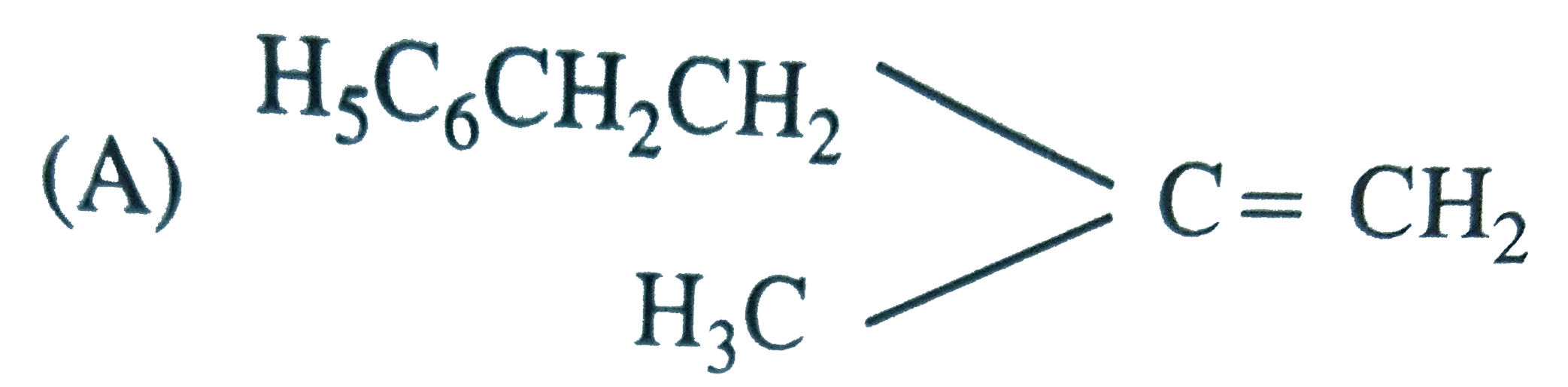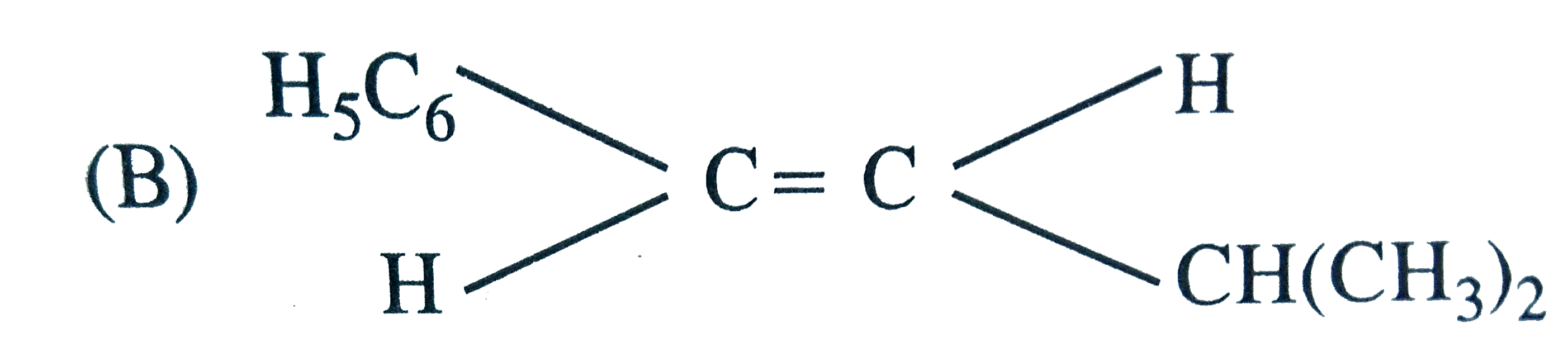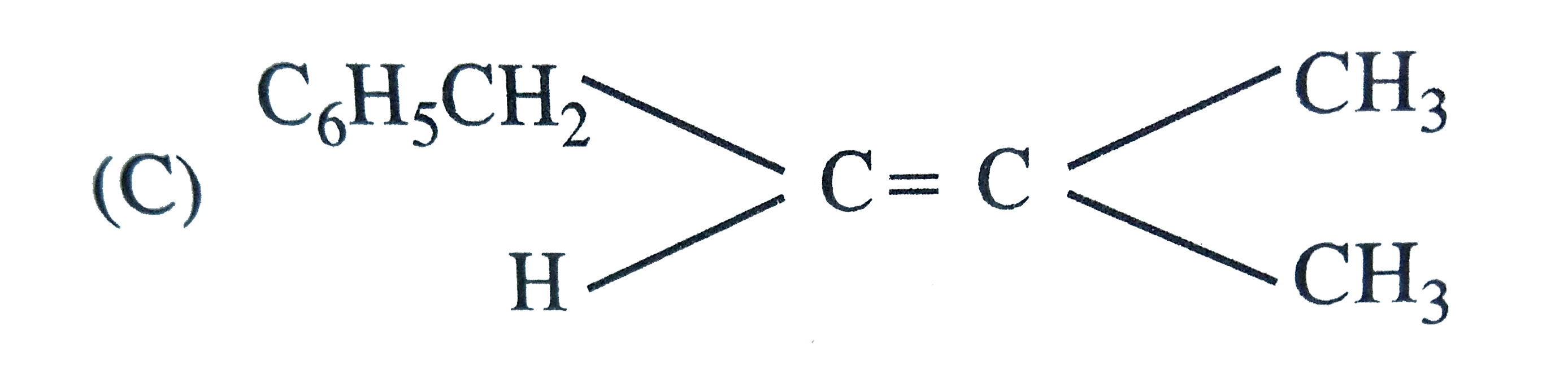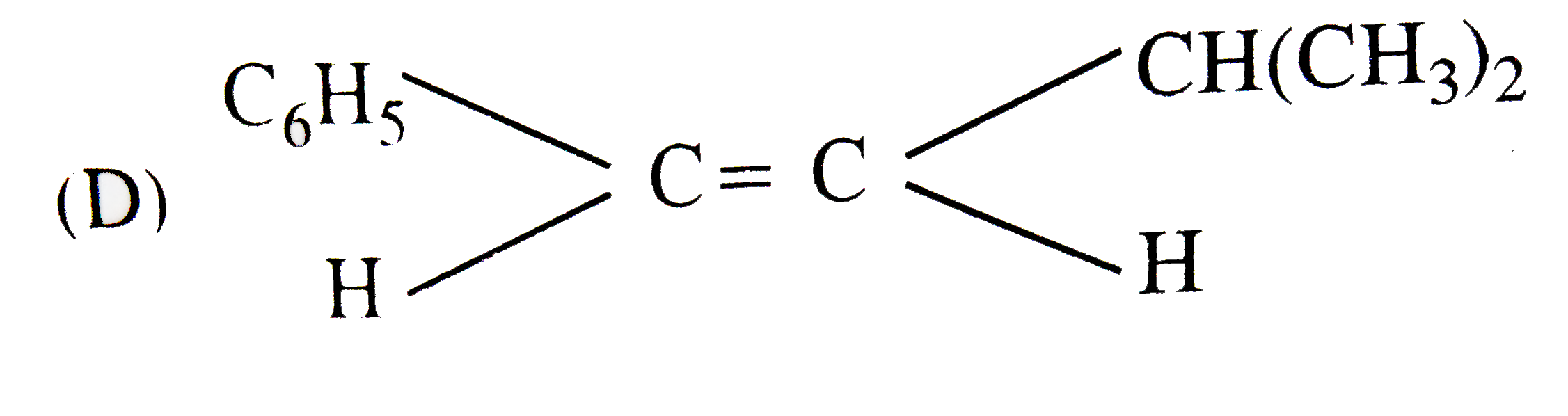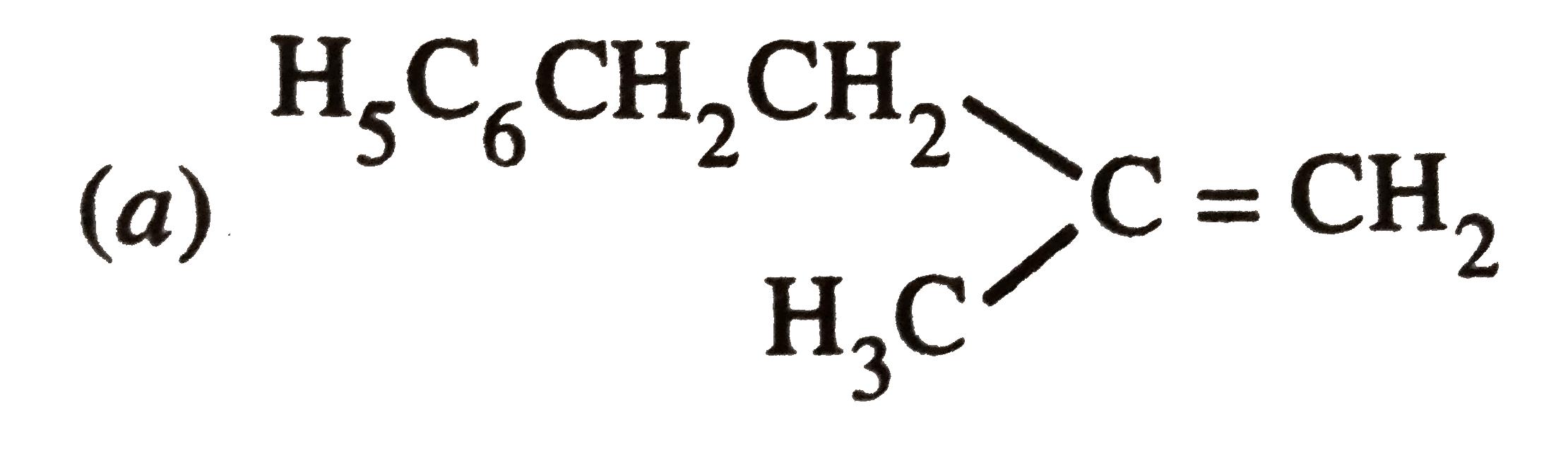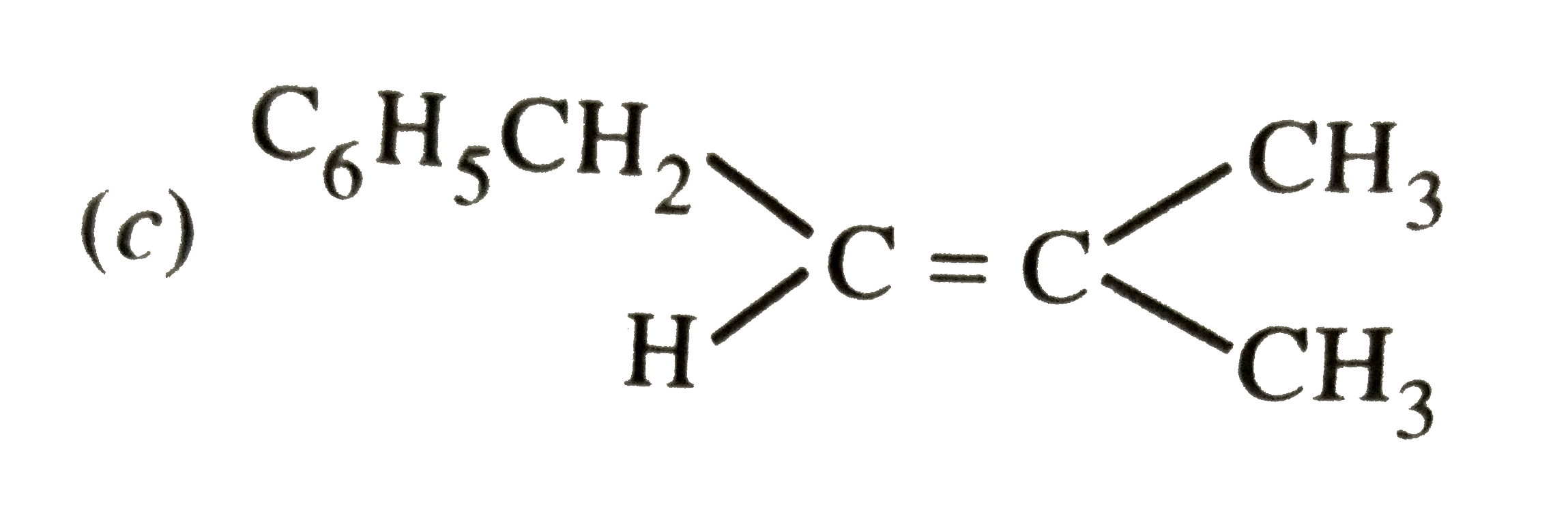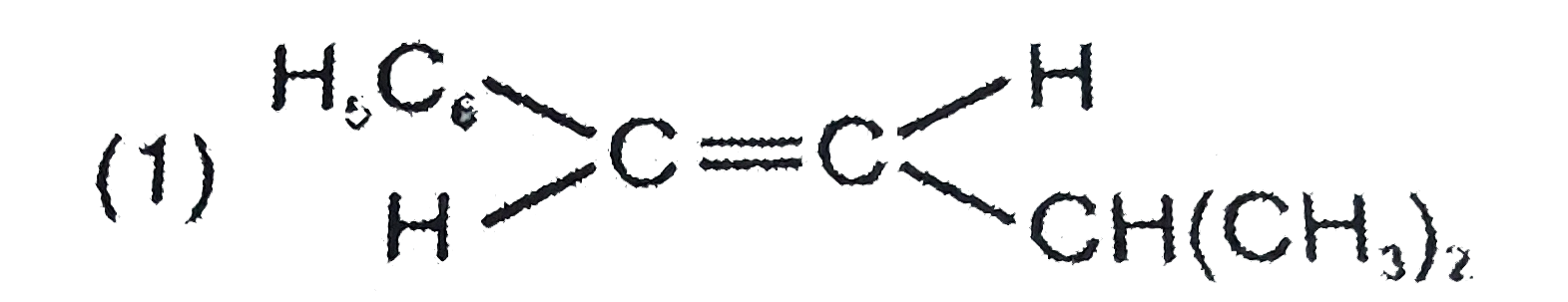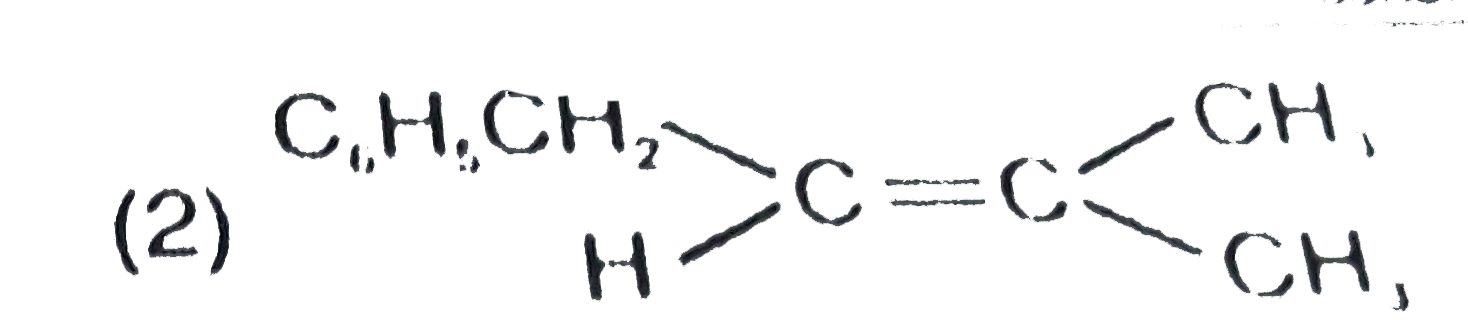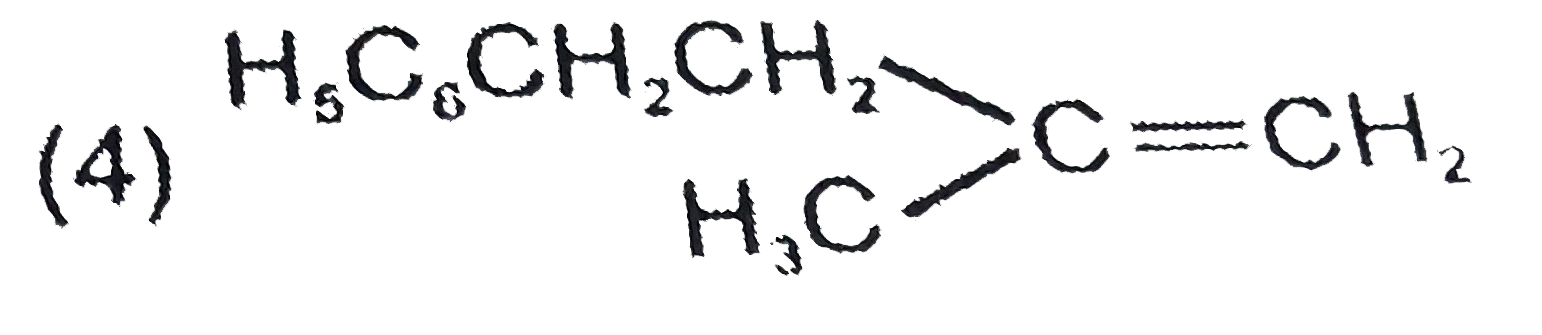Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
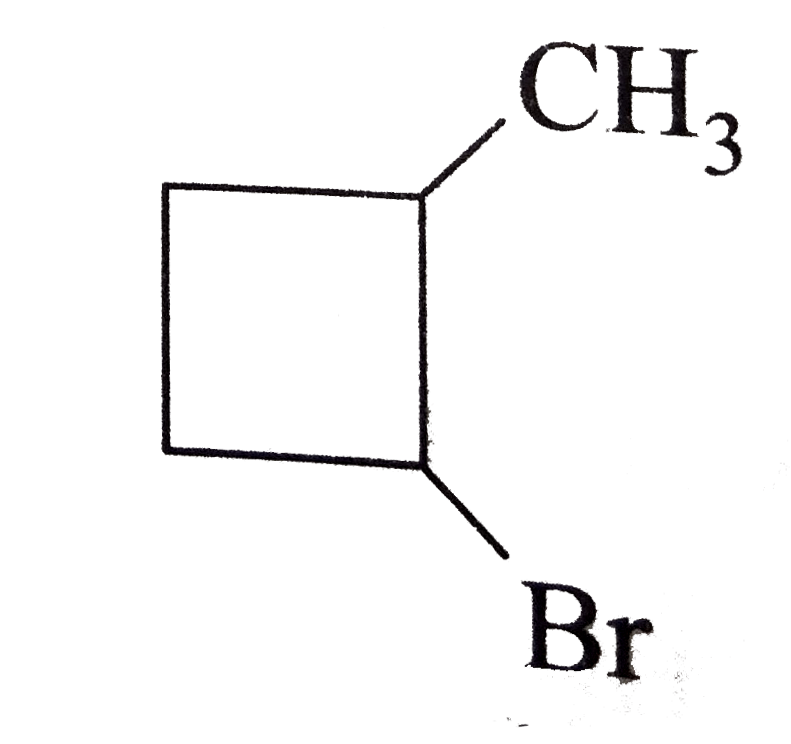
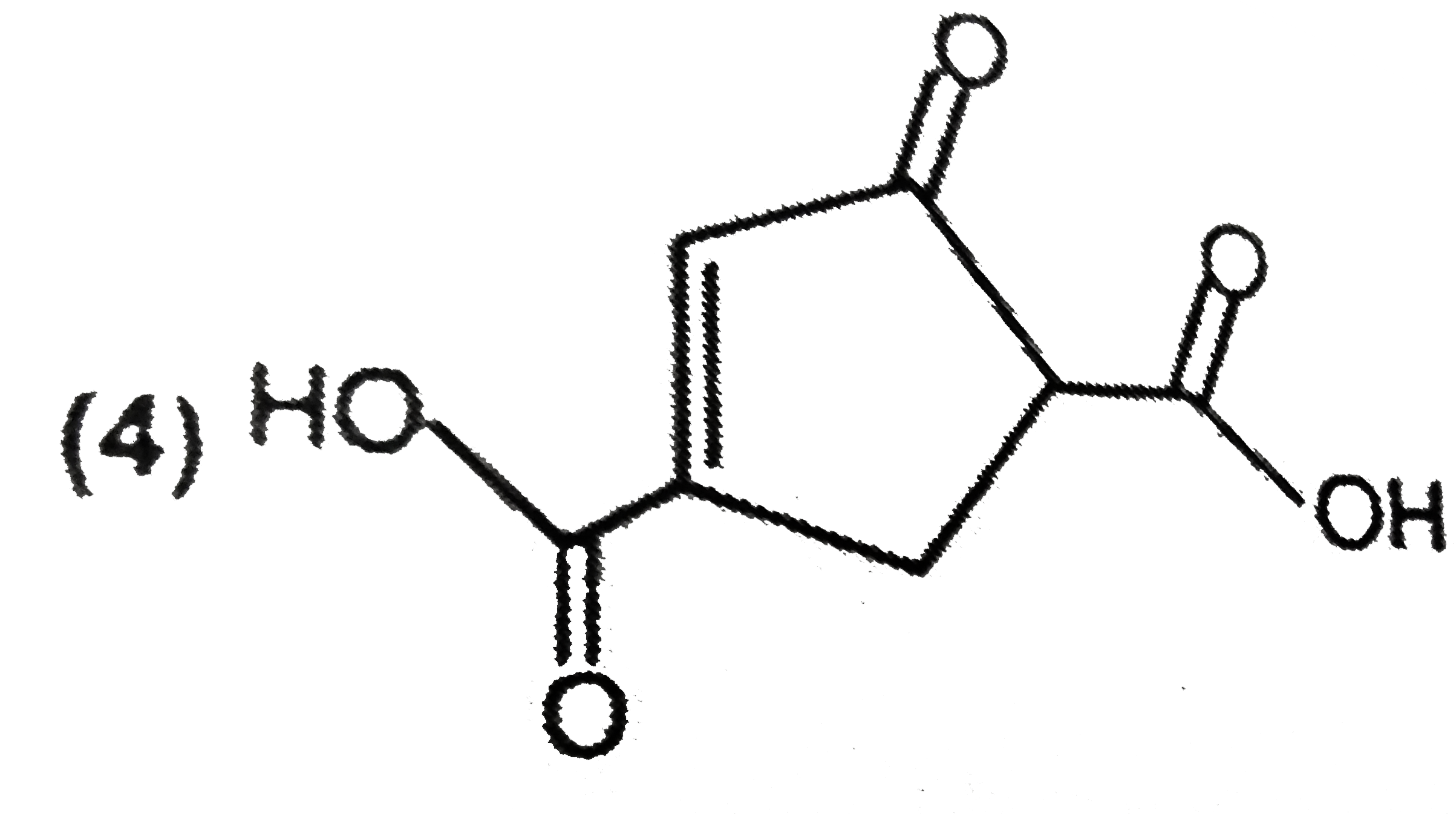
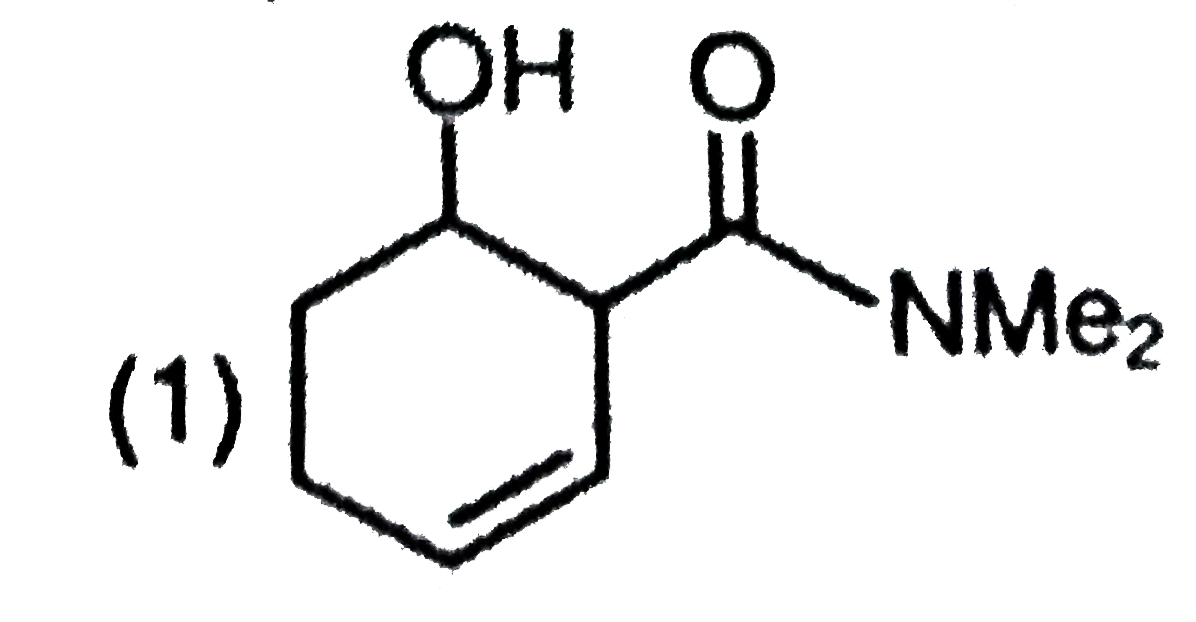
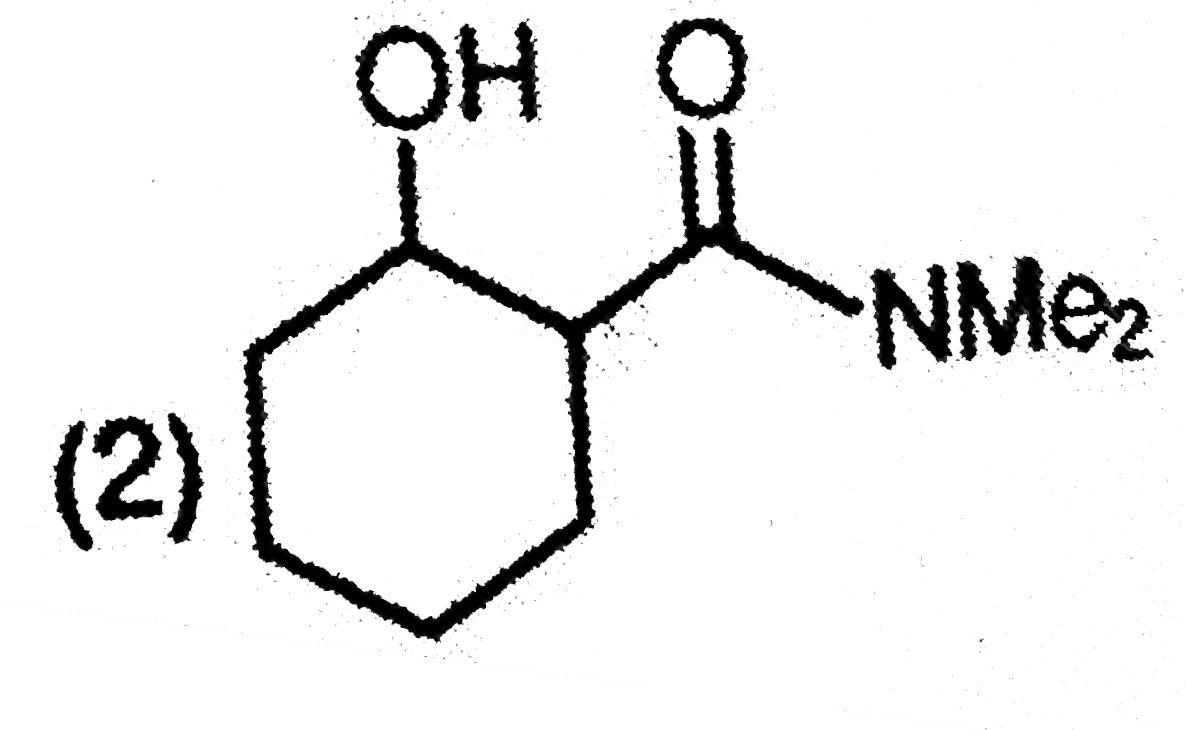
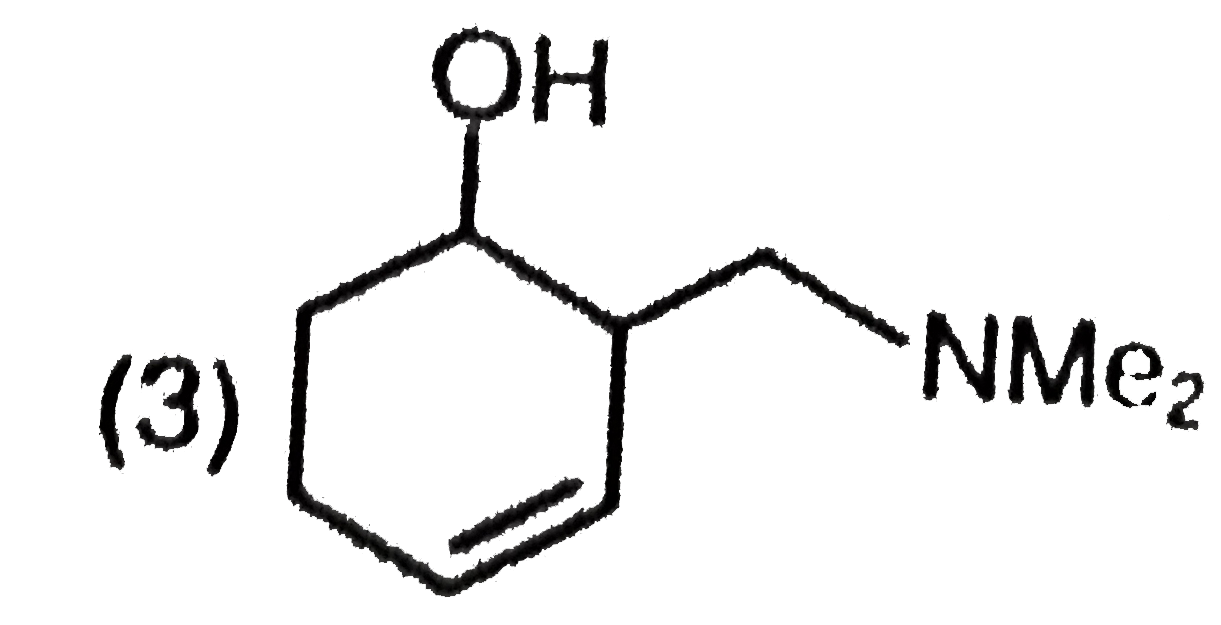
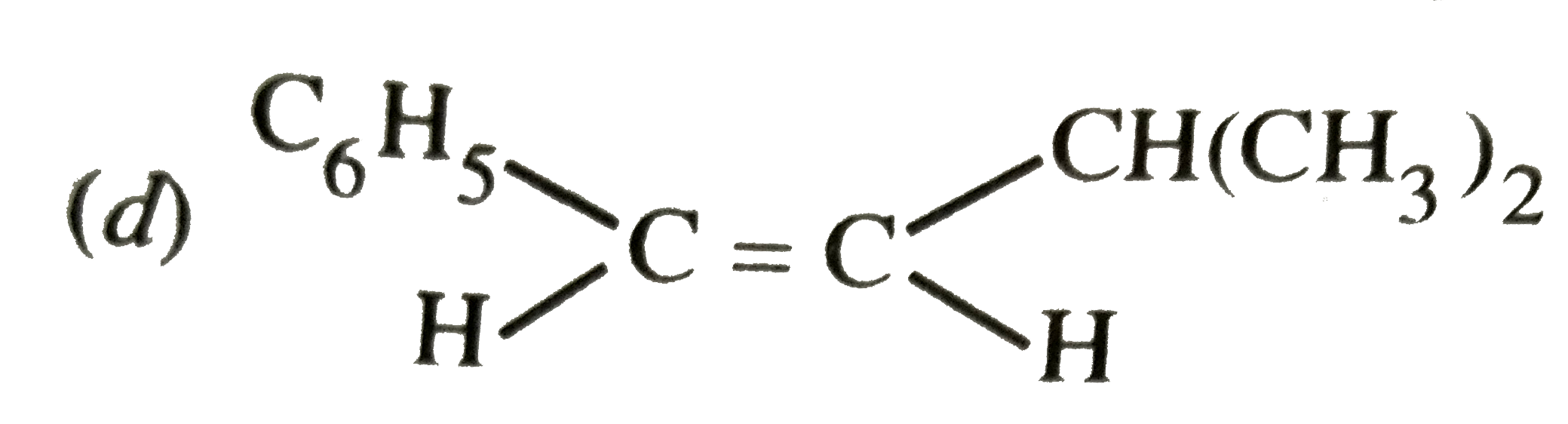
| 1. |
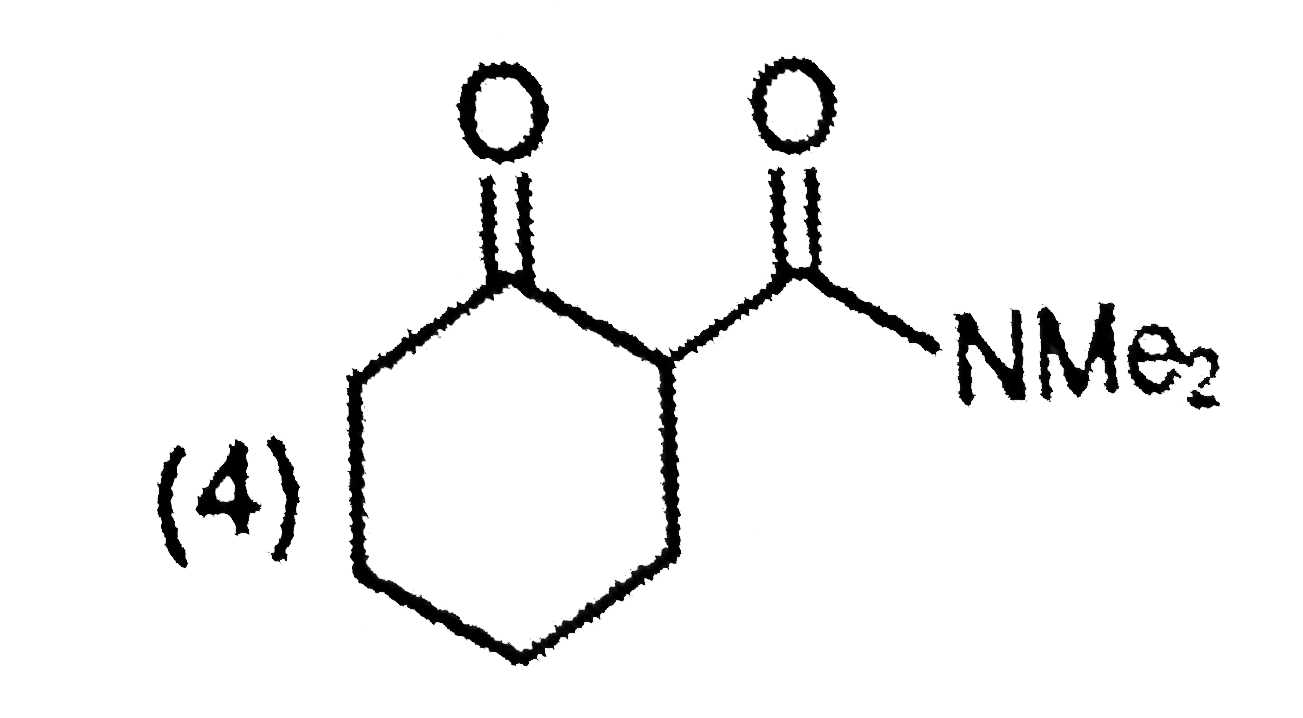
The major product formed in the following reaction is |
|
Answer»
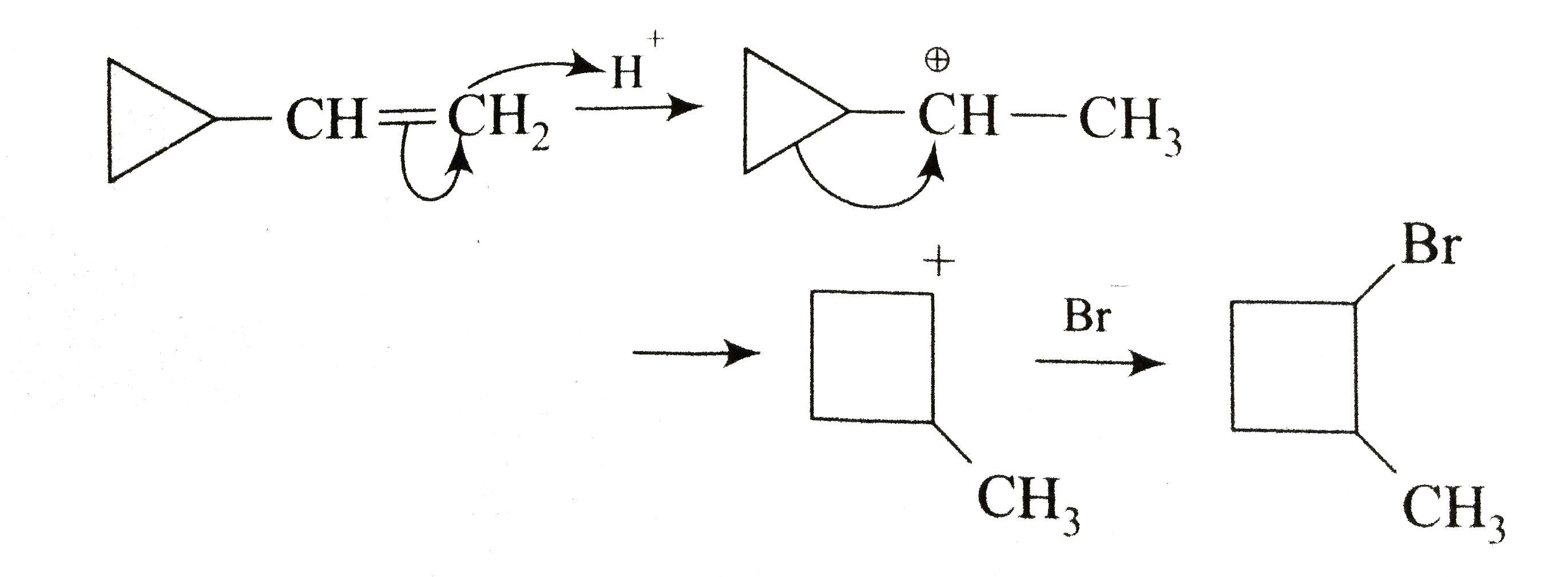
|
|
| 3. |
The major organic compound formed by the reactions of 1,1,1-trichloroethane with silver powder is |
|
Answer» 2-butene |
|
| 4. |
The major constituent of cement is ……… . |
| Answer» SOLUTION :tricalcium aluminate , `Ca_(3)SiO_(5)` | |
| 5. |
The main source of phosphate pollution in water is |
|
Answer» OIL pollutant |
|
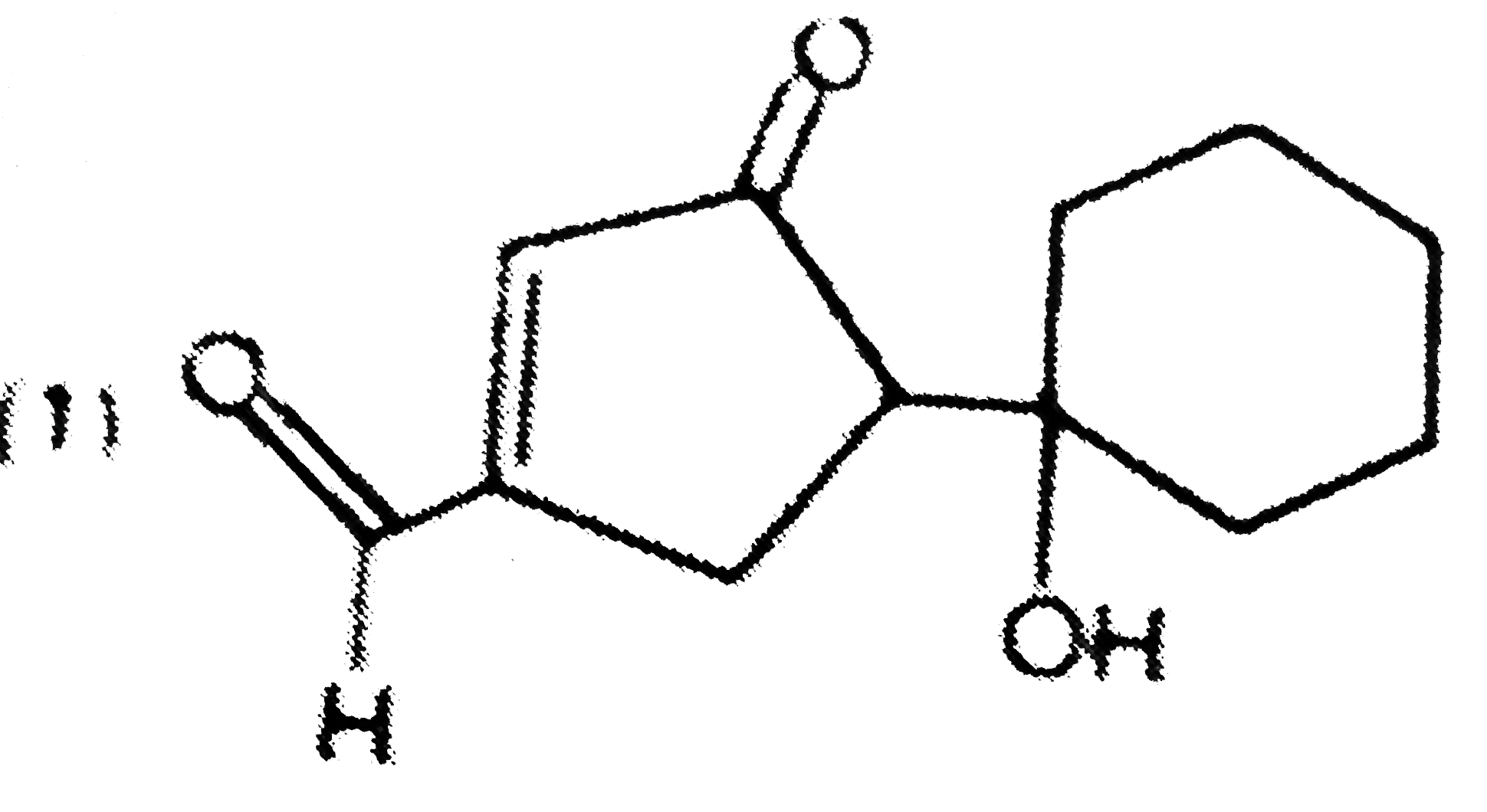
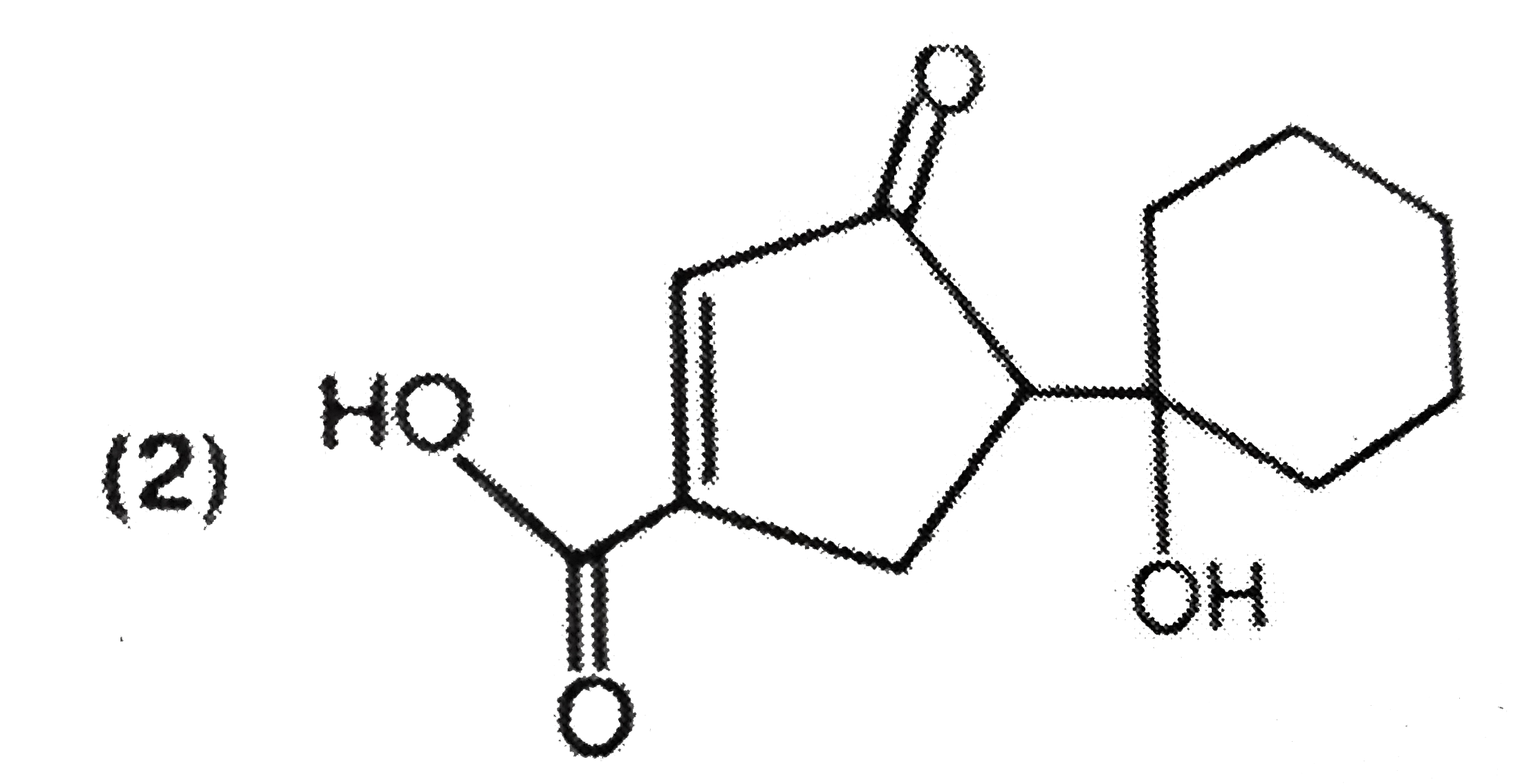
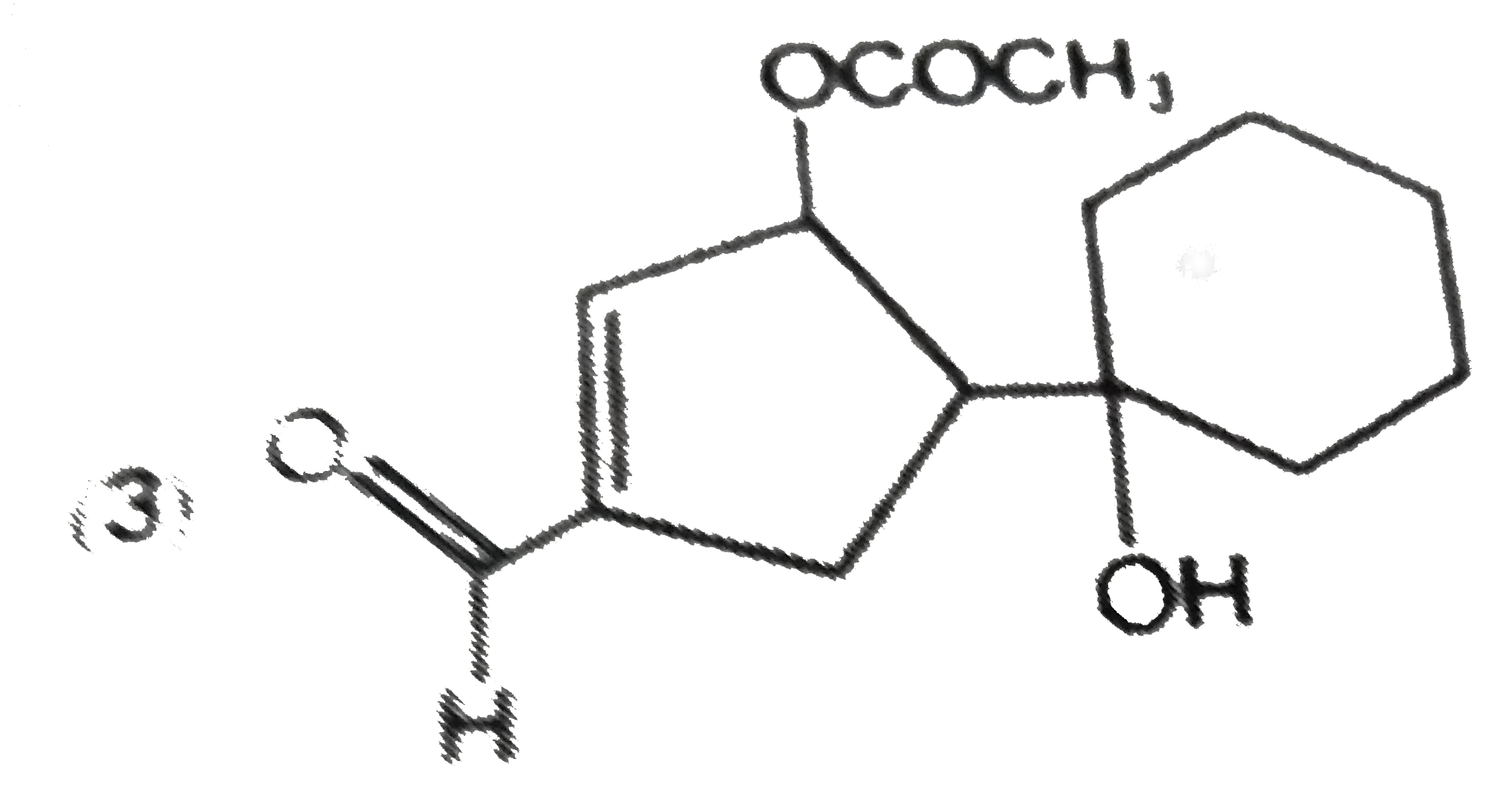
| 6. |
The main reduction product of the following compound with NaBH_(4) in methanol is: |
|
Answer»
|
|
| 7. |
The main reason for using a mercury electrolytic cell in NaOH manufacture is that |
|
Answer» HG is toxic |
|
| 8. |
The main reason for the fact that carboxylic acids can undergo ionisation |
|
Answer» absence of `alpha - H` |
|
| 9. |
The main reason for deviation of gases from ideal behaviour is few assumptions of kinetic theory . These are(i) there is no force of attraction between the molecules of a gas(ii) volume of the molecules of a gas is negligibly small incomparison to the volume of the gas(iii)particles of a gas are always in constant random motion . |
|
Answer» (i) and (II) |
|
| 10. |
The main product of the following reaction is C_(6)H_(5)CH(OH)CH(CH_(3))_(2) overset(conc. H_(2)SO_(4))rarr |
|
Answer»
(Benzyl CARBOCATION more STABLE due to resonance 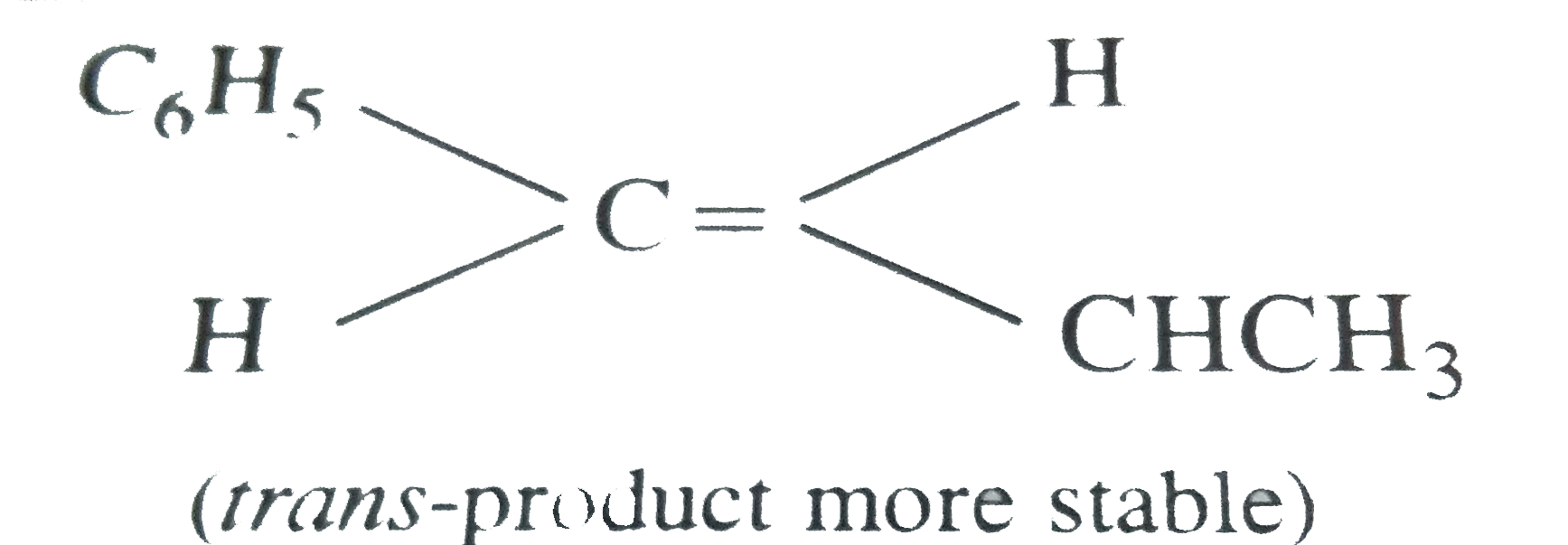
|
|
| 11. |
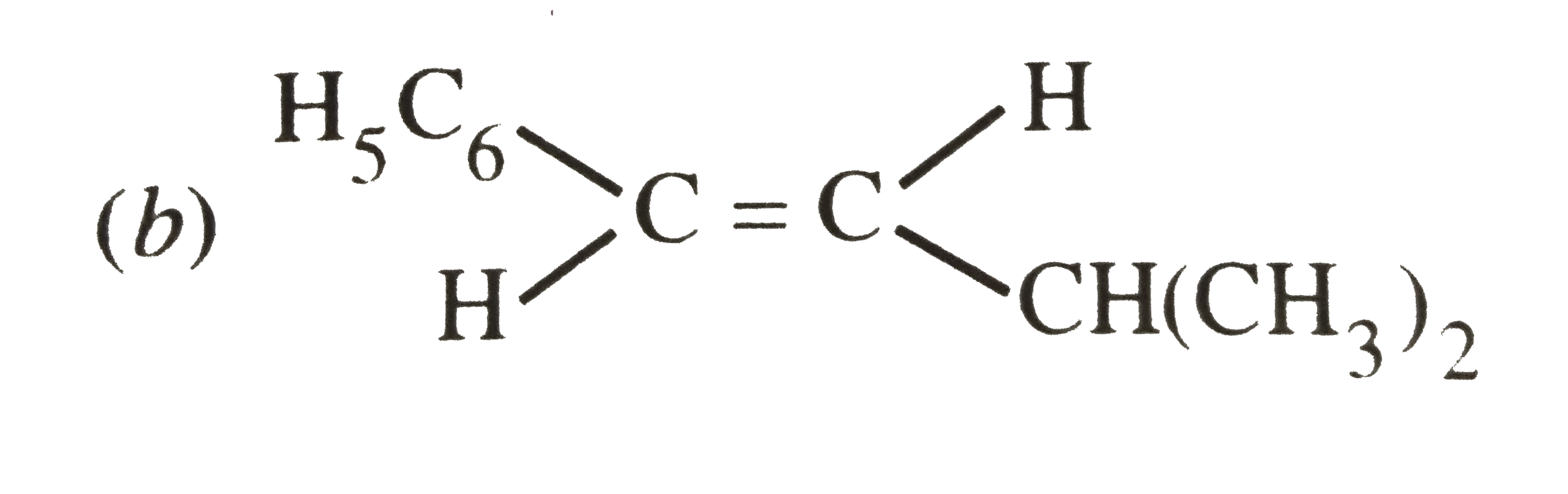
The main product of the following reaction is C_6H_5CH_2CH(OH)CH(CH_3)_2overset(Conc. H_2SO_4)to |
|
Answer»
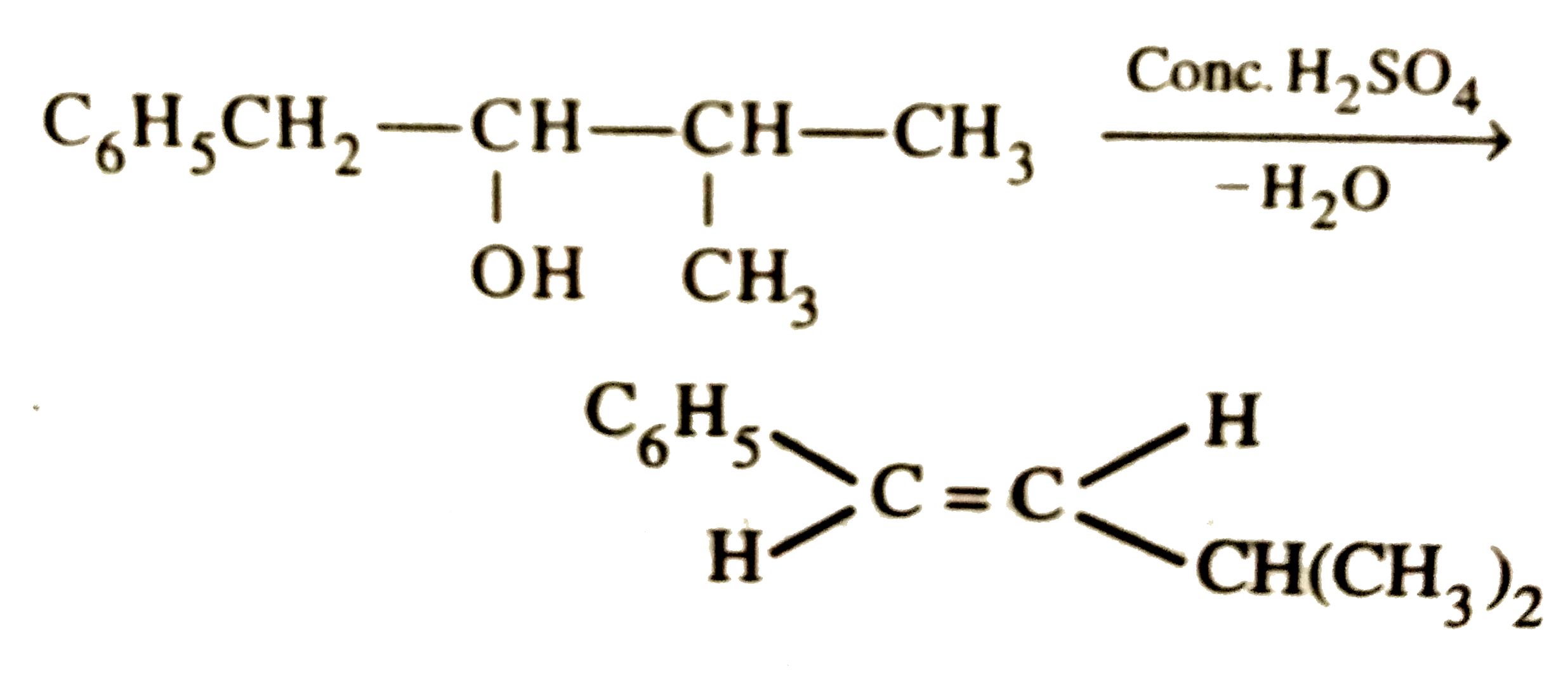 DEHYDRATION occurs in such a way that the double BOND comes in CONJUGATION with the `C_6H_5` GROUP and further since trans-alkenes are more stable than the cis-alkenes, therefore , OPTION (b) is correct. DEHYDRATION occurs in such a way that the double BOND comes in CONJUGATION with the `C_6H_5` GROUP and further since trans-alkenes are more stable than the cis-alkenes, therefore , OPTION (b) is correct.
|
|
| 12. |
The main product of the following reaction is , C_6H_5CH_2CH(OH)CH(CH_3)_2overset(conc. H_2SO_4)to |
|
Answer»
|
|
| 13. |
The main product obtained on reducing boron trichloride with lithium aluminium hydride is ……………………. . |
| Answer» SOLUTION :DIBORANE | |
| 14. |
The product obtained at anode when 50% H_(2)SO_(4) aqueous solution is electrolysed using platinum electrodes, is |
|
Answer» Sulphurous ACID |
|
| 16. |
The main oxides formed on combustion of Li, Na and K in excess of air respectively are - |
|
Answer» `LiO_2, Na_2O and KOH` `K+ O_2 to KO_2` |
|
| 17. |
The main oxides formed on combustion of Li , Na , and K in excess of air are respectively |
|
Answer» `Li_(2)O , Na_(2)O and KO_(2)` |
|
| 18. |
The main factor responsible for weak acidic nature of B - F bonds in BF_(3) is |
|
Answer» large electronegativity of F |
|
| 19. |
The main constituent of layer responsible for global warming is ........... |
| Answer» SOLUTION :`CO_(2)` | |
| 20. |
The main common constituent of producer gas and water gas is |
|
Answer» `N_2` |
|
| 21. |
The magnitude of surface tension of liquid depends on the attractive forces between the molecules. Arrange the following in increasing order of surface tension :Water, alcohol (C_(2)H_(5)OH) and hexane [CH_(3)(CH_(2))_(4)CH_(3))]. |
|
Answer» Solution :In the above given molecules, only hexane is non - polar molecules in which only London DISPERSION forces PRESENT. These forces are very weak while both water and alcohol are polar molecules in which H - bonding as well as DIPOLE - dipole interactions are present. However, H - bonding interactions are too stronger in water than therefore, it possesses much stronger intermolecular than alcohol and hexane. So, the INCREASING order is hexane `lt` alcohol `lt` water Higher is the attractive forces between the molecules, higher is the magnitude of surface tension of liquid. |
|
| 22. |
The magnitude of surface tension of liquid depends on the attractive forces between the molecules. Arrange the following in increasing order of surface tension : water, alcolhol (C_(2)H_(5)OH) and hexane [CH_(3)(CH_(2))_(4)CH_(3)]. |
| Answer» Solution :Attractive forces are MINIMUM in hexane (only London forces). Hydrogen bonding in `H_(2)O` is STRONGER than that in `C_(2)H_(5)OH`. Hence the order of surface tension is : hexane `lt` alcohol`lt`water. | |
| 23. |
The magnitude of enthalpy changes for irreversible adiabiatic expansion of a gas from 1 L to 2L is DeltaH_(1) and for reversible adiabatic expansion for the same expansion is DeltaH_(2). Then : |
|
Answer» `DeltaH_(1)gtDeltaH_(2)` |
|
| 24. |
The magnetic quantumnumber of an atom is reletedto the |
|
Answer» size of the ORBITAL |
|
| 25. |
The magentic quantum number, m for the outermost electron in the sodium atom is |
| Answer» SOLUTION :`3s^1 , m_l =0` | |
| 26. |
The magnetic ore is |
|
Answer» FERROUS oxide |
|
| 27. |
The magnetic nature of atoms (or) ions of IIA elements is |
|
Answer» Paramagnetic |
|
| 28. |
The magnetic moment of KO_(2) at room temperature is |
|
Answer» 1.41 B.M |
|
| 29. |
The magnetic moment of KO_(2) at room temperature is …… BM. |
|
Answer» 1.41 electron . Hence `mu= sqrt(n (n + 2) )BM= sqrt(1(1+2))` `sqrt(3) = 1.73` BM . |
|
| 30. |
The magnetic moment of complex 'C' is: |
|
Answer» `1.7 B.M.` |
|
| 31. |
The magnetic moment of ._(25)Mn in ionic state is sqrt(15)B.M, then Mn is in: |
|
Answer» `+2` STATE |
|
| 32. |
The M mass of NaOH is 40.50 mL of a solution containing 2 g of NaOH in 500 mL will require for complete neutralisation : |
|
Answer» 10 ML DECINORMAL HCl |
|
| 33. |
The 'm' vlaue for an electron in an atom is equal to the number of m values for l=1. the electron may be present in |
|
Answer» `3d_(X^(2)-y^(2))` |
|
| 34. |
The lowest region of atmosphere in which all living beings including human beings reside is known as ........ |
|
Answer» TROPOSPHERE |
|
| 35. |
The lowest region of atmosphere is |
|
Answer» Stratosphere |
|
| 36. |
The lowest first ionization energy would be associated with which of the following configurations. |
|
Answer» `1s^(2)2s^(2)2P^(6)3s^(1)` |
|
| 37. |
The lowest layer of atmosphere is the_____> |
| Answer» | |
| 38. |
The lower area of atmosphere, where all living thing including human can stay is known as Troposphere. |
| Answer» SOLUTION :TRUE STATEMENT | |
| 39. |
The low solubility of LiF and that of CsI in water are respectively due to which of the properties of the alkali metal ions ? |
|
Answer» Higher hydration ENTHALPY of `Li^(+)` , higher lattice enthalpy of `CS^(+)` |
|
| 40. |
The loss or reduction of chlorophyll in the leaves is termed as |
| Answer» Solution :Chlorosis | |
| 41. |
The loss of green colour in plants due to presence of even a very low concentration of SO_2 is called ________ |
|
Answer» |
|
| 42. |
The longest wavelength doublet absorption transition is oberved at 58 and 589-6 nm. Calculate the frequency of each transition and the energy difference between the two excited states |
|
Answer» SOLUTION :`lamda_(1) = 589 nm = 589 xx 10^(-9) m " " :. v_(1) = (C)/(lamda_(1)) = (3.0 xx 10^(8) ms^(-1))/(589 xx 10^(-9) m) = 5.093 xx 10^(14) s^(-1)` `lamda_(2) = 589.6 nm = 589.6 xx 10^(-9)m :. v_(2) = (c)/(lamda_(2)) = (3.0 xx 10^(8) ms^(-1))/(589.6 xx 10^(-9) m) = 5.088 xx 10^(14) s^(-1)` `DELTA E = E_(2) - E_(1) = h (v_(2) - v_(1)) = (6.626 xx 10^(-34) Js) (5.093 - 5.088) xx 10^(14) s^(-1) = 3.31 xx 10^(-22) J` |
|
| 43. |
The longest wavelength of He^(+) in Paschen series in 'm'. Then what will be the shortest wavelength of Be^(3+) in Paschen series in terms of m ? |
|
Answer» Solution :`(1)/(lamda_(He^(+))) = RZ^(2) ((1)/(n_(1)^(2)) - (1)/(n_(2)^(2)))` `(1)/(m) = R xx4 ((1)/(3^(2) ) - (1)/(4^(2))) = 4R xx (7)/(144) = (7R)/(36)`...(i) (`:'` For Paschen series, `n_(1) = 3` and for longest WAVELENGTH, `n_(2) = 4`. For He Z = 2) `(1)/(lamda_(Be^(3+))) = R xx 16 ((1)/(3^(2)) - (1)/(oo)) = (16)/(R)`...(ii) ( `:'` For SHORTEST wavelength, `n_(2) = oo` and for Be, Z = 4) Dividing eqn. (i) by eqn. (ii), we get `:. (lamda_(Be^(3+)))/(m) = (7 xx 9)/(16 xx 36) or lamda_(Be^(3+)) = (7)/(64) m` |
|
| 44. |
The lormula of Freon 12 is …………….. . |
| Answer» SOLUTION :`CF_2Cl_2` | |
| 45. |
The longest wavelengthdouble absorptiontransitionis observedat 589and 589nmCalculate the frequencyof eachtransitionandenergydifferencebetweentwoexcitedstates. |
|
Answer» Solution :Firstlinewavelength `lambda_(1)= 589nm = 589 xx 10^(9)m= 589 xx 10^(7) m` frequency`v_(1) = (c )/(LAMBDA) = (3.0 xx 10^(8)MS^(_1))/(5.89 xx 10^(7) m)` `=5.0934 xx 10^(14) s^(-1)` Secondlinewavelength `lambda_(2)= 589 NM = 589 .6 xx 10^(9) nm =5.896 xx 10^(7) m` Differenceof doubletfrequency`(v_(1)-v_(2)) Delta v` andenergydifference`Delta E= hDelta E` `(6.626 xx 10^(34 ) js) ( 0.0052 xx 10^(14)s^(-1))` `=0.03446 xx 10^(20)J = 3.446 xx 10^(22) J` |
|
| 46. |
The longest form of periodic table was constructed by…….. |
| Answer» Solution :Henry Moseley | |
| 48. |
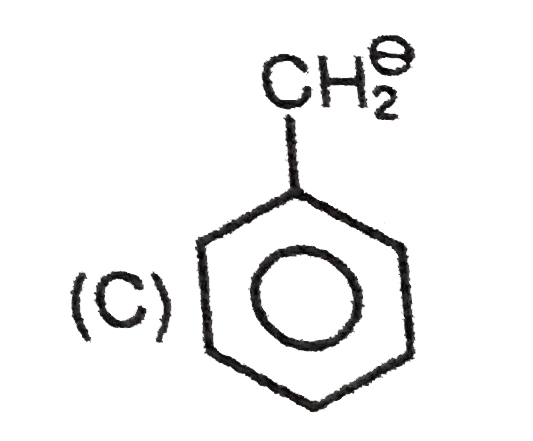
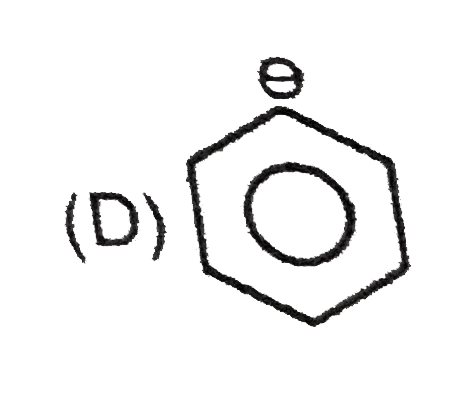
The lone pair of amines makes them basic. They react with acids to form acid-base salts. Amines are more basic than alcohols, ethers and water. When an amine is dissolved in water, an equilibrium is established , where water acts as an acid and transfer a proton to the amine. The basic strength of an amine can be measured by basicity constant K_(b). Arylamines are less basic than alkylamines because the lone pair of nitrogenis delocalised with the aromatic ring and are less available for donation. Substituted arylamines can be either more basic or less basic than aniline , depending on the substituted. ERG substituents, such as -CH_(3), -NH_(2) and -OCH_(3) increases the basicity and EWG substituents , such as -Cl, -NO_(2) and -CN decreases basicity. While sp^(2)- hybridized nitrogen atom in pyridine is less basic then the sp^(3) -hybridized nitrogen in an alkylamine. The most basic carbanion is : |
| Answer» ANSWER :D | |
| 49. |
The lone pair of amines makes them basic. They react with acids to form acid-base salts. Amines are more basic than alcohols, ethers and water. When an amine is dissolved in water, an equilibrium is established , where water acts as an acid and transfer a proton to the amine. The basic strength of an amine can be measured by basicity constant K_(b). Arylamines are less basic than alkylamines because the lone pair of nitrogenis delocalised with the aromatic ring and are less available for donation. Substituted arylamines can be either more basic or less basic than aniline , depending on the substituted. ERG substituents, such as -CH_(3), -NH_(2) and -OCH_(3) increases the basicity and EWG substituents , such as -Cl, -NO_(2) and -CN decreases basicity. While sp^(2)- hybridized nitrogen atom in pyridine is less basic then the sp^(3) -hybridized nitrogen in an alkylamine. Select the correct order of K_(b) |
|
Answer» `CH_(3)NH_(2) GT NAOH` |
|
| 50. |
The lone pair of amines makes them basic. They react with acids to form acid-base salts. Amines are more basic than alcohols, ethers and water. When an amine is dissolved in water, an equilibrium is established , where water acts as an acid and transfer a proton to the amine. The basic strength of an amine can be measured by basicity constant K_(b). Arylamines are less basic than alkylamines because the lone pair of nitrogenis delocalised with the aromatic ring and are less available for donation. Substituted arylamines can be either more basic or less basic than aniline , depending on the substituted. ERG substituents, such as -CH_(3), -NH_(2) and -OCH_(3) increases the basicity and EWG substituents , such as -Cl, -NO_(2) and -CN decreases basicity. While sp^(2)- hybridized nitrogen atom in pyridine is less basic then the sp^(3) -hybridized nitrogen in an alkylamine. pK_(b) order of the following compound is : (I) NH_(2)OH""(II) NH_(2)NH_(2)""(III) NH_(3)""(IV)H_(2)O |
|
Answer» `IV GT I gt II gt III` |
|