Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The only electron in the hydrogen atom resides under ordinary conditions on the first orbit. When energy is supplied, the electron moves to higher energy orbit depending on the amount of energy absorbed. When this electron returns to any of the lower orbits, it emits energy. Lyman series is formed when the electron returns to the lowest orbit while Balmer series is formed when the electron returns to second orbit. Similarly, Paschen, Brackett and Pfund series are formed when electron returns to the third, fourth orbits from higher energy orbits respectively (as shown in figure) Maximum number of lines produced when an electron jumps from nth level to ground level is equal to (n(n-1))/(2). For example, in the case of n = 4, number of lines produced is 6. (4 rarr 3, 4 rarr 2, 4 rarr 1, 3 rarr 2, 3 rarr 1, 2 rarr 1). When an electron returns from n_(2) to n_(1) state, the number of lines in the spectrum will be equal to ((n_(2) - n_(1))(n_(2)-n_(1) +1))/(2) If the electron comes back from energy level having energy E_(2) to energy level having energy E_(2) then the difference may be expressed in terms of energy of photon as E_(2) - E_(1) = Delta E, lambda = (h c)/(Delta E). Since h and c are constant, Delta E corresponds to definite energy, thus each transition from one energy level to another will prouce a higher of definite wavelength. THis is actually observed as a line in the spectrum of hydrogen atom. Wave number of the line is given by the formula bar(v) = RZ^(2)((1)/(n_(1)^(2)) - (1)/(n_(2)^(2))) Where R is a Rydberg constant (R = 1.1 xx 10^(7)) (i) First line of a series : it is called .line of logest wavelength. or .line of shortest energy.. (ii) Series limit of last of a series : It is the line of shortest wavelength or line of highest energy. In a single isolated atom in electron makes transition from 5^(th) excited state to 2^(nd) state the maximum number of different types of photons observed as |
|
Answer» 3 |
|
| 2. |
The only electron in the hydrogen atom resides under ordinary conditions on the first orbit. When energy is supplied, the electron moves to higher energy orbit depending on the amount of energy absorbed. When this electron returns to any of the lower orbits, it emits energy. Lyman series is formed when the electron returns to the lowest orbit while Balmer series is formed when the electron returns to second orbit. Similarly, Paschen, Brackett and Pfund series are formed when electron returns to the third, fourth orbits from higher energy orbits respectively (as shown in figure) Maximum number of lines produced when an electron jumps from nth level to ground level is equal to (n(n-1))/(2). For example, in the case of n = 4, number of lines produced is 6. (4 rarr 3, 4 rarr 2, 4 rarr 1, 3 rarr 2, 3 rarr 1, 2 rarr 1). When an electron returns from n_(2) to n_(1) state, the number of lines in the spectrum will be equal to ((n_(2) - n_(1))(n_(2)-n_(1) +1))/(2) If the electron comes back from energy level having energy E_(2) to energy level having energy E_(2) then the difference may be expressed in terms of energy of photon as E_(2) - E_(1) = Delta E, lambda = (h c)/(Delta E). Since h and c are constant, Delta E corresponds to definite energy, thus each transition from one energy level to another will prouce a higher of definite wavelength. THis is actually observed as a line in the spectrum of hydrogen atom. Wave number of the line is given by the formula bar(v) = RZ^(2)((1)/(n_(1)^(2)) - (1)/(n_(2)^(2))) Where R is a Rydberg constant (R = 1.1 xx 10^(7)) (i) First line of a series : it is called .line of logest wavelength. or .line of shortest energy.. (ii) Series limit of last of a series : It is the line of shortest wavelength or line of highest energy. The difference in the wavelength of the 2^(nd) line of Lyman series and last line of Bracket series in a hydrogen sample is |
|
Answer» `(119)/(8R)` |
|
| 3. |
The only electron in the hydrogen atom resides under ordinary conditions on the first orbit. When energy is supplied, the electron moves to higher energy orbit depending on the amount of energy absorbed. When this electron returns to any of the lower orbits, it emits energy. Lyman series is formed when the electron returns to the lowest orbit while Balmer series is formed when the electron returns to second orbit. Similarly, Paschen, Brackett and Pfund series are formed when electron returns to the third, fourth orbits from higher energy orbits respectively (as shown in figure) Maximum number of lines produced when an electron jumps from nth level to ground level is equal to (n(n-1))/(2). For example, in the case of n = 4, number of lines produced is 6. (4 rarr 3, 4 rarr 2, 4 rarr 1, 3 rarr 2, 3 rarr 1, 2 rarr 1). When an electron returns from n_(2) to n_(1) state, the number of lines in the spectrum will be equal to ((n_(2) - n_(1))(n_(2)-n_(1) +1))/(2) If the electron comes back from energy level having energy E_(2) to energy level having energy E_(2) then the difference may be expressed in terms of energy of photon as E_(2) - E_(1) = Delta E, lambda = (h c)/(Delta E). Since h and c are constant, Delta E corresponds to definite energy, thus each transition from one energy level to another will prouce a higher of definite wavelength. THis is actually observed as a line in the spectrum of hydrogen atom. Wave number of the line is given by the formula bar(v) = RZ^(2)((1)/(n_(1)^(2)) - (1)/(n_(2)^(2))) Where R is a Rydberg constant (R = 1.1 xx 10^(7)) (i) First line of a series : it is called .line of logest wavelength. or .line of shortest energy.. (ii) Series limit of last of a series : It is the line of shortest wavelength or line of highest energy. The wave number of electromagnetic radiation emitted during the transition of electron in between two levels of Li^(2+) ion whose principal quantum numbers sum if 4 and difference is 2 is |
|
Answer» R |
|
| 4. |
The only e^- in the H-atom resides under ordinary conditions on the first orbit when energy is supplied, the e^- moves to higher energy shells depending upon the amount of energy absorbed. When an e emits energy i.e., the e^- returns to the lowest energy state, from this Lyman, Balmer, Paschen, Bracket, Pfund series are there, so different spectral lines in the spectra of atoms correspond to different transitions of e^- s from higher to lower energy levels: In an hydrogen atom which of the following transition should be associated with highest absorption of energy |
|
Answer» n = 1 to n = 4 |
|
| 5. |
The only e^- in the H-atom resides under ordinary conditions on the first orbit when energy is supplied, the e^- moves to higher energy shells depending upon the amount of energy absorbed. When an e emits energy i.e., the e^- returns to the lowest energy state, from this Lyman, Balmer, Paschen, Bracket, Pfund series are there, so different spectral lines in the spectra of atoms correspond to different transitions of e^- s from higher to lower energy levels: The ratio of number of spectral lines obtained when an e^- s jumps from 7^(th) to ground to 6^(th) to 3^(rd) |
|
Answer» 7 `IMPLIES((7(7-1))/(2))/(((6-3)(6-3+1))/(2)) = (21)/6 = 3.5` |
|
| 6. |
The only e^- in the H-atom resides under ordinary conditions on the first orbit when energy is supplied, the e^- moves to higher energy shells depending upon the amount of energy absorbed. When an e emits energy i.e., the e^- returns to the lowest energy state, from this Lyman, Balmer, Paschen, Bracket, Pfund series are there, so different spectral lines in the spectra of atoms correspond to different transitions of e^- s from higher to lower energy levels: If the shortest wavelength of H atom in Lyinan series x, then longest wavelength in Balmer series of He^+ is |
|
Answer» `(36x)/(5)` For Balmer SERIES of `He^(+2)` LONGEST `implies1/lambda = R_H [1/(2^2) - 1/(3^2)] xx 4 implieslambda implies(9x)/(5)` |
|
| 7. |
The only correct combination in which the reaction does not proceed through redox mechanism. |
|
Answer» (I),(ii),(R) |
|
| 8. |
The only correct combination in which product gives position test with sodium bicarbonate is - |
| Answer» Answer :D | |
| 9. |
The only correct combination for benzyl cation is - |
|
Answer» <P>(P) (II), (II) |
|
| 10. |
The only correct combination for pyrrole is |
|
Answer» (P) (ii), (II) |
|
| 11. |
The only correct combination for pyridine is - |
|
Answer» (Q) (i), (II) |
|
| 12. |
The only cations present in a slightly acidic are Fe^(3+), Zn^(2+), and Cu^(2+). The reagent that when added in excess to this solution would identify and separate Fe^(3+) ions in one step is |
|
Answer» 2 M HCI |
|
| 13. |
The only alcohol that can be prepared by the indirect hydration of alkene is |
|
Answer» ethyl ALCOHOL |
|
| 14. |
The one with maximum enol content is |
|
Answer»
|
|
| 15. |
The one which is most basic out of the following |
|
Answer» `CO_(2)` |
|
| 16. |
The one which has least iondine value |
|
Answer» SUNFLOWER OIL |
|
| 18. |
The one electron species having ionization energy of 54.4 eV is |
|
Answer» H `= 13.6 Z^(2)` for one-electron SPECIES `:. 13.6 Z^(2) = 54.4 or Z^(2) = 4 or Z = 2, " i.e., " He^(+)` |
|
| 19. |
The O.N of P in Ba(H_(2)PO_(2))_(2) is: |
|
Answer» Solution :`Ba(H_(2)OVERSET(x)PO_(2))_(2)` +2 + 4 (+1) + 2 x + 4 (-2)=0 2x-2=0 or x=+1 |
|
| 20. |
The O.N of N in (NH_(4)_(2) SO_(4)) is : |
|
Answer» Solution :In `NH_(4)^(+)` ION : `OVERSET(x+1)[NH_(4)]^(+)` x+4(1)=+1,x=-3 O.N of N=-3 |
|
| 22. |
The [OH]^(-)] of 0.005 M H_(2)SO_(4) is |
|
Answer» `2xx 10^(-12)M` ` 10 ^(-2)=[OH^(-) ] = 10 ^(-14)rArr [OH^(-)] = 10 ^(-14)rArr [OH^(-) ]10 ^(-12)M ` |
|
| 23. |
The OH group of an alcohol or the -COOH group of a carboxylic acid can be replaced by -Cl using |
|
Answer» chlorine |
|
| 24. |
The -OH group in alcohols may be replaced by Cl is we use |
|
Answer» `Cl_(2)` |
|
| 25. |
The % of copper and oxygen in samples of CuO obtained by different methods were found to be the same. This proves the law of: |
|
Answer» Constant Proportion |
|
| 26. |
The % of copper and oxygen in samples of CuO obtained by different methods were found to be the same. This proves the law of : |
|
Answer» CONSTANT Proportion |
|
| 27. |
The % of Hydrogenin water and Hydrogen peroxide is 11.2% and 5.94% respectively. This illustrates the law of |
|
Answer» Constant Proportions so answer is 1 (one). |
|
| 28. |
The % of p character in hybrid orbital of the central atom of water molecule |
|
Answer» 0.25 |
|
| 29. |
The ....... of a gas is a measure of the average kinetic energy of its molecules. |
| Answer» SOLUTION :ABSOLUTE TEMPERATURE | |
| 30. |
The observed wavelengths in the line spectrum of hydrogen atom were first expressed in terms of a series by Johann Jakob Balmer, a Swiss teacher. Balmer's empirical formula is (1)/(lambda) = R_(H)[(1)/(2^(2)) - (1)/(n^(2))] = n = 3, 4,5… R_(H) = 109678 cm^(-1) is the Rydberg constant. Niels Bohr derived this expression theoretically in 1913. The formula is a generalised to any on electron atom/ion. The wavelength of first line of Balmer spectrum of hydrogen will be : |
|
Answer» 4340 Å |
|
| 31. |
The observed wavelegnth in the line spectrum of hydrogen atom were first expressed in terms of a series by Johann Jakob Balmer, a Swiss teacher. Balmer's emipirical formula is (1)/(lamda)=R_(H)[(1)/(2^(2))-(1)/(n^(2))]n=3,4,5 . . . R_(H)=109678cm^(-1) is the Rydberg constant. Niels Bohr derived this expression theoretically in 1913. The formula is generalised to any one electron atom/ion. Which of the following is not correctly matched? |
|
Answer» `H_(ALPHA)-`6569Ã…(Red) |
|
| 32. |
The observed wavelegnth in the line spectrum of hydrogen atom were first expressed in terms of a series by Johann Jakob Balmer, a Swiss teacher. Balmer's emipirical formula is (1)/(lamda)=R_(H)[(1)/(2^(2))-(1)/(n^(2))]n=3,4,5 . . . R_(H)=109678cm^(-1) is the Rydberg constant. Niels Bohr derived this expression theoretically in 1913. The formula is generalised to any one electron atom/ion. Q.In which region of electromagnetic spectrum does the Balmer series lie? |
|
Answer» UV |
|
| 33. |
The observed wavelengths in the line spectrum of hydrogen atom were first expressed in terms of a seriesby Johamn Balmer a swiss teacher. Balmer.s empirical formula is : (1)/(lambda) = R_(H)[(1)/(2^(2))-(1)/(n^(2))]n=3,4,5…. R_(H) = 109678 cm^(-1) is the Rydberg constant How many lines in the spectrum will be observed when electrons return from 7^(th) shell to 2^(nd) shell ? |
|
Answer» 13 |
|
| 34. |
(A) Balmer series lies in the visible region of electromagnetic spectrum (R): (1)/(lambda) = R((1)/(2^(2)) - (1)/(n^(2))) where n = 3,4,5 |
|
Answer» 2651 Å |
|
| 35. |
The observed value of dipole moment of H_(2)O molecule is found to be 1 .84 D. Calculate the H-O-H bond angle in H_(2)O molecule , given that the bond moment of O-H bond is 1.5 D. |
|
Answer» Solution :As dipole monent of `H_(2)O`is the resultant of the two vactors(O-H BONDS), therefore ,if `ALPHA ` is the angle between the two vactors , then `mu=sqrt(mu_(1)^(2) + mu_(2)^(2) + 2mu_(1) mu_(2) cos alpha )` `mu_(H_(2)O=sqrt(mu_(OH)^(2) + mu_(OH)^(2) + 2mu_(OH)^(2) cos alpha ))` `1.84 = sqrt((1.5)^(2) + (1.5)^(2)+2 (1.5)^(2) cos alpha)` or`3.3856 = 2.25 + 2.25 + 4.50 cos alpha or cos alpha= -0.2476` or `alpha = 104^(@) 20'[cos (180 - THETA )= - cos theta, i.e.,cos (180 - alpha ) = 0.2476 or 180 - alpha = 75^(@)40']` 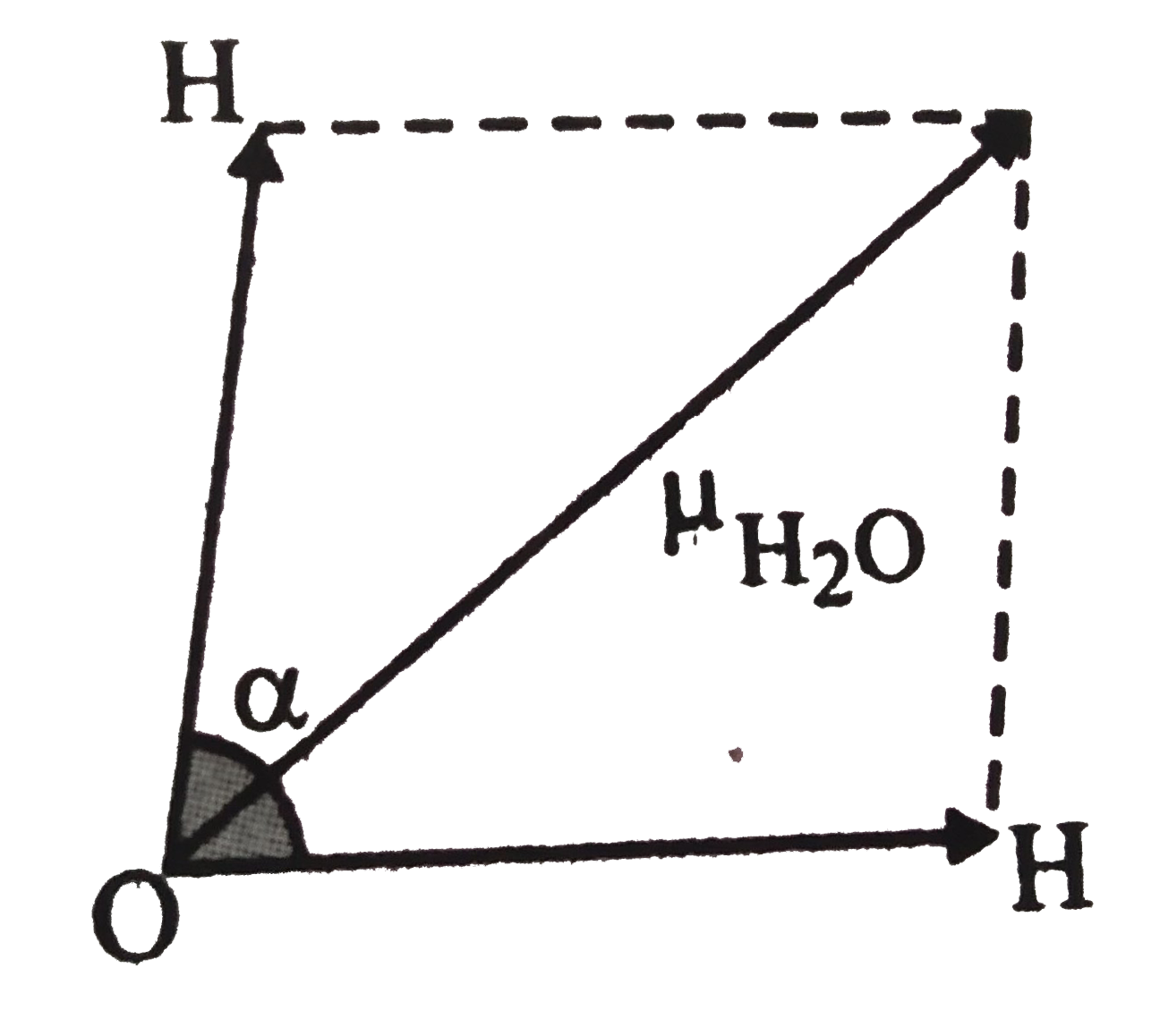
|
|
| 36. |
The observed rotation of 10ml of a solution containing 2g of a compound when placed in 25cm long polarimeter tube is +13.4^@. What is the specific rotation of the compound? |
| Answer» SOLUTION :`alpha_(OBS) = + 13.4^(@)` ,C `=0.2gcm^(-3)` ,Specific rotation ,`alpha = (+13.4)/(0.2 xx 2.5) = + 26.8^(@)` | |
| 37. |
The observed depression in freezing point of water for a particular solution in0.093^(@) kcalculate the concentration of the solution inmolality Given that molal depression constant for water is1.86 K KG mol ^(-1 ) |
|
Answer» Solution :`Delta T _(f) = 0.093^(@)C = 0.093 K, m = ` ? `K _(f) = 1. 86 K kg MOL ^(-1)` ` Delta T _(f) = K _(f) .m` `THEREFORE m = (Delta T _(f))/( K _(f)) = (0.093K)/(1.86 K kg mol ^(-1)) = 0.05 mol kg ^(-1) = 0.05 m` |
|
| 38. |
The observed bond length of N_(2)^(+) is larger than N_(2) while the bond length in NO^(+) is less than in NO. Why? |
|
Answer» Solution :(a) By molecular orbital theory , the bond order of both `N_(2)^(+)` is `2.5` where as `N_(2) " is " 3`. `N_(2)` has `5e^(-)` in the anti - bonding molecular where as `N_(2)^(+)` has `4e^(-)` in the anti - bonding molecular orbital so `N_(2)^(+)` will make a STRONGER and shorter bond length . More the bond order and bond STRENGTH and lesser will be the bond length. So we can easily CONCLUDE `N_(2)` has more bond length that `N_(2)`. Bond order in `= N_(2) = (N_(b) - N_(a))/2 = (10 - 4)/ 2= 6/2 = 3` Bond order in ` = N_(2) = (N_(b) - N_(a))/2 = (9 - 4)/ 2 = 5/2 = 2.5` So `N_(2)` is more stable that `N_(2)^(+)` but bond length `N_(2)^(+)` is greater than `N_(2)`. (b) ` NO^(+) & NO ` : Bond order of NO = `2.5` Bond order of `NO^(+) = 3` Due to lesser bond order in NO the bond length is greater than `NO^(+) `. So `NO^(+)` bond length is shorter than NO bond length. |
|
| 39. |
The observed bond length of N_(2)^(+) is larger than N_(2) while the bond length in NO^(+) is less than in NO. Why ? |
|
Answer» Solution :(a) (i) By molecular orbital theory, the bond order of both `N_(2)^(+)` is 2.5 whereas `N_(2)` is 3. (II) `N_(2) has 5e^(-)` in the antibonding molecular orbital whereas `N_(2)^(+) has 4E^(-)` in the antibonding molecular orbital. So `N_(2)^(+)` will make a stronger and shorter bond lenght. 9iii) More the bond order and bond strength, and lesser will be the bond length So we can easily conclude `N-(2)` has more bond length than `N_(2)` Bond order in `N_(2) = (N_(b)-N_(a))/(2)=(10 -4)/(2)=(6)/(2)=3` Bond order in `N_(2)^(+)=(N_(b)-N_(a))/(2)=(9-4)/(2)=(5)/(2)=2.5` So, `N_(2)` ismore stable than `N_(2)^(+)`. But bond length `N_(2)^(+)` is greater than `N_(2)`. (b) `NO^(+)` & NO Bond order of NO = 2.5 Bond order of `NO^(+) =3` Due to lesser bond order in NO, the bond length is greater than `NO^(+)` So, `NO^(+)` bond lenght is shorter than NO bond lenght. |
|
| 40. |
The O_(2) gas is collected over water at 400 K temperature in 2 L vessel. If the pressure of dry O_(2) gas is 32.20 bar bar then Find the vapour pressure of water under the same conditions ?(R=8.314xx10^(-2)" L bar mol"^(-1)K^(-1)) |
|
Answer» |
|
| 42. |
The O-O-H bond angle in H_(2)O_(2) is: |
|
Answer» `106^(@)` |
|
| 43. |
The O-O-H bond angle in molecule H_(2)O_(2) is |
|
Answer» `106^(@)` |
|
| 44. |
The O-H bond length in H_2 O is xA^@ . The o-H bond length in H_2 O_2 is |
|
Answer» `LT xÅ` |
|
| 45. |
The O-H bond energy in water when compared to O-D bond energy in heavy water is |
| Answer» ANSWER :B | |
| 46. |
The nun1ber of water molecules is maximum in : |
|
Answer» 18 GRAM of water `because` 1 mole water = `6.02 xx 10^(23)` molecules `thereofore` 18 mole water = `18 xx 6.02 xx 10^(23)` moleculesso, 18 mole water has maximum number of molecules. |
|
| 47. |
The value of the universal gas constant R depends upon the |
|
Answer» the NATURE of the gas |
|
| 48. |
The numerical value of solubility product of magnesium hydroxide at 25^@ C is 3.2 xx 10^(-11). What is its solubility ? |
|
Answer» Solution :The solubility equilibrium of magnesium hydroxide, `Mg(OH)_2 hArrMg^(2+) + 2OH^(-)` If the solubility is x for `AB_2` type SUBSTANCE, solubility product is `4x^3`. `4x^3 = 3.2 xx 10^(-11) , x = 8 xx10^(-12)` and hence, `x = 2 xx 10^(-4)` Solubility of `Mg(OH)_2= 2 x x 10^(-4)MOL L^(-1)` |
|
| 49. |
The numbers of significant figures in 0.0500 are |
|
Answer» One |
|



