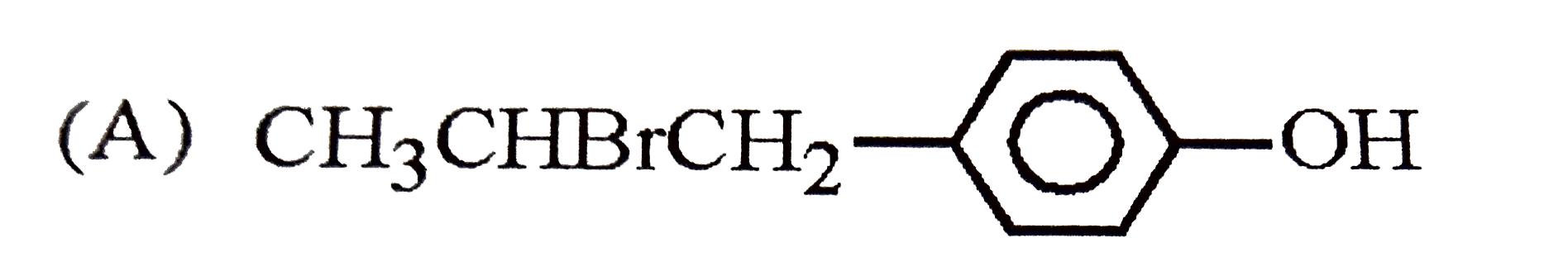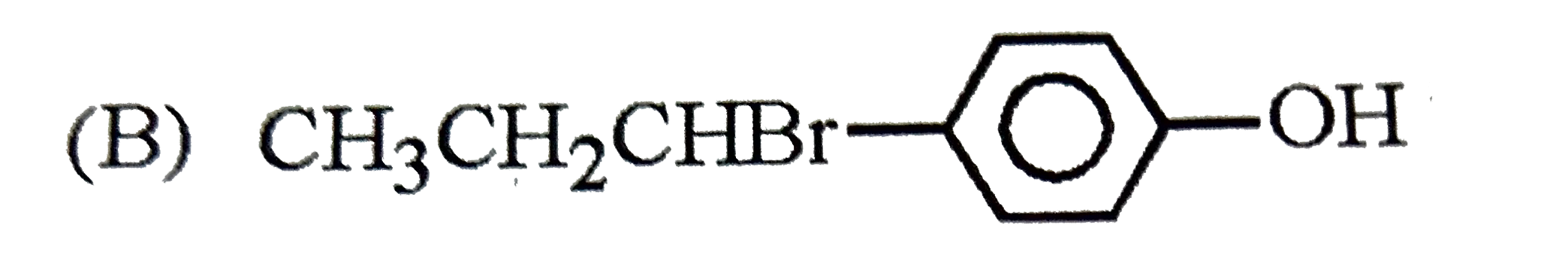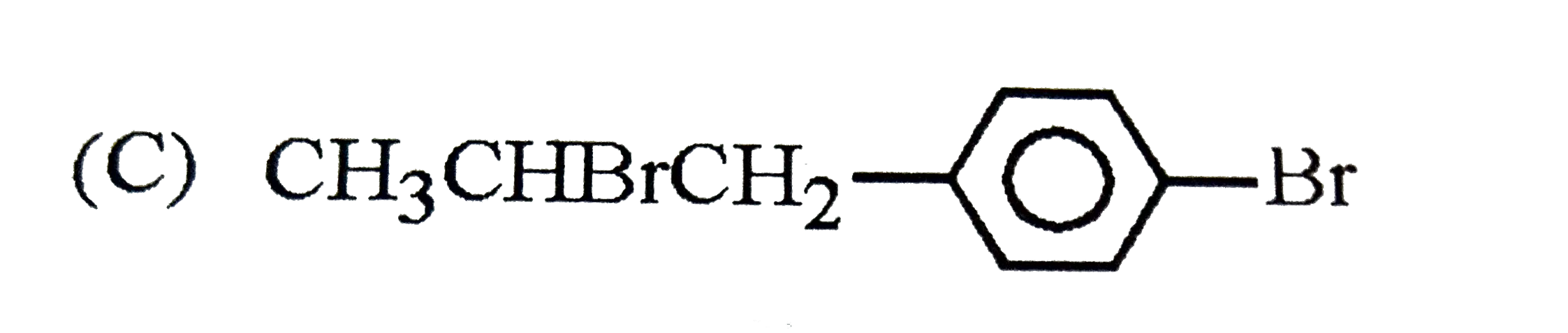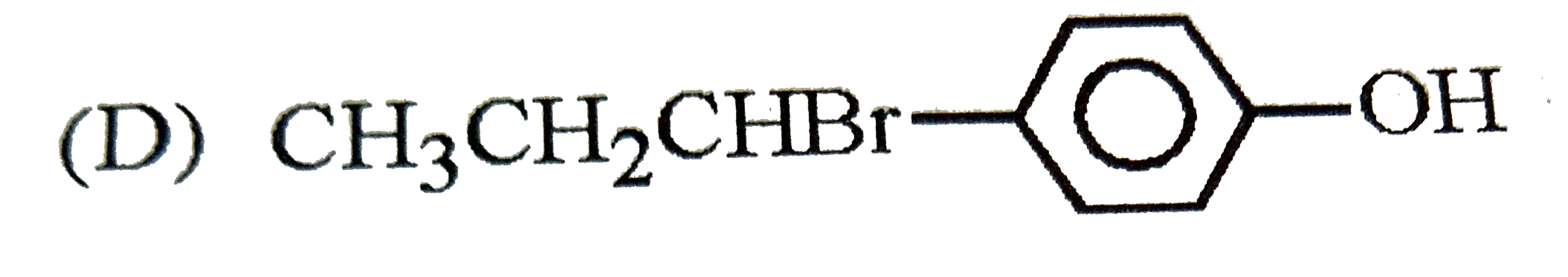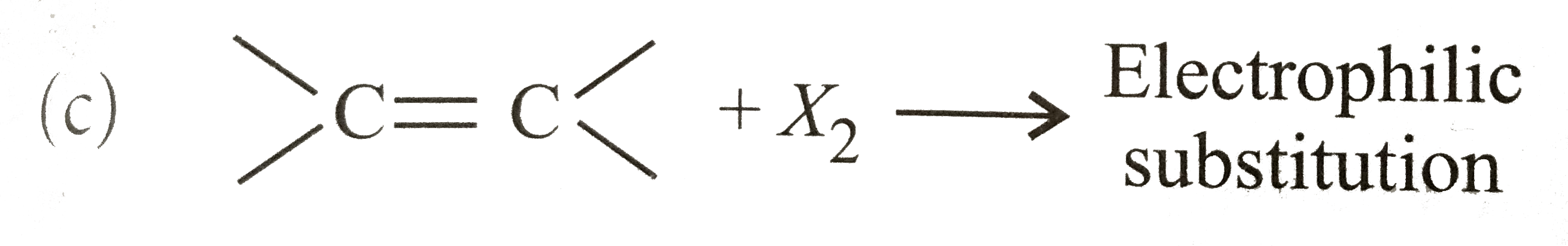Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The reaction of neopentyl alcohol with concentrated HCl gives |
|
Answer» neopentyl CHLORIDE |
|
| 2. |
The reaction of NaCl and K_(2)Cr_(2)O_(7) mixture with conc. H_(2)SO_(4) result in the formation of: |
|
Answer» `CrO_(2)Cl_(2)` |
|
| 3. |
The reaction of n-hexane in presence of catalyst at 773 K and 10-20 bar pressure to form benzene is known as ………reaction. |
|
Answer» Hydration |
|
| 4. |
The reaction of methyl trichloroacetate (C Cl_(3)CO_(2)Me) with sodium methoxide (NaOMe) generates |
|
Answer» carbocation 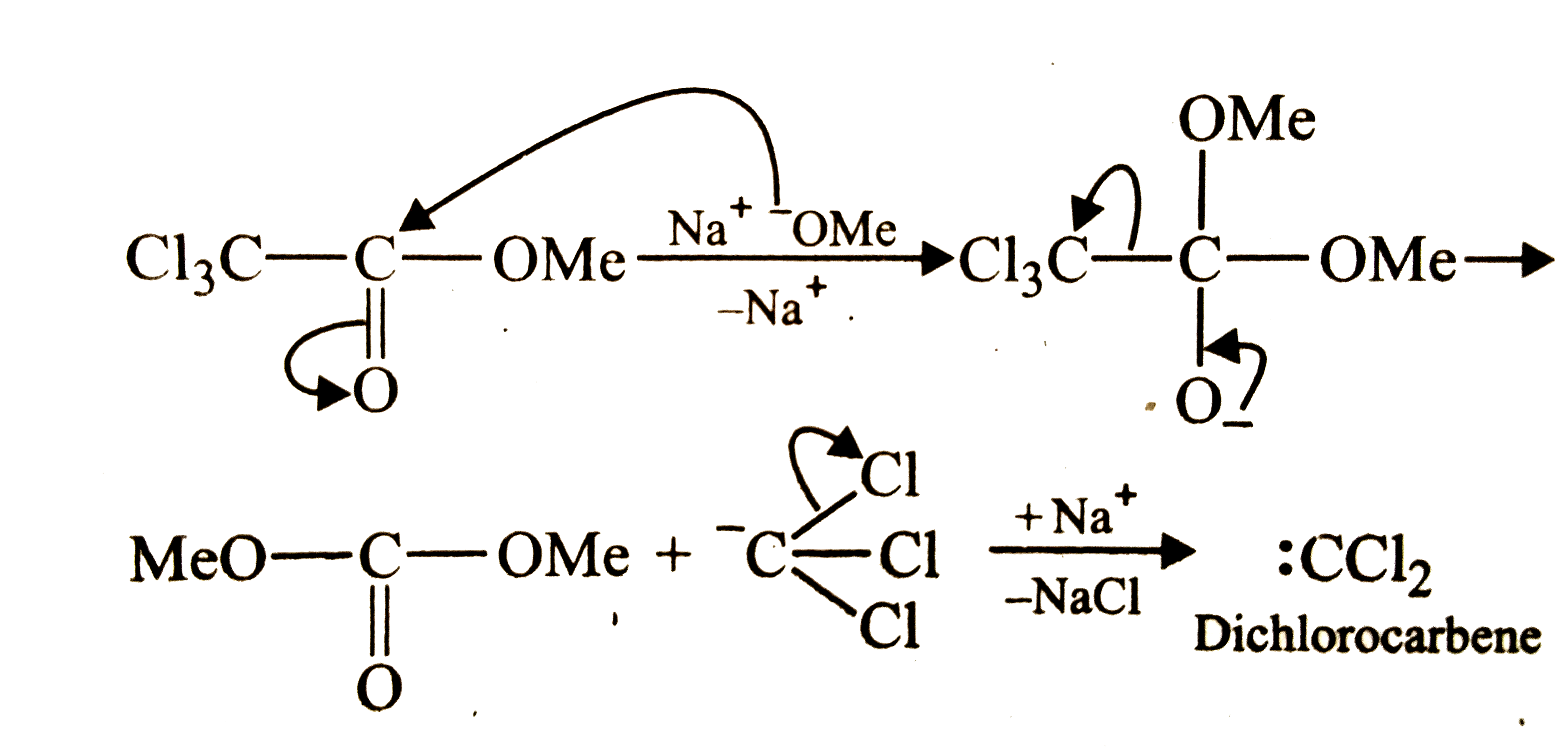
|
|
| 5. |
The reaction of methyl magnesium bromide with ethyl alcohol gives |
|
Answer» METHYL alcohol |
|
| 6. |
The reaction of H_2O_2 with X does not liberate gaseous product. Which of the following is X ? |
|
Answer» `PbO_2` |
|
| 7. |
The reaction of HBr with ethene is an example of reaction |
|
Answer» Polymerisation |
|
| 8. |
The reaction of Ethanol with H_(2)SO_(4) does not give |
|
Answer» Ethylene |
|
| 9. |
The reaction of cynamide, NH_(2)CN(s), with dioxygen was carriedout in a bomb calorimeter, and DeltaU was found to be - 742.7 k J mol^(-1) at 298 K. Calculate the enthalpy change of the reaction at 298 K NH_(2)CN(s) + (3)/(2)O_(2)(g)rarrN_(2)(g)+CO_(2)(g) +H_(2)O(l) |
|
Answer» <P> Solution :`Deltan_(g)=(n_(p) -n_(p))_(g)= 2-(3)/(2) = +(1)/(2)` mol`DeltaH =DeltaU +Deltan_(g) RT= - 742.7 KJ mol^(-1) +(+(1)/(2)mol)(8.314xx10^(-3)kJ K^(-1) mol^(-1))(298K)` `= - 742.7 +1.2kJ mol^(-1) = - 741.5 kJ mol^(-1)` |
|
| 10. |
The reaction of cyanamlde, NH_(2) CN_((s)), with dioxygen was carried out in a bomb calorimeter, and DeltaU was found to be -742.7 "kJ mol"^(-1) at 298 K. Calculate enthalpy change for the reaction at 298 K. NH_(2) CN_((g)) + (3)/(2) O_(2(g)) to N_(2(g)) + CO_(2(g)) + H_(2) O_((l)) |
|
Answer» SOLUTION :`Deltan_((G)) = n_(p) - n_(R)` `=2-(3)/(2) = +(1)/(2) "mol"` `Deltan_((g)) = Delta U + Deltan_((g)) "RT"` `= -742.7 + (+(1)/(2) ) (8.314 xx 10^(-3) ) (298)` `= -742.7 + 1.2` `= -741.5 "kJ mol"` |
|
| 11. |
The reaction of cyanamide, NH_2CN(s) with O_2 was carried out in a bomb calorimeter and Delta U was found to be -742.7 kJmol^-1 at 298 K. Calculate enthalpy change for the reaction at 298K.NH_2CN(s)+3/2O_2(g)rarrN_2(g)+CO_2(g)+H_2O(l) |
|
Answer» SOLUTION :`Delatan(G)=(1+1)-3/2=+1/2` `DeltaU=-742.7 KJ mol^-1 R=8.314xx10^-3 kJ mol^-1k^-1DeltaH=DeltaU+DeltanRT=-742.7+1/2xx8.314xx10^-3xx298=-741.5 kJ` |
|
| 12. |
The reaction of cyanamide (s), with dioxygen was carried out in a bomb calorimeter and DeltaU was found to be -742.7 kJ mol^(-1) at 298 K. Calculate the enthalpy change for the reaction at 298 K. NH_(2)CN_((g))+(3)/(2)O_(2)(g)toN_(2(g))+CO_(2(g))+H_(2)O_(l) |
| Answer» SOLUTION :`-743.94 KJ "MOLE" ^(-1)` | |
| 13. |
The reaction of Cl_(2) with X gives bleaching powder . X is |
|
Answer» `CAO` |
|
| 14. |
The reaction of (CH_(3))_(2)C(OH)CH_(2)CH_(3) with copper at 300^(@)C gives |
|
Answer» `CH_(3)CO(CH_(3))_(2)` and `H_(2)` `CH_(3)-underset(CH_(3))underset(|)overset(OH)overset(|)(C)-CH_(2)CH_(3) underset(300^(@)C)overset(Cu)rarr CH_(3)-underset(CH_(3))underset(|)(C)=CHCH_(3)+H_(2)O` |
|
| 15. |
The reaction of C_6H_5CH=CHCH_3 with HBr produces |
|
Answer» `C_6H_5CH_2CH_2CH_2Br`<BR> `C_6H_5-overset1CH=overset2CHCH_3overset(H^+)to underset"(Stabilized by resonance over the benzene ring)"(C_6H_5-overset+CH-CH_2CH_3) overset(Br^-)to underset"1-Bromo-1-phenylpropane"(C_6H_5-undersetunderset(Br)|CH-CH_2CH_3)` |
|
| 16. |
The reaction of C_(6) H_(5) CH= CHCH_(3) with HBr porduces : |
|
Answer» `C_(6) H_(5) CH UNDERSET(Br)(CH_(2)) CH_(3)` 
|
|
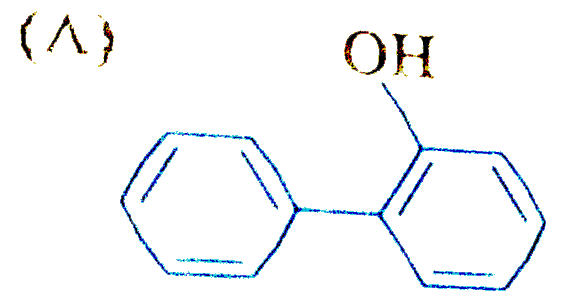
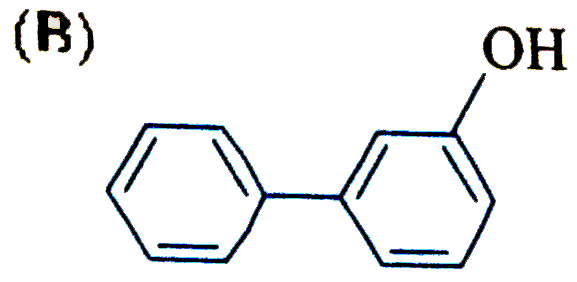
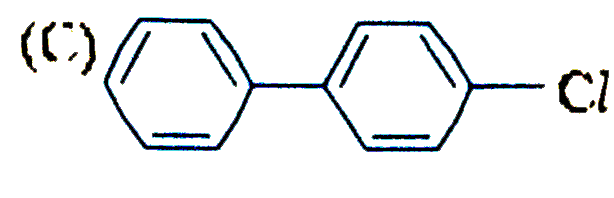
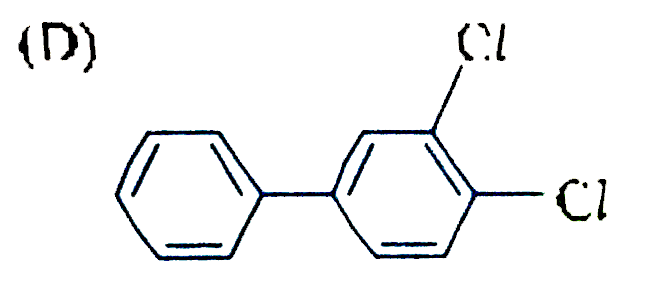
| 17. |
The reaction of biphenyl with HOCl in the presence of a strong acid gives |
|
Answer»
|
|
| 18. |
The reaction of aromatic acyl chloride and phenol in the presence of a base NaOH or pyridine is called |
|
Answer» Kolbe's reaction |
|
| 19. |
The reaction of aqueous KMnO_(4) with H_(2)O_(2) in acidic conditions gives |
|
Answer» `MN^(4+) and O_(2)` |
|
| 20. |
The reaction of aqueous KMnO_4 with H_2O_2 in acidic conditions gives : |
|
Answer» `MN^(4+)` and `O_2` |
|
| 21. |
The reaction of acetyleneand propyne with HgSO_4 in presence of H_2SO_4 produces respectively |
|
Answer» acetone and acetladehyde |
|
| 22. |
The reaction of acetaldehyde with HCN followed by hydroysis gives a product which exhibits |
|
Answer» Metamerism |
|
| 23. |
The reaction of |
|
Answer»
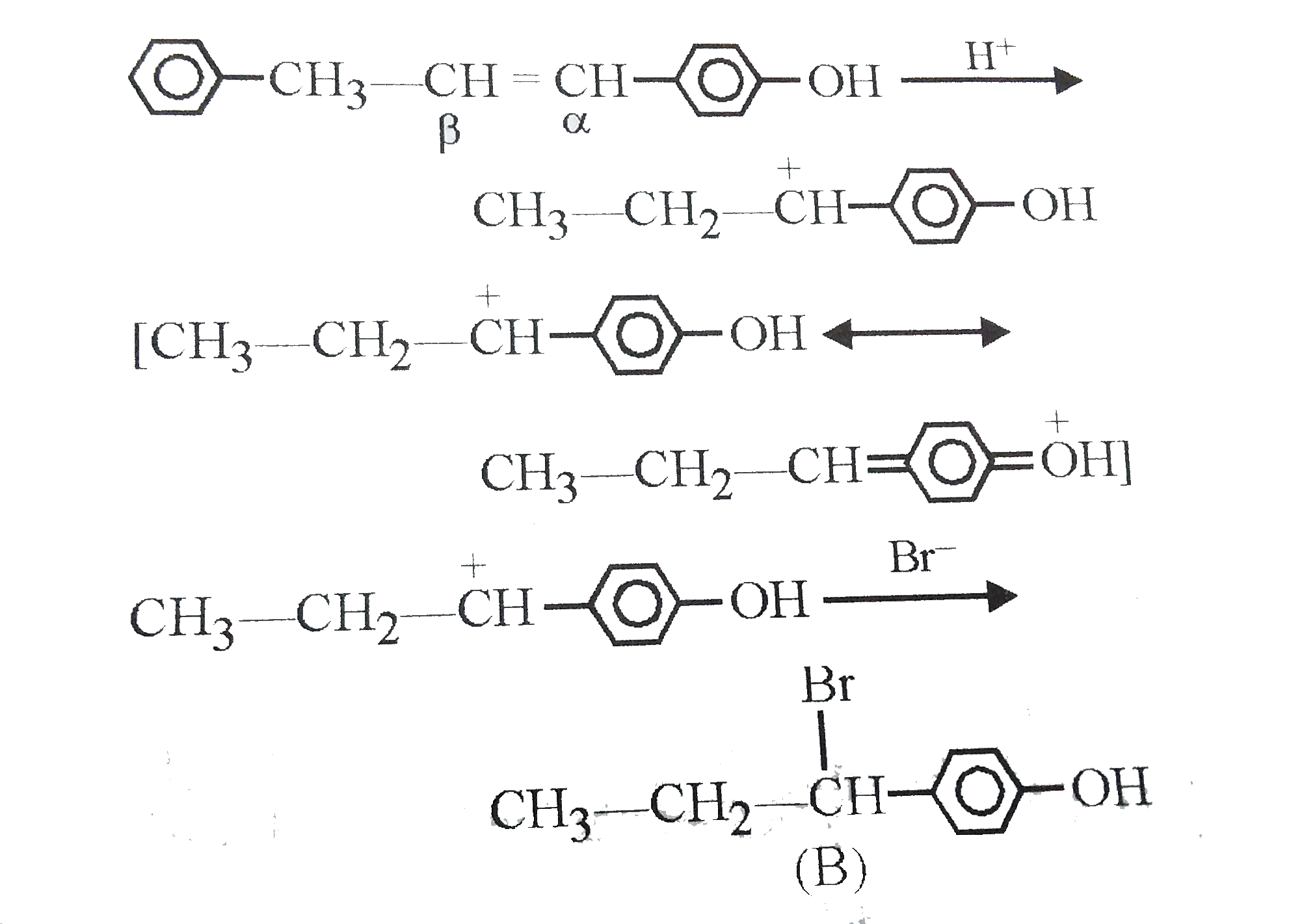 The intermediate carbocation I FORMED is resonance STABILISED DUE to the electron donating effect of `-OH` group at p-position. The addition of `Br^(-)` of this carbocation gives the addition product (B). |
|
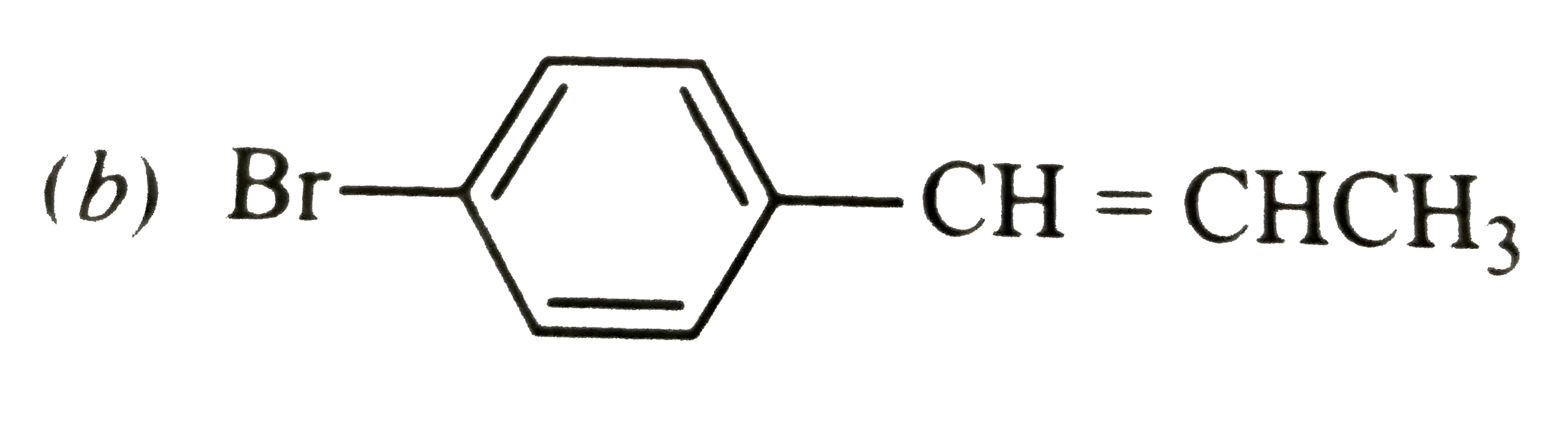
| 24. |
The reaction of 1-phenylpropane with limited amount of chlorine in the presence of light gives mainly . |
|
Answer» 4-chloropropylbenzene |
|
| 25. |
The reaction occurs between H_2 and I at 731 K temperature equilibrium concentration of H_2, I_2 and HI are as under in Table Calculate "[HI]"/([H_2][I_2]) and [HI]^2/([H_2][I_2]) and discuss of obtainedresults and derive chemical equilibrium rule. |
Answer» Solution : The result of A is different but B is same, so expression of B explain the law of chemical EQUILIBRIUM. At definite temperature, the multiple of active mass of product and multiple of active mass of reactant is constant. OR The ratio of multiple of concentration of product with PROPER POWER of MOLES of the multiple of concentration of reactant with proper power of moles is constant. |
|
| 26. |
The reactionNa_(2)S+I_(2)+Na_(2)SO_(3)rarrNa_(2)S_(2)O_(3)+2NaIis called |
|
Answer» SPRINGS REACTION |
|
| 27. |
The reaction M2A(g) + B (g) hArr 3 C (g) + D (g)is begun with the concentrationsof A and B both at intial value of 1.00 M. Whenequilibrium is reached , the concentrations of D is measured and is reached , the concentration of D is measured and found to be 0*25 M . The value for the equilibriumconstant for this reaction is given by the expresion |
|
Answer» `[(0*75)^(3)(0*25)] div [(1.00)^(2)(1.00)]` `{:(,2A(g),+,B(g),hArr,3 C(g),+,D(g)),("Intial",1.0 M,,1.0 M,,0,,0),("conc.",,,,,,,),("Eqm.",1-(2xx0.25),,1-0.25,,3xx0.25,,0.25),("conc".,0.5 M,,0.75 M,,0.75 M,,):}` ` K=([C]^(3) [D])/([A]^(2) [B])= ((0.75)^(3) (0.25))/((0.5)^(2) (0.75))` |
|
| 28. |
The reaction is oxidation - reduction type and takes place in presence of 50% aqueous orethanolic alkali One molecule of the aldehyde (missing of a- hydrogen atom )oxidizes, to corresponding carboxlic acid and the other is being reduced to the corresponding alcohol. Mechanism : the mechanism involves two distinct steps Nucleophilic attack and hydride ion transfer. A. The A is |
|
Answer» LACTIC acid 
|
|
| 29. |
The reaction is oxidation - reduction type and takes place in presence of 50% aqueous orethanolic alkali One molecule of the aldehyde (missing of a- hydrogen atom )oxidizes, to corresponding carboxlic acid and the other is being reduced to the corresponding alcohol.Mechanism : the mechanism involves two distinct steps Nucleophilic attack and hydride ion transfer. |
|
Answer»

|
|
| 30. |
The reaction is oxidation - reduction type and takes place in presence of 50% aqueous orethanolic alkali One molecule of the aldehyde (missing of a- hydrogen atom )oxidizes, to corresponding carboxlic acid and the other is being reduced to the corresponding alcohol. Mechanism : the mechanism involves two distinct steps Nucleophilic attack and hydride ion transfer. |
|
Answer»

|
|
| 31. |
The reaction is fastest when X is |
|
Answer» `Cl` |
|
| 32. |
The reaction is Decomposition but it's not redox reaction |
|
Answer» `2HgO to 2H_(2)+O_(2)` |
|
| 33. |
The reaction is CH_(2) = CH- CH_(3) + HBr rarr CH_(3) - CHBr-CH_(3) |
|
Answer» NUCLEOPHILIC addition |
|
| 34. |
The reaction is balanced if, 5H_(2)O_(2) + XClO_(2) +2OH^(-) rarr XCl^(-) YO_(2)+6H_(2)O |
|
Answer» `X = 5, Y = 2` |
|
| 35. |
The reaction involves in the oil of winter green test is Salicylic acid underset(conc. H_(2)SO_(4))overset(Delta)rarr Product The product is treated with Na_(2)CO_(3) solutions. The missing reagent in the above reaction is |
Answer» Solution :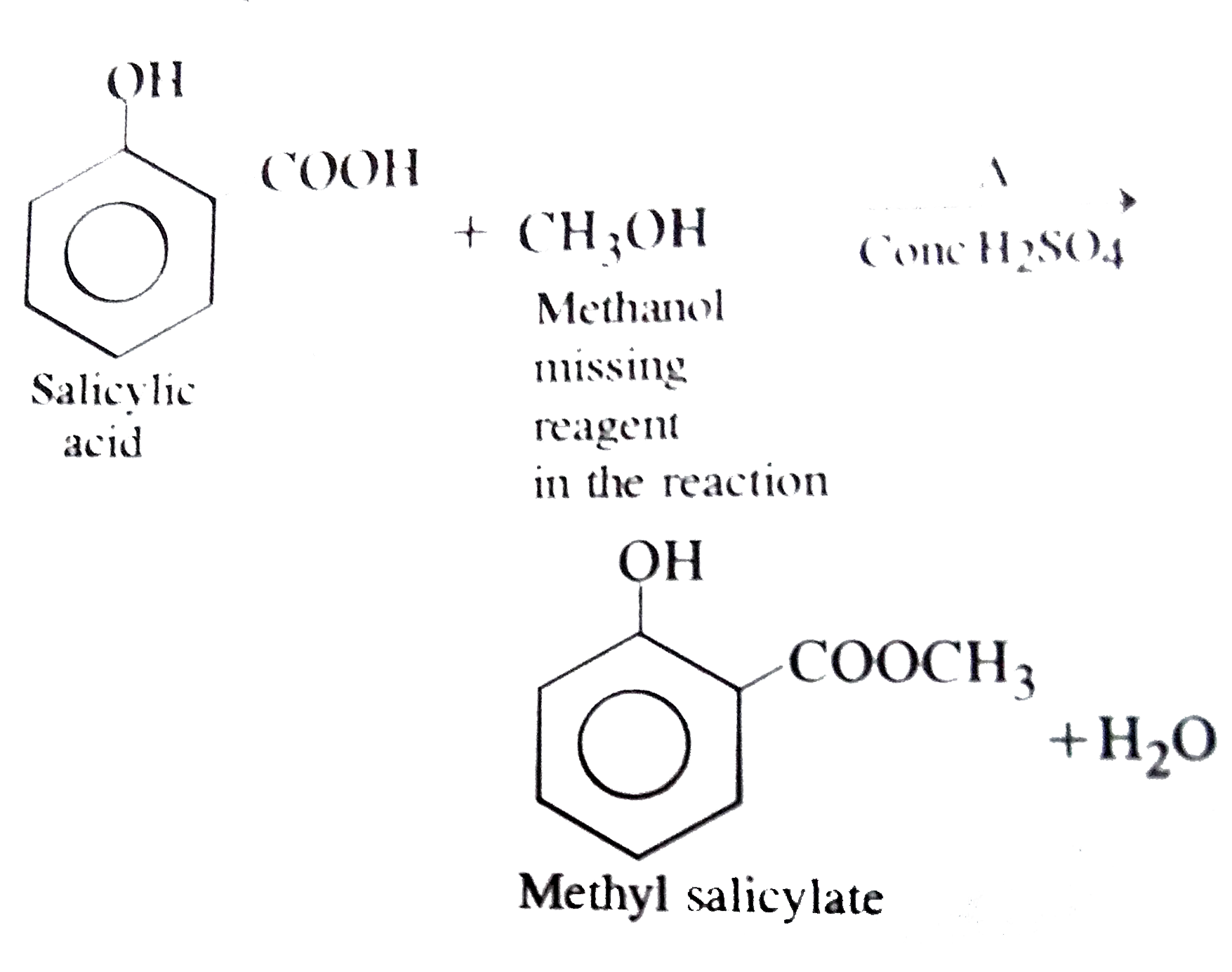
|
|
| 36. |
The reaction involved during the removal of temporary hardness of water is |
|
Answer» `Ca(HCO_(3))_(2)overset(Delta)toCaCO_(3)+H_(2)O+CO_(2)uarr` |
|
| 37. |
The reaction in which sodium alkoxide react with haloalkane to form ether in called …………………… . |
| Answer» SOLUTION :Williamson.s SYNTHESIS | |
| 38. |
The reaction in which hydrogen poeroxide is a reducing agent is: |
|
Answer» `Pbs+4H_(2)O_(2)rarrPbSO_(4)+4H_(2)O` |
|
| 39. |
The reaction in which hydrogen peroxide acts as a reducting agent is . |
|
Answer» `PBS+ 4H_(2)O_(2) rarr PbSO_(4) + 4H_(2)O` |
|
| 40. |
The reaction in which electrons are transferred from one reactant to another is called a .......... |
| Answer» SOLUTION :REDOX REACTION | |
| 41. |
The reaction in terms of intermediates and type of reaction is given below. Mark the incorrect option. |
|
Answer» `CH_(3)-overset(CH_(3))overset(|)(C)=CH_(2)+HB R to` Carbocation intermediate 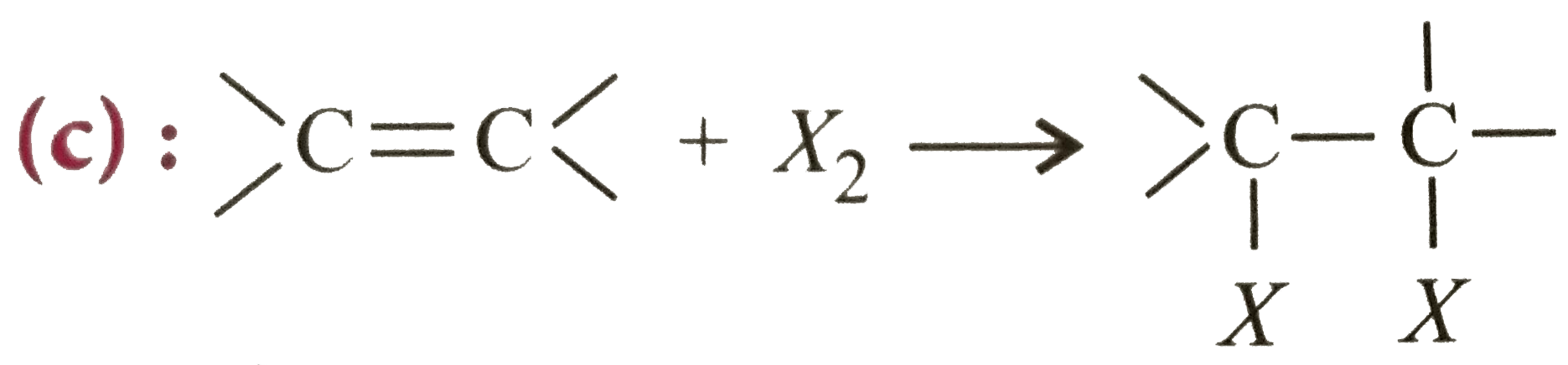 of electrophilic addition reaction. |
|
| 42. |
The reaction H_3PO_2+D_2OtoH_2DPO_2+HDO indicates that hypo-phosphorus acid is |
|
Answer» TRIBASIC acid |
|
| 43. |
The reaction H_2O_2toH_2O+O_2 represents, |
|
Answer» OXIDISING of `H_2O_2` |
|
| 44. |
The reaction H_(2(g))+I_(2(g)) harr 2HI_((g)) is carried out in a 1 litre flask.If the same reaction is earned out in a 2 litre flask at the same temperature, the equilibrium constant will be |
|
Answer» same |
|
| 45. |
The reaction, H_(2(g)) + 1/2O_(2(g)) to H_2O_((l)), is spontaneous. The DeltaS^(@) = - 163.1 J mol^(-1) K^(-1). The absolute entropies of H_(2(g)) and O_(2(g)) are 130.6 JK^(-1)mol^(-1) and 205 JK^(-1) mol^(-1) respectively. Calculate the absolute entropy of water. |
|
Answer» Solution :CHANGE in STANDARD entropy, `DeltaS^(@) = S_(H_2O_(l))^(@) - [S_(H_2)^(@) + 1/2S_(O_2)^(@)]` Absolute entropy of WATER = `69.9 JK-mol^(-1).` |
|
| 46. |
The reaction for which K_(C) lt K_(P) at a given temperature ( gt 50 k)is/are: |
|
Answer» `PCI_5(G)hArrPCI_3(g)+CI_2(g)` |
|
| 47. |
The reaction condtions leading to best yields of C_2H_5CI are |
|
Answer» `C_6H_6(EXCESS)+CI_2overset(UV" "light)to` |
|
| 48. |
The reaction conditions used for converting 1,2-dibromoethane to ethylene are |
|
Answer» Zn, ALCOHOL, `Delta` |
|
| 49. |
The reaction, CO_((g)) + 3H_(2(g)) hArr CH_(4(g)) + H_2O_((g))is at equilibrium at 1300K in a 1L flask. It also contain 0.30 mol of CO, 0.10 mol of H_2 and 0.02 mol of H_2O and an unknown amount of CH_4 in the flask. Determine the concentration of CH_4 in the mixture. The equilibrium constant, K_c for the reaction at the given temperature is 3.90. |
|
Answer» Solution :`{:("EQUILIBRIUM reaction :",CO_((g))+,3H_(2(g)) HARR , CH_(4(g))+ , H_2O_((g))),("Mol at equilibrium :",0.30,0.10,x ,0.02),("Volume = 1L So, mol L"^(-1):,0.30,0.10,x,0.02):}` `K_c`=3.90, where, x=Concentration of `CH_4` equilibrium According to chemical equilibrium constant, `K_c=([CH_4][H_2O])/([CO][H_2]^3)` `therefore 3.90=((x)(0.02))/((0.30)(0.10)^3)` `therefore x=((3.9)xx(0.30)(1xx10^(-3)))/0.02` `=58.5xx10^(-3)` `=5.85xx10^(-2) "mol L"^(-1)` |
|
| 50. |
The reaction , CO(g) + 3 H_(2) (g) hArr CH_(4) (g) + H_(2)O (g),is at equilibrium at 1300 K in a 1 L flask. It also contains 0*30 mol of CO, 0*10 mol of H_(2) and 0*02 " mol of " H_(2)O and an unknown amount of CH_(4) " in the flask. Determine the concentration of " CH_(4)in the mixture. The equilibrium constant, K_(c), for the reaction at the given temperature is 3*90. |
|
Answer» Solution :` K_(c) = ([ CH_(4)][H_(2)O])/([CO][H_(2)]^(3))` ` :. 3* 90 = ([CH_(4)][H_(2)O])/([CO][H_(2)]^(3)) "" ("Molar conc = No. of moles because volume of flask = 1 L")` `[CH_(4)] = 0*0585 M = 5*85 xx 10^(-2) M` |
|