Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 2. |
Sulphur dioxide and hydrogen peroxide can act as an oxidising as well as a reducing agents in their reactions, while ozone and nitric acid act only as an oxidants. Why? |
|
Answer» Solution :(a) `SO_(2)` : In `SO_(2)`, oxidation NUMBER of S is +4. Oxidation number of S ranges from -2 to +6. Therefore, in `SO_(2)` oxidation number of S can vary. S can acts as an oxidation agent or as a reducing agent. (b) `H_(2)O_(2)` : In `H_(2)O_(2)` oxidation number of O is -1. GENERALLY minimum oxidation number of oxygen is -2 and maximum oxidation number of oxygen is 0. (Exception : In `O_(2)F_(2)` oxidation number of oxygen is +1.) So, oxidation number of oxygen of `H_(2)O_(2)` is increasing by -1 to 0 and decreases by -1 to -2. So, `H_(2)O_(2)` act as oxidising agent and reducing agent. ( c) `O_(3)` : Oxidation number of oxygen in `O_(3)` is zero. Here oxidation number of oxygen reduced from zero to -1 or -2. Therefore, it acts as an oxidizing agent. (d) `HNO_(3)` : Nitrogen is `HNO_(3)` oxidation number is +5. Therefore, its oxidation number it can only decreased. Therefore, `HNO_(3)` can only used as an oxidizing agent. |
|
| 3. |
Sulphur colloid is prepared by |
|
Answer» mechanical dispersion `Br_(2)+H_(2)Sto2HBrunderset((sol))(S)` |
|
| 4. |
Sulphur and fluorine form SF_(6) and S_(2)F_(10), both of which are gases at 30^(@)C. When an equimolar mixture of them is allowed to effuse through a pinhole, what is the ratio SF_(6)//S_(2)F_(10) in the first sample that escapes? {:("Molar mass",g"mol"^(-1)),(SF_(6),146),(S_(2)F_(10),254):} |
|
Answer» `(1.32)/(1)` |
|
| 5. |
Sulphur atom of which oxo acid have non bonding electron pair ? |
|
Answer» SULPHUROUS ACID |
|
| 6. |
Sulphonation of phenol with conc. H_(2)SO_(4) at 288-293 K gives |
|
Answer» o-Phenolsulphonic ACID |
|
| 7. |
Sulphide ores of metals are usually concentrated by froth floatation process. Which one of the following sulphide others offers an exception and is concentrated by chemical leaching ? |
|
Answer» Sphalerite |
|
| 8. |
Sulphide lon reacts with solid sulphur S_((aq))^(2-)+S_((s))hArrS_(2(aq))^(2-)," "K_(1)=10 S_((aq))^(2-)+2S_((s))hArrS_(3(aq))^(2-)," "K_(2)=130 The equilibrium constant for the formation of S_(3)^(2-)(aq) from S_(2)^(2-)(aq) and sulphur is |
|
Answer» 10 |
|
| 9. |
Sulphide minerals are converted to corresponding oxides by "___________________". |
|
Answer» CRACKING |
|
| 10. |
Sulphates of which of the following does not form alum |
|
Answer» Cs |
|
| 11. |
Sulphide ioninalkaline solutionreacts withsolidsulphur toform polysulphide ions havingformulae S_2^(2-), S_3^(2-), S_4^(2-) and so on. The equilibrium constant for the formationof S_2^(2-) is 12(K_1) andfor the formation of S_3^(2-) is 132(K_2) both from S and S^(2-). What is the equilibrium constant for the formation of S_3^(2-) from S_2^(2-) and S ? |
|
Answer» Solution :`S+S^(2-) harr S_(2)^(2-), K_(1)=12` `2S+S^(2-) harr S_(3)^(2-), K_(2)=132` `S_(2)^(2-)+S harr S_(3)^(2-), K=(K_(2))/(K_(1))=(132)/(12)=11` |
|
| 12. |
Sulphanilic acid is |
|
Answer» Arrhenius acid  It CONTAINS acidic `-SO_(3)H` group which makes it Arrhenius acid. Also, it contains `-NH_(2)` group which can a PAIR of electrons and hence is a Lewis base |
|
| 13. |
Sulphadrugs are : |
|
Answer» salvarsan |
|
| 14. |
Sulhuyr dioxide affects |
|
Answer» CELL WALL |
|
| 15. |
Sulfonation like iodination is reversible and is believed to take place in concentrated sulfuric acid via the following pathway Find the correct statement(s) for above given reaction |
|
Answer» BENZENE is sulfonated fairly slowly by hot concentrated sulphuuric acid but rapidly by oleum. (RATE than being related to `SO_3` content) |
|
| 16. |
Suggest why there is no hydrogen (H_(2)) in our atmosphere. Why does the moon have no atmosphere? |
|
Answer» Solution :Hydorgen is the third most abundant element in the earth's surface, HOWEVER, hydrogen gas is very rare in the earth's atomsphere due to its light weight, which enables it to escape from earth's gravity easily than heavier GASES. MOON has low gravity and so have low escape velocitly. The atmospheric gases on the surface of the moon have thermal VELOCITIES greater than the escape velocity. That is the reason why molecules of gases eacapes and there is no atmosphere on moon. |
|
| 17. |
Suggest the suitable reagent for the following transformation. |
|
Answer» meta-chloroperbenzoic ACID |
|
| 18. |
Suggest the spontaneity for the thermal decomposition of lime stone and discuss. |
| Answer» SOLUTION :SPONTANEOUS at HIGH TEMPERATURE | |
| 19. |
Suggest the product of following reaction: |
|
Answer»

|
|
| 20. |
Suggest the route for the preparation of the following from benzene. 1.3-chloro-nitrobenzene2. 4-chlorotoluene 3. Bromobenzene4. m - dinitrobenzene |
Answer» SOLUTION :Preparation of 3-chloronitrobenzene from BENZENE: Benzene under ONS and FOLLOWED by chlorination and it leads to ination and it leads to the formation of 3-chloronitrobenzene. 
|
|
| 21. |
Suggest theroute for the preparation of the followingfrom benzene 1 3-chloro nitrobenzene 2 4-chlorotoluene 3 Bromobenzene 4 m-dinitrobenzene |
Answer» Solution :Preparation of 3-chloronitro - BENZENE from benzene : Benzene undergoes nitration and followed by chlorination and it LEADS to the formation of 3-chloronitrobenzene . 2. Preparation 4-chlorotoluence from benzene : Benzene undergoes FRIEDEL cradf.s alkylation followed by chlorination and it leads to formation of 4-chlorotoluene.  3. Preparation of Bromobenzene from benzene : Benzene undergo bromination to give bromobenzene .  4. preparation of m-dintrobenzene from benzene : Benzene undergo twice the TIME nitration to give m-dinitrobenzene. 
|
|
| 22. |
Suggest the name of a Lewis acid other than anhydrous aluminium chloride which can be used during ethylation of benzene. |
| Answer» SOLUTION :`FeCl_3, BF_3, ETC` | |
| 23. |
Suggest the conditions for the spontaneity the thermal decomposition of lime stone. |
|
Answer» |
|
| 24. |
Suggest reasons why the B–F bond lengths in BF_(3) (130 pm) and BF_(4 )^(– )(143 pm) differ. |
| Answer» Solution :DUE to `P pi-P pi` back bonding in `BF_3` molecule (for details, (3), Figs.B-F BOND acquires some double bond character. This shortens the B-F bond LENGTH. In `BF_4^-`, the B-F bond is a pure single bond and has a LARGER bond length. This is why B-F bond length in `BF_3` is shorter as compared to that in `BF_4^-` . | |
| 25. |
Suggest reasons why the B_F bond length in BF_(3)(130 pm) and BF_(4)^(ө)(143 pm) differ. |
|
Answer» Solution :In `BF_(3)`,B is `sp^(2)` hybridised and has only six electrons in the valence shell and behaves as an electron-deficient compound. To reduce its electron deficiency, `B` accept a PAIR of electron from filled `2p` orbital present on it. This is known as `ppi-ppi` back bonding which happens due to compatibility in the size and energy of `2p`orbital on `F` and `2p` orbital on `B` atom in `BF_(3)`. Due to back-bonding in `BF_(3)` `B-F` bond ACQUIRES some double bond character resulting in shortening of the bond. However, in `[BF_(4)](ө)`,B is `sp^(3)` hybridised and hence has eight electrons in its valence shell. Since there is an EMPTY `p`-orbital available in `B` in `[BF_(4)](ө)` ION, back-bonding is not possible and `B-F` is purely single bond. Since double bonds are shorter than single bond, the `B-F` bond length in `BF_(3)` is shorter than in `[BF_(4)](ө)`. 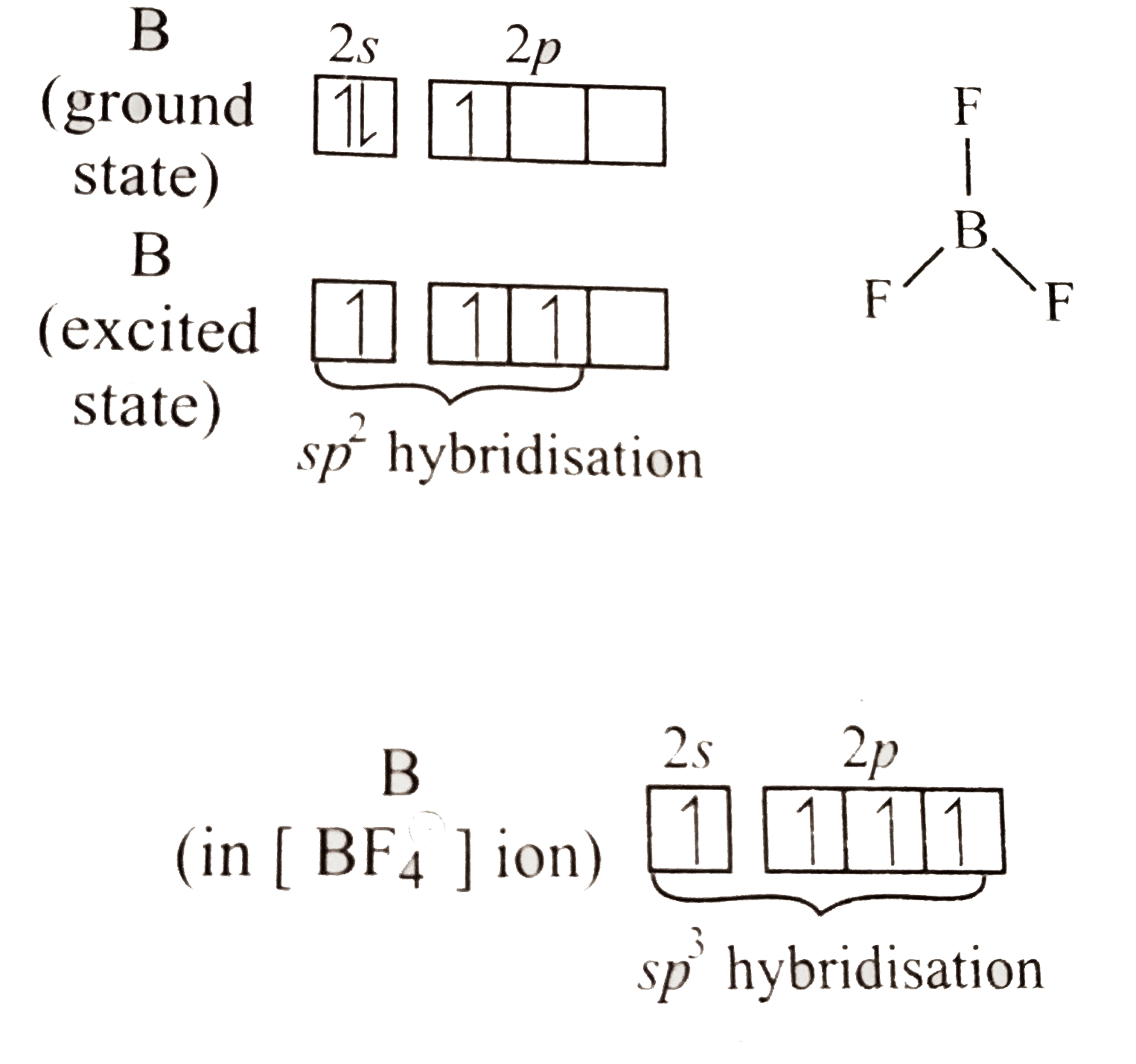 
|
|
| 26. |
Suggest reasons why the B-Fbond lengths in BF_(3) (130 pm) and BF_(4)^(-) (143 pm) differ. |
|
Answer» Solution :`BF_(3)` is a planar MOLECULE in which B is `sp^(2)`-hybridized. It has an empty 2p-orbital F-atom has threelone pairs of electronin the 2p-orbitals. Because of similar sizes, `ppi-ppi` back bondingoccurs in whicha lone pair of electrons is transferredfrom F to B as shownbelow : As a result of this back bonding , `B-F`bond acquiressome doublebond character . In contrast, in `[BF_(4)]^(-)`ION, B is `sp^(3)`-hybridizedand hencedoes not havean empty p-orbitalsavailableto acceptthe electronsdonatedby the F atom. Consequently, in `[BF_(4)]^(-), B-F` is a purely SINGLE bond. Sincedoublebonds areshorter than singlebonds,therefore, the B-F bond length in `BF_(3)` is SHORTER (130 pm) than `B-F` bond length(143) pm in `[BF_(4)]^(-)` 
|
|
| 27. |
Suggest reasons why the B - F bond lengths in BF_3 (130 pm) and BF_4^- (143 pm) differ. |
|
Answer» Solution :The B-F bond length in `BF_3` is shorter than the B-F bond length in `BF_4^-`. `BF_3` is an electron deficient species. With a vacant p-orbital on boron, the FLUORINE and boron atoms undergo `ppi-ppi` back-bonding to remove this deficiency. This imparts a double bond character to the B-F bond.  This double-bond character causes the bond length to shorten in `BF_3` (130 pm). HOWEVER, when `BF_3` coordinates with the FLUORIDE ion, a change in HYBRIDISATION from `sp^2` (in `BF_3`) to `sp^3` (in `BF_4^-`) occurs. Boron now forms 4`sigma`- bonds and the double-bond character is lost. This accounts for a B-F bond length of 143pm in `BF_4^-` ion. 
|
|
| 28. |
Suggest name of another Lewis acid instead of anhydrous aluminium chloride which can be used during ethylation of benzene. |
| Answer» SOLUTION :`FeCl_3` can ALSO be USED as a LEWIS ACID. | |
| 29. |
Suggest methods for the separation of the following mixtures. (i) A mixture of liquid A (b.p. 365 K) and liquid B (b.p. 356 K). (ii) A mixture of liquid C (b.p. 353 K) and liquid D (b.p. 413 K). |
|
Answer» Solution :(i) Fractional distillation because the boiling POINTS of the two liquids differ by just `9^(@)`. (II) SIMPLE distillation since the boiling points of the two liquids are widely APART. |
|
| 30. |
Suggest methods for the separation of following mixtures: (i) A mixture of liquid A (b.p. = 356 K) and liquid B (b.p. = 365 K) (ii) A mixture of liquid C (b.p. = 360 K) and liquid D (b.p. = 395 K) (iii) A liquid 'E' (b.p. = 563 K) having decomposition temperature 450 K. |
| Answer» SOLUTION :(i) Fractional distillation (II) Simple distillation (iii) Distillation under reduced PRESSURE. | |
| 31. |
Suggest appropriate structures for the missing compounds. (The number of carbon atoms remains the same throughout the reactions). Compound (A),(B) & ( C) can be |
|
Answer»

|
|
| 33. |
Suggest and explain an indirect method to calculate lattice enthalpy of sodium chloride crystal. |
|
Answer» Solution :Let us use the Born- Haber cycle for determining the LATTICE enthalpy of Nacl as follows : Since the reaction is carried out with REACTANTS in elemental forms and products in their standard states, at I bar, the overcall enthalpy change of the reaction is also the enthalpy of formation for NaCl. Also , the formation of NaCl can be considered in 5 steps . Thesum of the enthalpy changes of these steps is EQUAL to the enthalpy change for the overall reaction from which the lattice enthalpy of NaCl is calculated. Let us calculate the lattice energy of sodium CHLORIDE using Born- Haber cycle  `Delta^(@)H_(f)`=heat of formation of sodium chloride `= -411.3kJmol^(-1)` `Delta^(@)H_(1)`= Heat of sublimation of `Na_((S))=108.7kJ mol^(-1)` `Delta^(@)H_(2)` = ionisation energy of `Na_((g))=495.0 kJ mol^(-1)` `Delta^(@)H_(3)` = dissociation energy of `Cl_(2_((g))) =244kJ mol^(-1)` `Delta^(@)H_(4)` = Electron AFFINITY of `Cl_((g))=-349.0 kJ mol ^(-1)` U = Lattice energy of NaCl `Delta^(@)H_(f) = Delta^(@)H_(1) +Delta^(@)H_(2)+1//2Delta^(@)H_(3)+Delta^(@)H_(4)+Delta^(@)H_(5)` `:. Delta^(@)H_(5)=(Delta^(@)H_(f))-(Delta^(@)H_(1)+Delta^(@)H_(2)+1//2Delta^(@)H_(3)+Delta^(@)H_(4))` `implies Delta^(@)H_(5)=(-441.3)-(108.7+495.0+122-349)` `Delta^(@)H_(5)=(-441.3)-(376.7)` `Delta^(@)H_(5)=-788kj mol^(-1)` The negative p sign in lattice energy indicates that the energy is released when sodium is formed from its constituent gaseous ions `Na^(+) and Cl^(-)` |
|
| 34. |
Suggest and explain an indirect method to calculate lattice enthalpy of magnesium bromide. |
|
Answer» Solution :Born Haber.s cycle method: `Mg(s)+Br_(2)(l) to MgBr_(2)(s) "" DeltaH_f^0` Sublimation : `Mg_((s)) to Mg_((g)) "" DeltaH_1^0` Ionisation : `Mg_((g)) to Mg_((g))^(2+) + 2e^(-) "" DeltaH_2^0` Vapourisation : `Br_(2(l)) to Br_(2(g)) "" DeltaH_3^0` Dissociation : `Br_(2(g)) to 2Br_((g)) "" DeltaH_4^0` Electron affinity : `2Br_((g)) + 2e^(-) to 2Br_((g))^(-) "" DeltaH_5^0` LATTICE ENTHALPY : `Mg_((g))^(2+) + 2Br_((g))^(-) to MgBr_((2)s) "" DeltaH_6^0` =? `DeltaH_f^0 = DeltaH_1^0+DeltaH_2^0 + DeltaH_3^0+ DeltaH_4^0 + DeltaH_5^0 + DeltaH_6^0` `DeltaH_6^0=DeltaH_f^0 -(DeltaH_1^0+DeltaH_2^0+DeltaH_3^0+DeltaH_4^0+DeltaH_5^0)` If we KNOW the VALUES of `DeltaH_f^0,DeltaH_1^1,DeltaH_2^0, DeltaH_3^0, DeltaH_4^0` and `DeltaH_5^0` . We can calculate the value of `DeltaH_6^0` by indirect method. |
|
| 35. |
Suggest a scheme of classification of the following redox reactions. (a) N_(2(g))+O_(2(g))to2NO_((g)) (b) 2Pb(NO_(3))_(2(s))to2PbO_((s))+2NO_(2(g))+1/2O_(2(g)) ( c) NaH_((s))+H_(2)O_((l))toNaOH_((aq))+H_(2(g)) (d) 2NO_(2(g))+2OH_((aq))^(-)toNO_(2(aq))^(-)+NO_(3(aq))^(-)+H_(2)O_((l)) |
| Answer» Solution :In reaction (a), the COMPOUND nitric oxide is formed by the combination of the elemental substances, nitrogen and oxygen, therefore, this is an example of combination redox reactions. The reaction (B) involves the breaking down of lead nitrate into three components, ( c), hydrogen of water has been DISPLACED by hydride ion into DIHYDROGEN gas. Therefore. this may be called as displacement redox reaction. The reaction (d) involves disproportionation of `NO_(2)` (+4 state) into `NO_(2)^(-)` (+3 state) and `NO_(3)^(-)` (+5 state). Therefore reaction (d) is an example of disproportionation redox reaction. Therefore, this is categorised under decomposition redox reaction. | |
| 36. |
Suggest a simple chemical test to distinguish propane and propene |
|
Answer» Solution :Chemicaltest todistinguishbetweenpropaneand propene (i) Brominewatertest :Propenecontainsdoublethereforewhen wepourthebrominesaturatedhydrocarbondoesnot decolourisethe brominewater . (ii)Baeyer.stestWhenpropenereactswith Bayer.sreagentit gives `1,2`dihyroxypropenepropanedoesnot reactwith Baeyer.sreagent. `(a) CH_(3)-CH=CH_(3)UNDERSET(H_(5)O)overset(KMnO_(4))(to)CH- CH- CH_(3)` `(b)CH_(3) - CH_(2)- CH_(4)underset( H_(2)O) overset(KMnO_(4))(to)` NOREACTION Propene |
|
| 37. |
Suggest a scheme ofclassification of the following redox reaction (a)N_(2)(g)+O_(2)(g)rarr2NO(g),(b)Pb(NO_(3))_(2)rarrPbo(s)+2NO_(2)(g)+1/2O_(2)(g) (c )NaH(s)+H_(2)O(l)rarrNaOH(aq)+H_(2)(g) (d) 2NO_(2)(g)+2OH^(-)(aq)rarrNO_(2)^(-)(aq)+H_(2)O(l) |
|
Answer» Solution :(a) ` oveset(N)_(2)+overset(0)O_(2)(g)rarr2 overset(+2)Boverset(-2)O(g)` in this REACTION the compound nitric oxide is formed combination of elemental substances like nitrogen and oxygen therefore it isa combination reaction since in this reactionnitrogen increase form 0 in `N_(2)` to +2 NO and that of oxygen decrease from 0 in `O_(2)` gas to -2 NO therefore it is combination redox reaction (b) `overset(+2)Pboverset(+5)NO_(3)_(2)overset(triangle)rarrPbO(s)+2NO_(2)+1/2O_(2)(g)` In this reaction lead nitrate decomposes to from three product viz oxide nitrogen dioxide and oxygen therefore it is DECOMPOSTION reaction since in this reaction the oxidation number of nitrogen and that of oxygen increase from -2 in lead nitrate to 0 in `O_(2)` therefore it is a decompostion redox reaction (c ) `overset(+)Naoverset(-1)H(s)+overset(+1)H_(2)overset(-2)O(l)rarroverset(+1)Naoverset(-2)Ooverset(+1)H_(2)(g)` in this reaction hydrogen of water has been displaced by hydride ion to from dihydrogen gas therefore it is a displacement reaction since in this reaction the oxidation number of hydrogen increase from -1 in hydride ion to 0 in the displacement redox reaction (d)  this is a disproportionation reaction since here the oxidatoin of nitrogen decreases from +4 in `NO_(2)` to +3 in `NO_(2)^(-)` ion as well as increase from +4 `NO_(2)` to +5 `NO_(3)^(-)` ion |
|
| 38. |
Suggest a route to prepare ethyl hydrogensulphate (CH_3-CH_2-OSO_2-OH) starting from ethanol (C_2H_5OH). |
|
Answer» Solution :When ETHANOL is heated with conc. `H_2SO_4` at 383 K (i.e. , `110^@` C), ETHYL hydrogen sulphate is formed. `H_2SO_4 to H^(+) + underset"Hydrogen sulphate ion"(.^-OSO_2OH)` 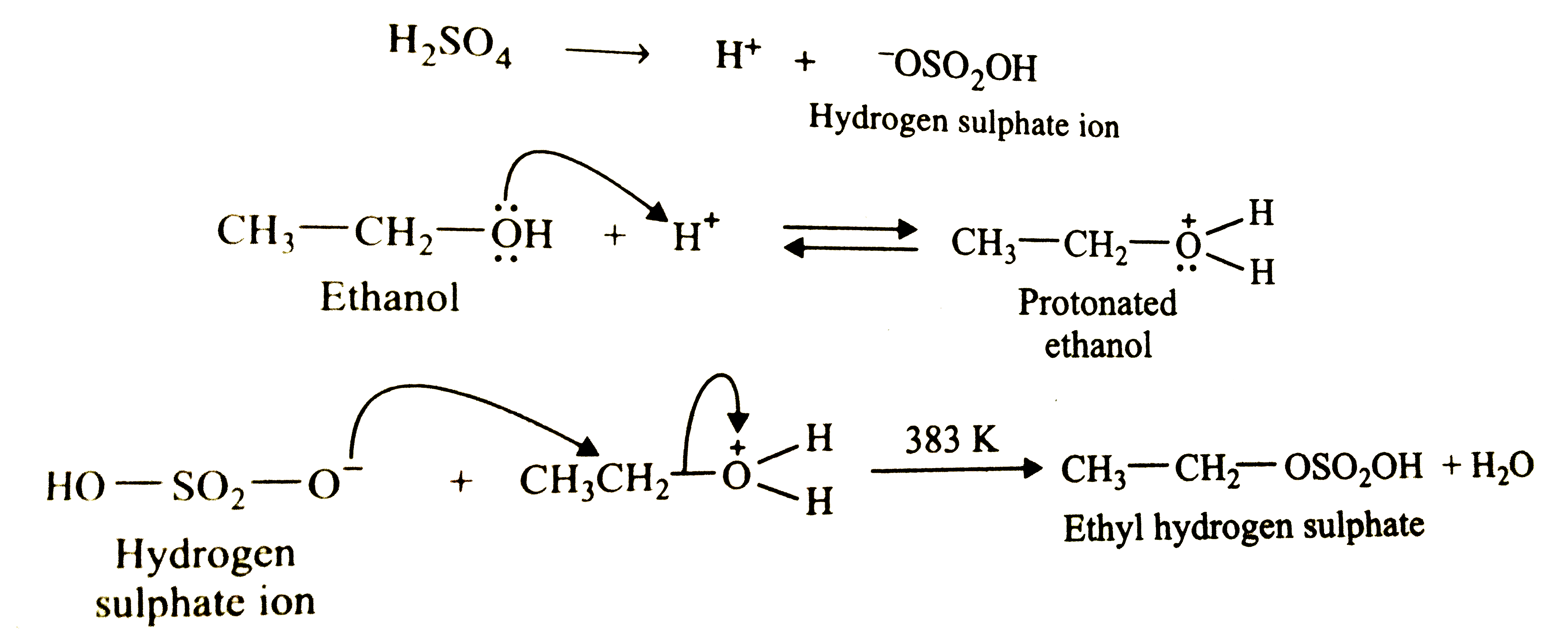 Temperature should not be allowed to rise above 383 K, otherwise diethyl ETHER will be obtained at 413 K or ethane at 433-443 K. |
|
| 39. |
Suggest a route to prepare ethyl hydrogen sulphate (CH_(3)-CH_(2)-OSO_(2)-OH) starting from ethanol (C_(2)H_(5)OH). |
|
Answer» Solution :Preparation of ethyl HYDROGEN sulphate `[CH_(3)CH_(2)-OSO_(2)-OH]` starting from ethanol. STEP -1 : Protonation of alcohol and FORMATION of carbocation. `CH_(3)-CH_(3)-O-H^(+) rarr CH_(3)-CH_(2)-""^(+)OH_(2)` `CH_(3)-Cl-H_(2)SO_(4) rarr H^(+) + ""^(-)OSO_(2)OH` Step - 2: Attack of NUCLEOPHILE `HO-SO_(2)-O^(-) + ""^(+)CH_(2) - CH_(3) rarr CH_(3)-CH_(3)-CH_(2)-O-SO_(2)-OH` |
|
| 40. |
Suggest a reason why B-F bond length in BF_3 (130 pm) and BF_4^(-) (143 pm) differ? |
|
Answer» |
|
| 41. |
Suggest a route for the preparation of nitrobenzene starting from acetylene ? |
Answer» SOLUTION :Ethane when passed through red hot iron tube at 873 K undergoes cyclic POLYMERIZATION to GIVE benzene which upon subsequentritration gives NITRO benzene. 
|
|
| 42. |
Suggest a reason as to why CO is poisonous. |
|
Answer» Solution :The haemoglobin present in blood is responsible to TRANSFER oxygen inhaled by a PERSON to different tissues of the body. Carbon monoxide combines with the haemoglobin present in the blood to FORM carboxyhaemoglobin which is about 300 times more stable than oxyhaemoglobin complex. This destroys the oxygen carrying capacity of haemoglobin to VARIOUS organs of the body. As a result the person dies of suffocation. `Haemoglobin + CO to underset("300 times more stable than oxyhaemoglobin")("Carboxyhaemoglobin")` |
|
| 43. |
Suggest a method to separate the two allotropic forms of hydrogen. |
| Answer» Solution :The ordinary hydrogen at room temperature is a mixture of 75% ortho and 25% para hydrogen. On passing through activated charcoal kept 20K, the para variety is adsorbed LEAVING behind the ortho variety. From the charcol surface, para hydrogen can be RELEASED by reducing the pressure, In this WAY, the allotropes of hydrogen can be released by reducing the pressure,. In this way, allotrpes . In this way, the allotropes of hydrogen are SEPARATED | |
| 44. |
Suggest a method to purify : (i) Camphor containing traces of common salt. (ii) Kerosene oil containing water. (iii) A liquid which decomposes at its boiling point. |
|
Answer» Solution :(i) Sublimation. Camphor sublimes while common salt remains as residue in the china dish. (II) Since the two liquids are immiscible, the technique of solvent extraction with a separating funnel is used. The mixture is thoroughly shaken and the separating funnel is allowed to stand. Kerosene being lighter tha water forms the upper layer while forms the layer. The lower water layer is run off when kerosene oil is obtained. It is dried over anhydrous`CaCl_(2)` or `MgSO_(4)` and then DISTILLED to give pure kerosene oil. (iii) Distillation under reduced pressure. Since the b.p. of a LIQUID depends upon the pressure acting on it. Therefore, a liquid which decomposes at its b.p. can be purified SAFELY at a lower temperature if the pressure acting on it reduced. |
|
| 45. |
Suggest a method to separate o-nitrophenol from p-nitrophenol. |
| Answer» SOLUTION :STEAM DISTILLATION. | |
| 46. |
Suggest a method to purify impure naphthalene. |
|
Answer» |
|
| 47. |
Suggest a method to separate the constituents from the following mixture : (i) Mixture of two miscible liquids. (ii) A mixture of oil and water. (iii) A mixture of plant pigments. (iv) A mixture of ether and water. (v) A mixture of solid nemzoic acid and sodium chloride. (vi) o-nitrophenol and p-nitrophenol present in a mixture. |
|
Answer» Solution :(i) SEPARATION can be DONE by fractional distillation. (ii) The two can be separated with a separating funnel. (iii) Plants pigments can be separated by adsorption chromatograpgy. (iv) Separation can be affected by separating funnel. (v) Sublimation can be USED for the separation. (VI) These can be sepated by steam distillation. |
|
| 48. |
Suggest a method to purify a mixture of two solids which are soluble in the same solvent. |
| Answer» SOLUTION :By FRACTIONAL CRYSTALLISATION. | |
| 49. |
Suggest a method to deparate a mixture of o-nitrophenol and p-nitrophenol. |
|
Answer» |
|