Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The litres of CO_(2) represented by 4.4 g of CO_(2) at S.T.P are : |
|
Answer» 2.4 litres = 22.4 L at S.T.P 4.4 of `CO_(2)` represent `= ((22.4L))/((44g))xx(4.4g)=2.24L`. |
|
| 2. |
The liquefaction behaviour of temporary gases like CO_(2) approaches that of N_(2),O_(2) (permanent gases ) as we go, |
|
Answer» below CRITICAL TEMPERATURE |
|
| 3. |
The liquefaction behaviour of temporary gases approacches that of perrmanent gases as we go |
|
Answer» below CRITICAL temperature |
|
| 4. |
The linear structure is assumed by |
|
Answer» `SnCl_(2)` while `SnCl_(2)` is ANGULAR . |
|
| 5. |
The linear structure is assumed by : |
|
Answer» `SnCI_(2)` |
|
| 6. |
The linearshape of CO_(2) is due to "……………." |
|
Answer» `SP^(3)` hybridisation of carbon |
|
| 7. |
The linear shape of CO_2 is due to …… |
|
Answer» `sp^3` hybridization of carbon. Structure of `CO_2` is of following TYPE: `O{:(ul(p-p(pi))),(bar(sp-p(6))):}C{:(ul(p-p(pi))),(bar(sp-p(6))):}O` Here , `2 sigma`-bonds and `2 pi` - bonds HYBRIDISATION of `CO_2` is sp so its shape is linear. |
|
| 10. |
The light oil fraction of coal tar mainly contains..........,..............., and .......... |
| Answer» SOLUTION :BENZENE, TOLUENE, XYLENE | |
| 11. |
The Li^(2+) ion is a hydrogen like ion that can BE DESCRIBED BY THE Bohr model.Calculate the Bohr radius of the third orbit and calculate the energy of an electron in 4^(th) orbit. |
|
Answer» Solution :`Li^(2+)`=HYDROGEN LIKE ion. Bohr radius of the third orbit =`r_3`=? `r_3=((0.529)xx3^(2)N^(2))/(Z)`A where n=shell number,Z= atomic number. `r_3=(0.529xx3^(2))/(2)`[because for lithliumZ=3,n=3] `=(0529xx9)/(3)``r_3+1.587A` `E_n=((-13.6)Z^(2))/(n^(2))eV at OM^(-1)` `E_4`=Energy of the fourth orbit=? `E_4=((-13.6)Z^(2))/(4^(2))` `=(-13.6xx9)/(16)=-7.65eVatom^(-1)` `E_4=-7.65eVatom^(-1)` |
|
| 12. |
The Li^(2+) ion is a hydrogen like ion that can be described by the Bohr model. Calculate the Bohr radius of the thired orbital and calculate the energy of an electron in 4^(th) orbital. |
|
Answer» SOLUTION :`r_(n) = ((0.529)n^2)/(z) Å ""E_(n) = (-13.6 (z^2))/(n^2) ev "atom"^(-1)` for `Li^(2+) z = 3` Bohr RADIUS for the THIRD orbit `(r_3)` `=((0.529)(3)^2)/(3)` `= 0.529 xx 3` `= 1.587 Å` Energy of an electron in the fourth orbit `(E_4) = (-13.6(3)^2)/((4)^2)` `= -7.65 eV atom^(-1)`. |
|
| 13. |
The Lewis dot structure of carbonate ion is ………….. |
Answer» SOLUTION :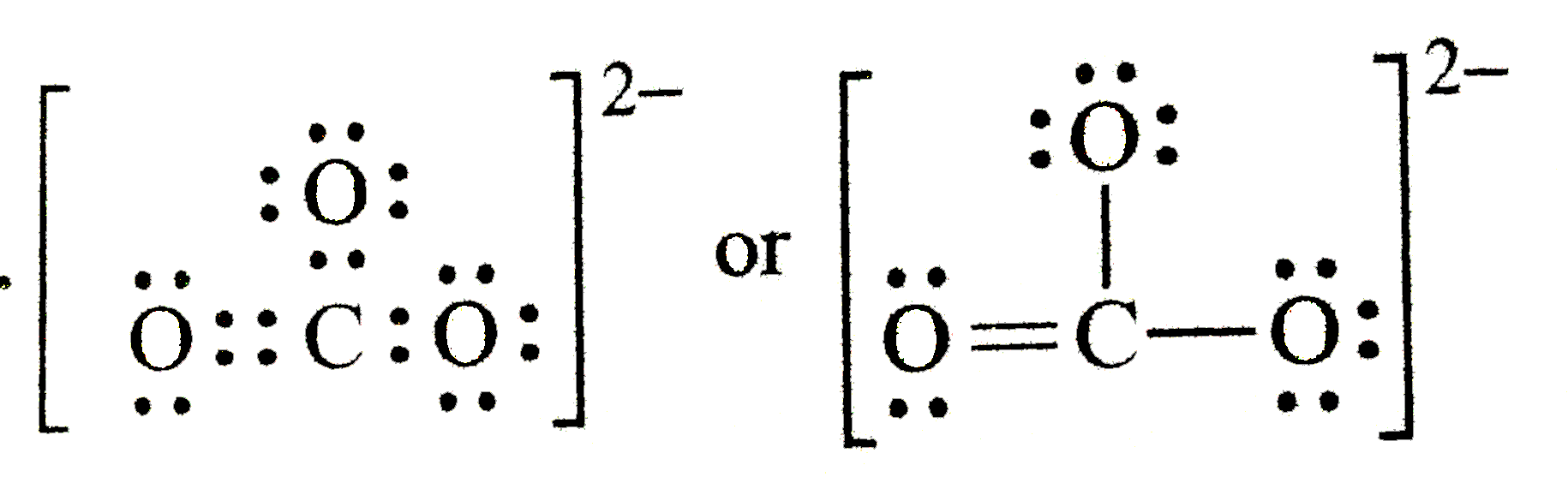
|
|
| 14. |
The Lewis dot structure can be written by following the steps given below. Which one is incorrect? |
|
Answer» DRAW the skeletal structure of the molecule. |
|
| 15. |
Why does the Lewis acid strength of boron halides follow the order B Br_3gt BCl_3 gtBF_3 ? |
|
Answer» `BF_3 GT BCl_3 gt B Br_3` |
|
| 17. |
The level of thermal energy in a substance is known as ...... . |
|
Answer» Entropy |
|
| 18. |
The letters n, l and m proposed by Bohr, Sommerfeld and Zeeman respectivelt for quantization of angular momentum in classical physics were later on obtained as the results of solution of Schrodinger wave equation based on quantum mechanics. The term n, l, m are named as principal quantum number, azimuthal quantum number and magnetic number respectively. The fourth quantum number i.e., spin quantum number (s) was given by Uhlenback on the basis of two spins of electrons. The first two quantum number also decides the nodes of an orbital. Which statement about energy level in H-atom is correct ? |
|
Answer» Only n and L DECIDE ENERGY level |
|
| 19. |
The level of CO gas in air that causes immediate death is |
| Answer» Solution :1000 ppm | |
| 20. |
The letters n, l and m proposed by Bohr, Sommerfeld and Zeeman respectivelt for quantization of angular momentum in classical physics were later on obtained as the results of solution of Schrodinger wave equation based on quantum mechanics. The term n, l, m are named as principal quantum number, azimuthal quantum number and magnetic number respectively. The fourth quantum number i.e., spin quantum number (s) was given by Uhlenback on the basis of two spins of electrons. The first two quantum number also decides the nodes of an orbital. The numerical value Psi_(4,3,0) denotes |
|
Answer» 3d-ORBITAL |
|
| 21. |
The lengthof a unit cell in the NI Crystal is 0.352 nm. The diffraction of X-rays of 0.154 nm wavelength (lambda) from a Ni crystal occurs at 22.2^(@), 55.9^(@) and 38.2^(@) . By using Bragg's law, (nlambda=2d "sin"theta) , and assuming that the diffractions are first order (n=1), the distance are calculated to be 0.204 nm , 0.176 nm and 0.124 nm The various structures for the Ni crystal are represented as: Which of the following statements is correct? |
|
Answer» I represents sc- type structure |
|
| 22. |
The lengthof a unit cell in the NI Crystal is 0.352 nm. The diffraction of X-rays of 0.154 nm wavelength (lambda) from a Ni crystal occurs at 22.2^(@), 55.9^(@) and 38.2^(@) . By using Bragg's law, (nlambda=2d "sin"theta) , and assuming that the diffractions are first order (n=1), the distance are calculated to be 0.204 nm , 0.176 nm and 0.124 nm The various structures for the Ni crystal are represented as: The distance (d) of 0.176 nm represent which structure? |
|
Answer» I |
|
| 23. |
The lengthof a unit cell in the NI Crystal is 0.352 nm. The diffraction of X-rays of 0.154 nm wavelength (lambda) from a Ni crystal occurs at 22.2^(@), 55.9^(@) and 38.2^(@) . By using Bragg's law, (nlambda=2d "sin"theta) , and assuming that the diffractions are first order (n=1), the distance are calculated to be 0.204 nm , 0.176 nm and 0.124 nm The various structures for the Ni crystal are represented as: This distance (d) of 0.124 nm represent which structure? |
|
Answer» I |
|
| 24. |
The lengthof a unit cell in the NI Crystal is 0.352 nm. The diffraction of X-rays of 0.154 nm wavelength (lambda) from a Ni crystal occurs at 22.2^(@), 55.9^(@) and 38.2^(@) . By using Bragg's law, (nlambda=2d "sin"theta) , and assuming that the diffractions are first order (n=1), the distance are calculated to be 0.204 nm , 0.176 nm and 0.124 nm The various structures for the Ni crystal are represented as: The distance (d) of 0.204 nm represent which structure? |
|
Answer» I |
|
| 25. |
The length of a rectangle is 15 inches and the breadth 12 inches. Calculate its area in SI units. |
|
Answer» |
|
| 26. |
The length of a rectangle is 10.4 cm and the breadth is 5.661 cm. Find its area. |
|
Answer» |
|
| 27. |
Length cannot be measured by …………… |
|
Answer» SPECTROSCOPIC method |
|
| 29. |
The least stable conformer of ethane is …………….form |
| Answer» SOLUTION :clipsed | |
| 30. |
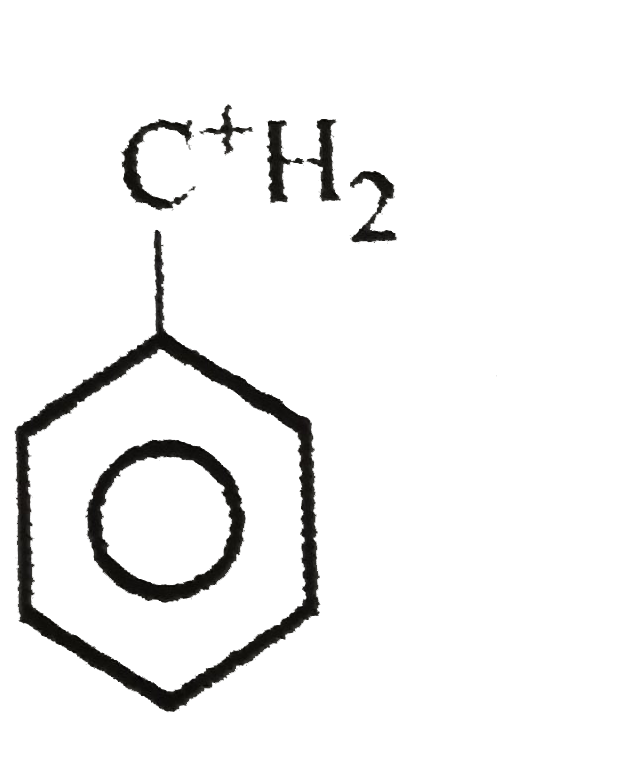
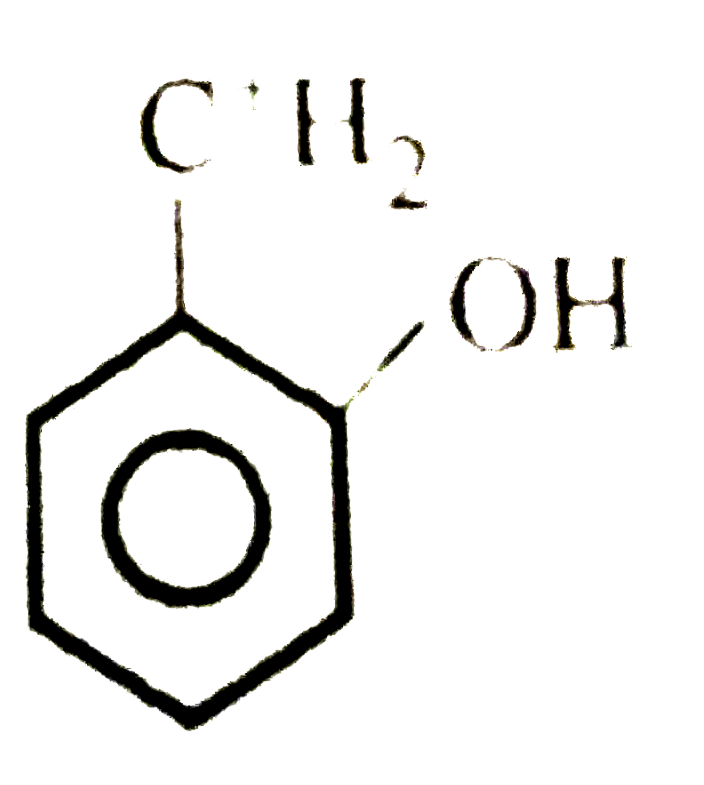
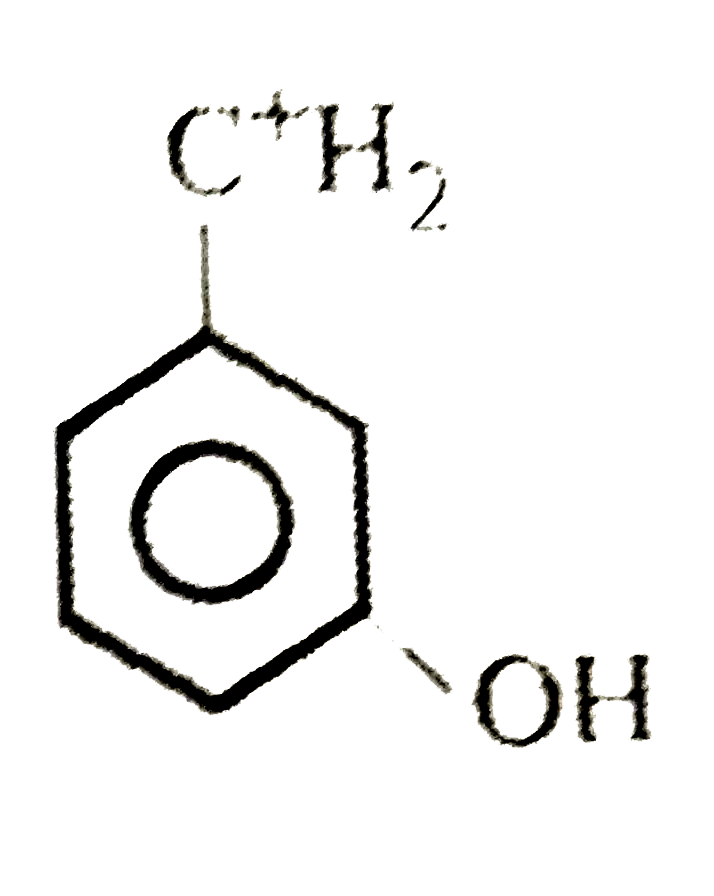
The least stable carbonium ion is |
|
Answer»
|
|
| 31. |
The least stable carbonate of alkali metal is |
|
Answer» `Cs_(2)CO_(3)` |
|
| 32. |
The least soluble in water among the following is:- |
|
Answer» `MgCrO_(4)` |
|
| 34. |
Theleast soluble compound (salt)of the following is |
|
Answer» `CSCL(K_(sp)=10^(-12))` |
|
| 36. |
The least soluble carbonate among the following is |
|
Answer» `Na_(2)CO_(3)` |
|
| 37. |
The least soluble alkali metal carbonate is |
|
Answer» `K_(2)CO_(3)` |
|
| 38. |
The least reactive alkene towards dil. HCl is |
|
Answer»
 COMPOUND I FORMS most stable CARBONIUM ion |
|
| 39. |
The least random state of H_(2)O system is |
|
Answer» Ice |
|
| 40. |
The least priority functional group among the following is |
|
Answer» `-OH` |
|
| 41. |
The least number of carbon atoms in alkane forming chain isomers is |
|
Answer» 3 |
|
| 42. |
The least ionic chloride is formed by |
| Answer» Solution :Being small in size and more ELECTRONEGATIVE , Be forms least ionic or more covalent CHLORIDE . | |
| 43. |
The lead of leadpencils melts at |
|
Answer» `2000^(@)C` |
|
| 44. |
The laws of chemical combination are the basis of the atomic theory. State and explain the law of conservation of mass |
| Answer» SOLUTION :Matter can neither be created nor be DESTROYED `CaCo_3overset"heat"toCaO+CO_2` 100 g of `CaCO_3` on the heating given 56 GRAM of CAO and 44 gram of `CO_2` | |
| 45. |
The laws of chemical combination are the basis of the atomic theory. Name the law of chemical combination illustrated by the pair of compounds, CO and CO_2 |
| Answer» SOLUTION :LAW of MULTIPLE PROPORTION | |
| 46. |
The laws of chemical combination are the basis of the atomic theory Calculate the molarity of a solution containing 8g of NaOH in 500 mL of water |
| Answer» SOLUTION :MOLARITY `(M)=(W_Bxx1000)/(M_BxxV)=(8xx1000)/(40xx500)=8000/20000=0.4M` | |
| 47. |
The law that relates the pressure and volume of gases is |
|
Answer» BOYLE's |
|
| 50. |
The law of multiple proportions was observed for the pair |
|
Answer» `CO_(2),NO_(2)` |
|