Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The maximum number of electrons that can be present in an orbit with s = +1/2 and l=2 |
|
Answer» 1 |
|
| 2. |
The maximum number of electrons that can be accommodated in N shell is……………….. |
|
Answer» SOLUTION :NUMBER of electrons in the SHELL`=2n^(2)` n=4,for N shell `THEREFORE` Maximum number of electrons in N shell=`2(4)^(2)=32` |
|
| 3. |
The maximum number of electrons that can be accommodated in F shell is……………….. |
|
Answer» Solution :14 f orbital-l=3 Maximum NUMBER of ELECTRONS in subshell=2(2l+1) `therefore`For .f. orbital,the maximum number of electrons=`2(2xx3+1)=14` |
|
| 4. |
The maximum number of electrons that a p-orbital can accommodate is |
|
Answer» 6 |
|
| 5. |
The maximum number of electrons on a subshell is equal to ______where l = ______ |
| Answer» SOLUTION :2l + l , AZIMUTHAL QUANTUM NUMBERS | |
| 6. |
The maximum number of electrons in a sub shell is given by the expression………… |
| Answer» Solution :2(2l+1)=4l+2 | |
| 7. |
The maximum number of electrons in a sub-shell is given by the expression. |
|
Answer» (l + 2) |
|
| 8. |
The maximum number of electrons accommodated in 5f orbitals are |
|
Answer» 5 |
|
| 9. |
The maximum number of electrons accomodated in 5f orbitals |
|
Answer» 5 |
|
| 10. |
The maximum number of electronin subshellis givenby expression . |
|
Answer» 4l -2 `=4(0) + 2=2` maximum no ofelectronin psubshell =4(1)+2 =6 maximum no ofelectronin dsubshell maximum no ofelectronin f subshell `=4(3) + 2 =14` |
|
| 11. |
The maximum number of carbon atoms arranged linearly in the molecule, CH_(3)-C-=C-CH=CH_(2) is |
|
Answer» 5 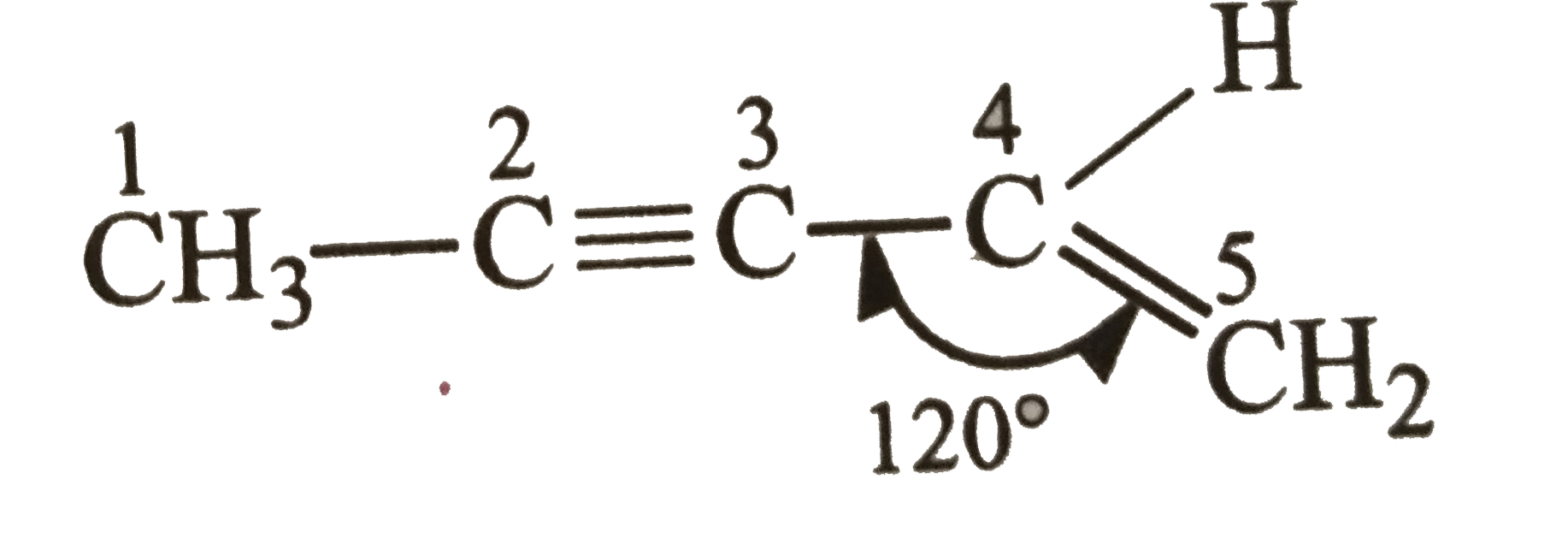
|
|
| 12. |
Themaximum number of 90^(@)angles between bond pair-bond pari of electrons is observed in |
|
Answer» `DSP^(3)` HYBRIDISATION 
|
|
| 13. |
The maximum inductive effect of -Cl on which carbon in overset(4)(C )H_(3)- overset(3)(C )H_(2) - overset(2)( C)H_(2)- overset(1)( C)H_(2)-Cl? |
|
Answer» 1 |
|
| 14. |
The maximumelectron occupy in f orbital are |
|
Answer» 10 |
|
| 15. |
The maximum ease of abstraction of a hydrogen atom by a chlorine atom is given by: |
|
Answer» `(CH_3)_3C-CH_3` |
|
| 16. |
The maximum difference in oxidation state of central atom of anionic part between [Y] and [Z] is: |
|
Answer» |
|
| 17. |
The maximum density of water is observed at ___________ |
|
Answer» `0^(@)`C |
|
| 18. |
The maximum covalency of aluminium is 6 where as that of boron is '4' because |
|
Answer» ALUMINIUM is more electropositive than BORON |
|
| 19. |
The maximum covalency of Be and Mg respectively |
|
Answer» 4, 6 |
|
| 20. |
The maximum amount of BaSO_(4) precipitated on mixing equal volumes of BaCl_(2) (0.5 M) with H_(2)SO_(4) (1 M) will correspond to |
|
Answer» `0.5N` Eq. `H_(2)SO_(4)=Vxx1xx2=2V` `therefore` Max eq. of `BaSO_(4)=V` `[BaSO_(4)^(-)]=(V)/(2V)=0.5N` |
|
| 21. |
The maximum amount of BaSO_(4) precipitated on mixing BaCl_(2)(0.5 M) with H_(2)SO_(4) (1M) will correspond to : |
|
Answer» 0.05 M `BaCl_(2_` is the LIMITING reactant. |
|
| 22. |
The mathematical from of Chart.s law is given under V_(t)=V_(0)((273.15+t^(@)C)/(273^(@)C)), From this equation explain V to T in Kelvin. |
|
Answer» Solution :`V_(t)=V_(0)((273.15+t)/(273.15)) ""` ….(Eq. -i) `therefore V_(t)=(V_(0)(273))/(273)+(V_(0)t^(@)C)/(273)` `therefore V_(t)=V_(0)+((t^(@)C)/(273))V_(0) ""`…..(Eq. -II) As per equation `Y=C+mx` `therefore` The graph of VOLUME `(V) to` Temperature in Kelvin. Where, SLOPE `= tan theta = (V_(0))/(273)`, INTERCEPT `= V_(0)` Upward graph is straight. 
|
|
| 23. |
Give the mathematical exprssion that relates gas volume and moles. |
|
Answer» BOYLE's LAW |
|
| 24. |
The mathematical expression of Raoult's law is ………………….. |
|
Answer» <P> SOLUTION :`P_(A)=P_(A) ^(0). X_(A)` |
|
| 26. |
The material possessing superconducting properties is |
|
Answer» `YbBa_(2)Cu_(2)O_(8)` |
|
| 27. |
The masses of carbon, hydrogen and oxygen in ann organic compound are in the ratio 6:1:8 respectively. Which of the following pairs of formulae correspond to above information? |
|
Answer» `CH_(3)O and CH_(3)CHO` mass of 3 O-atoms`=16xx3=48` `THEREFORE`Ration of MASSES of `C:H:O` is 36:6:48 or 6:1:8, In HCHO, mass of 1C-atom=12 mass of 2 H-atoms=2 mass of 1 O-atom=16 `therefore`Ratio of masses of `C:H:O` is 12:2:16 orr 6:1:8 |
|
| 28. |
The mass of zinc (Zn=65) required to produce 224 ml of H_(2) at STP on treatement with dilute H_(2)SO_(4) is |
|
Answer» 6.5 g |
|
| 29. |
The mass of water (in grams) in one mole of crystalline hypo is |
|
Answer» 18 |
|
| 30. |
The mass of the atom is determined by ________. |
|
Answer» neutrons |
|
| 31. |
The mass of piece of paper is 0.02g and the mass of a solid substance along with the same piece of paper is 20.036g. If the volume of the solid is 2.16 cm^(3), its densify to the proper number of significant digits will be |
|
Answer» `9.27 G CM^(-3)` |
|
| 32. |
The mass of oxygen that a 11.2L vessel can hold at 380 torr and 0^@C is |
| Answer» Answer :B | |
| 33. |
The mass of oxygen in N_(2)O_(3) and N_(2)O_(5)is respectively ............ |
|
Answer» 48,48 |
|
| 35. |
The mass of oxygen present in 0.5 mole of P_(4)O_(10) is (at. wt. P=31, O=16) |
|
Answer» 160 g |
|
| 36. |
The mass of oxygen required for the rusting of 4.2g of iron is (Fe = 56) |
|
Answer» 1.2 g |
|
| 37. |
The dot at the end of this sentence has a mass of about one microgram. Assuming that black stuff is carbon, calculate approximate atoms of carbon needed to make such a dot. |
|
Answer» |
|
| 38. |
The mass of one unit cell of NaCl is |
|
Answer» 234amu |
|
| 40. |
The mass of one molecule of AgCl in grams is |
|
Answer» 108g |
|
| 41. |
The mass of one mole of CaCl_(2) is ................ . |
|
Answer» |
|
| 42. |
The mass of nitrogen per gram in hydrazine is exactly one and half the mass of nitrogen in the compound ammonia. The fact illustrates the |
|
Answer» LAW of CONSERVATION of mass |
|
| 43. |
The mass of one atom of hydrogen is 1.008 amu. Calculate the mass of 26 atoms of hydrogen. |
|
Answer» Solution :The mass of ONE atom of hydrogen = 1.008 amu `therefore`The mass of 26 atoms of hydrogen would be `= 1.008 xx 26 = 26.208 = 26.21` amu In this calculation, the least PRECISE number is 1.008 which CONSISTS of four significant figures. 26 is an exact number and MAY be supposed to have INFINITE number of significant figures. Therefore, the final result is reported upto four significant figures only. |
|
| 44. |
The mass of one atom of C^(12)=........ |
|
Answer» `1.992648 xx 10^(23) gm` |
|
| 45. |
The mass of molecule A is twice the mass of molecule B. The rms speed of A is twice the rms speed of B. If two samples of A and B contain same number of molecules, what will be the ratio of pressures of two samples in separate containers of equal volume.________ |
|
Answer» SOLUTION :`(C_1)/(C_2) = SQRT((P_1)/(M_1) xx (M_L)/(P_L)) IMPLIES 2/1 = sqrt((P_1)/(P_2) xx m/(2m))` `implies (P_1)/(P_2) = 4 xx 2 = 8`. |
|
| 46. |
The mass of molecule A is twice that of molecule B. The root mean square velocity of molecule A is twice that of molecule B. If two containers of equal volume have same number of molecules the ratio of pressure (P_(A))/(P(B)) will be: |
|
Answer» `8:1` |
|
| 47. |
The mass ofCO_(2) obtained when 2g of pure limestone is calcined is |
|
Answer» `44G` `100gmCaCO_(3)rarr44gmCO_(2)` `2gmrarr? =0.88gm` |
|
| 48. |
The mass of CaCO_(3) produced when carbon dioxide is bubbled through 500 mL of 0.5 M Ca(OH)_(2) solution will be : |
|
Answer» 25 g Molarity `= (("Mass of "Ca(OH)_(2))/("MOLECULAR mass"))/("Volume of solution in litres")` `0.5=("Mass of "Ca(OH)_(2))/(74)xx(1000)/(500)` `=0.5xx39 = 18.5g` `{:(Ca(OH)_(2)+CO_(2),rarr,CaCO_(3)+H_(2)O),(74g,,100G),(18.5g,,(100)/(74)xx18.5=25g):}`. |
|
| 49. |
The mass of an oxygen atom is half that of a sulphur atom. Can we decide on this basis that the density of sulphur vapour relative to oxygen is 2? (a) Yes, (b) No |
|
Answer» Yes |
|