Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Some meta -directing substituents in aromatic substitution are given .Which one is most deactivating ? |
|
Answer» `-COOH` |
|
| 2. |
Some meta directing substituents in aromatic substitution are given which one is mostdeactivating |
|
Answer» `-COOH` |
|
| 3. |
Somemercury is poured into a capillary tube of uniform bore and one end closed. When the tube is held horizontally, mercury column has a length of 4 cm and air enclosed in the closed end has a length of 10 cm. If the tube is now held vertically, with open end upwards, the length of the air column will be (take atmospheric pressure at the place of experiment to be 760 mm) |
|
Answer» <P>10.5 cm 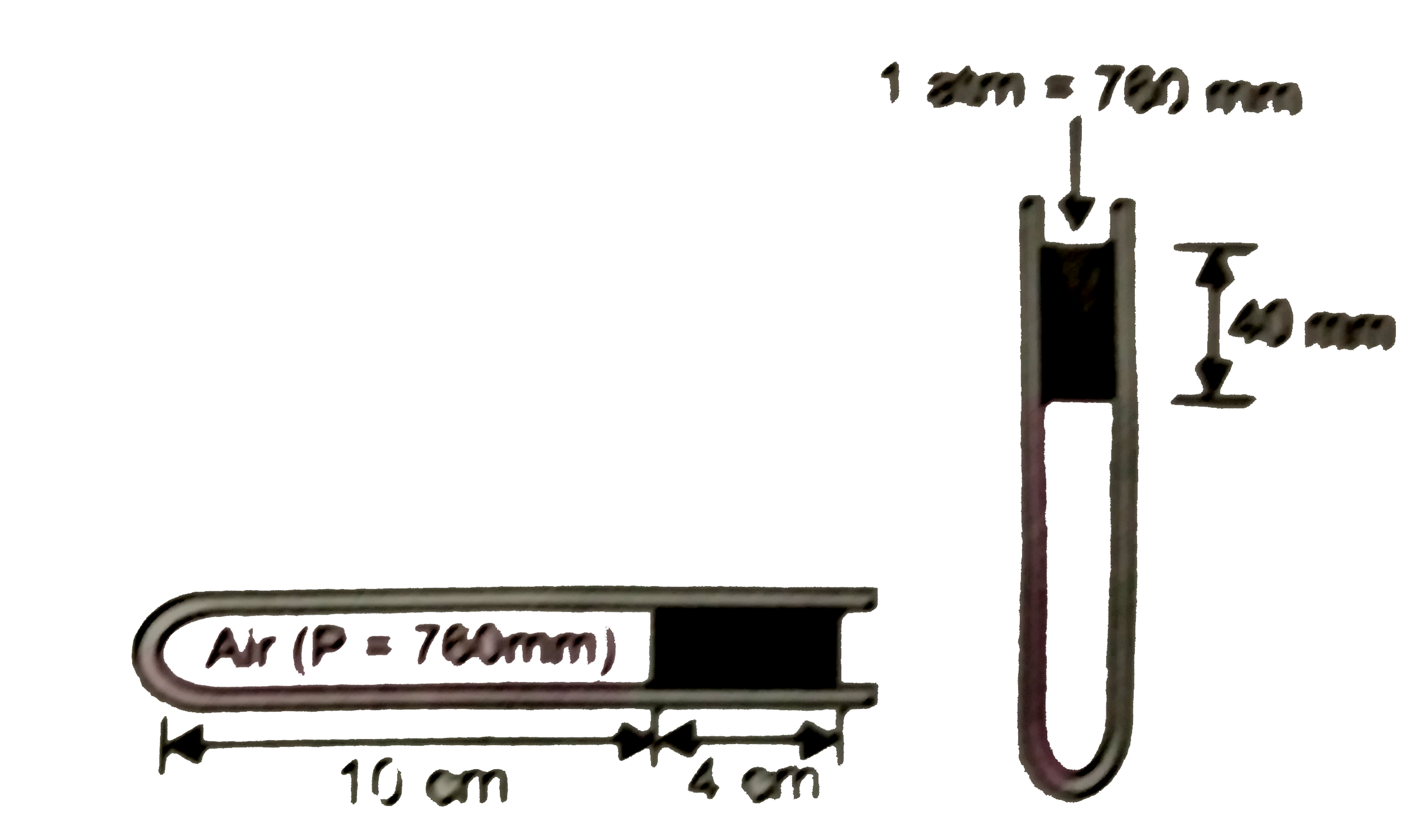 When held horizontally, pressure of enclosed air =760 mm When held vertically, pressure increases by 4 cm, i.e., 40 mm Hence, now pressure=760+40=800 mm Applying `P_(1)V_(1)=P_(2)V_(2)` `P_(1)xx(Axxl_(1))=P_(2)xx(Axxl_(2))` (A=area of cross-section of the capillary TUBE) or `P_(1)xxl_(1)=P_(2)xxl_(2)` `760xx10=800xl_(2)"or"_(2)=9.5" cm"` |
|
| 4. |
Some meta- directing subsituents in aromatic substitution are given. Which one is most deactivating ? |
|
Answer» `-SO_(3)H` |
|
| 5. |
Some ether is added to anb aqueous soluetion of a mixtrue of LiCl, NaCl and AlCl_(3) . Which will be extracted into ether ? |
|
Answer» `LiCl, NACL` hence EXTRACTED into ETHER. |
|
| 6. |
Some elements are wrongly placed in the decreasing orderof the property mentioned. Rectifying the fault, place them in correct order of the property . Also furnish reason for the correction done. NagtMggtAlgtSi ( Firstionization potential ) . |
| Answer» Solution :`SI gt Al gt "Mg "gt NA` , silicon has HIGHEST first ionization potential due to smallest size , Mg is exception. i.e., has HIGHER ionization potential than ALUMINUM because of `ns^(2)` configuration which is stable. | |
| 7. |
Some elements are wrongly placed in the decreasing orderof the property mentioned. Rectifying the fault, place them in correct order of the property . Also furnish reason for the correction done. NgtSi gt C gt P (electronegative of the elements ). |
| Answer» Solution :`NgtCgtPgtSi` , nitrogen has smallest SIZE and so has hight tendency to ATTRACT SHARED PAIR of electrons. | |
| 8. |
Some compounds are given below : How many compounds are chiral and also have chiral centre? |
|
Answer» 5 |
|
| 9. |
Some compounds are given below : How many compounds are chiral ? |
|
Answer» 5 |
|
| 10. |
Some compounds are given below : How many compounds are achiral but have chiral centre ? |
|
Answer» 5 |
|
| 12. |
Some amount of NH_(4)Cl was boiled with 50 mL of 0.75N NaOH solution till the reaction was complete. After the completion of the reaction, 10 mL of 0.75 N H_(2)SO_(4) were required for the neutralisation of the remaining NaOH. Calculate the amount of NH_(4)Cl taken. |
|
Answer» |
|
| 13. |
Some amount of ..20V..H_(2)O_(2) is mixed with excess of acidifeid solution KI. The iodine so liberated required 200ml of 0.IN Na_(2)S_(2)O_3for tritration. The mass of K_(2)Cr_(2)O_7 needed to oxidises the above volume of H_2O_2 solution : |
|
Answer» 3.6 G WT of `K_2Cr_2O_7=0.98` g |
|
| 14. |
Some amount of ..20V..H_(2)O_(2) is mixed with excess of acidifeid solution KI. The iodine so liberated required 200ml of 0.IN Na_(2)S_(2)O_3for tritration. The volume of O_2 at STP that would be linerated above H_2O_2 solution : |
|
Answer» Solution :VOLUME of `O_2` liberated = Volume strength `xx` vol.of `H_2O_2` solution `=20xx5.6 =112`ml |
|
| 15. |
Some amount of ..20V..H_(2)O_(2) is mixed with excess of acidifeid solution KI. The iodine so liberated required 200ml of 0.IN Na_(2)S_(2)O_3for tritration. The volume of H_2O_2 solution is |
|
Answer» 11.2 ml `V_1N_1 =V_2N_2` `V_(1) XX(20)/(5.6) = 200 xx0.1` VOL. of `H_2O_2` solution = 5.6 ml |
|
| 16. |
Solvay ammonia soda process is used to prepare ......... industrially. |
|
Answer» `NaHCO_(3)` |
|
| 17. |
Solvay process can be represented by the following scheme Identify A, B, C and D : |
|
Answer» |
|
| 18. |
Solvay process is used in the manufacture of |
|
Answer» `K_(2)CO_(3)` |
|
| 19. |
Solutions of sodium azide (NaN_(3)) and iodine (as KI_(3)) do not react but on addition of a trace of 'X' ion, which acts as a catalyst there is an immediate vigorous evolution of nitrogen. Then 'X' may be: |
|
Answer» `S_(2)O_(3)^(2-)` |
|
| 20. |
Solutions containing 23 g HCOOH is/are : |
|
Answer» `46 G " of " 70%((w)/(V))HCOOH(d_("solution" )=1.40g//mL)` |
|
| 21. |
Solution ''X'' contains Na_(2)CO_(3) and NaHCO_(3),20 ml of X when titrated using methyl organge indicator consumed 60 ml of 0.1M HCI solution. In another experiment, 20 ml of X solution when titrated using phenolphthalein consumed 20 ml of 0.1 M HCl solution. The concentraions ("in mol lit"^(-1)) of Na_(2)CO_(3) and NaHCO_(3) in X are respectively) |
|
Answer» 0.01, 0.02 |
|
| 22. |
Solutions A and Bare both clear and colourles. When solution A is mixed with solution B, the temperature of the mixture increases and a yellow precipitate is observed. What can be concluded from these observations? |
|
Answer» The reaction is thermodynamically favoured ( spontaneous) at all temperatures `DeltaG= DELTAH-T DeltaS`. For reaction to be spontaneous, `DeltaG` should be `-ve`. As `DeltaH` is`-ve` and`DeltaS `is `-ve`, `DeltaG` can be -ve only if T is low.Hence, reaction will be spontaneous at low temeprature. |
|
| 23. |
Solution of X is being titrated against a solution of Y. If phenolphathalen is found to be a suitabe indicator. X and Y could be respectively. |
|
Answer» `NAOH` and `HCL` |
|
| 24. |
Solution ofFeCl_(3) " ( yellow )" and NH_(4)SCN " (colourless) "were mixed in a beaker. Red colour was obtained . On adding H_(g)Cl_(2) to the solution, the intensity of colour will ………. . |
|
Answer» |
|
| 26. |
Solution of an acid and it's anion (that is it's conjugate base) or of a base and it's common cation are buffered. When we add a small amount of acid or base to any one of them, the pH of solution changes very little pH of buffer solution can be compoted as , for , acidic , buffer pH = pK_(a) + "log"(["Conjugate base"])/(["Acid"]) for basuc buffer pOH = pK_(b) + "log" (["Conjugated base"])/(["base"]) it is generally accepteed that a solution has useful buffer capacity (pH change resistng power) provided that the value of [salt or conjugate base]/[acid] for acidic acid buffer lies within the range of 1:10 to 10:1. Buffer capacity is max. When [conjugate base]=[acid] Select correct statement: |
|
Answer» When we add shall amount of `NAOH` inacidicbuffer SOLUTION, pOH of the solution is increases |
|
| 27. |
Solution of an acid and it's anion (that is it's conjugate base) or of a base and it's common cation are buffered. When we add a small amount of acid or base to any one of them, the pH of solution changes very little pH of buffer solution can be compoted as , for , acidic , buffer pH = pK_(a) + "log"(["Conjugate base"])/(["Acid"]) for basuc buffer pOH = pK_(b) + "log" (["Conjugated base"])/(["base"]) it is generally accepteed that a solution has useful buffer capacity (pH change resistng power) provided that the value of [salt or conjugate base]/[acid] for acidic acid buffer lies within the range of 1:10 to 10:1. Buffer capacity is max. When [conjugate base]=[acid] Calculate the pH of the solution made by adding 0.01 mole of HCl in 100 ml. of solution which is 0.2 M in NH_(3)(pK_(b) = 4.74) and 0.3 M in NH_(4)^(+): |
|
Answer» `5.34` |
|
| 28. |
Solution of an acid and it's anion (that is it's conjugate base) or of a base and it's common cation are buffered. When we add a small amount of acid or base to any one of them, the pH of solution changes very little pH of buffer solution can be compoted as , for , acidic , buffer pH = pK_(a) + "log"(["Conjugate base"])/(["Acid"]) for basuc buffer pOH = pK_(b) + "log" (["Conjugated base"])/(["base"]) it is generally accepteed that a solution has useful buffer capacity (pH change resistng power) provided that the value of [salt or conjugate base]/[acid] for acidic acid buffer lies within the range of 1:10 to 10:1. Buffer capacity is max. When [conjugate base]=[acid] Useful buffer range of weak acid HA(k_(a) =10^(-5)) is : |
| Answer» Solution :N//A | |
| 29. |
Solution of an acid and it's anion (that is it's conjugate base) or of a base and it's common cation are buffered. When we add a small amount of acid or base to any one of them, the pH of solution changes very little pH of buffer solution can be compoted as , for , acidic , buffer pH = pK_(a) + "log"(["Conjugate base"])/(["Acid"]) for basuc buffer pOH = pK_(b) + "log" (["Conjugated base"])/(["base"]) it is generally accepteed that a solution has useful buffer capacity (pH change resistng power) provided that the value of [salt or conjugate base]/[acid] for acidic acid buffer lies within the range of 1:10 to 10:1. Buffer capacity is max. When [conjugate base]=[acid] One litre of an aqueous solution contaion 0.15 mole of CH_(3)COOH(pK_(a) = 4.8) and 0.15 mole of CH_(3)COONa. After the addition of 0.05 moe of solid NaOH to this solution, the pH will be: |
|
Answer» `4.5` after addition of `0.05` MOLES of `NaOH` `{:(CH_(3)COOH,+,NaOH,RARR,CH_(3)COONa),(0.05,,0.05,,0.05):}` LEFT finally `{:(CH_(3)COOH,rarrCH_(3)COONa),(0.1 ""0.2,):}` `:. pH = 4.8 + "log" (0.2)/(0.1) = 5.1` |
|
| 30. |
Solution of 0.1 N NH_4 OH and 0.1 N NH_4 Cl "has "P^(H)9.25. ThenP^(K_b)of NH_4 OHis |
|
Answer» ` 9.25` ` 14- 9. 25 =PK_b +log "" (0.1)/( 0.1 ) rArr PK_b =4.75` |
|
| 31. |
……….solution is added for produce the precipitate of halide for estimation of halogen |
| Answer» SOLUTION :`AgNO_(3)` | |
| 32. |
………solution is added for produce the precipitate of BaSO_(4) in estimation of sulphur. |
| Answer» SOLUTION :`BaCl_(2)` | |
| 33. |
Solution containing 23 g HCOOH is/are : |
|
Answer» `46 g` of `70% ((w)/(v)) HCOOH (d_("SOLUTION") = 1.40 g//mL)` `= 70= (70)/(140)xx46=23 g` `(b) 10M=("Mass of solute" //46)/((50)/(1xx1000))` Mass of solute `=23 g` `(c ) 100 g` solution contain `25 g` of solute mass of solute `=(25)/(100)xx50=12.5` `(d) 5 M =("Mass of solute"//46)/(46//1000)` Mass of solute `=10.58 g` |
|
| 34. |
Solublility product constant of a sparingly soluble salt MCl_(2) is 4 xx 10^(-12) at 25^(@)C. Also, at 25^(@)C , solublity of MCl_(2) in an aquoneous solutoion of CaCl_(2), is 4 xx 10^(8) times less compared to its solubility in pure water. Hence, concentration (molarity) of CaCl_(2) solution is |
|
Answer» |
|
| 35. |
Soluble complex of 'R' Compound 'P' may be: |
|
Answer» `CuSO_(4)` |
|
| 36. |
Solubility products of ACO_(3), BSO_(4), and ASO_(4), are 4 xx 10^(-10), 6 xx 10^(-10) and 8 xx 10^(-10) respectively. The solubility product of BCO_(3), is x x 10^(-10), What is x ? |
|
Answer» ` [B^(+2) ][SO_4^(-2) ] = 6 xx 10 ^(-10) ` ` [B ^(+2) ] 2 [CO_3^(-2) ]=6 xx 10 ^(_10) ` ` [B^(+2) ][CO_3^(-2) ]=3xx 10 ^(-10) ` |
|
| 37. |
Solubility products of ACO_3 , BSO_4 and ASO_4 are 4xx10^(-10), 6xx 10^(-10) and 8xx 10^(-10)respectively. The solubility product of BCO_3 is x xx 10^(-10)What is x? |
|
Answer» ` [B^(+2) ][SO_4^(-2) ] = 6 xx 10 ^(-10) ` ` [B ^(+2) ] 2 [CO_3^(-2) ]=6 xx 10 ^(_10) ` ` [B^(+2) ][CO_3^(-2) ]=3XX 10 ^(-10) ` |
|
| 38. |
Solubility products of Ag_2 CrO_4and AgCl are 9 xx 10^(-12) and 1 xx 10^(-10)respectively at 298. Concentration of Ag^(+) is more in the saturated solution of which salt? |
|
Answer» Solution :If the concentration of `AG^(+) ` is .s. `4s^3` for `Ag_2 CrO_(4) =9 xx 10^(-12)s = 1.3 xx 10^(-4)MOL L^(-1)` `s^2` for `AgCl = 1 xx 10^(-10), s = 1 xx 10^(-5 )mol L^(-1)` Saturated `Ag_2 CrO_4` , has more concentration of `Ag^+` ion in solutions. |
|
| 39. |
Solubility products constants (K_(sp))of salts of types MX, MX_2,M_3Xat temperature T are 4xx 10^(-8)3.2 xx 10^(-14)and 27 xx 10 ^(-15)respectively. Solubility ("moldm" ^(-3) )of the salts at temperature T are in the order : |
|
Answer» `MX gt MX_2 gt M_3X ` ` K_(Sp)"of " MX_2 = 3.2 xx 10 ^(-14), S_2 =2xx 10 ^(-5)M` ` K_(sp)"of "M_3X =27xx 10 ^(-15), S_3 ~~ 10 ^(-4)M` ` S_1 gt S_3 gt S_2` |
|
| 40. |
The values of K_(sp) of two sparingly soluble salts Ni(OH)_(2) and AgCN are 2.0xx10^(-15) and 6xx10^(-17) respectively. Which salt is more soluble ? Explain . |
| Answer» SOLUTION :`NI(OH)_2` is more SOLUBLE | |
| 41. |
Solubility product of the hydroxide M(OH)_2" is "4 xx 10 ^(-12), Select the correct statement (s) among the following: |
|
Answer» The pH of its saturated solution will be 10. 3 ` [OH^(-) ] =2 xx 10^(-12), S = 10 ^(-4)M,` In pH `= 9, POH =10 ^(-5) ` ` i.e., [OH^(-) ] downarrow rArr " solubilitywill "uparrow ` In pH =1, POH =` 10 ^(-13)` ` i.e., [OH^(-)] downarrow rArr " Solubility will " uparrow ` |
|
| 42. |
Solubility product of silver bromide is 5.0xx10^(-13). The quantity of potassium bromide (molar mass taken as 120 g "mol"^(-1)) to beadded to 1 litre of 0.05M solution of silver nitrate to start the precipitation of AgBr is |
|
Answer» `6.2xx10^(-5) g` `K_(SP)(AgBr)=[Ag^(+)][Br^(-)]` `:. [Br^(-)]=(K_(sp))/([Ag^(+)])=(5.0xx10^(-13))/(0.05)=10^(-11)M` i.e., amount of KBR to be added`=10^(-11)` MOLE `=10^(-11)xx120 g = 1.2 xx 10^(-9) g`. |
|
| 43. |
Solubility product of barium sulphide (BaSO_(4)) is 2.4xx10^(-9). Calculate it's solubility. |
|
Answer» Solution :`BaSO_(4)` is AB TYPE of salt `K_(sp)=2.4xx10^(-9)` `S=sqrt(K_(sp))=sqrt(2.4xx10^(-9))=sqrt(0.24xx10^(-8))=0.4898xx10^(-4)"MOL"//dm^(3)`. |
|
| 44. |
Solubility product constants (K_(sp)) of salts of types MX, MX_(2) at M_(3)X at temperature T are 4.0xx10^(-8),3.2xx10^(-14) and 2.7xx10^(-15) respectively. Solubility (mol dm^(-3)) of the salts at temperature T are in the order |
|
Answer» `MX gt MX_(2) gt M_(3)X` or `s=sqrt(K_(sp))=sqrt(4xx10^(-8))=2XX10^(-4)M` `MX_(2)hArr underset(s)overset(2+)M + 2 underset(2s)X^(-), K_(sp)=s(2s)^(2)=4s^(3)` `:. S=((K_(sp))/(4))^(1//3)=((3.2xx10^(-14))/(4))^(1//3)` `=2xx10^(-5)M` `M_(3)X hArr 3 underset(3s)M^(+)+ underset(s)X^(3-)`, `K_(sp)=(3s)^(3)(s)=27S^(4)` `:. s=((K_(sp))/(27))^(1//4)=((2.7xx10^(-15))/(27))^(1//4)=10^(-4) M` Thus, the solubilities are in the order `2xx10^(-4) gt 10^(-4) gt 2 xx 10^(-5)`, i.e., `MX gt M_(3)X gt MX_(2)`. |
|
| 45. |
Solubility of the alkaline metal sulphates in water decreases in the sequence . |
|
Answer» `SR gt Ca gt Mg gt BA` |
|
| 46. |
Solubility of the alkaline earth's metal sulphates in water decreases in the sequence… |
|
Answer» `Mg gt Ca gt SR gt Ba` 
|
|
| 47. |
Solubility of silver chloride in pure water is 1 xx 10^(-5) M. What is its solubility in 0.001M sodium chloride solution ? |
| Answer» SOLUTION :`1 XX 10^(-7) M` | |
| 48. |
Solubility of Pb(OH)_2 in water is 6.7xx 10^(-6) M. Calculate its solubility in a buffer solution of pH value 8. |
| Answer» SOLUTION :`1.2 xx 10^(-3) mol L^(-1)` | |
| 49. |
Solubility of gas decreases in a liquid by |
|
Answer» INCREASE of TEMPERATURE |
|