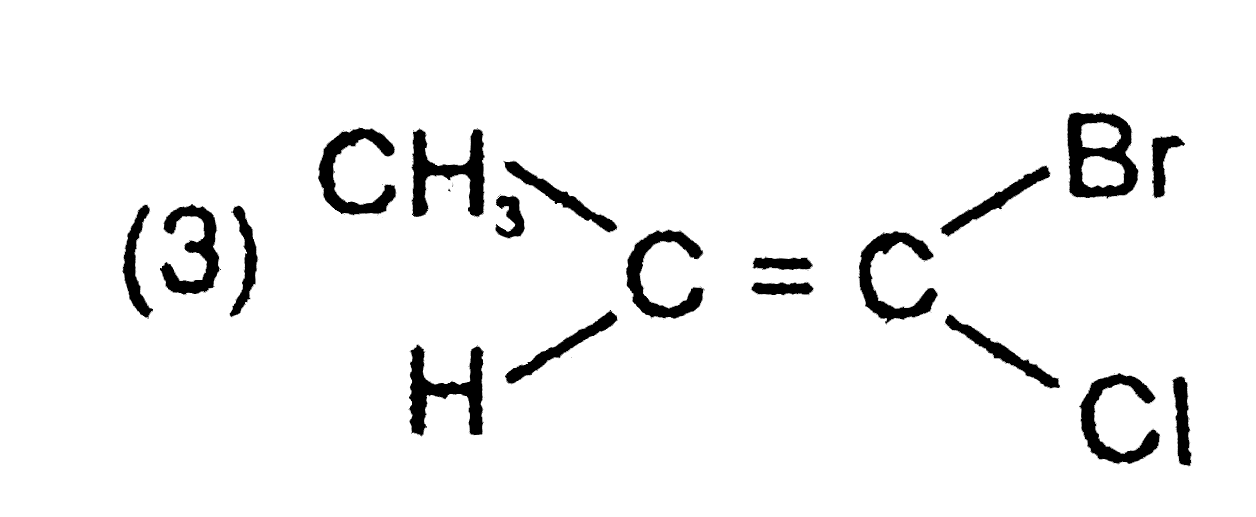
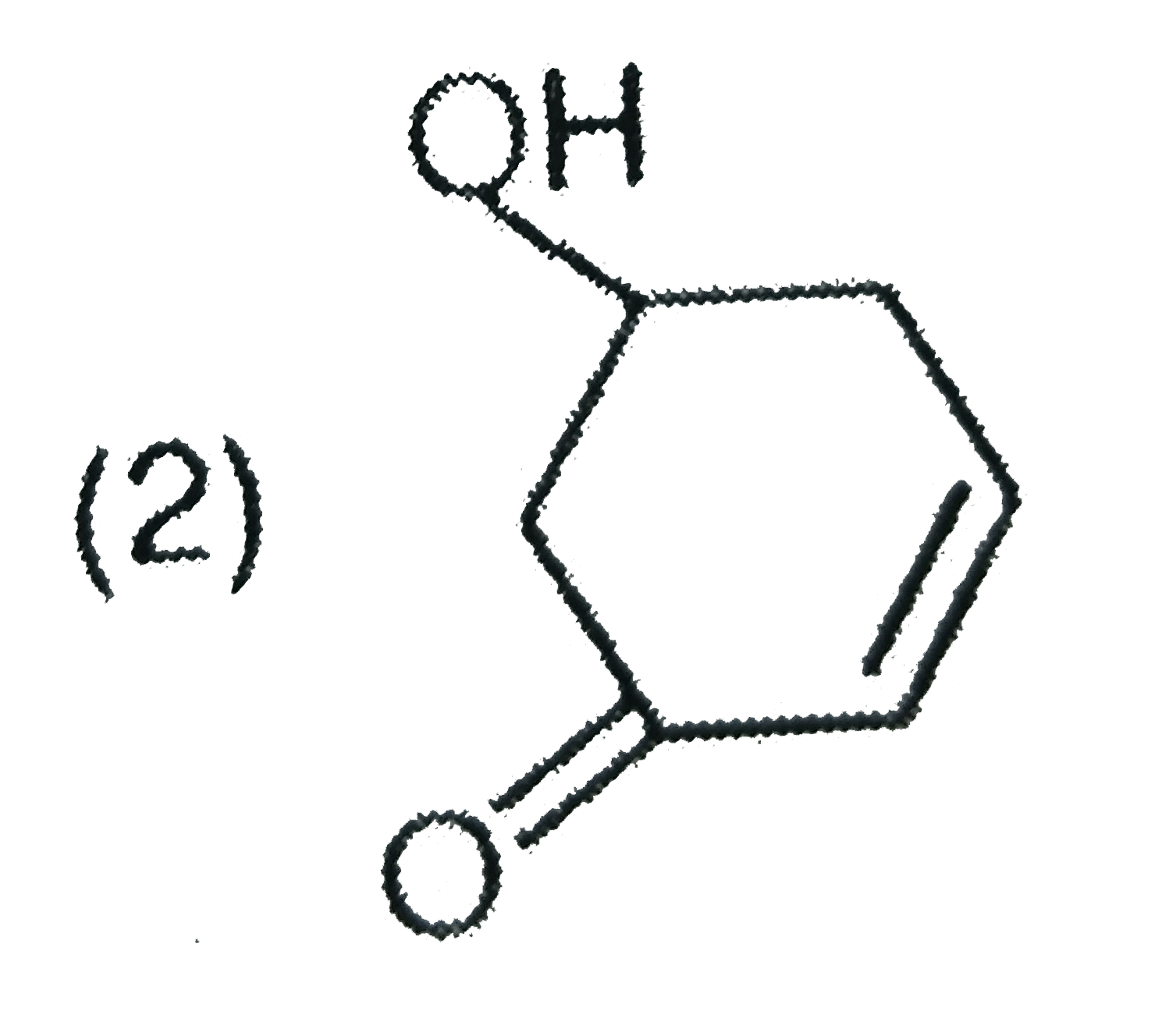
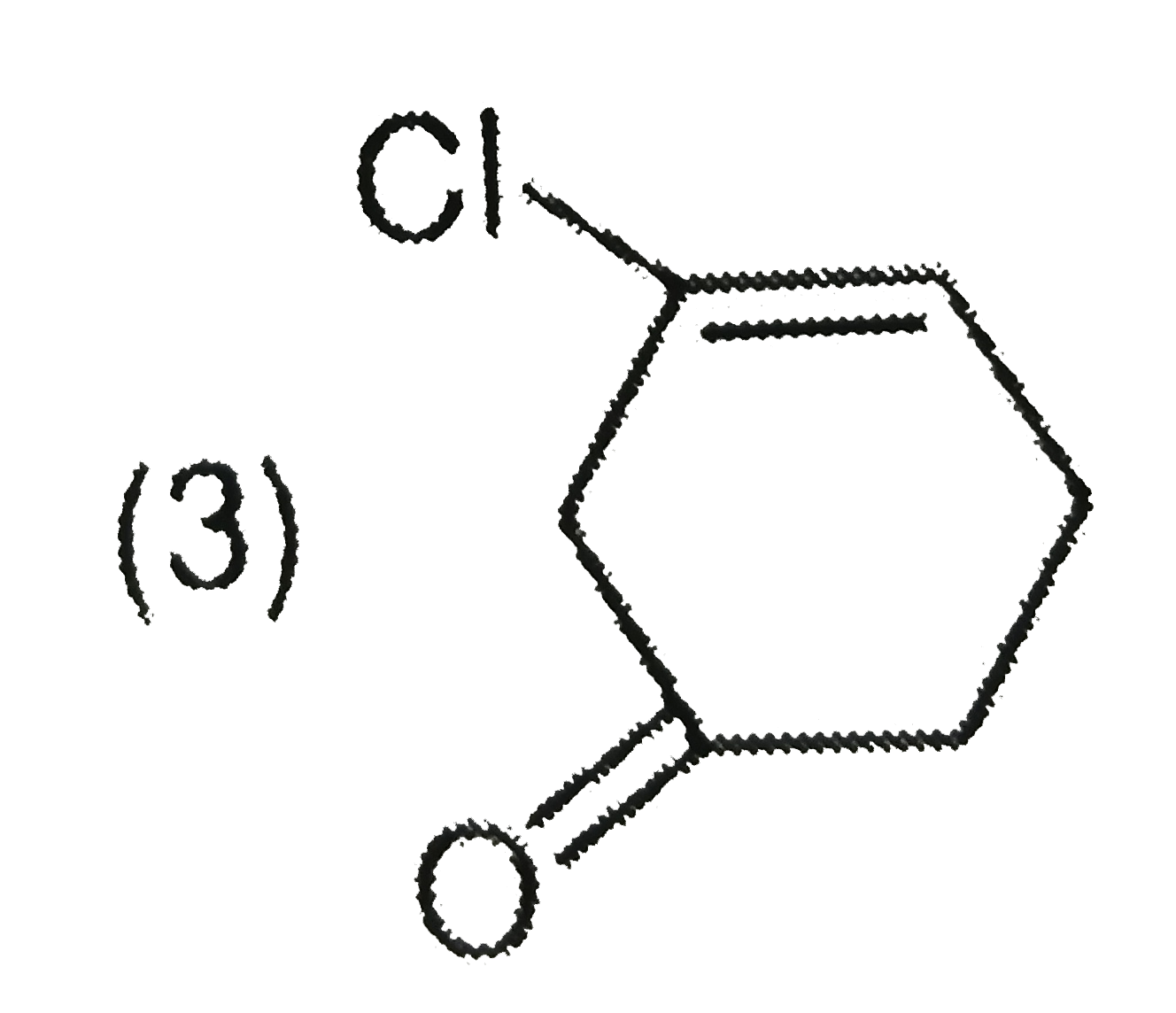
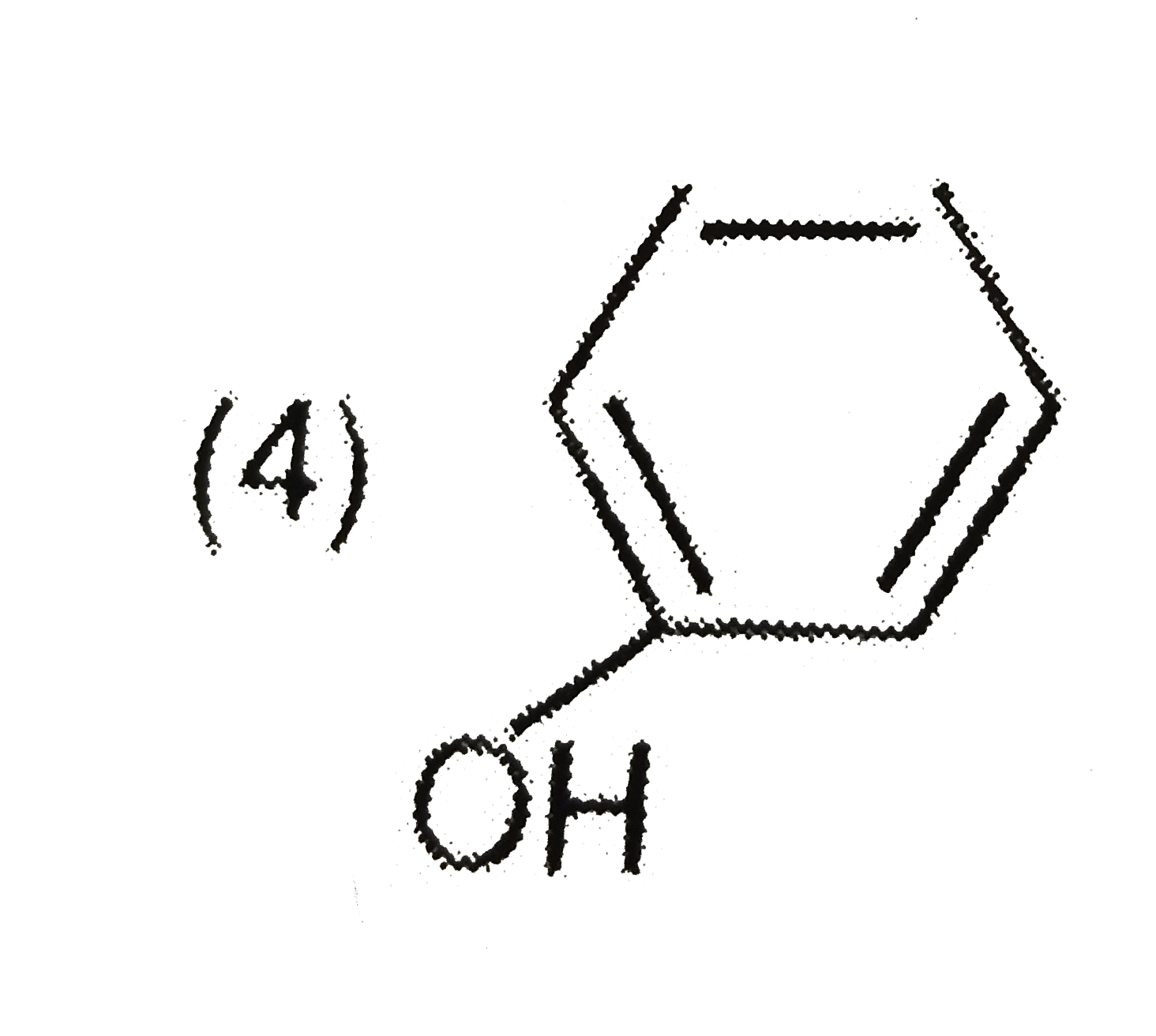
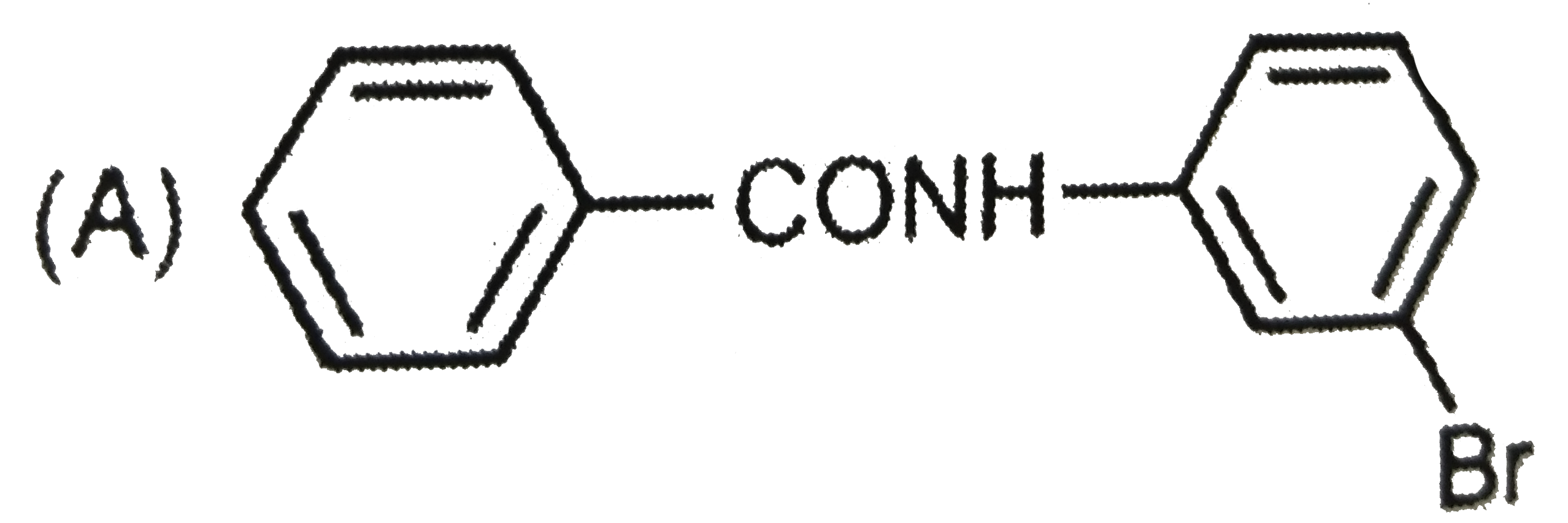
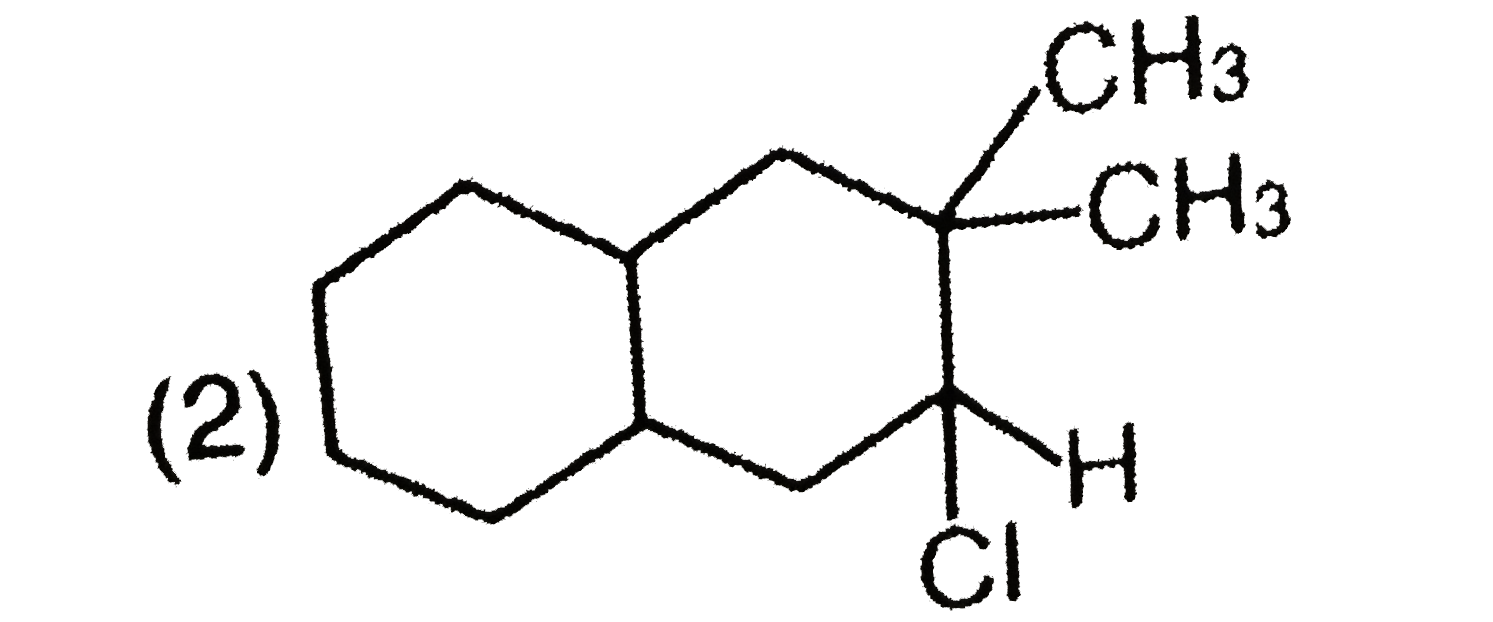
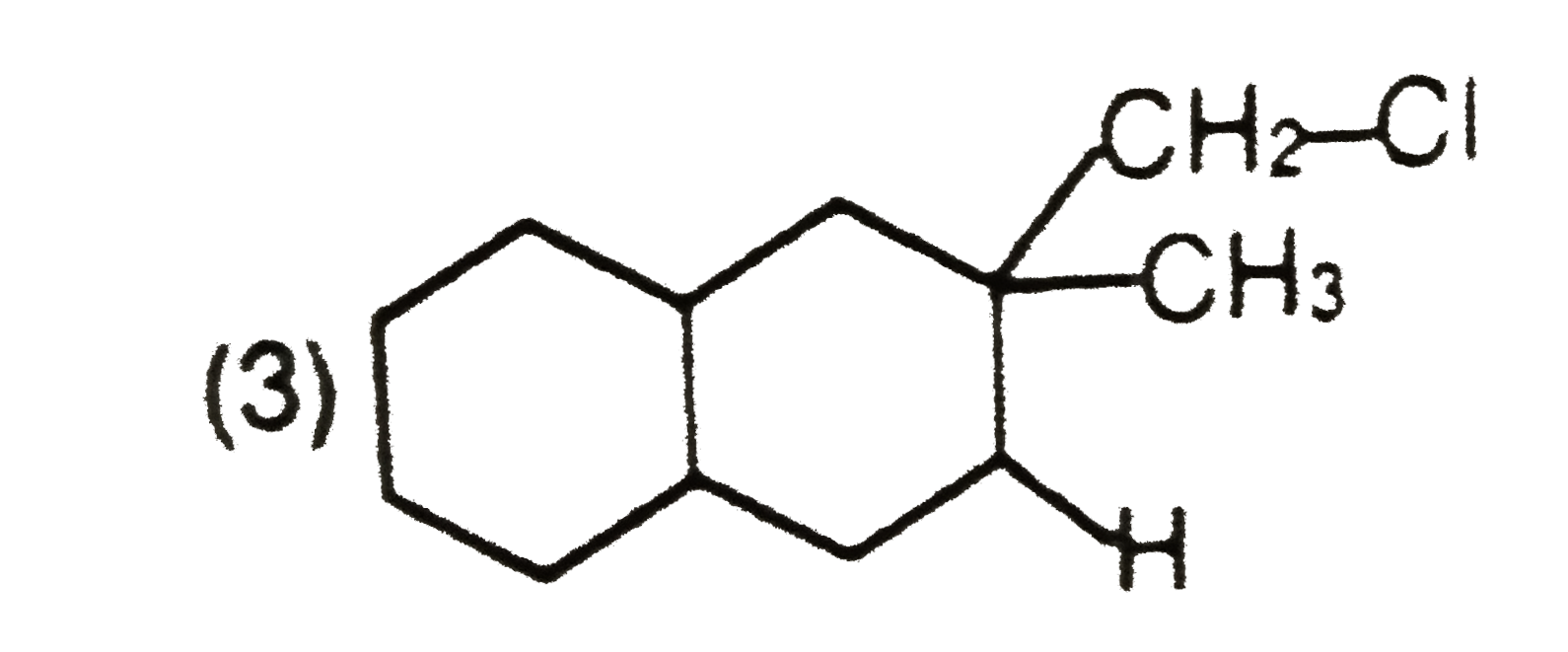
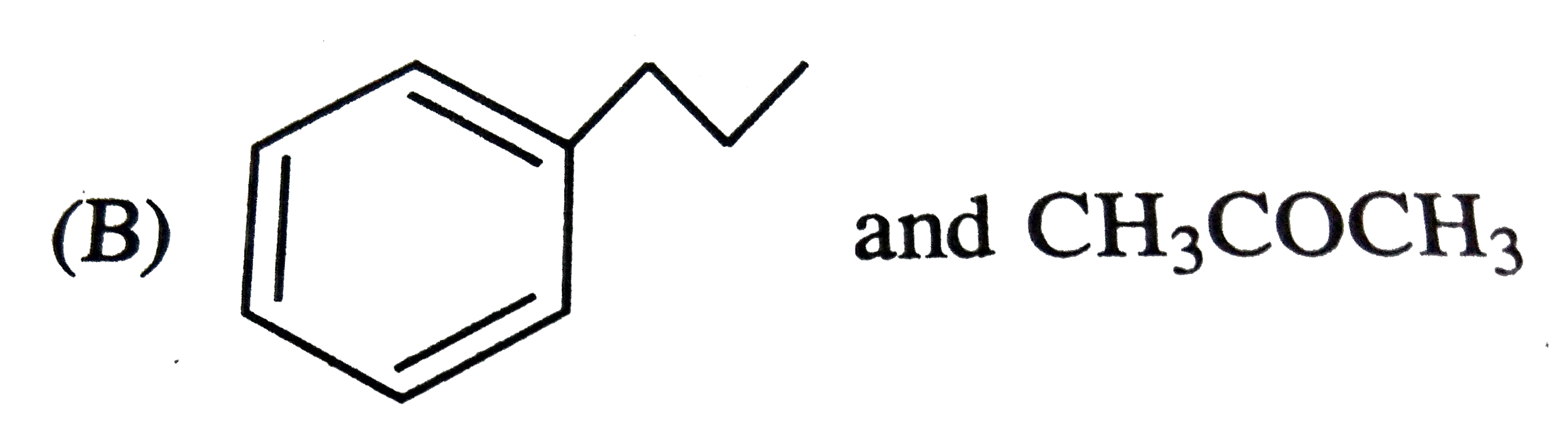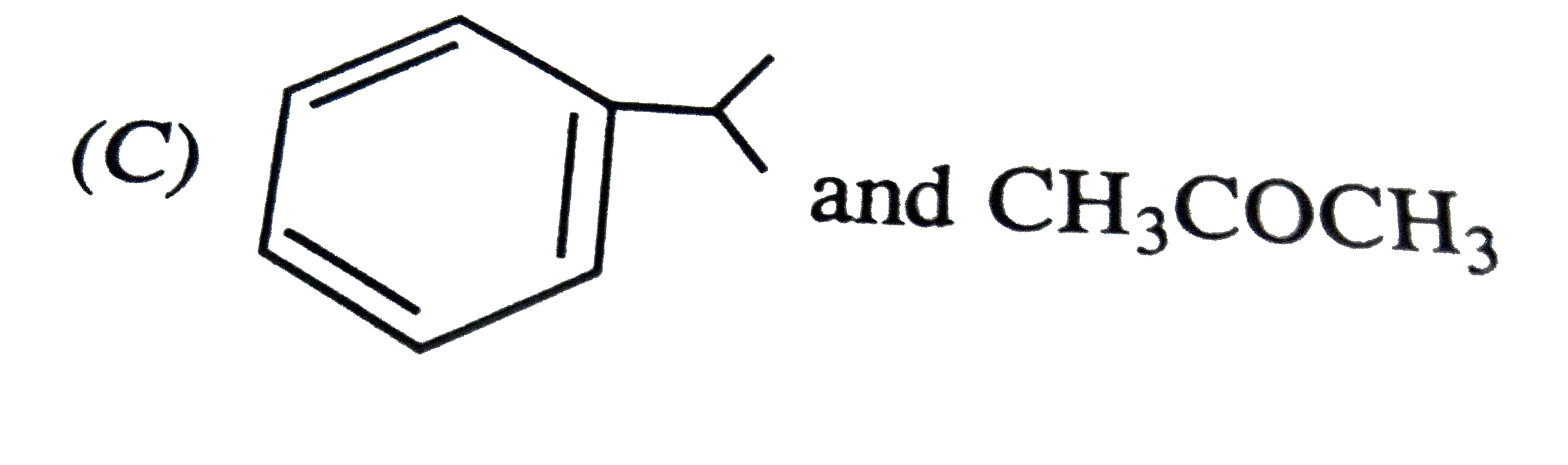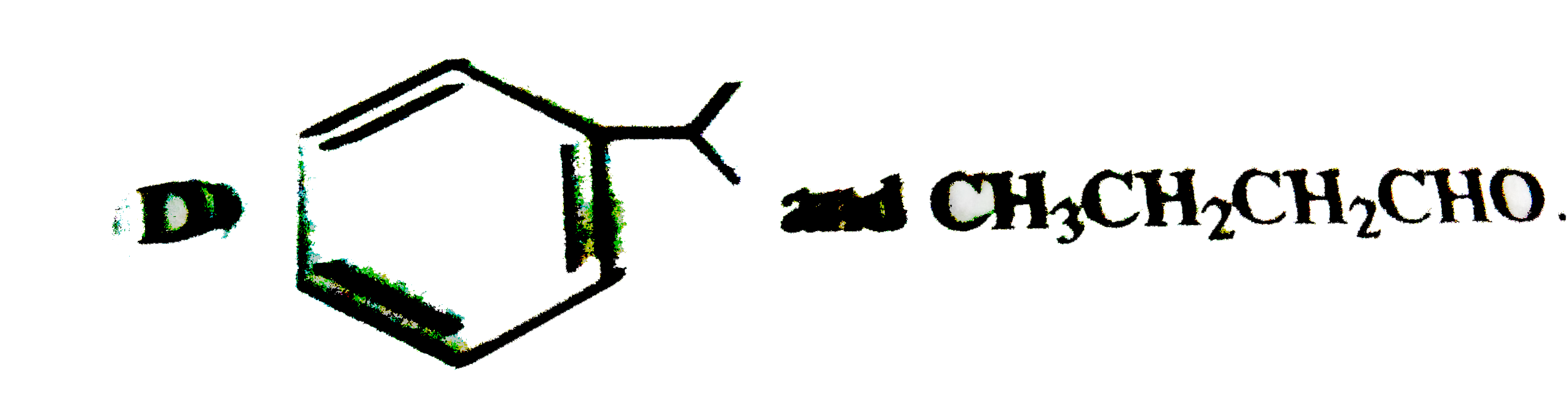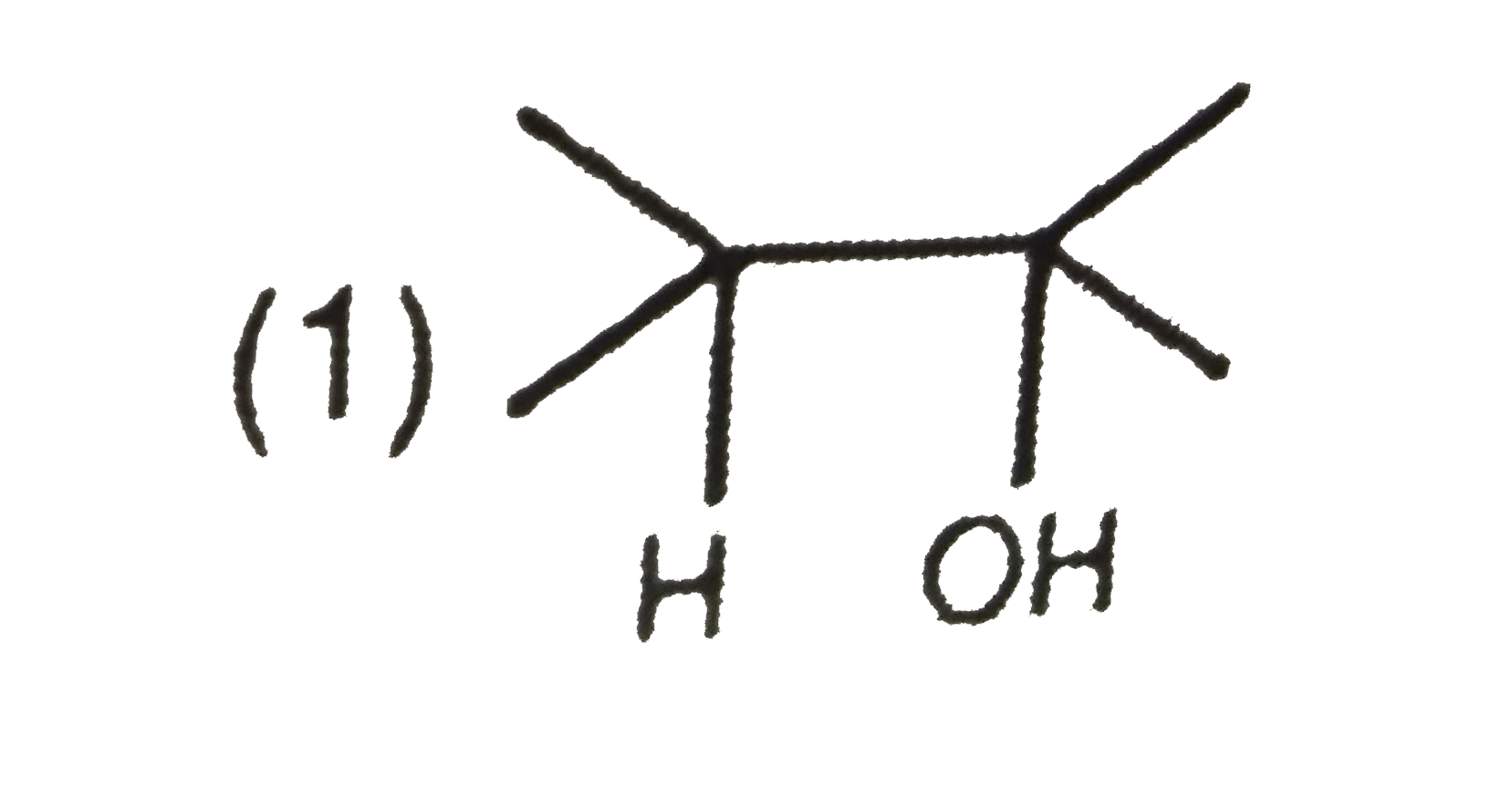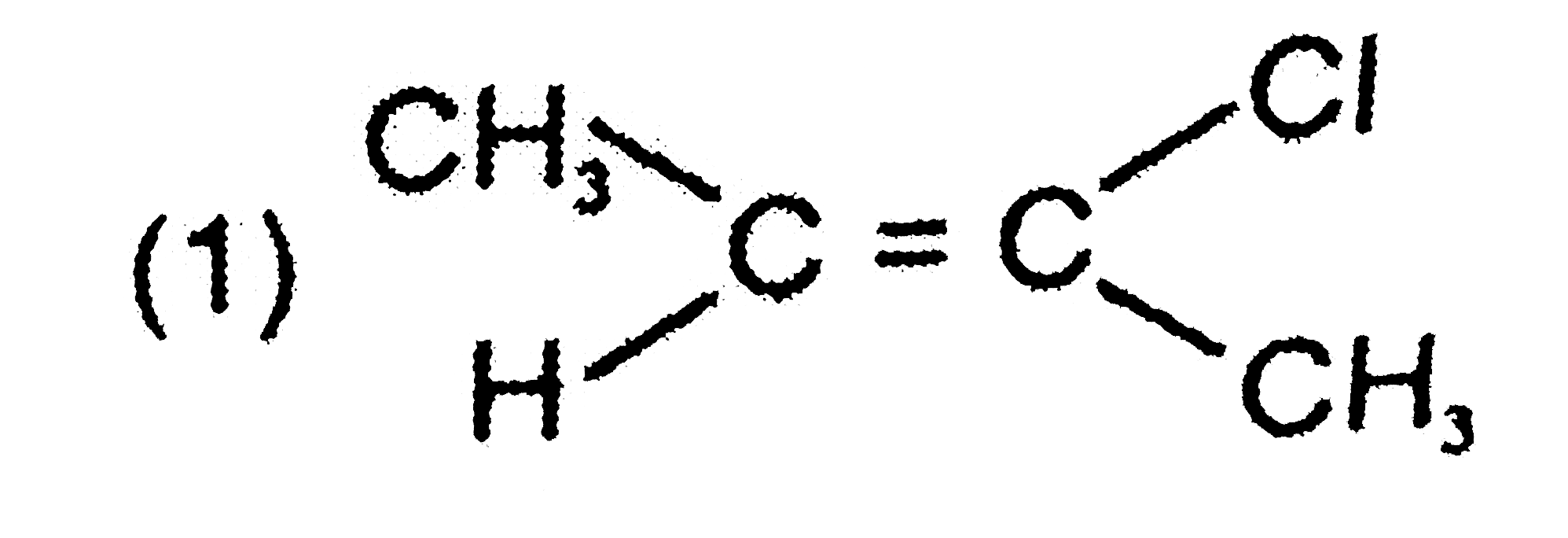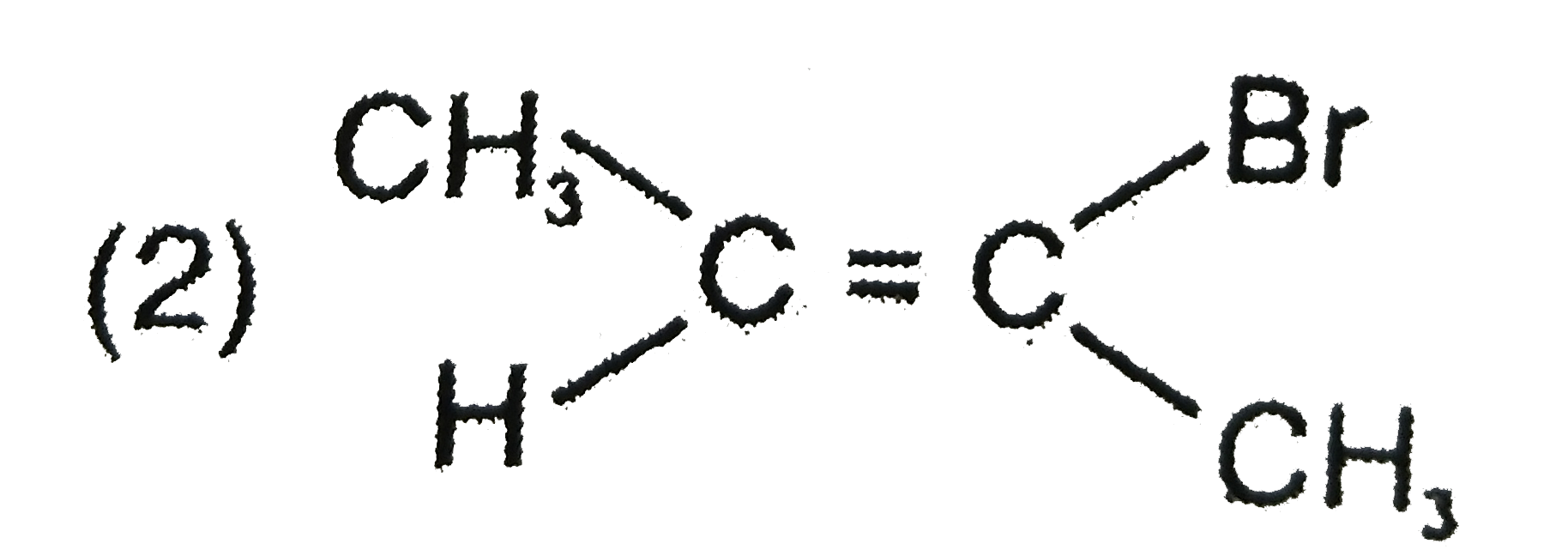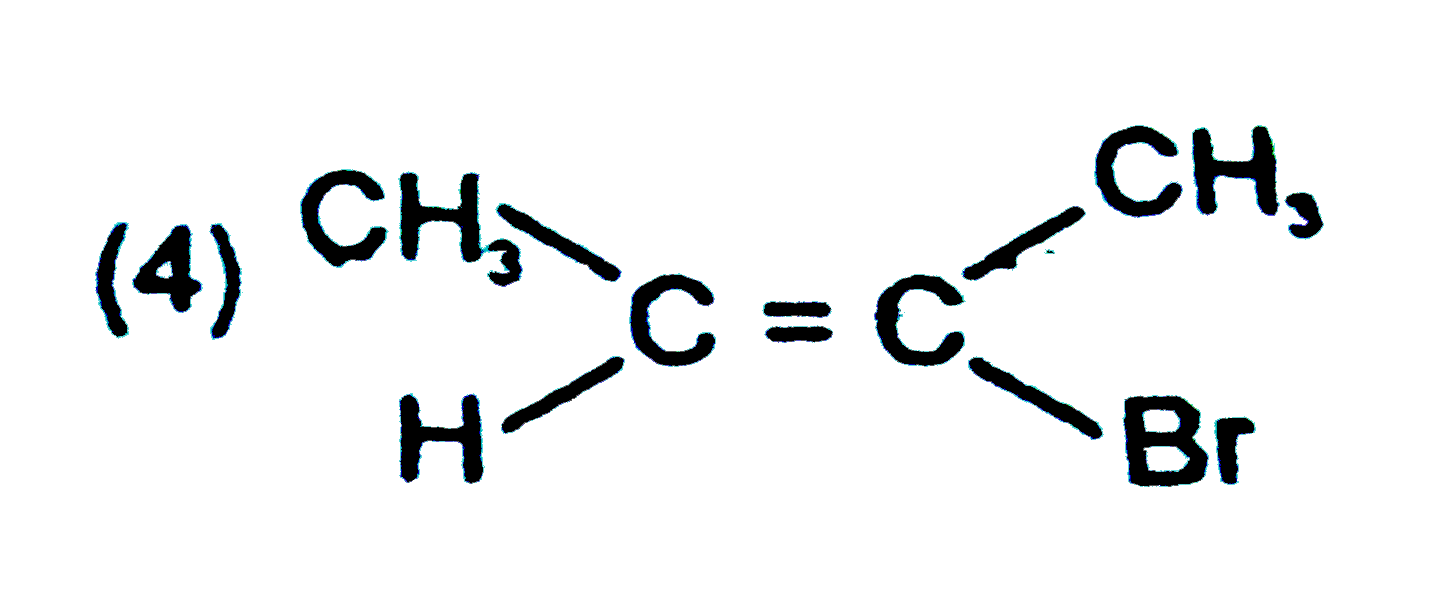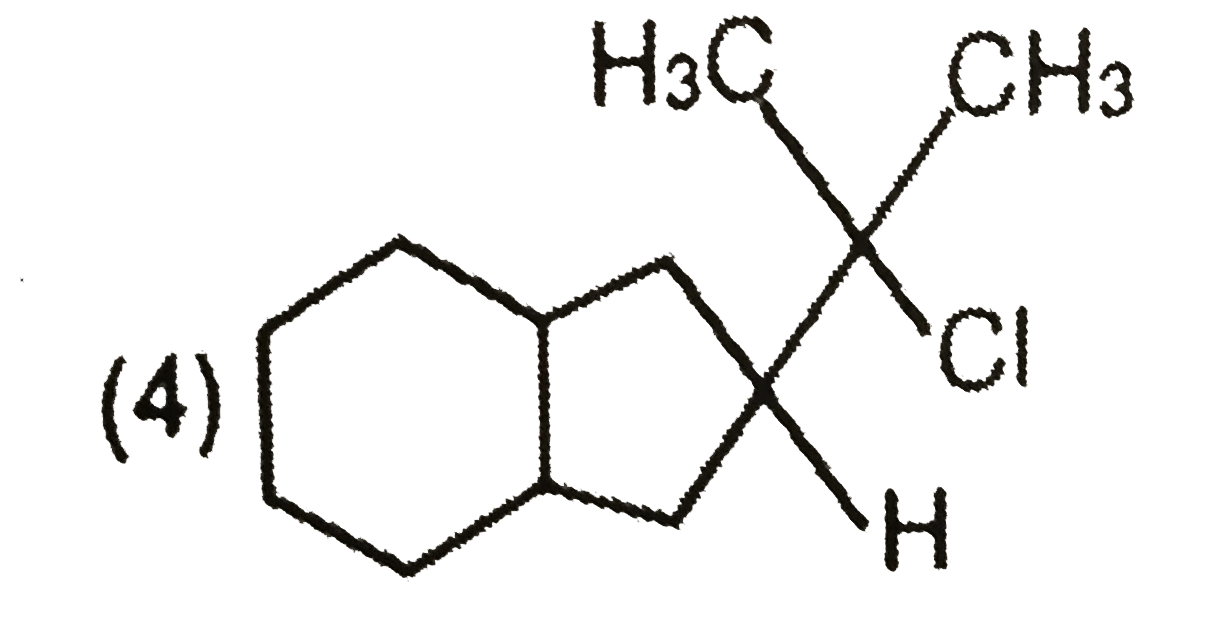Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The mass of an electron is 9.1xx10^(-31) kg. If its K.E. is 3.0xx10^(-25)J, calculate its wavelength. |
|
Answer» Solution :`K.E.=1/2 MV^(2)` `:. V=sqrt((2 K.E.)/m)=sqrt((2xx3.0xx10^(-25) J)/(9.1xx10^(-31) KG))=812 ms^(-1)""(1 J=1 kg m^(2) s^(-2))` By de Broglie EQUATION, `lambda=h/(mv)=(6.626xx10^(-34) Js)/((9.1xx10^(-31) kg)(812 ms^(-1)))=8.967xx10^(-7) m=8967 Å` |
|
| 2. |
The mass of an electron is 9.1 xx 10^(-31) kg. If its K.E. is 3.0 xx 10^(-25)J, calculate its wavelength |
|
Answer» Solution :Here, we are GIVEN: KINETIC energy, i.e., `(1)/(2) mv^(2) = 3.0 xx 10^(-25)J` `m = 9.1 xx 10^(-31) kg, h = 6.6 xx 10^(-34) kg m^(2) s^(-1)` `:. (1)/(2) xx (9.1 xx 10^(-31)) v^(2) = 3.0 xx 10^(25) or v^(2) = (3.0 xx 10^(-25) xx 2)/(9.1 xx 10^(-31)) = 659340 or v = 812 m s^(-1)` `:. lamda = (h)/(mv) = (6.626 xx 10^(-34) kgm^(2) s^(-1))/((9.1 xx 10^(-31) kg) xx 812 ms^(-1)) = 8967 xx 10^(-10) m = 896.7 nm` ALTERNATIVELY, K.E. `= (1)/(2) mv^(2) or v = sqrt((2K.E.)/(m))` `lamda = (h)/(mv) = (h)/(m) xx sqrt((m)/(2K.E.)) = (h)/(sqrt(2m xx K.E.)) = (6.626 xx 10^(-34) kg m^(2) s^(-1))/(sqrt(2 xx 9.1 xx 10^(-31) kg xx 3.0 xx 10^(-25) j)) = 8967 xx 10^(-10)m` |
|
| 3. |
The mass of an electorn is 9.1xx10^(-31) Kg.If its Kinetic energy is 3.0xx10^(-25)J,calculate its wavelength. |
|
Answer» Solution :Step I.Calculation of the velocity of elctron Kinetic energy`=1/2mv^(2)=3.0xx10^(-25)J=3.0xx10^(-25)Kgm^(2)s^(-2)` `v^(2)=(2xxK.E)/(M)=(2xx(3.0xx10^(-25)kGm^(2)s^(-2))/((9.1xx10^(-31)Kg))=65.9xx10^(4)m^(2)s^(-2))` `v=(65.9xx10^(4)m s^(-2))(1/2)=8.12xx10^(2)ms^(-1)` Step II.Calulation of wavelength of the ELECTRON according to de Broglie.s equation, `gamma=h/(mv)=((6.626xx10^(-34)kgm^(2)s^(-1)))/((9.1xx10^(-31)kg)XX(8.12xx10^(2)ms^(-1)))` `=0.08967xx10^(-5)m=8967xx10^(-10)m=8967A( :.1A=10^(-10)m)` |
|
| 4. |
The mass of an atom of oxygen is: |
|
Answer» 16 amu |
|
| 5. |
The mass of 1 atom of nitrogen is |
|
Answer» |
|
| 6. |
The mass of an atom of ""_(2)He^(4) is 4.0026 amu . The mass of a neutron and a proton are 1.0087 amu and 1.0078 amu respectively. The nuclear binding energy per nucleon is nearly |
|
Answer» Solution :Massdefect `=[2(1.0087+1.0078)-4.0026]` AMU =0.0304 amu Binding ENERGY per nucleon `=(28.32)/(4)` MeV `=7.089` MeV `~= 7` MeV |
|
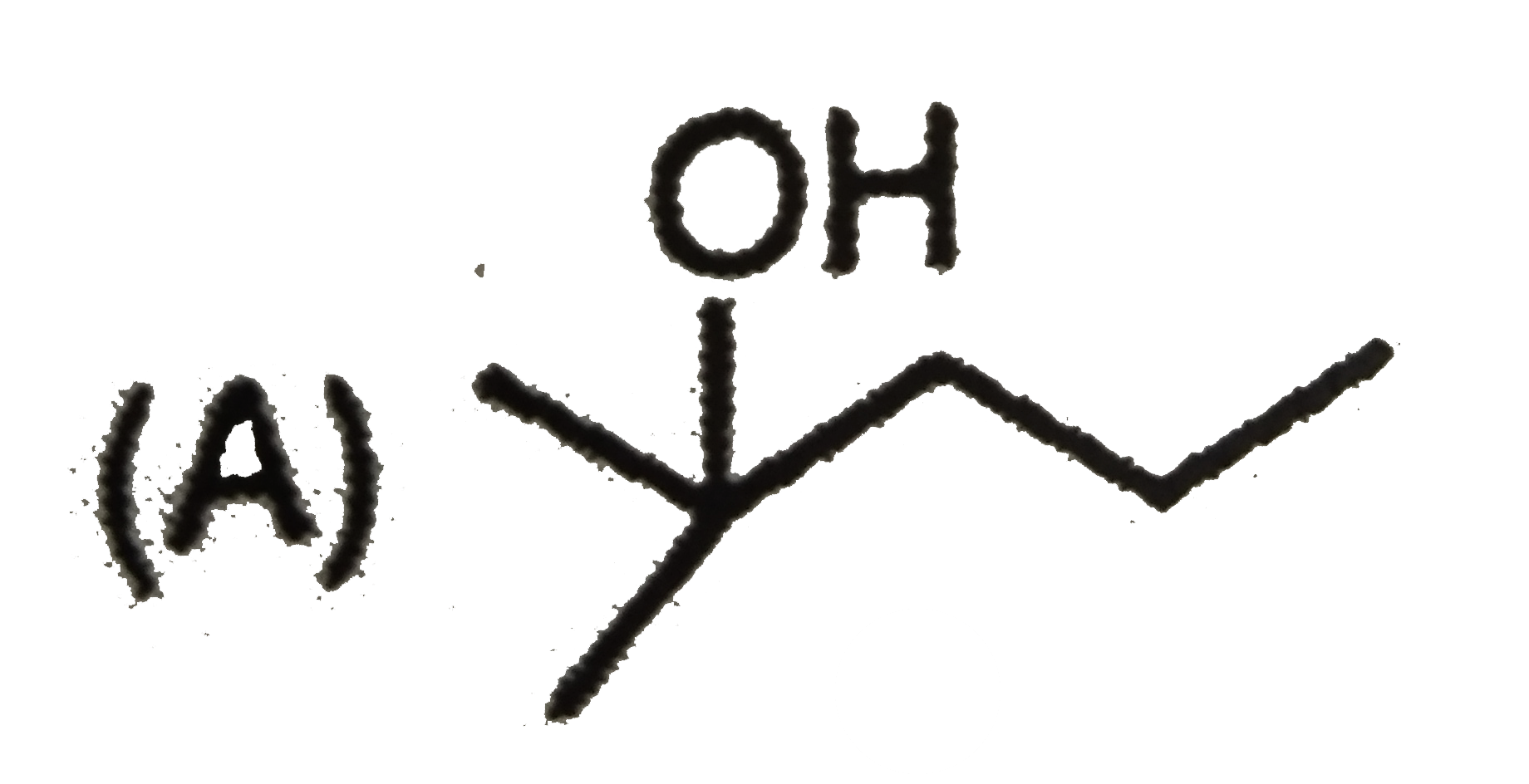
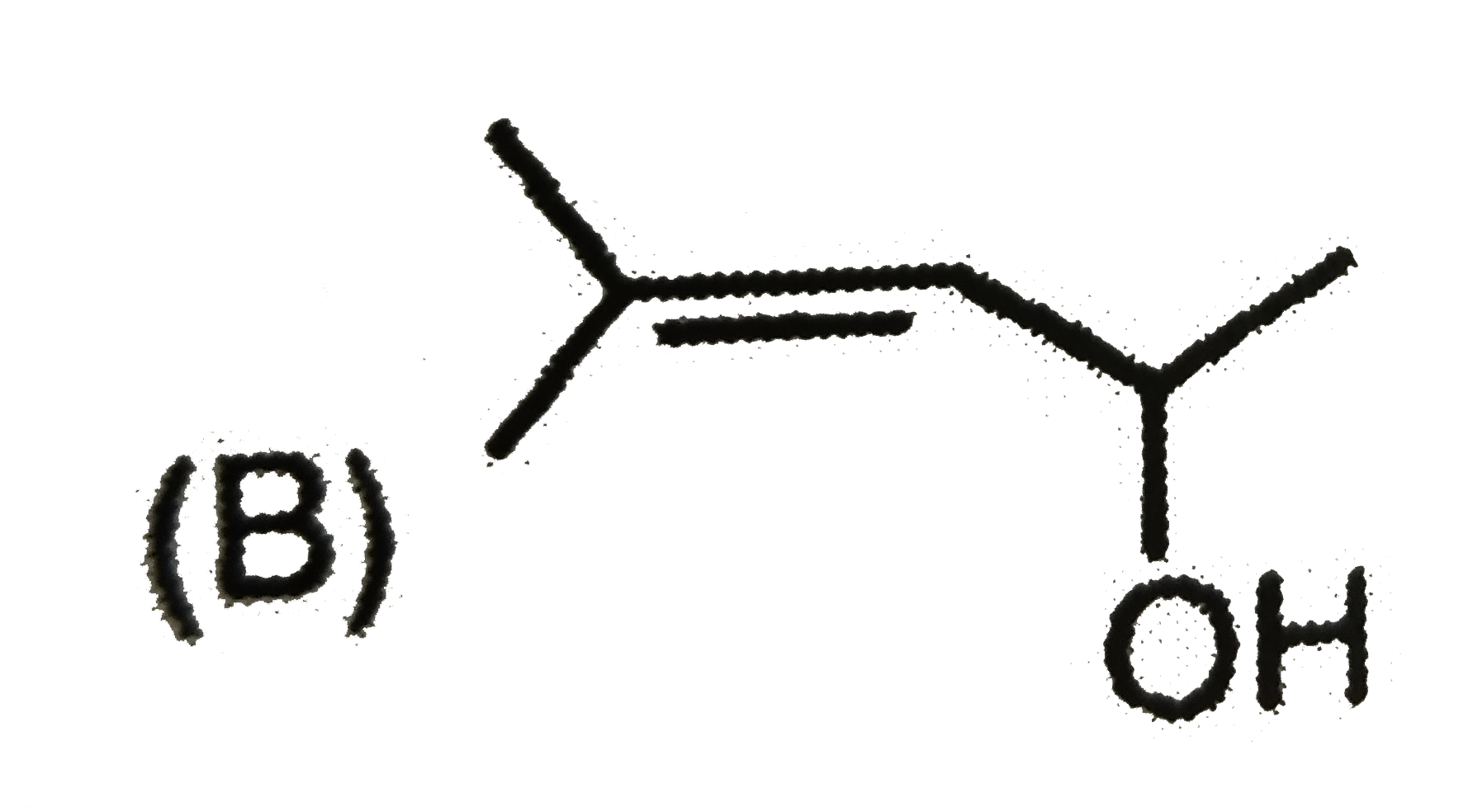
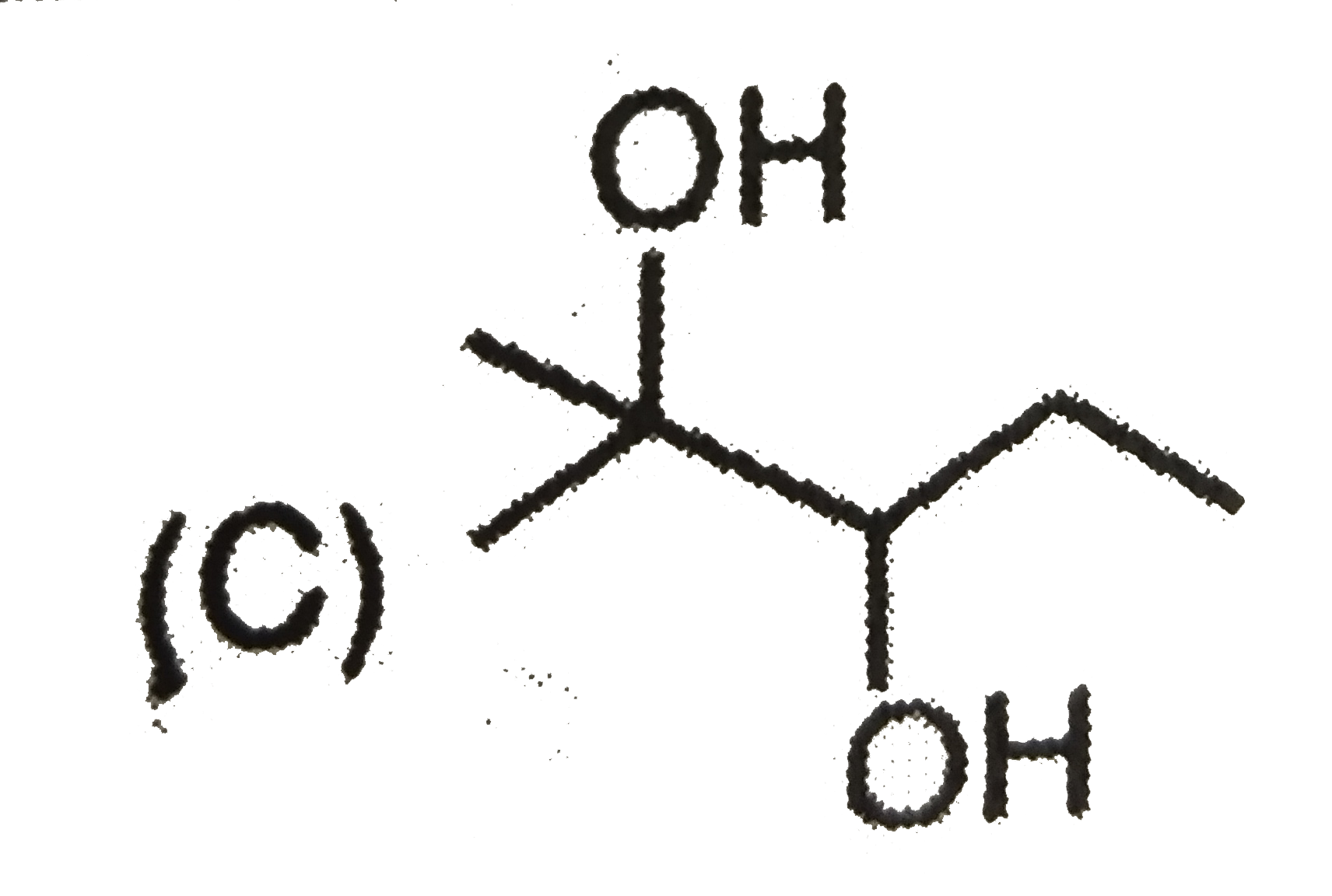
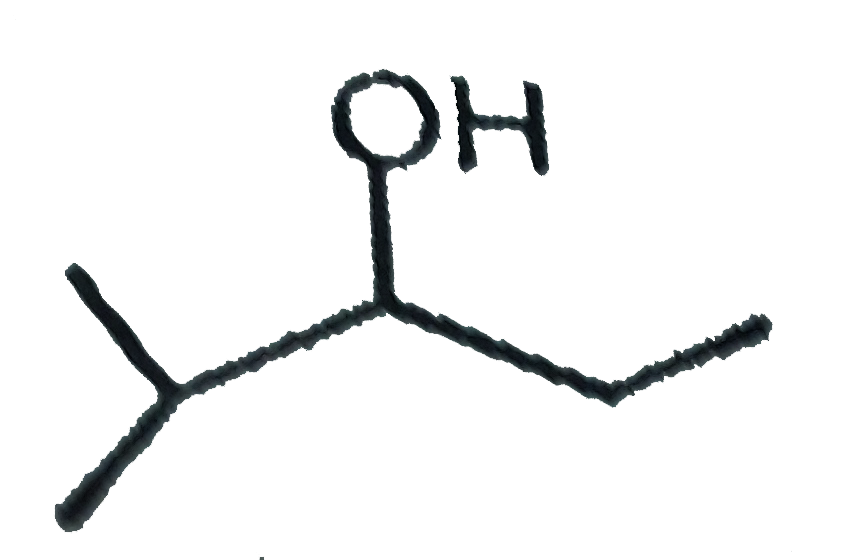
| 7. |
The mass of acetic acid present in 500 ml solution in which it is 1% ionised ( K_(a) of CH_(3) COOH = 1.8 xx 10^(-5)) |
|
Answer» `10.8` G |
|
| 8. |
The mass of a unit cell of element is the product of the atomic mass of the element and _______ divided further by __________ |
|
Answer» |
|
| 9. |
The mass of a silver coin is 10.0 g. A person can carry a load of 40 kg. How many persons will be required to carry one Avogadro number of such coins? |
|
Answer» |
|
| 10. |
The mass of a sample of metal is 8.3432 g. If the density of the metal is 19.3 g cm^(-3), what is the volume of the sample? |
|
Answer» |
|
| 11. |
The mass of a photon of wavelength 1.54 Å is |
|
Answer» `2.5xx10^(-32)` kg where `v=3xx10^(8)m//sh=6.625xx10^(-34)` Js `lambda =1.54xx10^(-10)`m `:. M=1.42xx10^(-32)` kg |
|
| 12. |
The mass of a particle is 10^(10)g and its radius is 2 xx 10^(-4)cm. If its velocity is 10^(-6) cm sec^(-1) with 0.0001% uncertainty in measurement, the uncertainty in its position is : |
|
Answer» `5.2 XX 10^(-8) m` |
|
| 13. |
The mass of a photon with a wavelength equal to 1.54xx10^(-8) cm is |
|
Answer» `0.8268xx10^(-34)` kg The velocity of photon (v)`=3xx10^8 msec^(-1)` `lambda=1.54xx10^(-8) cm =1.54xx10^(-10)` meter `therefore m=(6.626xx10^(-34)JS)/(1.54xx10^(-10) m XX 3xx 10^8 m sec^(-1))` `=1.4285xx10^(-32)` kg |
|
| 14. |
The mass of a non-voltalle solute (molar mass 80 g mol ^(-1) ) which should be dissolved in 92 g of toluene to reduce its vapour pressure to90 % |
| Answer» ANSWER :D | |
| 16. |
The mass of a gas that occupies a volume of 612.5 ml at room temperature and pressure (25^(@)C and 1 atm pressure) is 1.1 g. The molar mass of the gas is |
|
Answer» 66.25 G `mol^(-1)` `612xx10^(-3)L//24.5molL^(-1)` 0.025moles We know that, Molar mass=Mass no. /no. of moles 1.1g/0.025 mol=44g `mol^(-1)` |
|
| 17. |
The mass of a mole of electrons is:0.008 g0.184 g0.55 mg1.673 |
|
Answer» 0.008 G |
|
| 18. |
The mass of 750 mL of a gas collected at 25°Cand 716.2 mm pressure is found to be equal to 0.809 g. Calculate the molecular mass of the gas. |
|
Answer» Solution :Suppose, the volume of the gas at S.T.R is `V_2`. According to the gas equation, `(P_(1)V_(1))/T_(1) = (P_(2)V_(2))/T_(2)` In the PRESENT case, `P_(1) = 716.2 mm, V_(1) = 750 mL, T_(1) = 25^(@) C = 298 K` `P_(2) = 760 mm, V_(2) = ?, T_(2) = 273 K` SUBSTITUTING the values, we have `(716.2 xx 750)/298 = (760 xx V_(2))/273` or `V_(2) = (716.2 xx 750 xx 273)/(760 xx 298) = 647.48 mL` `therefore 22400` mL of the gas at S.T.P. will weigh `=0.809/(647.48) xx 22400 = 27.99 g` Hence, the molecules mass of the gas =27.99 |
|
| 19. |
The mass of 80% pure H_(2)SO_(4) required to completely neutralise 60g of NaOH is |
|
Answer» 92g |
|
| 20. |
The mass of 50% (mass/mass) solution of HCI required to react with 100g of CaCO_3would be |
|
Answer» 73 g `W/(36.5) =100/50 , W_(" HCI") = 73` gam 50 g of HCLIS present in 100GM of HCI solution 73g of HCI `rarr "?"`= 146 gmof HCl |
|
| 22. |
The mass of 216.5 mL of a gas at S.T.P. is found to be 0.6862 g. Calculate the molecular mass of the gas. Also calculate its atomic mass if the gas is diatomic. |
| Answer» Solution :Molecular MASS = 71 AMU, ATOMIC mass = 35.5 amu | |
| 23. |
The mass of 1.5xx10^(20) atoms of an element is 15mg. The atomic mass of the element is |
|
Answer» 60g `6XX10^(23)rarr?` `=(6xx10^(23)xx15xx10^(-3))/(1.5xx10^(20))=60` IMPLIES At. Mass = 60 amu |
|
| 24. |
The mass of 1.5 xx 10^(26 molecules of a substances is 16 kg . The molecular mass of the substance is |
|
Answer» 64 G |
|
| 25. |
The mass of 1.5 xx 10^(20) molecules of a substance is 20 mg . The molar mass of substance is |
| Answer» Answer :C | |
| 26. |
The mass in grams of 0.45 mole of Ca^(2+) ions |
|
Answer» ` Ca to Ca^(2+) + 2e^(-)` 41 g of Ca = 1 mole (for `Ca^(2+)` = Atomic mass remains same) 1 mole of `Ca^(2+)` = 40 g `:.` 0.45 mole of `Ca^(2+)=40/1xx0.45=18g` |
|
| 27. |
The Marsh test is described as follows: In this test, if SbH_(3) and BiH_(3) mixture is sent then which of the following statement is correct? |
|
Answer» SB will be DEPOSITED at the LEFT and BI at the right |
|
| 28. |
The mass and charge of one mole of electrons, respectively is |
|
Answer» `54.8 XX 10^(-7) Kg, 9.65 xx 10^(4)` Coulomb |
|
| 29. |
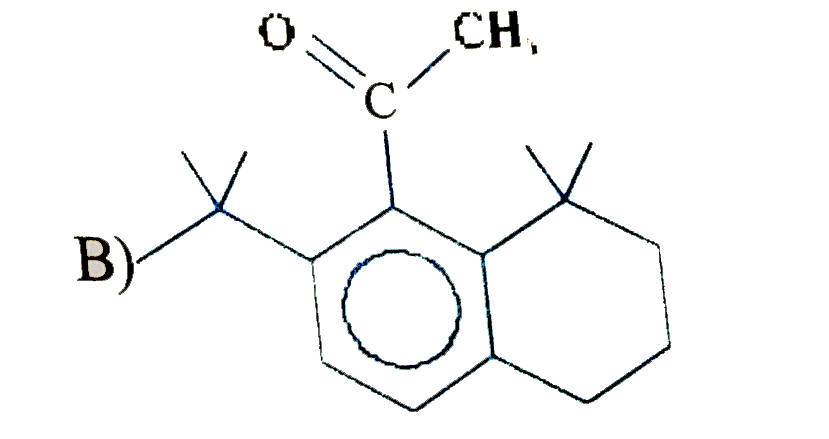
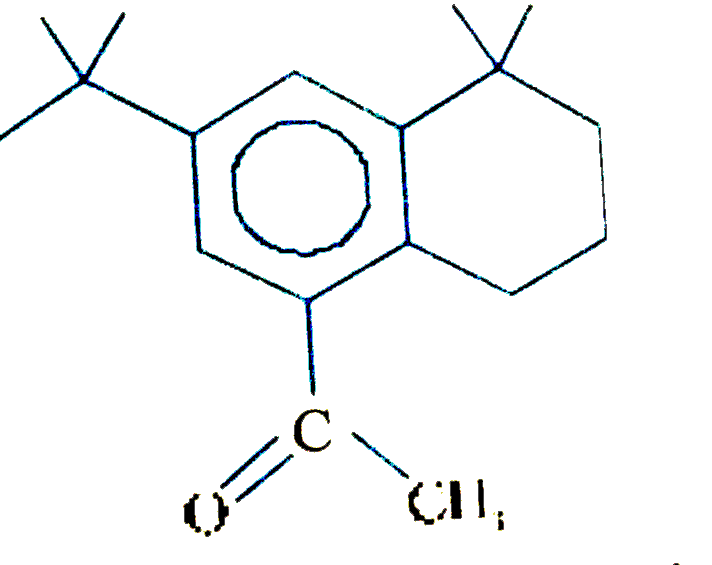
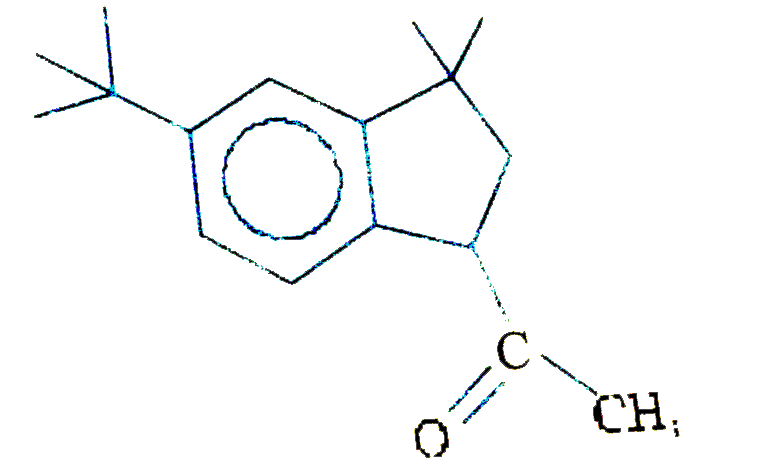
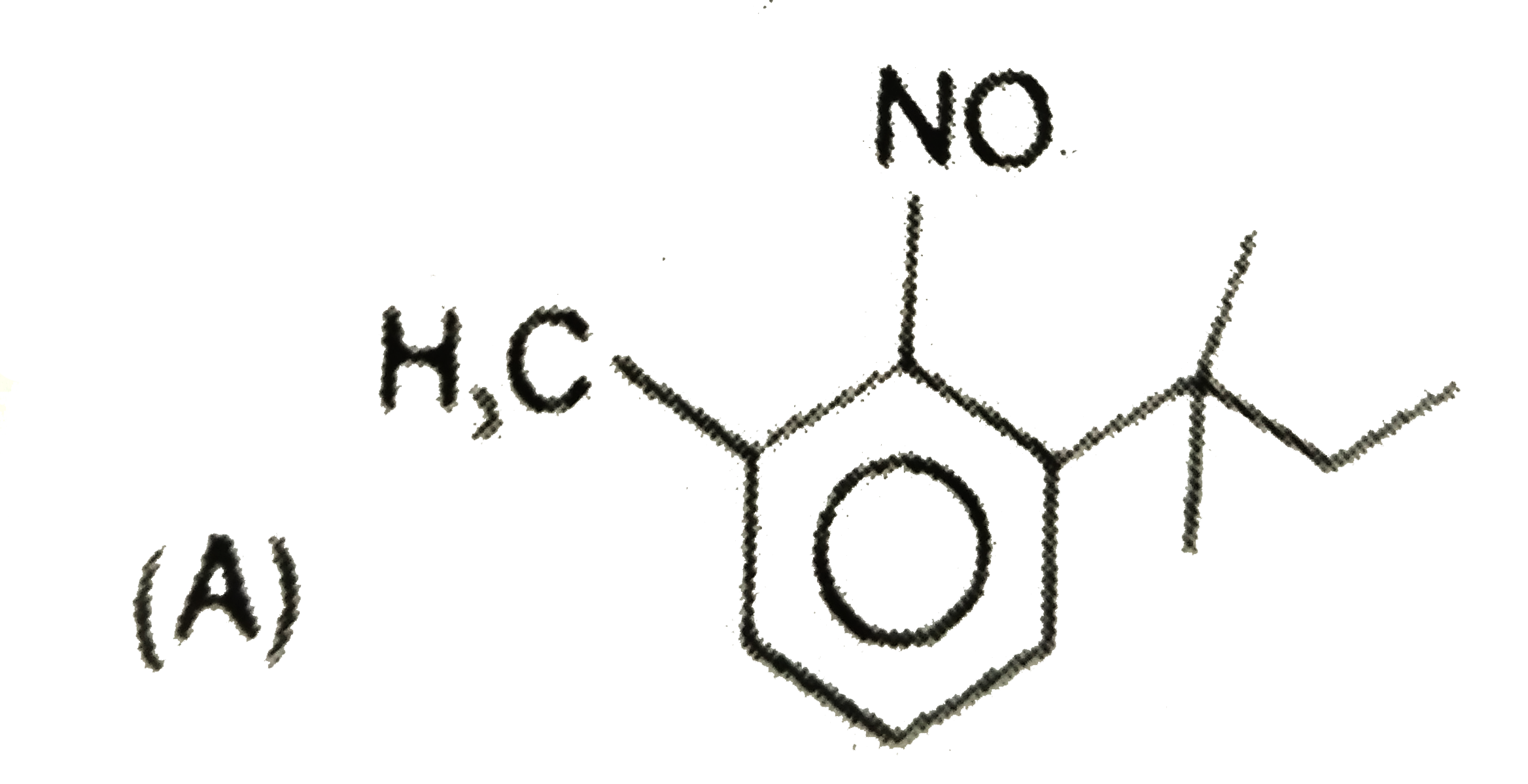
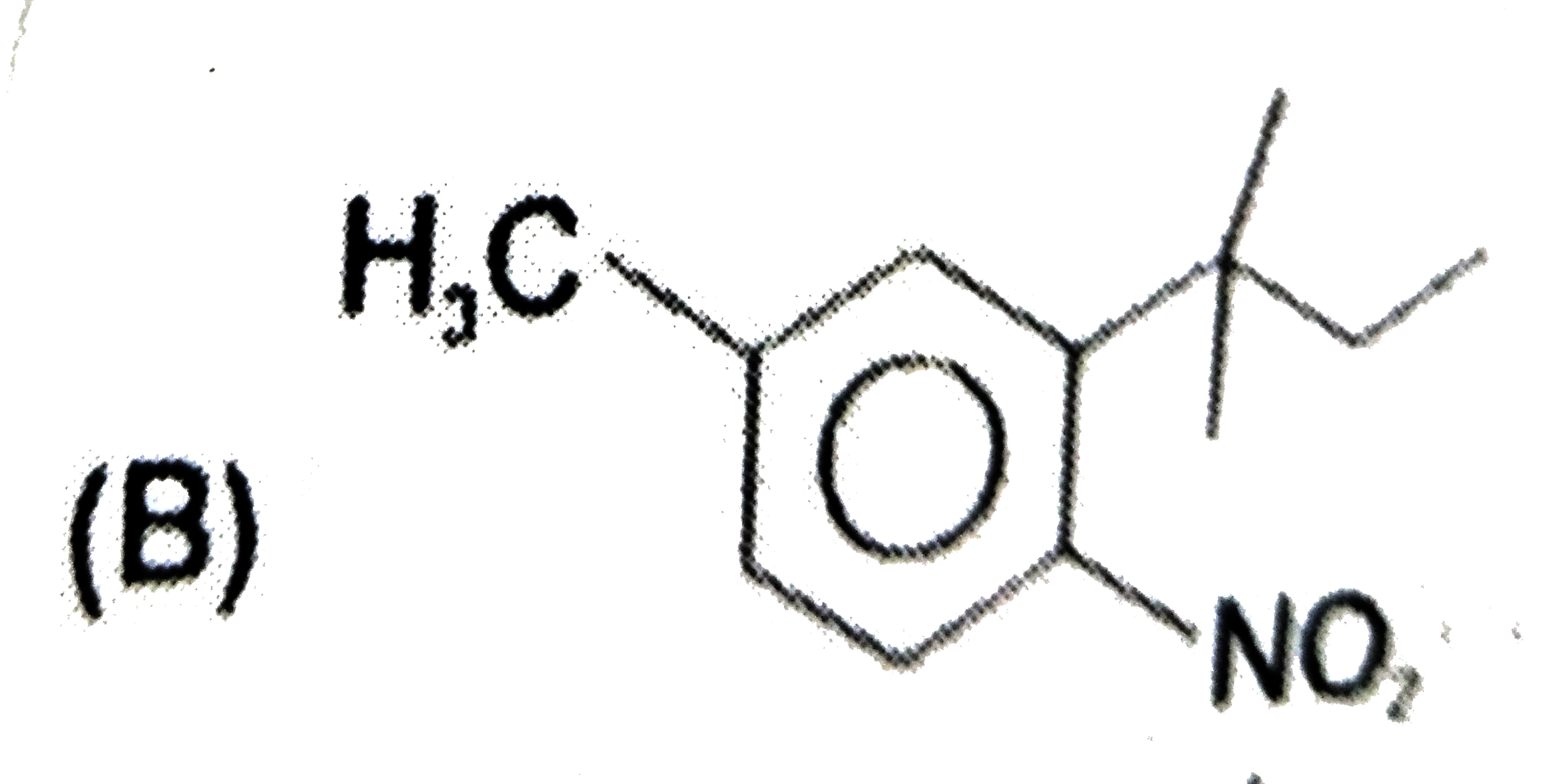
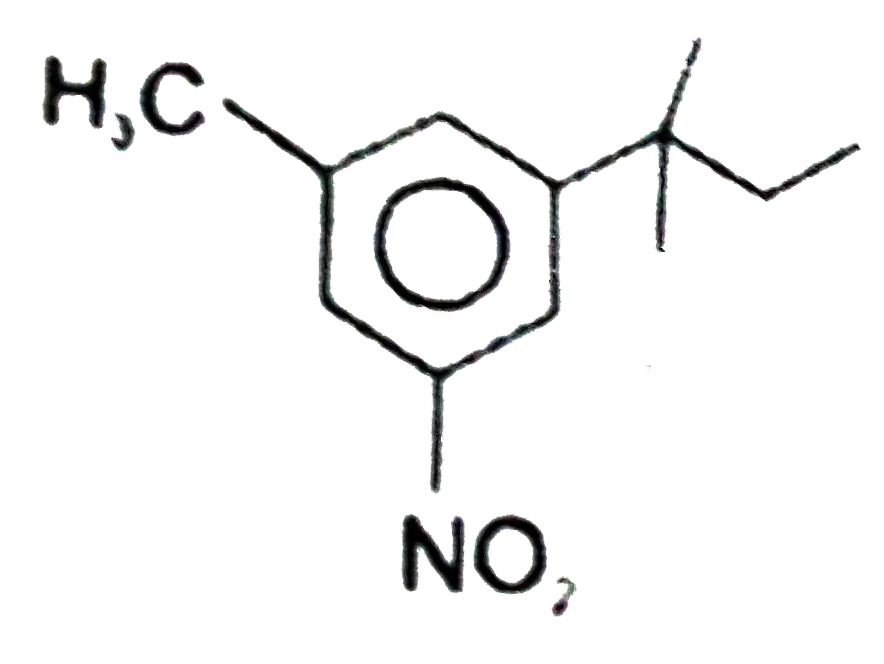
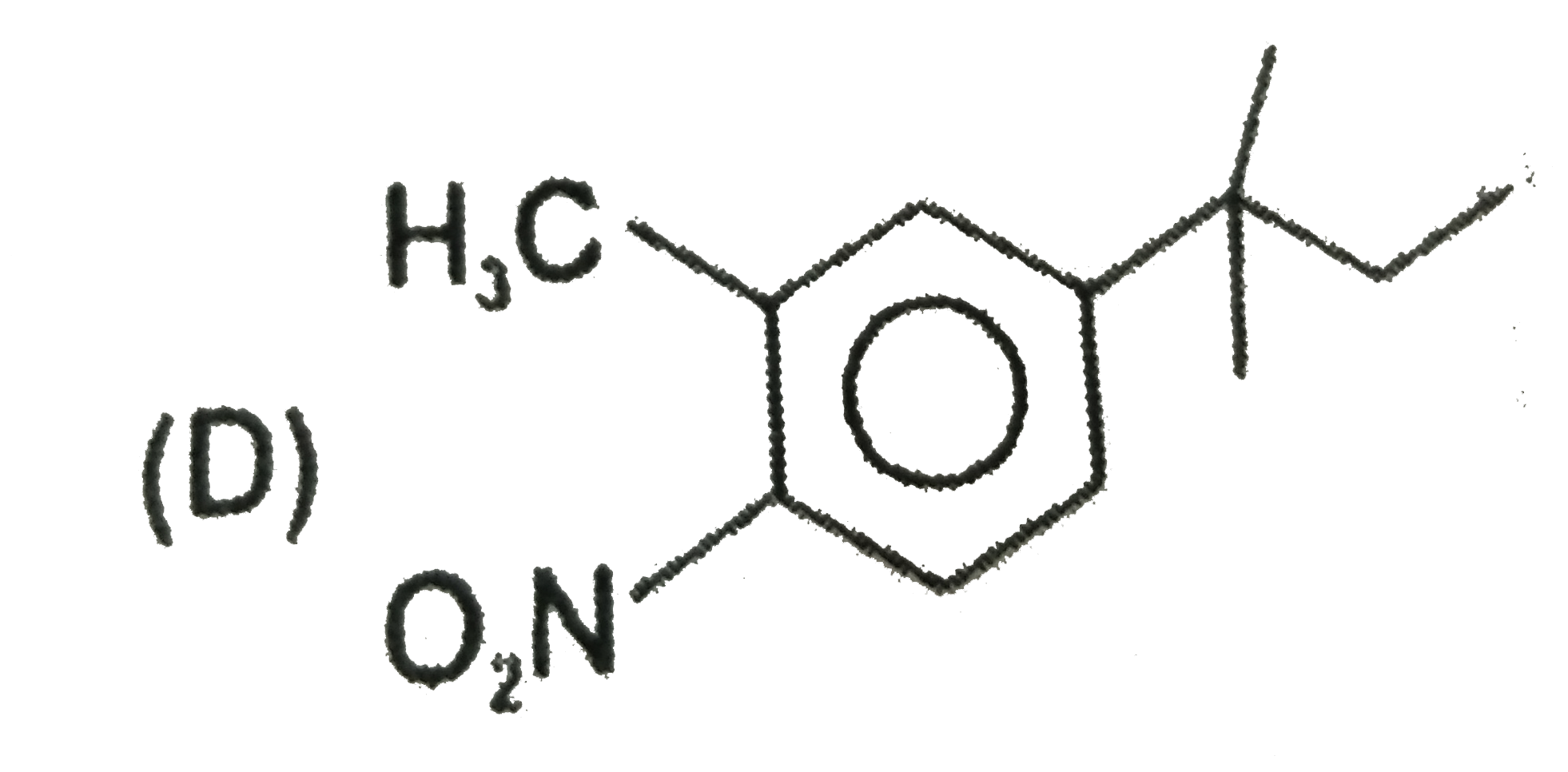
The major substitution product of the following reaction is |
|
Answer»
|
|
| 30. |
The major role of fluorspar, which is added in small quantities in the electrolytic reduction of Al_(2)O_(3) dissolved in fushed cryolite is |
|
Answer» as a catalyst |
|
| 31. |
The major source of CO pollution is : |
|
Answer» FOREST fire |
|
| 32. |
The major reagent present in corey house reaction is …………….. |
| Answer» SOLUTION :Lithiumdimethylcuparate | |
| 34. |
The major products obtained when chlorobenzene is nitrated with HNO_(3) and "con "H_(2)SO_(4) |
|
Answer» 1-chloro-4-nitrobenzene |
|
| 35. |
The major products obtained when chlorobenzene is nitrated with HNO_3 and conc. H_2SO_4 |
|
Answer» 1-chloro-4-nitrobenzene |
|
| 36. |
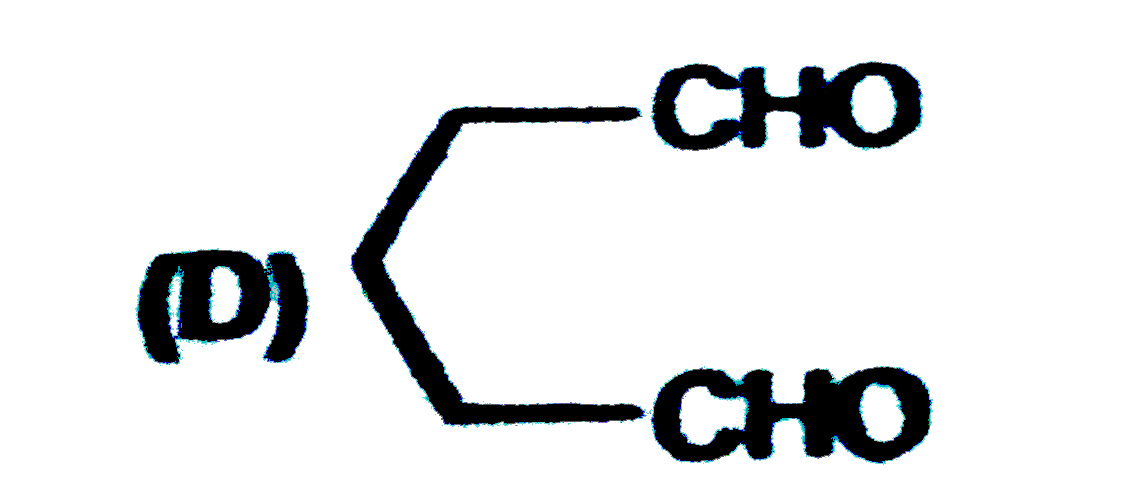
The major products obtained during ozonolysis of 2,3-dimethyl-1-butene and subsequent reductions with Zn and H_2O are |
|
Answer» methanoic ACID and 2-methyl-2-butanone 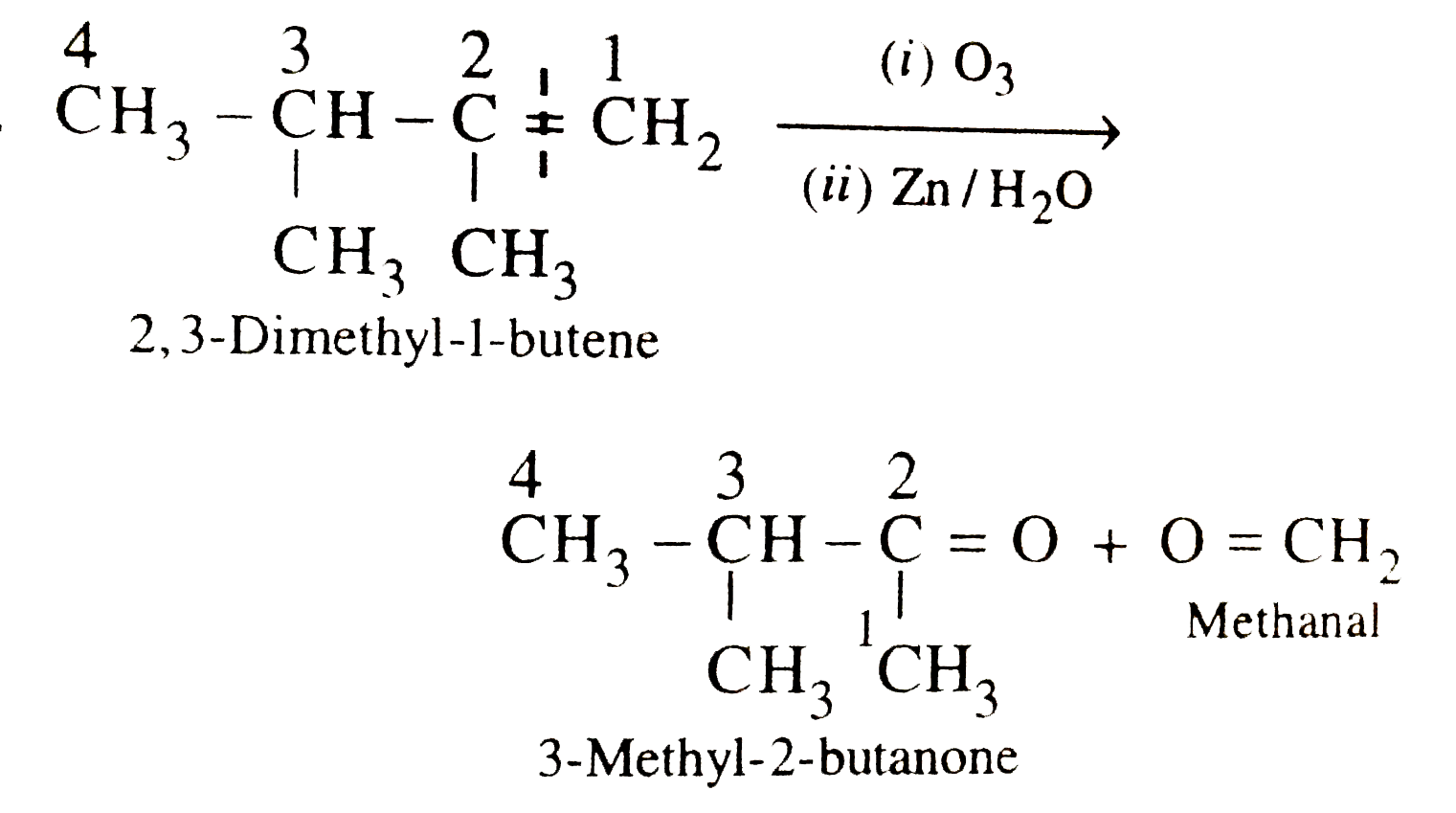
|
|
| 37. |
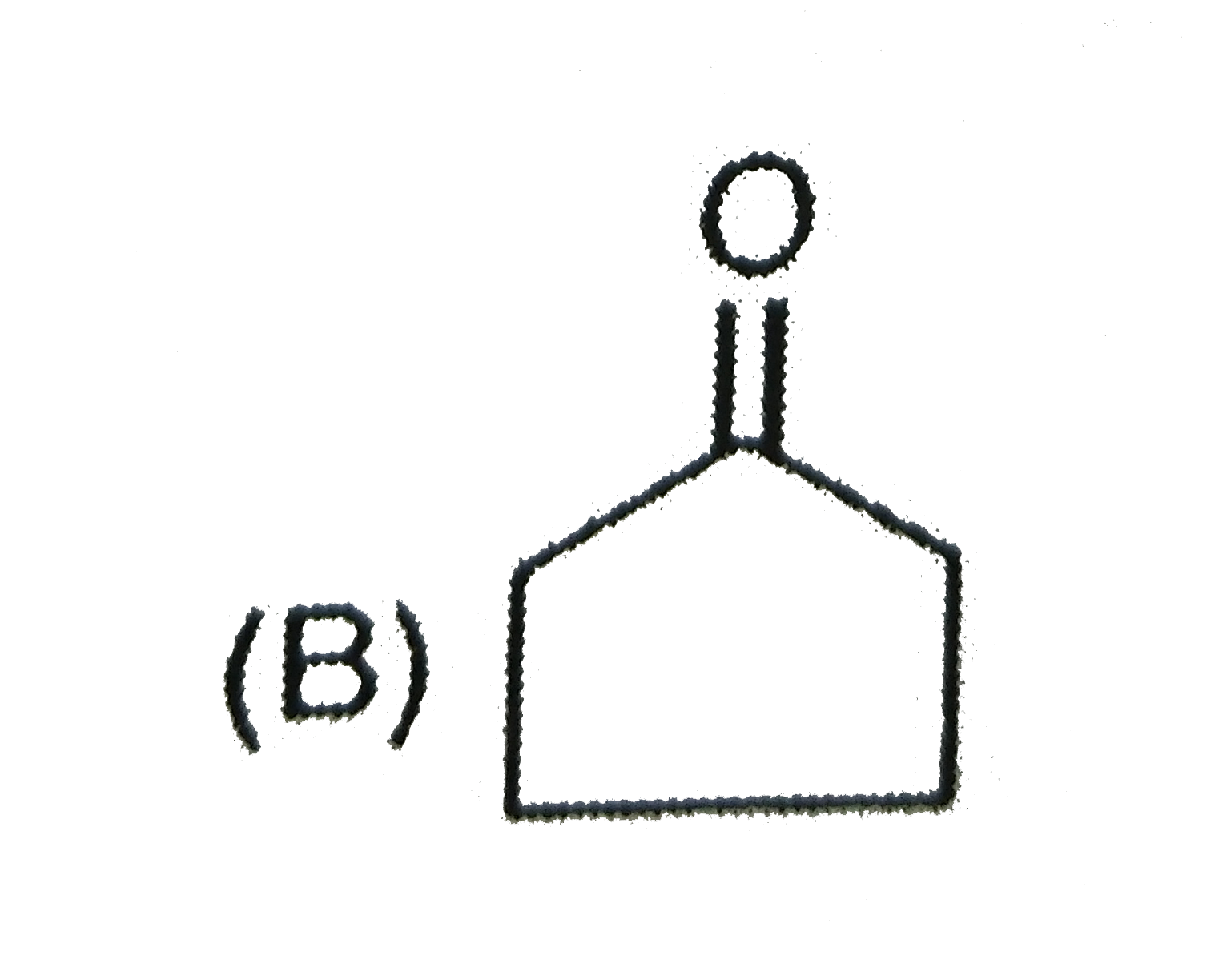
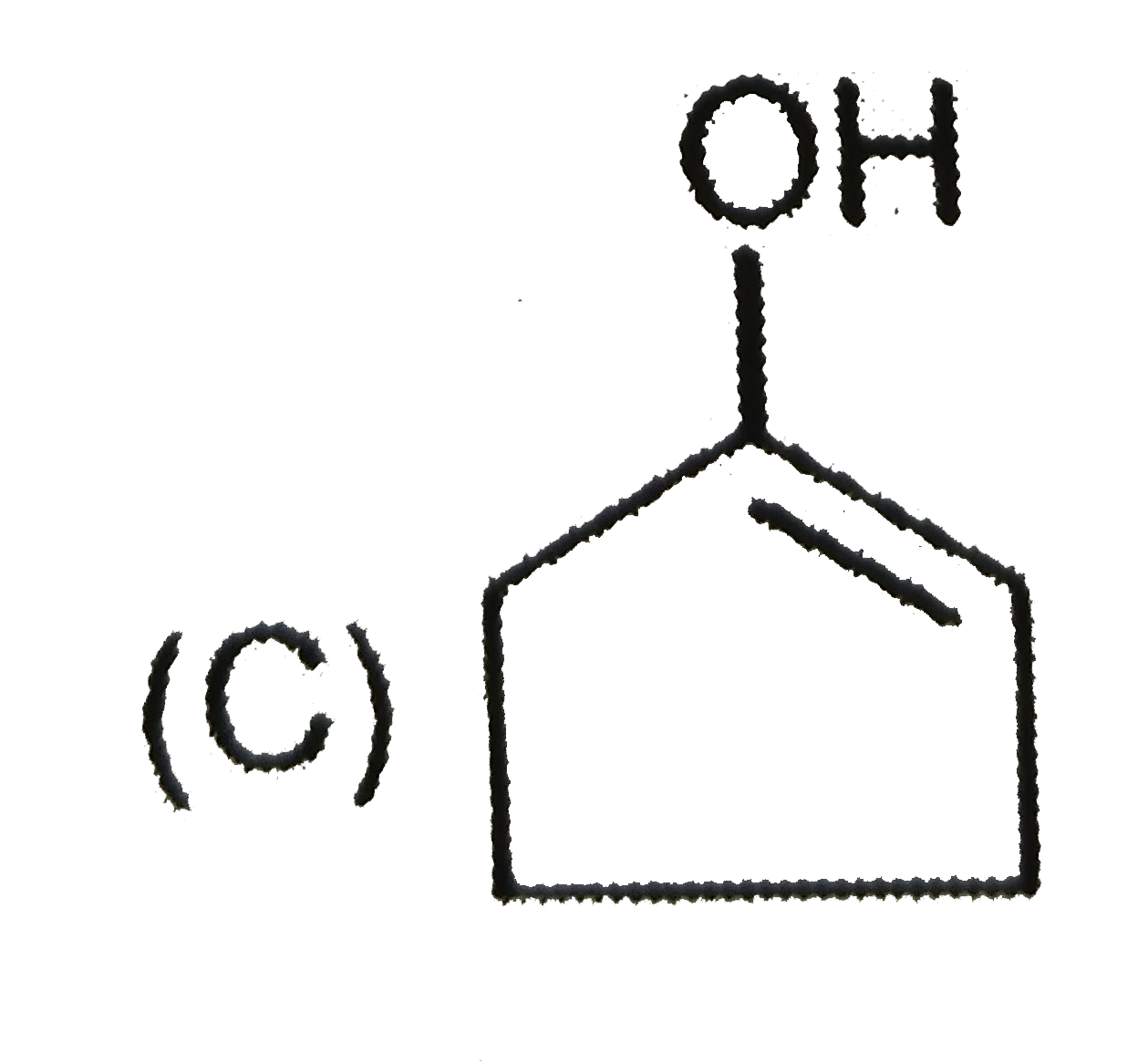
The major product when 3,3-dimethyl-butan-2-ol is heated with concentrated sulphuric acid is |
|
Answer» 2,3-dimethyl-2-butene 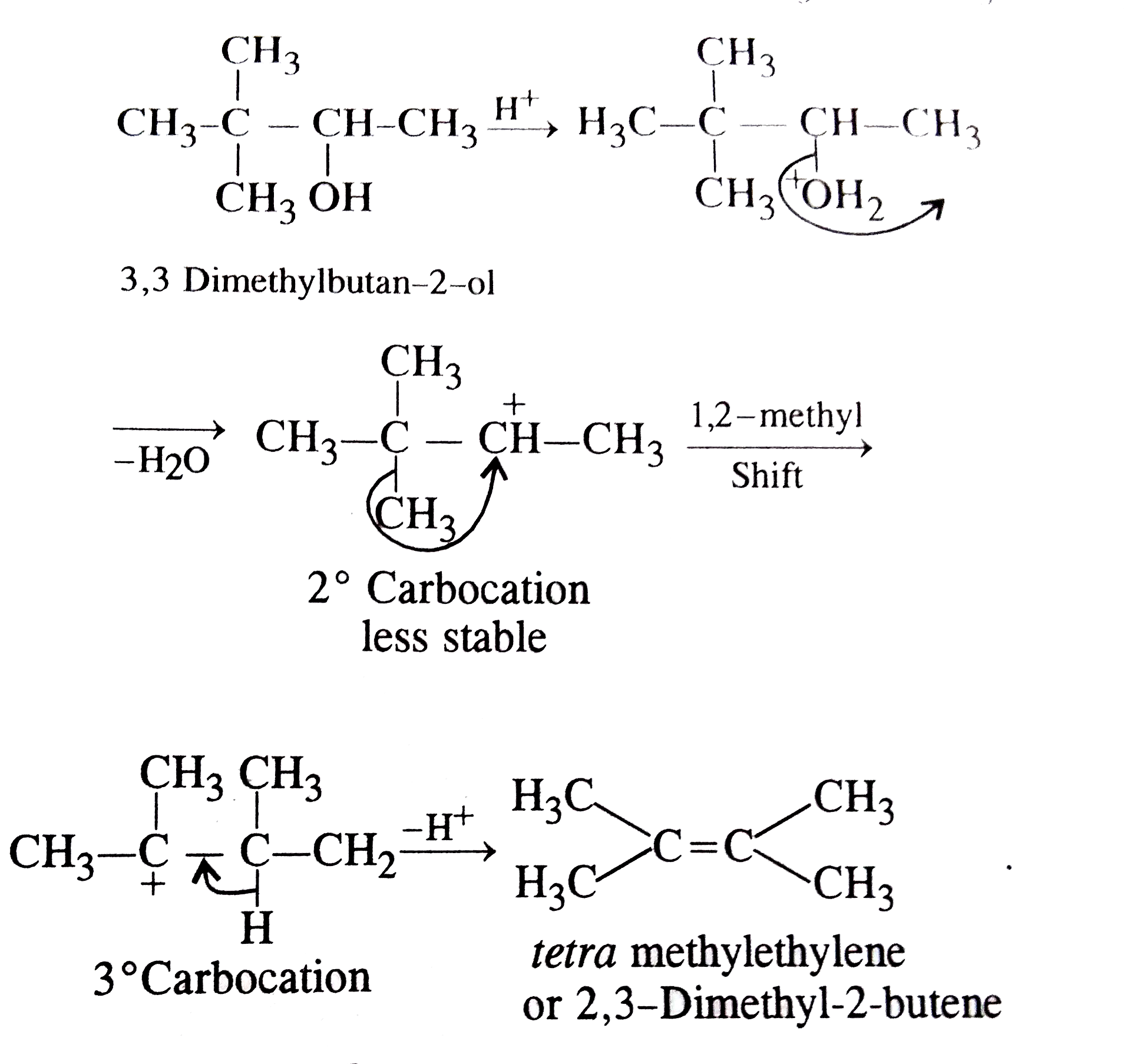
|
|
| 39. |
The major product of the reaction is |
|
Answer»
|
|
| 41. |
The major product of the given reaction is : |
|
Answer»
 
|
|
| 42. |
The major product of the following reactions is : |
|
Answer»
|
|
| 43. |
The major product of the following reaction is : CH_3CH_2undersetunderset(Br)(|)CH-undersetunderset(Br)(|)CH_2underset((ii)NaNH_2 "in liq." NH_3)overset((i)KOH alc.)to |
|
Answer» `CH_3CH=C=CH_2` |
|
| 44. |
The major product of the following reaction is : (CH_3)_2C=CH-CH_2-CH_3overset((i)B_2H_6,"ether"(ii)H_2O_2, "NaOH")to |
|
Answer»
|
|
| 45. |
The major product of the following reaction is : C_6H_5CH_2-undersetunderset(Br)(|)oversetoverset(CH_3)(|)C-CH_2CH_3underset(C_2H_5OH)overset(C_2H_5ONa)to |
|
Answer» `C_6H_5CH_2-undersetunderset(CH_3)(|)C=CHCH_3` |
|
| 46. |
The major product of the following reaction is |
|
Answer»
|
|
| 47. |
The major product of the following reaction is |
|
Answer»
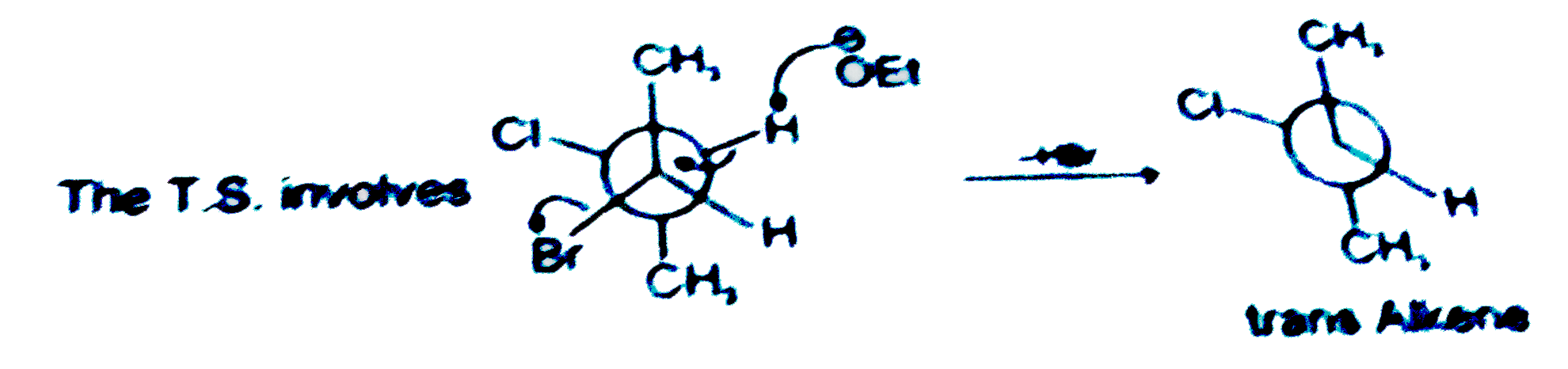
|
|
| 48. |
The major product of the following reaction is |
|
Answer»
|
|
| 50. |
The major product of the following reaction is |
|
Answer»
|
|