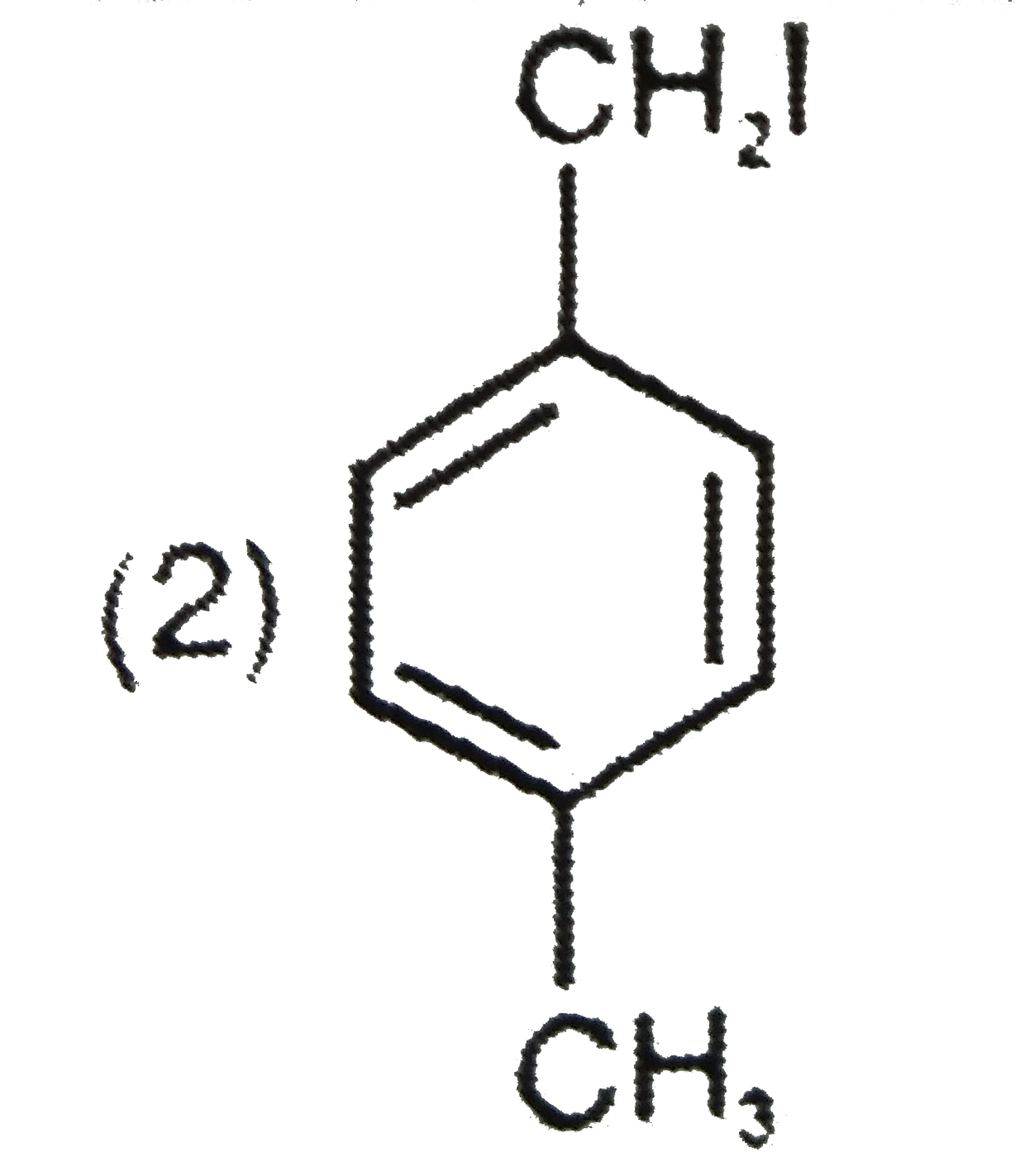
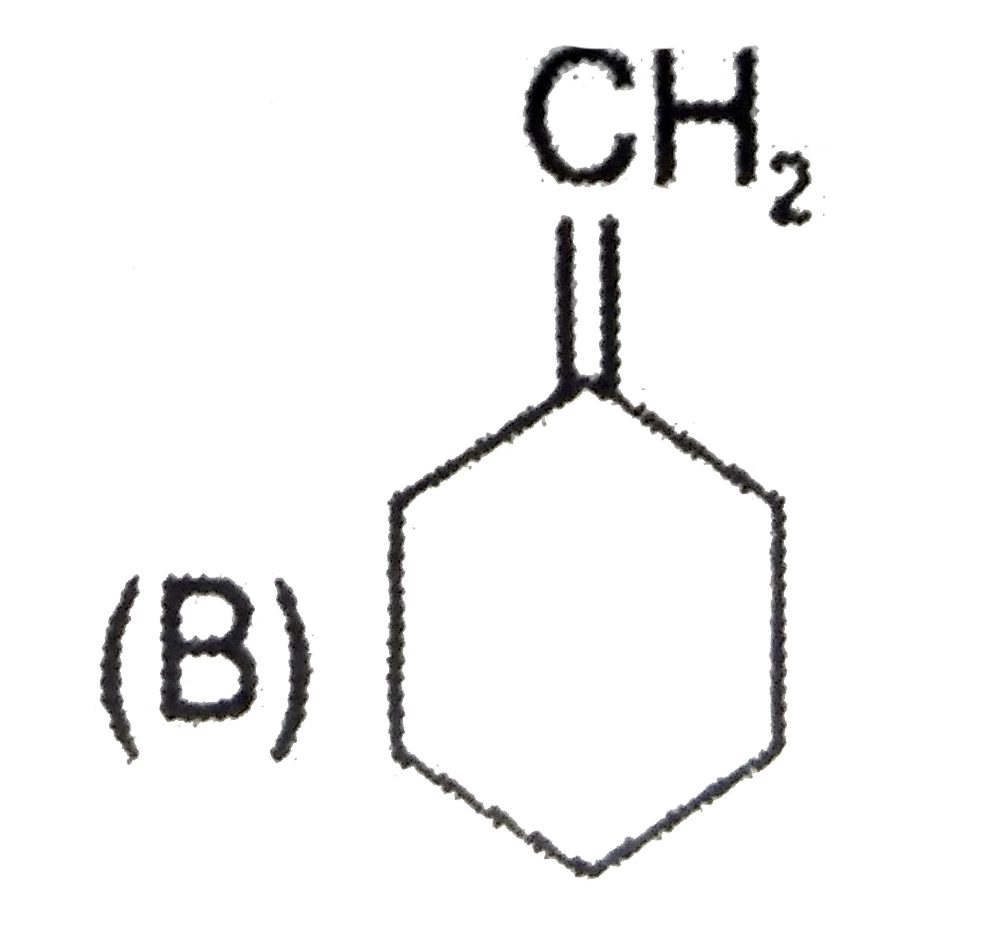
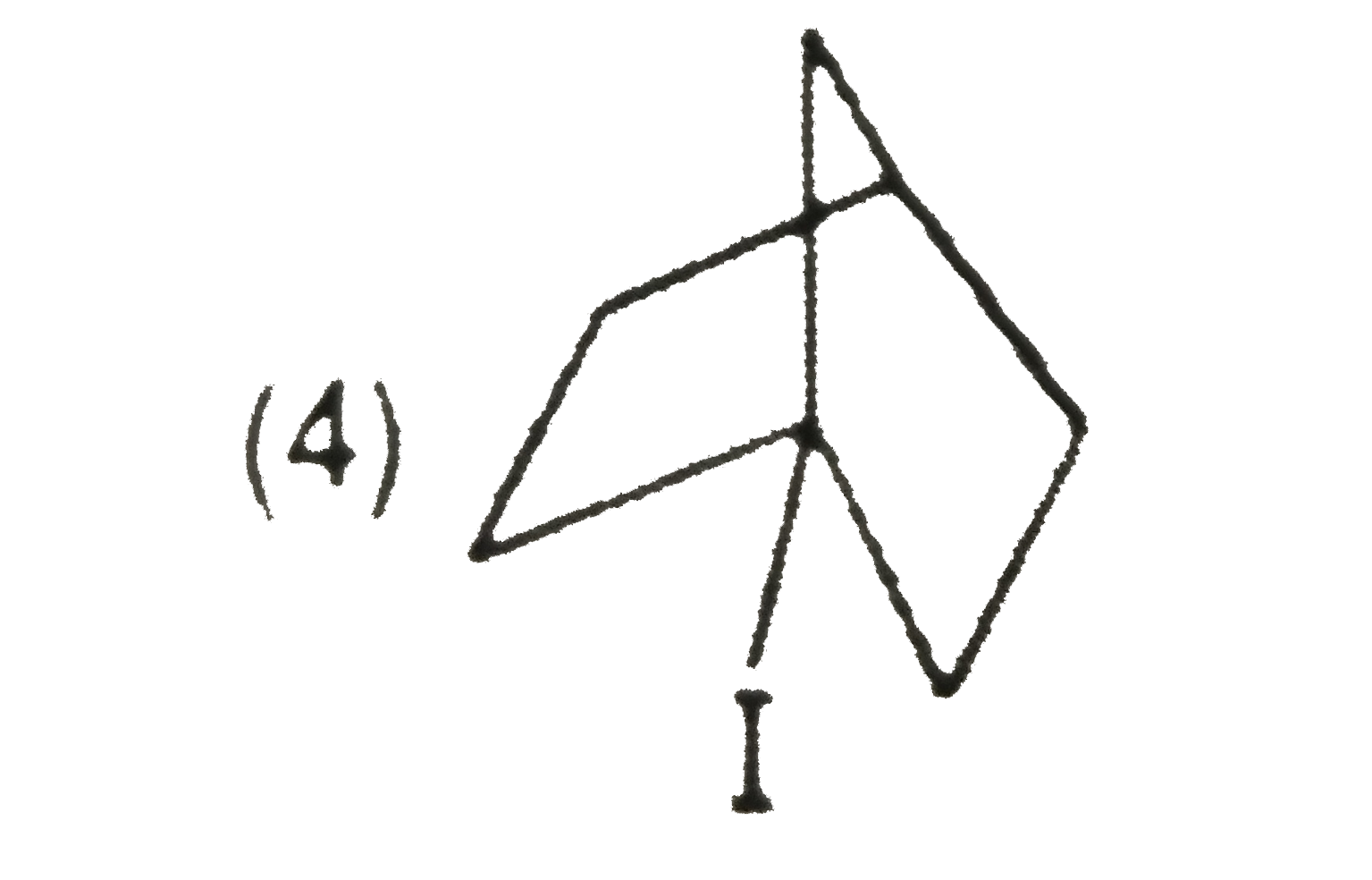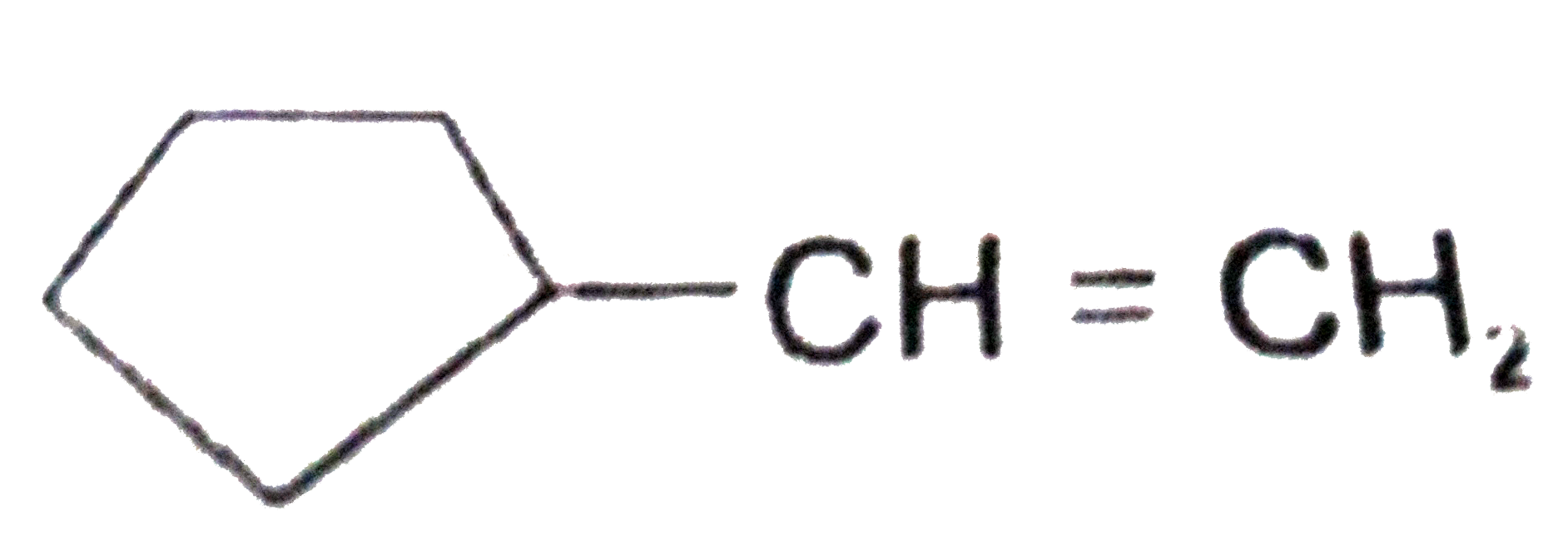Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Suggest a list of the substances where carbon can exhibit oxidation states from –4 to +4 and nitrogen from –3 to +5. |
Answer» SOLUTION :
|
|
| 2. |
Suggest a list of substacnes where carbon can exhibit oxidation state from -4 to +4 and nitrogen from -3 to +5 |
Answer» SOLUTION :
|
|
| 3. |
Suggest a list of the substances where carbon can exhibit oxidation states from -4 to +4 and nitrogen from –3 to +5. |
Answer» SOLUTION :
|
|
| 4. |
Sugar is obtained from its saturated solution by |
|
Answer» SUBLIMATION |
|
| 5. |
Sugar containing an impurity of common salt can be purified by crystallisation from |
|
Answer» Benzene |
|
| 6. |
Sufficient amount of a solid X is taken in a rigid vessel at T^(@)C where it attained the equilibrium: X(s)hArrY_((g))+2Z(g) Total pressure was measured. Now the vessel is evcuated and filled with sufficient amount of another solid V under same conditions where it attained theequilibrium: Total pressure measured now is found to be double that of previous value. Now, if both X "&" V solids are allowed to attain their respective equilibrium together in the same vessel at same temperature, select the correct staement(s): |
|
Answer» `K_(P)` for decompostion reaction of `V(s)=8xxK_(P)` for decomposition reaction of `X (s)`. |
|
| 7. |
Successive ionisation potentials of an element M are 8.3, 25.1, 37.9, 259.3 and 340.1ev. The formula of its bromide is |
|
Answer» `MBr_5` |
|
| 8. |
Substrate that show E1 reaction |
|
Answer» `CH_3CH_2I` |
|
| 9. |
Subtract : (i) 0.015 from 5.16 (ii) 16.92 from 237.5 (iii) 0.0604 from 0.08638 |
|
Answer» The correct answer 5.14 (Digit 5 is to be DELETED) (ii) Subtract : `{:(237.5),(16.92),(bar(220.58)):}` The correct answer is 220.6 (Digit 5 has been rounded off to 6) (iii) Subtract : `{:(0.08638),(0.0604),(bar(0.02598)):}` The correct answer is 0.0260 (Digit 5 has been rounded off to 6) |
|
| 10. |
Substract 24.5492 from 62.831 and report the answer in significant figures. |
|
Answer» |
|
| 11. |
Substantiate lithium fluoride has the lowest solubility among group one metal fluorides |
| Answer» Solution :Lithium fluoride has high LATTICE enthalpy due to the small SIZE of `Li^(+)` and `F^(-)` So, DU high lattice enthalpy, LiF is less soluble in WATER | |
| 12. |
Substances which show permanent magnetism even in the absence of magnetic field are called ________ |
|
Answer» |
|
| 13. |
Substances which readily undergo hydrolysis are |
|
Answer» `AlCl_(3)` |
|
| 14. |
Substances which can donate lone pairs of electrons to form a coordinate covalent bonds are called |
|
Answer» Acids |
|
| 15. |
Substance which is weakly repelled bya magnetic field is |
|
Answer» `O_(2)` |
|
| 16. |
Substance which is weakly repelled by a magnetic field is |
|
Answer» `O_2` |
|
| 17. |
Substance which is weakly rapelled by a magnetic field is |
|
Answer» `O_(2)` |
|
| 18. |
Substance A_(2)B(g) can undergo decomposition to form of set of products : if the molar ratio of A_(2)(g) to A(g) is 5 : 3 in a set of product gases, then energy involved in the decomposition of 1 mole of A_(2)B is : |
|
Answer» 43.75 KJ`//`mol |
|
| 19. |
Sublimation is a process in which a solid : |
|
Answer» CHANGES into ANOTHER ALLOTROPIC form |
|
| 20. |
SubQue : Whodiscoveredproton? |
| Answer» SOLUTION :Thesmallestand lightestpositiveion wasobtainedfromhydrogenand was calledproton `(H^(+))` | |
| 21. |
Sub. Que : Give number, bond angle and bond length in ethyne. |
|
Answer» SOLUTION : In ethyne bond LENGTH, angle, shape and type of bonds : Two C -H sigma bond in it. In triple bond two `PI` and one `sigma`-bond. So total three bond.  In it C - C bond length 120 pm and C - H bond length 106 pm. Both carbon have sp hybridization . So, H - C - C - H is linear and H- C - II and C - C - H bond angle `180^(@)`. |
|
| 22. |
Sub. Que. : Explainpi -bond formation in ethyne, |
|
Answer» Solution :Each of the two unhybridised p ORBITALS of both CARBON atoms overlap SIDEWISE to form two `pi`-bond between the carbon atoms. So the triple bond between the carbon atoms is MADE up of one sigma and two `pi` BONDS. 
|
|
| 23. |
Sub Que : Give Bond length, Bond angle in ethene. |
|
Answer» Solution :Bond length ANG angle in ETHENE : C - C Bond length is 134 pm C - H Bond length is 108 pm H- C-H : `117.6^(@) , "" H-C-C : " 121^(@)` |
|
| 24. |
Sub Que. : Explain the formation pi-bond in ethene. |
|
Answer» <P> Solution :`pi` BOND structured : In ETHENE the non HYBRID p and FORM `pi`-bond between two carbon atom. In ethene `sigmaand pi` bnd can be presented as follows. 
|
|
| 25. |
Sub. Que. : Explain sigma-bond formation in ethene. |
Answer» Solution :Now one sp ORBITAL of carbon combine with sp orbital of other carbon and form sigma BOND. Remaining TWO `sp^(2)` orbital. Combine with is orbital of H and form four C-H sigma bond. In this way four C-H sigma bond and two carbon & four Hydrogen are in one plane  Now the each carbon has one non hybride 2p orbital which side by side OVERLAP and form `pi`-bond. in ethene both carbon has `sp^(2)` hybridization . So ethene is planar. 
|
|
| 26. |
Sub que : Explain sigma-bond formation in ethyne . |
|
Answer» Solution : In ethyne one sp hybrid orbital of one carbon atom OVERLAPS axially with sp hybrid orbital of the other carbon atom to form C-C SIGMA bond. While the other hybrids of orbital of each carbon atom overlaps axially with half filled s orbital of hydrogen atoms forming `sigma` honds. sp orbital are linear so all the four atoms H-C-C- H arrange in linear SHAPE figure of `sigma`-bond   Still 2PX and 2py orbital are perpendicular to H-C-C-H bond AXIS. |
|
| 27. |
Styrene (C_(6)H_(5)CH=CH_(2)) is treatedwith HBrpresenceof perxides. |
|
Answer» Solution :Additionoceursaccording toanti- Markovnikov's rule . `C_(6)H_(5)CH =CH_(2)+ HBroverset("PEROXIDES ")UNDERSET("(Anti- Mark adition)") (to) underset(1-"Bromo-2- phenylethane ")(C_(6)H_(5)CH_(2)CH_(2)Br)` |
|
| 28. |
Styphinic acid |
|
Answer» 2,4,6-Trinitrophenol 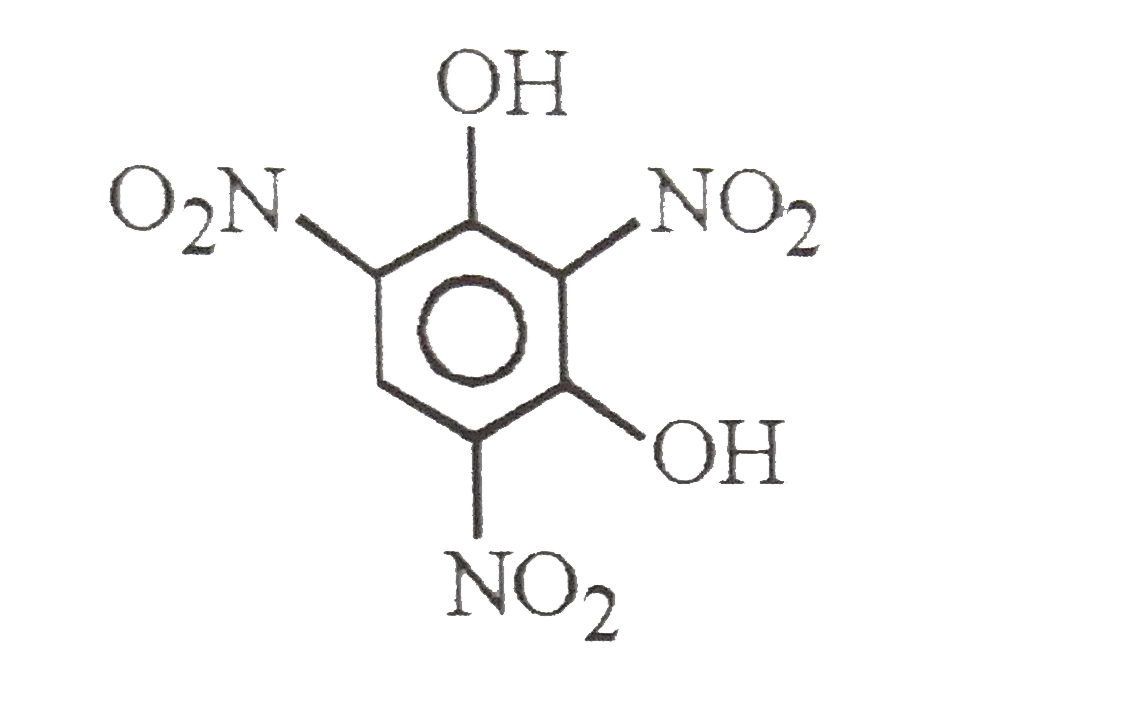
|
|
| 29. |
Study the following reactions and mark the correct properties shown by water. (i) SO_(3) + H_(2)O to H_(2)SO_(4) (ii) CI_(2)O_(7) + H_(2)O to 2HCIO_(4) (iii)CaO + H_(2)O to Ca(OH)_(2)(iv) Na_(2)O + H_(2)O to 2NaOH |
|
Answer» All oxides react with water to give hydroxides. |
|
| 30. |
Study the following reactants// product and choose the correct option of reagents for path Xand Y respectively. |
|
Answer» |
|
| 31. |
Study the following isotherms of carbondioxide at various temperature and answer the following questions based on that: At 21.5°C decrease in volume(B-C) doesn't result into increase in pressure because |
|
Answer» The compression CAUSES increase in solidification |
|
| 32. |
Study the following isotherms of carbondioxide at various temperature and answer the following questions based on that: Carbondioxide present at F can be liquified |
|
Answer» At CRITICAL volume by compression To LIQUIFY, T should be dec and then should be inc. |
|
| 33. |
Study the following isotherms of carbondioxide at various temperature and answer the following questions based on that: Choose the temperature above which CO_(2)cannot be liquified whatsoever applied pressure is |
|
Answer» 290K `T_C = 30.98^@C = 303.98 K`. |
|
| 34. |
Studiedtheradioactivityand developed it ._(6) ^(14)C._(6)^(13) C ._(7) ^(14) N._(6)^(12) C |
|
Answer» SOLUTION :ISOBARS`._(6)^(14) C._(7)^(14)N ` …. Samemassnumber Isotops `._(6)^(14)C ._(6)^(15) N ,_(6)^(12) C`….. Sameatomicnumber |
|
| 35. |
Structures of molecules of two compounds are given below : (a) Which of the two compounds will have intennolecular hydrogen bonding and which compound is expected to show intramolecular hydrogen bonding. (b) The melting point of a cornpound depends on, among other things, the extent of hydrogen bonding. On this basis explain which of the above two cornpounds will show higher melting point. (c) Solubility of compounds in water depends on power to form hydrogen bonds with water. Which of the above compounds will form hydrogen bond with water easily and be more soluble in it. |
|
Answer» Solution :(a) In compound (I) Intramolecular H-bonding is formed Here H-atom, in between the two HIGHLY ELECTRONEGATIVE atoms, is PRESENT within the same molecule. In ortho-nitrophenol (compound n. H-atom is in between the two oxygen atoms.  Compound (II) forms intermolecular H- bonding. In para-nitrophenol cm there is a gap between `NO_(2)` and OH group. So, H-bond exists between H-atom of one molecule and O -atom of another molecule as shown below.  (b) Compound (ID Possess higher melting point because large number of molecules are JOINED together by H-bonds. (c) Due to intramolecular H bonding, compound (I) is not able to form H-bond with water, so it is less soluble in water. While molecules of compound II form H-bonding with H20 easily, so it is soluble in water. |
|
| 36. |
Structures and IUPAC names of some hydrocarbons are given below. Explain why the names given in the parenthesis are incorrect. (i) {:(CH_(3)-CH-CH_(2)-CH_(2)-CH-CH-CH_(2)-CH_(3)),("|""|""|"),(""CH_(3)""CH_(3)" "CH_(3)),("2,5,6-Trimethyloctane"),("(and not 3,4,7-Trimethyloctane)"):} (ii) {:(CH_(3)-CH_(2)-CH-CH_(2)-CH-CH_(2)-CH_(3)),("|""|"),(""CH_(3)CH_(2)""CH_(3)),("3-Ethyl-5-methylheptane"),("(and not 5-Ethyl-3-methylheptane)"):} |
|
Answer» Solution :(i) The set of locants (2,5,6) is lower than the set of locants (3,5,7). (ii) When substituents are at equivalent positions, lowest LOCANT is given to that substituents which comes first in the alphabetical ORDER. Therefore, lower locant 3 is assigned to ETHYL and HIGHER locant 5 is assigned to methyl GROUP. |
|
| 37. |
Structures and IUPAC names of some hydrocarbons are given below. Explain why the names given in the parentheses are incorrect. (a) underset("2, 5,6-Trimethyloctane (and not 3,4,7-Trimethyloctane)")(CH_(3) - underset(underset(CH_(3))(|))(CH)- CH_(2) - CH_(2) - underset(underset(CH_(3))(|))(CH)-underset(underset(CH_(3))(|))(CH)-CH_(2) - CH_(3)) (b) underset("3-Ethyl-5-methylheptane (and not 5-Ethyl-3-methylheptane)")(CH_(3) - CH_(2) - underset(underset(CH_(2)CH_(3))(|))(CH)- CH_(2) - underset(underset(CH_(3))(|))(CH)- CH_(2) - CH_(3)) |
|
Answer» Solution :(a) LOWEST locant number, 2, 5, 6 is lower than 3, 5, 7 (b) Substituents are in equivalent position, lower number is given to the one that comes FIRST in the name according to alphabetical order |
|
| 39. |
Structure of the compound whose IUPAC name is 5, 6-dimethylhept-2-ene is ........... . |
|
Answer»
|
|
| 40. |
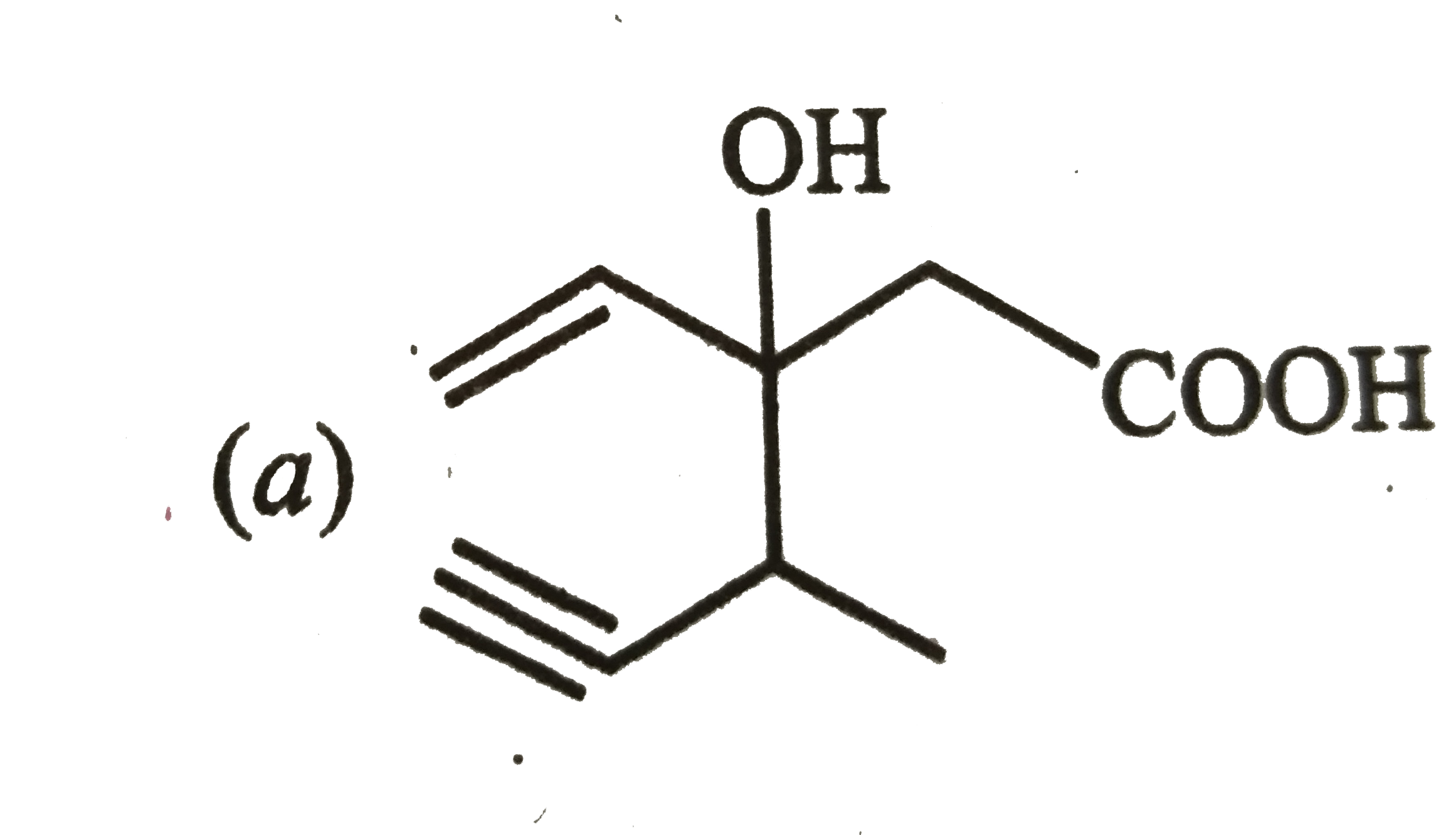
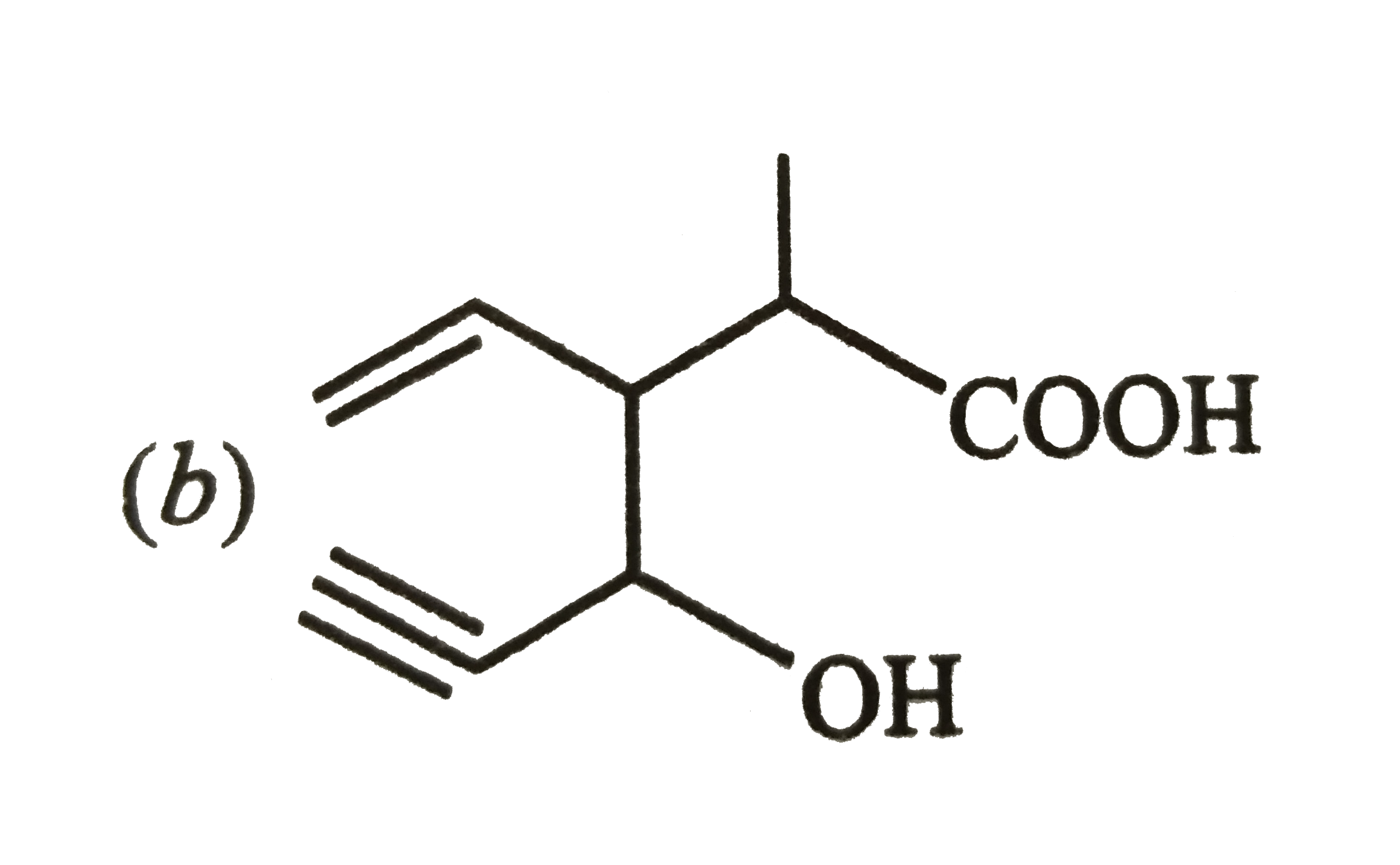
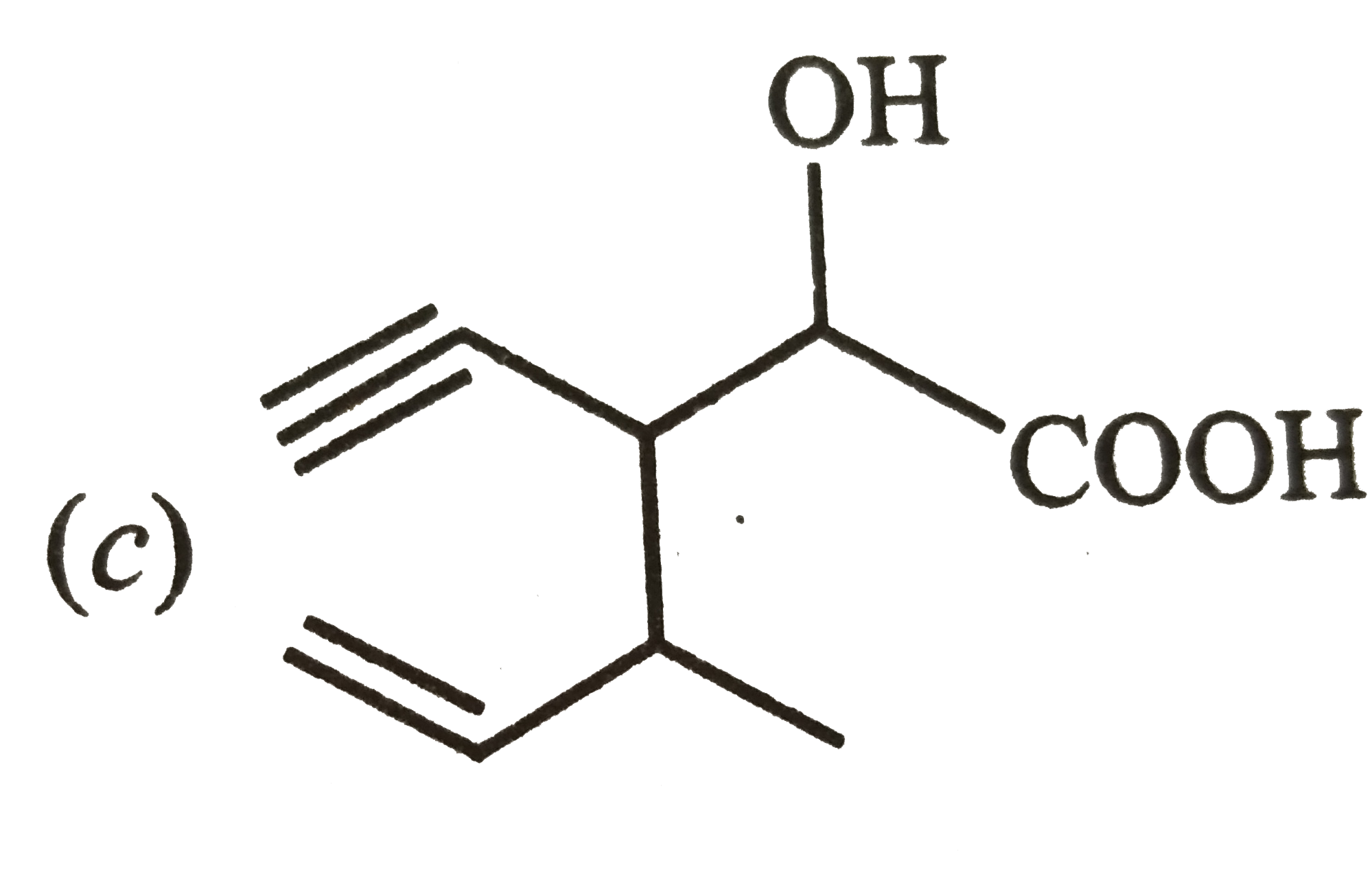
Structure of the compound whose IUPAC name is 3-ethyl-2-hydroxy-4-methylhex-3-en-ynoic acid is |
|
Answer»
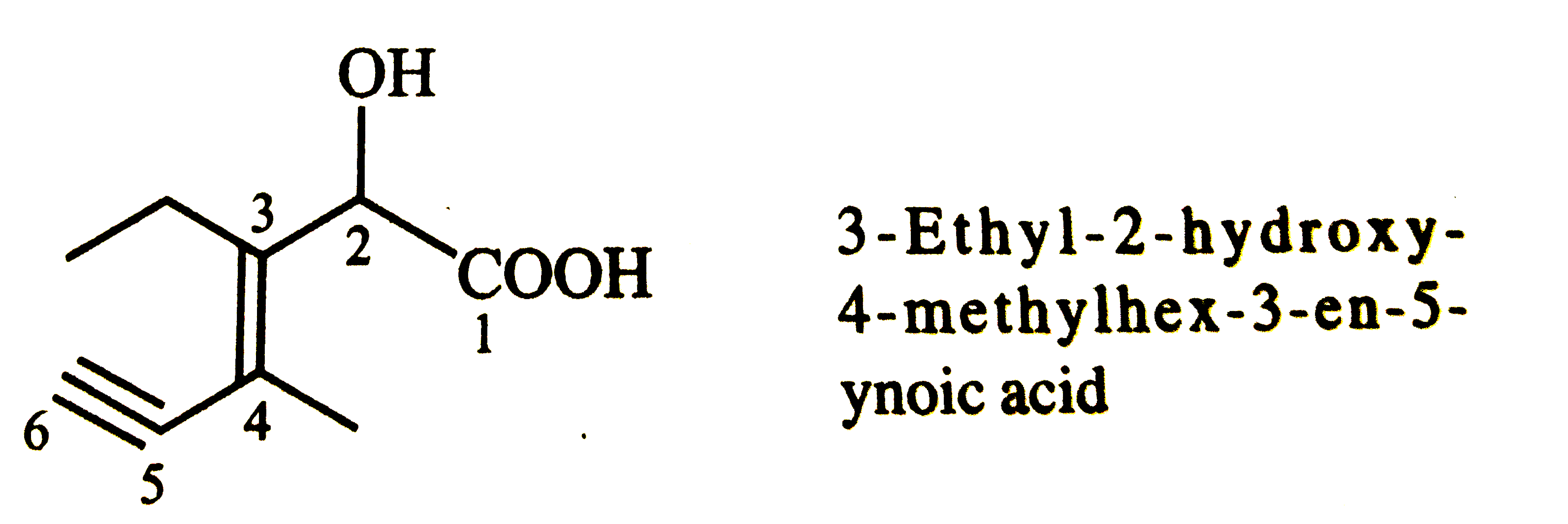
|
|
| 41. |
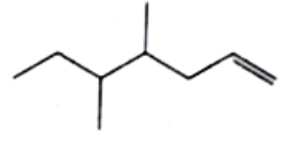
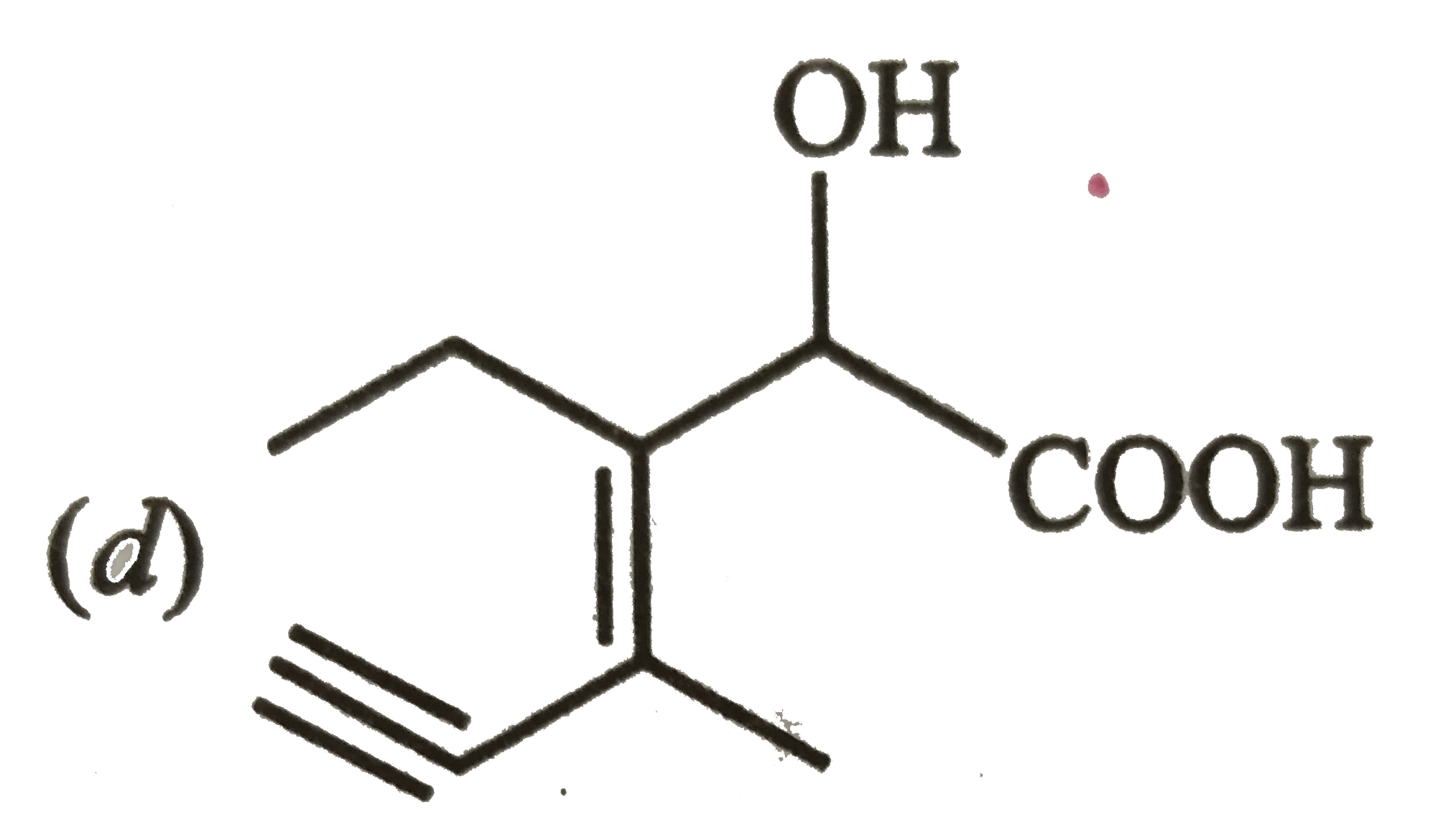
Structure of the compound whose IUPAC name is 3-Ethyl -2-hydroxy -4-methylhex-3-en-5-ynoic acid is |
|
Answer»
|
|
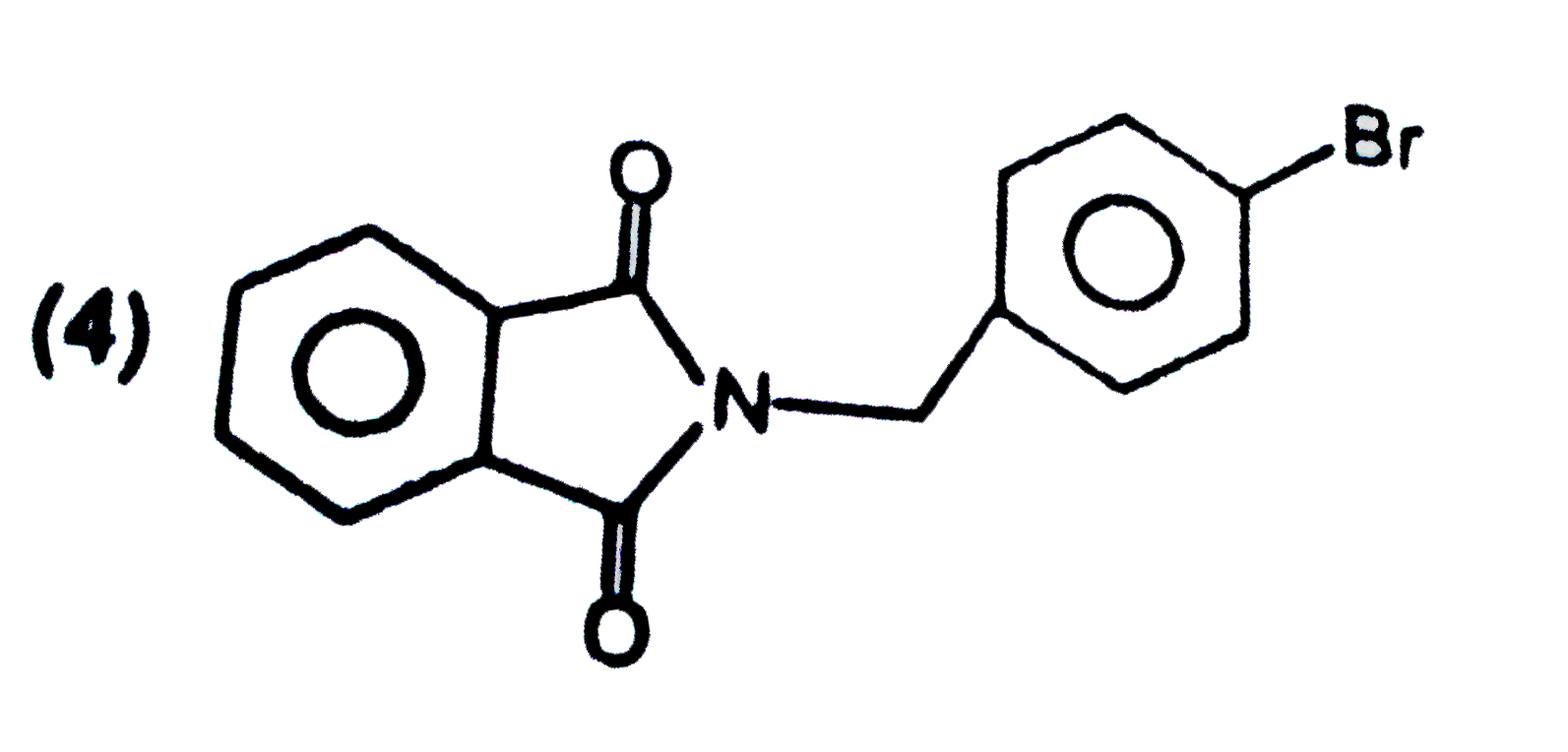
| 42. |
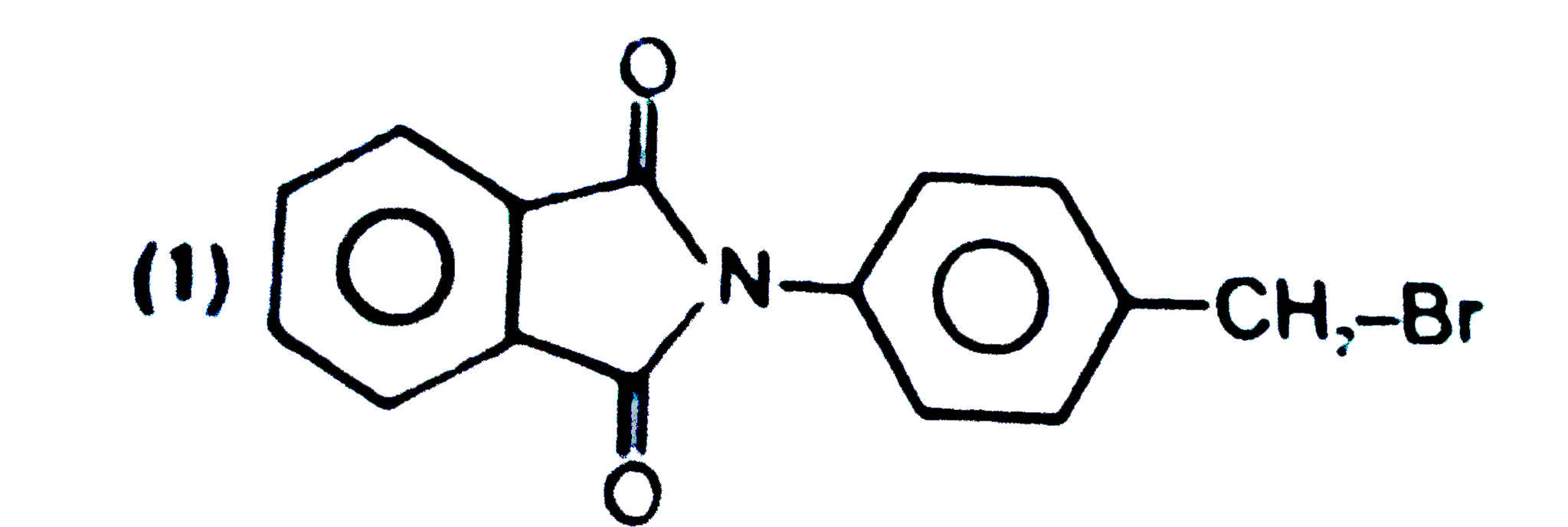
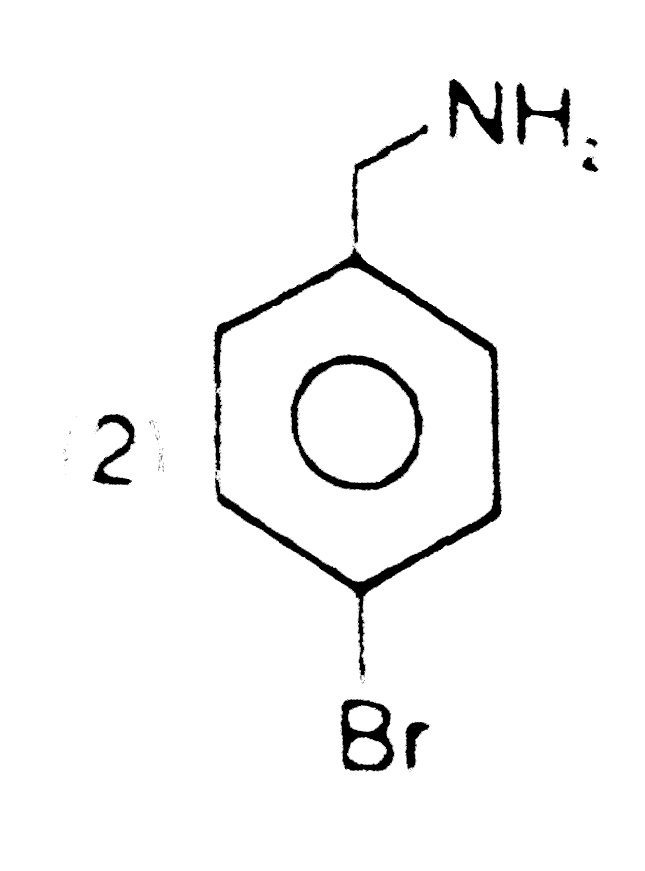
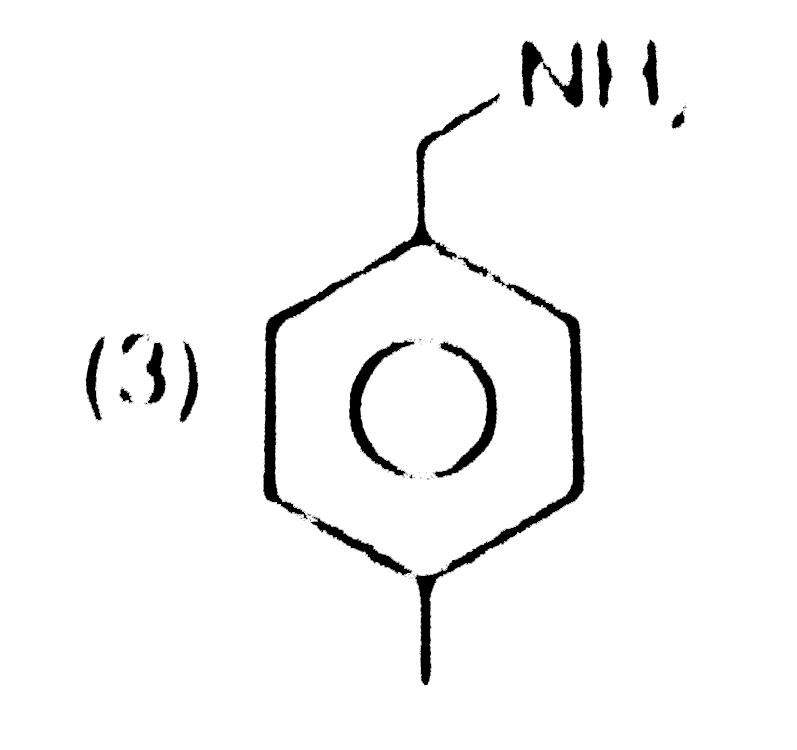
Structure of product is |
|
Answer»
|
|
| 43. |
Structure of is : |
|
Answer»

|
|
| 46. |
Structure of a mixed oxide is cubic close-packed (c.c.p.) The cubic cell of mixed oxide is composed of oxide ions. One fourth of the tetrahedral voids are occupied by divalent metal A and the octahedral voids are occupied by a monovalent metal B . The formula of the oxide is : |
|
Answer» `A_2B_3O_4` `therefore` No. of octahedral voids=n No. of TETRAHEDRAL voids=2n Divalent metal ions A present =`"2n"/4=n/2` Monovalent Metal ions B present =n `therefore` Ratio `A:B:O^(2-)=n/2:n:n=1/2:1:1` HENCE, formula =`AB_2O_2` |
|
| 47. |
Structure of a mixed oxide is cubic close-packed (c.c.p)the cubic unit cell of mixed oxide is composed of oxide ions. One fourth of the tetrahral voids are occupied by divalent metal A and the octahedral voids are occupied by a monovalent metal B. the formula of the oxide is : |
|
Answer» `A_(2)B_(3)O_(4)` No. of octahedral VOIDS = n No. of tetradral voids = 2n Divalent METAL ions A present = `(2n)/4 = n/2 ` Monovalent metal ions B present =n Ratio `A :B : O^(2-) = n/2 : n :n = 1/2 :1:1 ` Hence, formula = ` AB_(2)O_(2)` |
|
| 48. |
Structural of Ascorbic acid is represented as follows How many of the following reagents can gives positive test with ascorbic acid. {:(Cu_(2)Cl_(2)+NH_(4)OH,"2,4"-DNP,"Na Metal",HCL+ZnCl_(2),FeCl_(3)),("(I)","(II)","(III)","(IV)","(V)"),("NAOH+Phenophthalein",dil. KMNO_(4),Br_(2)//H_(2)O,AgNO_(3)+NH_(4)OH,I_(2)+NaOH),("(VI)"," (VII)"," (VIII)","(IX)","(X)"):} Number of compound which gives positive Haloform test=(x) Number of compound which can gives positive Lucas reagent test=(y) Report your answer (x+y) |
|
Answer» |
|
| 49. |
Structural isomers have different covalent linkage of atoms. Stereoisomers are compounds that have same sequence of covalent bonds but differ in the relative dispositions of their atoms in space. Geometrical and optical isomers are the two important types of configurational isomers. The compound with double bonds or ring structure have restricted rotation, so exist in two geometrical forms. The double bonds in larger rings (ring size 10 carbon large) can also cause geometrical isomerism. The optical isomers rotate the plane of plane-polarised light. A sp^3 -hybridised carbon atom bearing four different types of substituents is called an asymmetric centre or chiral centre. A chiral object or molecule cannot be superimposed on its mirror image. Stereoisomers that are mirror images of each other are called enantiomers. The stereosomers that the pot mirror images of each other are called diastereomers. Diasteremers have different physical properties.A racemic mixture is optically inactive and contains equal amounts of both the enantiomers. Resolution refers to method of separating a racemic mixture. Into two pure enantiomers. A meso compound is an optically inactive stereoisomer, which is achiral due to the presence of an internal plane of symmetry or centre of symmetry within the molecule.The pair of showing identical species is |
|
Answer»

|
|
| 50. |
Structural isomers have different covalent linkage of atoms. Stereoisomers are compounds that have same sequence of covalent bonds but differ in the relative dispositions of their atoms in space. Geometri cal and optical isomers are the two important types of configurational isomers. The compound with double bonds or ring structure have restricted rotation, so exist in two geometrical forms. The double bonds in larger rings (ring size 10 carbon large) can also cause geometrical isomerism. The optical isomers rotate the plane of plane-polarised light. A sp^(3)-hybridised carbon atom bearing four different types of substituents is called an asymmetric centre or chiral centre. A chiral object or molecule cannot be superimposed on its mirror image. Stereoisomers that are mirror images of each other are called enantiomers. The stereosomers that the pot mirror images of each other are called diastereomers. Diasteremers have different physical properties. A racemic mixture is optically inactive and contains equal amounts of both the enantiomers. Resolution refers to method of separating a racemic mixture. Into two pure enantiomers. A meso compound is an optically inactive stereoisomer, which is achiral due to the presence of an internal plane of symmetry or centre of symmetry within the molecule. The pair of the following that showing identical species is |
|
Answer»

|
|