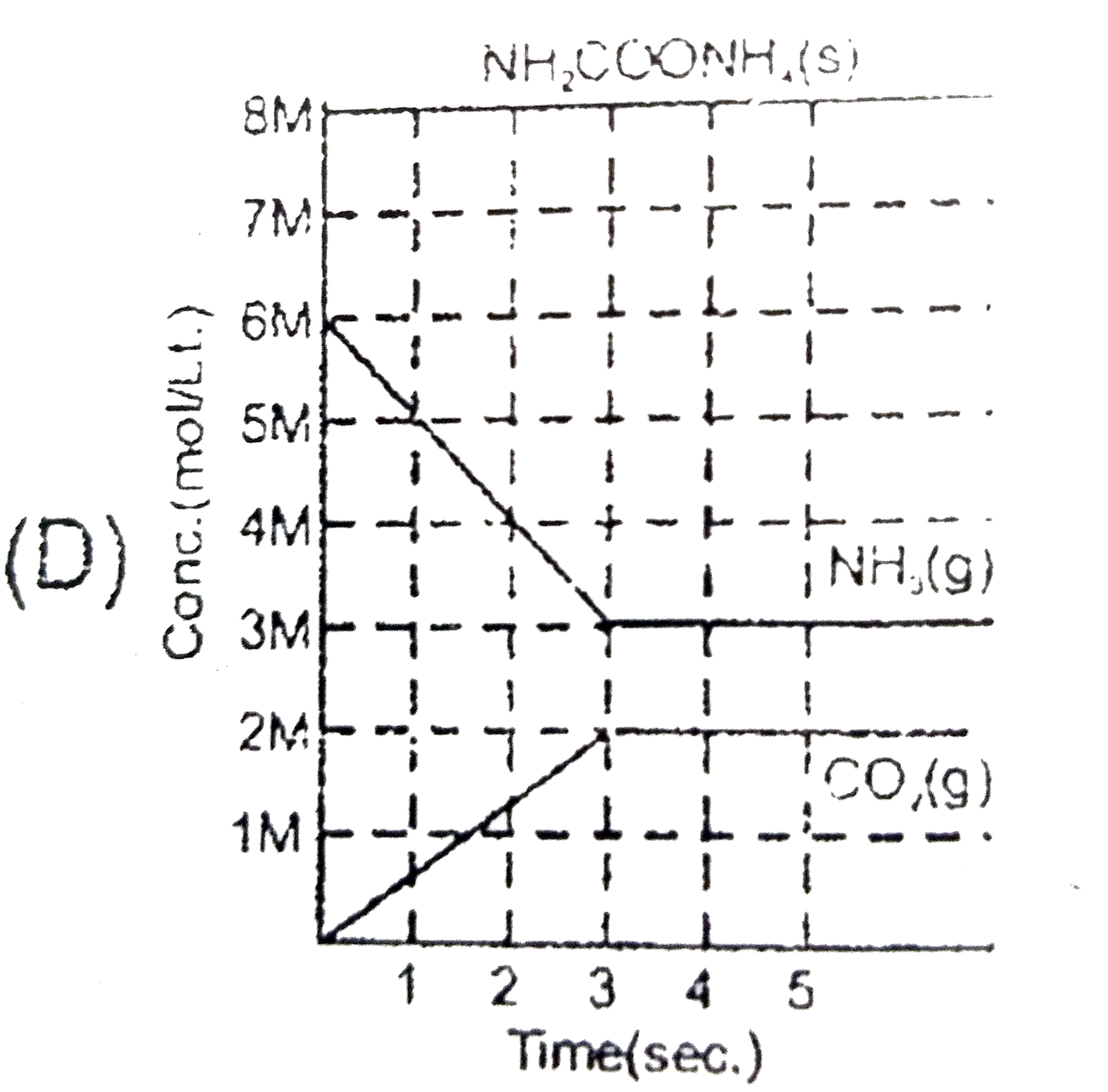
Solid ammonium cabamate dissociae to give ammonia and carbon dioxide as folows NH_(2)COOMH_(4)(s)hArr2NH_(2)(g)+CO_(2)(g) which of the following graph correctly represents the equilibrium. |
|
Answer»
|
|
| 24. |
Solid AgNO_(3) is added to a solution which 0.1 M in Cl^(-) and 0.1 M in CrO_(4)^(2-). K_(sp) values for AgCl and Ag_(2)CrO_(4) are 1.7 xx 10^(-10) and 1.9 xx 10^(-12) respectively. The concentration of Cl^(-) when Ag_(2)CrO_(4) starts precitating will be |
|
Answer» `3.9 xx 10^(-5) M` To precipitate `AgCl`, `:. [Ag^(+)] = (1.7 xx 10^(-10))/(0.1) = 1.7 xx 10^(-9)` `:' K_(sp) (Ag_(2)CrO_(4)) = [Ag^(+)]^(2)[CrO_(4)^(2-)]` To precipitate `Ag_(2)CrO_(4)` `:. [Ag^(+)] = SQRT((1.9 xx 10^(-12))/(0.1)) = 4.34 xx 10^(-6) M` Since `[Ag^(+)]` required to precinpitate `AgCl` is less than that required to precipitate `Ag_(2)CrO_(4), AgCl` will precipitate first. Now since `[Ag^(+)] = 4.34 xx 10^(-6)` when `Ag_(2)CrO_(4)` starts can be CALCULATED as `K_(sp)[AgCl] = [Ag^(+)] [Cl^(-)]` or `1.7 xx 10^(-10) = (4.34 xx 10^(-6))[Cl^(-)]` or `[Cl^(-)] = 3.9 xx 10^(-5) M`. |
|
| 25. |
Solid A overset("on")underset("heating")rarr anhydrous A A on strong heating produces compound(s) has/have: |
|
Answer» chain structure `{:(Na_(2)S_(2)O_(3).5H_(2)O OVERSET(Delta)rarr,Na_(2)S_(2)O_(3)+5H_(2)O),(,""darrDelta),(,Na_(2)SO_(4)+Na_(2)S_(5)):} ` `Na_(2)SO_(4): Na^(+) .^(-)O-overset(O)overset(||)underset(O)underset(||)(S)-O^(-)Na^(+)`, 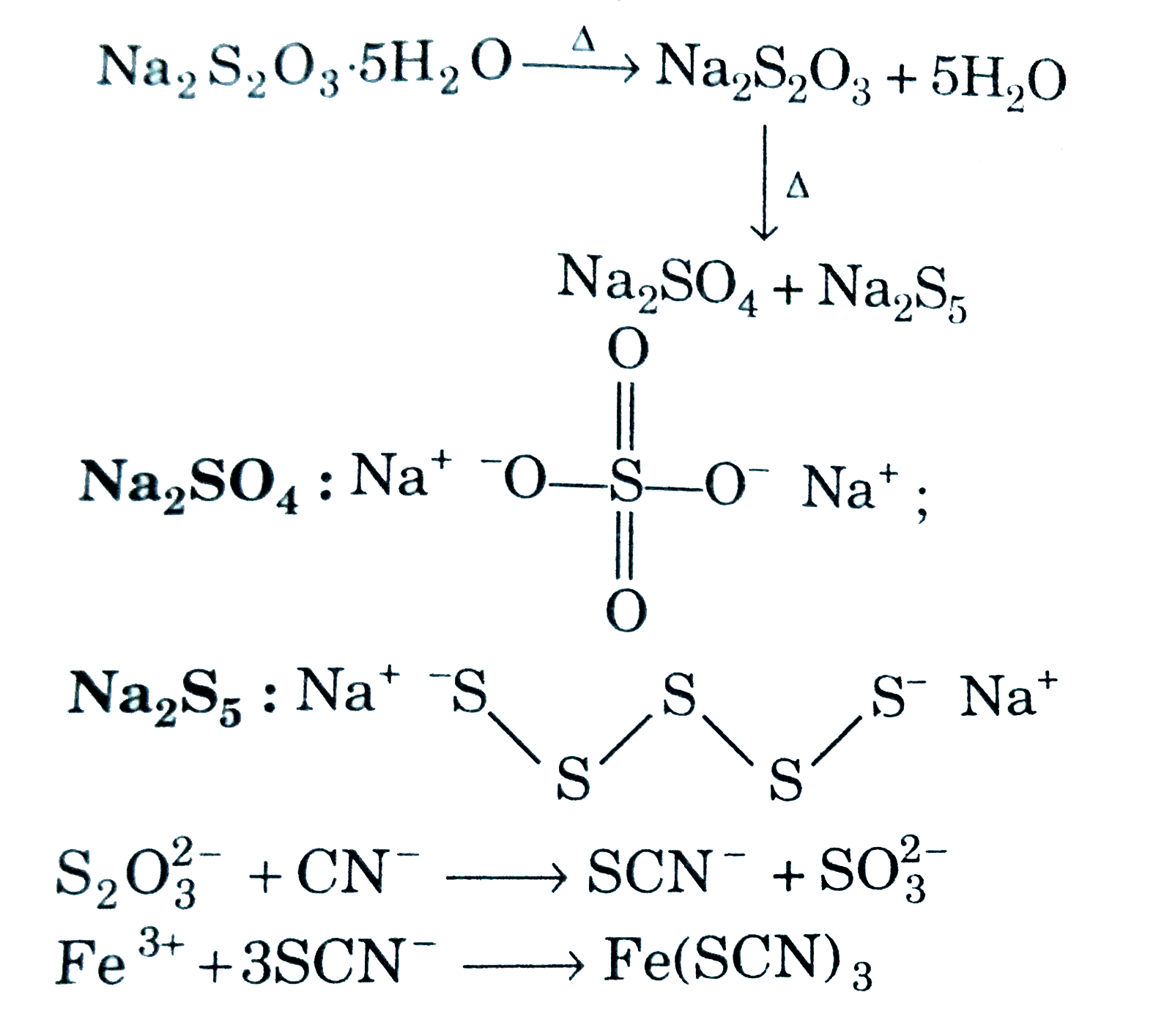 `S_(2)O_(3)^(2-) + CN^(-) rarr SCN^(-) +SO_(3)^(2-)` `Fe^(3+) +3SCN^(-) rarr Fe(SCN)_(3)` |
|
| 26. |
Solid A overset("on")underset("heating")rarr anhydrous A A is: |
|
Answer» `K_(2)S_(2)O_(3)` `{:(Na_(2)S_(2)O_(3).5H_(2)O overset(DELTA)rarr,Na_(2)S_(2)O_(3)+5H_(2)O),(,""darrDelta),(,Na_(2)SO_(4)+Na_(2)S_(5)):} ` `Na_(2)SO_(4): NA^(+) .^(-)O-overset(O)overset(||)underset(O)underset(||)(S)-O^(-)Na^(+)`, 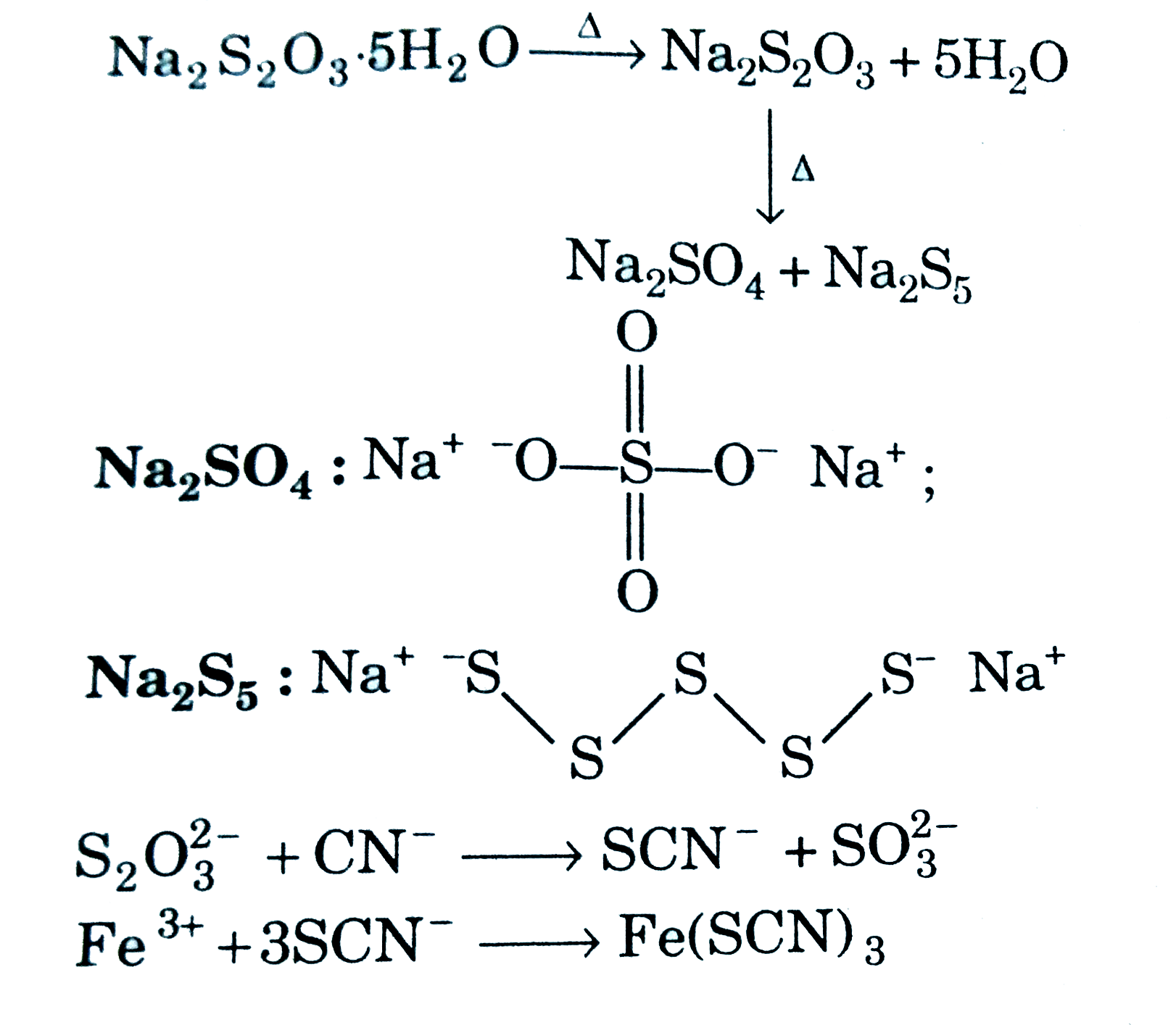 `S_(2)O_(3)^(2-) + CN^(-) rarr SCN^(-) +SO_(3)^(2-)` `Fe^(3+) +3SCN^(-) rarr Fe(SCN)_(3)` |
|
| 27. |
Solid A is very hard, electrical insulator in solid as well as molten state and melts at extremely high temperature. What type of solid is it? |
| Answer» Solution :Covalent Network SOLID like `SiO_2` (QUARTZ) or SIC or C (diamond) | |
| 28. |
Solid A is very hard, electrical insulator in solid as well as molten state and melts at extremely high temperature. What type of solid is it ? |
| Answer» SOLUTION :COVALENT Network solid like ` SiO_(2)` (quartz) or SIC or C(diamond). | |
| 29. |
Solid A "and" B are taken in a closed container at a certain temperature. These two solids decompose are following equilibria are established simultaneously A(s)hArrX(g)+Y(g) K_(P_(1)=250atm^(2) B(s)hArrY(g)+Z(g) K_(P_(2)=? If the total pressure developed over the solid mixture is 50 atm. Then the value of K_(P) for the 2^(nd) reaction |
|
Answer» Solution :`A(s)X+Y` `alpha alpha+beta` `B(s)hArrY+Z` `beta+alpha beta` `rArrK_(P_(1)=alpha(alpha+beta)` `K_(P_2)=beta(alpha+beta)` `P_(total)=(alpha+beta)+alpha+beta=2(alpha+beta)` `RARR 2(alpha+beta)=50rArr alpha+beta=25` `rArr 250=25alpha rArr alpha=10,beta=15` `rArr K_(P_(2)=beta(alpha+beta)=15xx25=375` |
|
| 30. |
Soil salinity can be measured by |
|
Answer» Calorimeter |
|
| 32. |
Soduim acetate can be converted to ethane by |
|
Answer» Heating with `LiAlH_(4)` |
|
| 33. |
In the reaction, I_(2)+2S_(2)O_(3)^(2-) rarr 2I^(-)+S_(4)O_(6)^(2-). |
|
Answer» |
|
| 34. |
Sodium thiosulphate reacts with iodine to give iodide and tetrathionate. In this reaction S_(2)O_(3)^(2-) undergoes |
|
Answer» Oxidation |
|
| 35. |
Sodium sulphate is soluble in water, whereas barium sulphate is sparingly soluble because |
|
Answer» The HYDRATION energy of sodium sulphate is more than its lattice energy. |
|
| 36. |
Sodium sulphate is soluble in water but barium sulphate is sparingly soluble because |
|
Answer» The hydration energy of `Na_(2)SO_(4)` is more than its LATTICE energy |
|
| 37. |
Sodium sulphate in soluble in water but barium sulphate is sparingly soluble because |
|
Answer» The hydration energy of `Na_(2)SO_(4)` is more than its LATTICE energy while the lattice energy of `BaSO_(4)` is more than its hydration energy |
|
| 38. |
Sodium street lamp gives off a characteristic yellow light of wavelength 588 nm. Calculate the energy mole (in kJ/mol) of these photons. |
|
Answer» `= 2.036 xx 10^(5) J mol^(-1)` |
|
| 39. |
Sodium salt of which acid will be needed for preparation of propane ? Write chemical equation for the reaction. |
Answer» SOLUTION :Since one carbon ATOM is LOST as `CO_2` during decarboxylation, therefore , the ACID needed must CONTAIN one carbon atom more than propane , i.e., butanoic acid or 2-methylpropanoic acid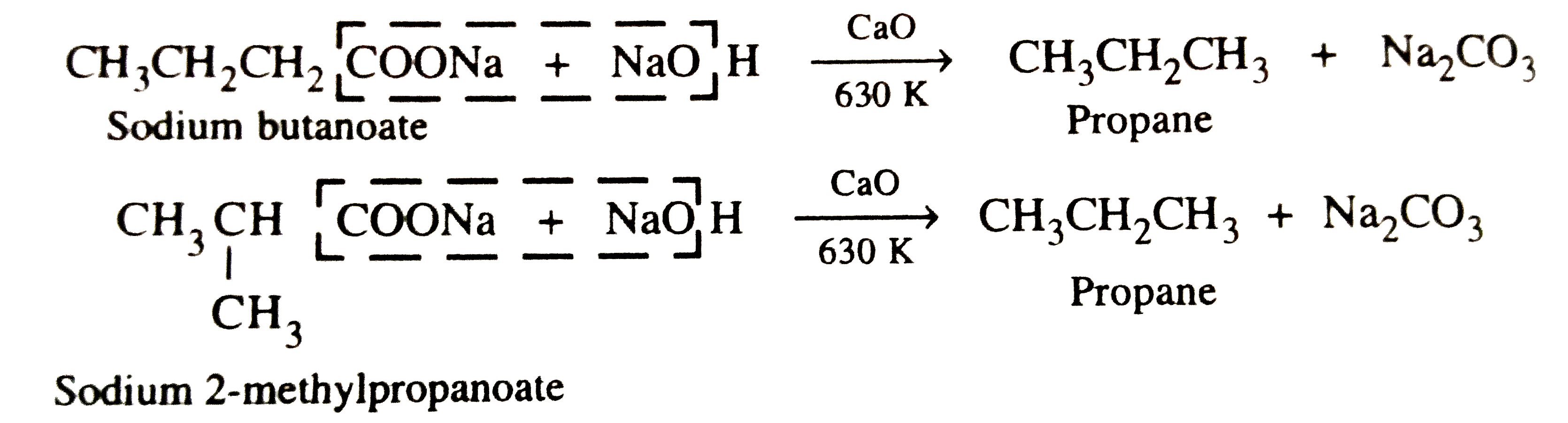
|
|
| 40. |
Sodium salt of which acid will be needed for the preparation of propane ? Write chemical equation for the reaction ? |
|
Answer» SOLUTION : `{:(""CH(CH_(3))_(2)),("|"),(OVERSET(1)CH_(3)-overset(2)CH_(2)-overset(3)CH_(2)-overset(4)CH-overset(5)CH-overset(6)CH_(2)-overset(7)CH_(2)-overset(8)CH_(2)-overset(9)CH_(2)-overset(10)CH_(3)),("|"),(""H_(3)C-CH-CH_(2)-CH_(3)):}` Name : 5 -SECONDARY -butyl-4-isopropyldecane (Where -`CH(CH_(3))_(2)` is isopropyl GROUP and `CH_(3)-underset(|)(CH)-CH_(2)-CH_(3)` means secondary butyl group) |
|
| 41. |
Sodium salt of benzoic acid when heated with which of the following gives benzene |
|
Answer» Sodamide |
|
| 42. |
Sodium reacts with water more vigorously than lithium because |
|
Answer» It has high atomic mass |
|
| 43. |
Sodium readily formsNa^(+)ion but never forms Na^(2+) ion. Explain. |
| Answer» Solution :Due to STABLE closed SHELL electronic CONFIGURATION of `Na^(+)` ion. | |
| 44. |
Sodium reacts more vigorously than lithium because, it..... |
|
Answer» has HIGHER ATOMIC weight. |
|
| 45. |
Sodium pyrophosphate is represented by which of the following formula |
|
Answer» `Na_(2)P_(2)O_(4)` `2H_(3)PO_(4) underset("Pyrophosphoric acid")(overset(-H_(2)O)toH_(4)P_(2)O_(7)"")` `underset("Sodium pyrophosphate")(O larr underset(NA)underset(|)underset(O)underset(|)overset(Na)overset(|)overset(O)overset(|)P-O-underset(Na)underset(|)underset(O)underset(|)overset(Na)overset(|)overset(O)overset(|)P RARRO)` |
|
| 46. |
Sodium phenoxide reacts with CO_(2) at 400 K and 4.7 atm pressure to give |
|
Answer» SODIUM salicylate |
|
| 47. |
Sodium peroxide which is yellow solid , when exposed to air becomes white due to the formation of |
|
Answer» `H_(2)O_(2)` `Na_(2)O_(2) + H_(2) O to 2 NaOH + H_(2)O_(2)` `2 Na_(2) O_(2) + 2 CO_(2) to 2 Na_(2) CO_(3) + O_(2)` or `Na_(2)O_(2) + H_(2)O +CO_(2) to Na_(2)CO_(3) + H_(2)O_(2)` |
|
| 48. |
Sodium peroxide which is yellow solid, when exposed to air becomes white due to the formation of |
|
Answer» `H_(2)O_(2)` |
|
| 49. |
Sodium peroxide which is a yellow solid, when exposed to air becomes white due to the formation of |
|
Answer» `Na_(2)O` and `O_(3)` |
|