Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following compounds is isomeric with 2,2,4,4-tetramethylhexane? |
|
Answer» 3-ethyl-2,2-dimethylpentane is isomeric with 4-isopropylheptane. |
|
| 2. |
Which pair of the following chlorides do not impart colour to the flame ? |
|
Answer» `BeCl_(2) and SrCl_(2)` |
|
| 3. |
The work function of a metal is 4.2eV. If the radiation of 2000Å falls on the metal, find the kinetic energy of the metal |
|
Answer» Solution :`"WORK FUNCTION "E_(0)=hv_(0)=4.2eV` `=4.2xx1.602xx10^(-19)J=6.7284xx10^(-19)J` `"Energy (E) is given as "` `E=hv=(hc)/(LAMBDA)=(6.625xx10^(-34)Jxx3.0xx10^(8)ms^(-1))/(2000xx10^(-10)m)` `"Threshold energy, "E_(0)=6.7284xx10^(-19)J` `"KINETIC energy, "KE=E-E_(0)` `=[9.939xx10^(-19)J-6.728xx10^(-19)J]` `=3.211xx10^(-19)J` |
|
| 5. |
Write the complete symbol for the atom with the given atomic number (Z) and atomic mass (A) (i) Z = 17, A = 35 (ii) Z = 92, A = 233 (iii) Z = 4, A = 9 |
| Answer» SOLUTION :(i) `._(17)^(35)CL` (ii) `._(92)^(233)U` (III) `._(4)^(9)Be` | |
| 6. |
What volume of carbonmonoxide at 2 atm and 273^(@)C is required in order to produce 5.6 grams of metal by the reduction of ferric oxide? |
|
Answer» SOLUTION :Reduction of FERRIC oxide with carbonmonoxide gives iron metal. `Fe_(2)O_(3)+3COrarr3CO_(2)+2Fe` 2 moles of `Fe -= 3` moles of CO 56 grams of `Fe -= 33.6` L of CO at STP 5.6 grams of `Fe -= 3.36` lit of CO at STP The volume of CO required at STP = 33.6 L `{:("STP CONDITIONS","Given conditions"),(P_(1)=1ATM,P_(2)=2atm),(T_(1)=273K,T_(2)=2xx273K),(V_(1)=3.36L,V_(2)=?):}` `V_(2)=(P_(1)V_(1))/(T_(1))xx(T_(2))/(P_(2))=(1xx3.36xx2xx273)/(273xx2)=3.36L` |
|
| 7. |
The valency shell configuration of IVA element is |
|
Answer» `ns^(2)np^(1)` |
|
| 8. |
What is the normality of sulphate ions in 1M Al_(2)(SO_(4))_(3) solution? |
|
Answer» |
|
| 9. |
Why are BeSO_(4) and MgSO_(4)readily soluble in water while CaSO_(4), SrSO_(4) and BaSO_(4) are insoluble? |
|
Answer» Solution : Solubility of sulphates of group 2 ELEMENTS depends on their hydration ENERGY which decreases down the group. The lattice energy of group 2 sulphates is almost same. Very high hydration enthalpy`Be^(+2)and Mg^(+2)`ions overcome the lattice enthalpy and their sulphates are soluble. However, the hydration enthalpy of`CA^(2+), Sr^(2+)and BA^(2+)`cannot overcome their lattice enthalpy and remain insoluble in water. |
|
| 10. |
Which one of the following statements regarding H_(2)O_(2) is false? |
|
Answer» It is more STABLE in a basic solution |
|
| 11. |
Which of the following gas is insoluble in water ? |
|
Answer» `SO_2` |
|
| 12. |
The volume in litres of CO_2 librated at STP when 10 g of 90% pure limestone is heated completely,is |
|
Answer» `2.016` `100grarr22.4` LIT `(90)/(100)xx10grarr?=2.016L` |
|
| 13. |
Van der Waal's real gas, acts as an ideal gas, at which conditions |
|
Answer» HIGH TEMPERATURE, LOW PRESSURE |
|
| 14. |
The repetition of physical and chemical properties at regular intervals is called_______. |
|
Answer» ATOMIC radii |
|
| 15. |
Two isomeric cyclic dienes X and Y having molecular formula C_6H_8 on hydrogenation give cyclohexane as the only product. A mixture of these two dienes on reductive ozonolysis gives succinaldehyde, propane-1,3-dial and glyoxal. Deduce the structures of dienes X and Y. |
Answer» Solution :Since , X and Y on hydrogenation GIVE cyclohexane as the only product, therefore X and Y MUST be cyclohexadienes . The two possible cyclohexadienes are : 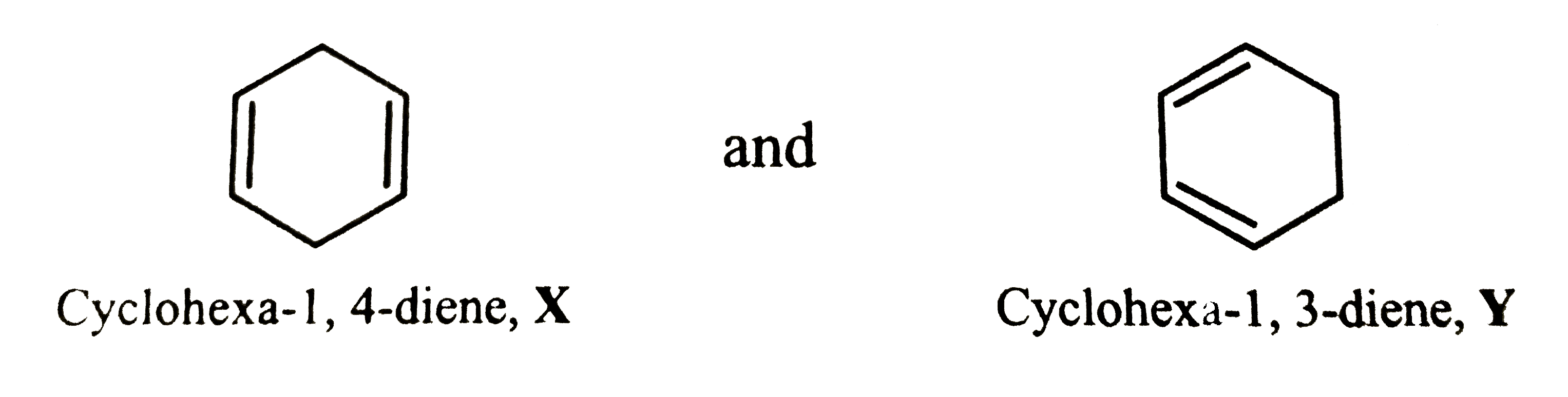 Whereas reductive ozonolysis of X will give two MOLECULES of propane-1,3-dial, reductive ozonolysis of Y will give succinaldehyde and glyoxal as shown below : 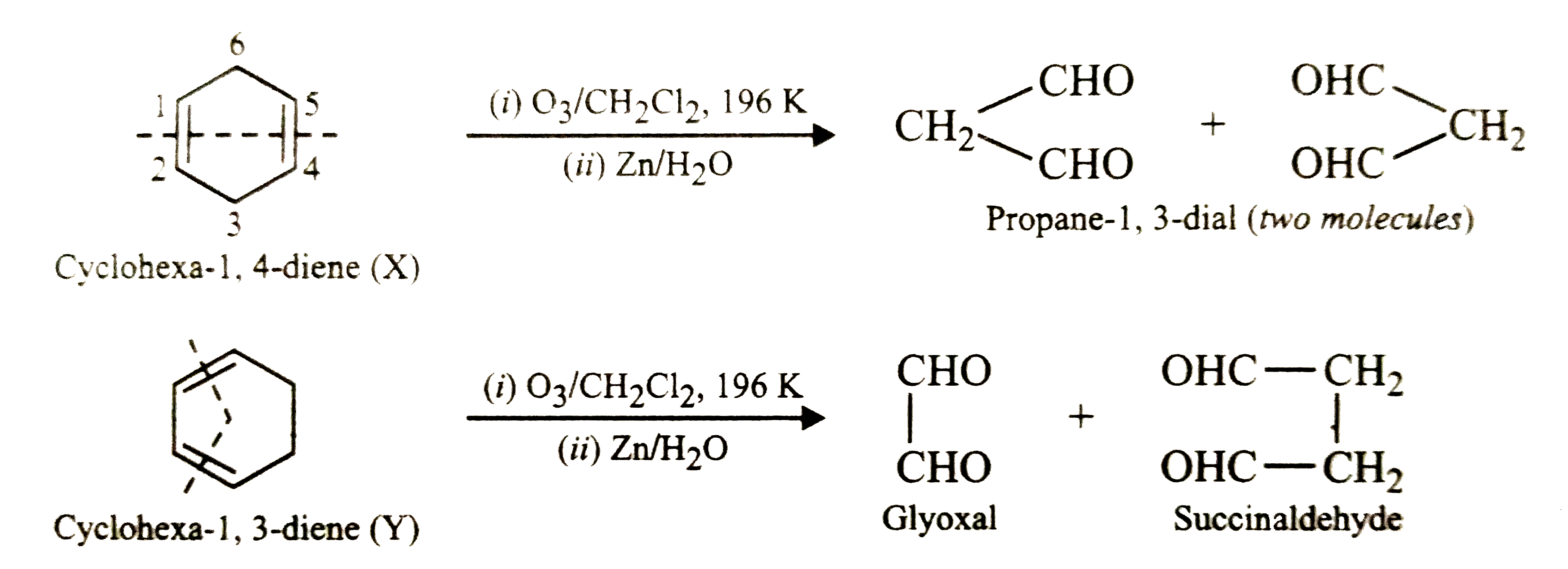
|
|
| 16. |
Whatis the effect of pressure on NaCl type crystals ? |
| Answer» Solution :On INCREASING pressure, the COORDINATION NUMBER of NaCl type crystals increases from 6 : 6 to 8 : 8. | |
| 17. |
When PbO_(2) reacts with conc. HNO_(3), the gas evolved is |
|
Answer» `NO_(2)` |
|
| 18. |
What are the possible values of principal (n) and azimuthal (l) quantum number for the unpaired electron in the atom of an element whose atomic number is 9 ? |
| Answer» SOLUTION :`._(9)F = 1s^(2) 2s^(2) 2p_(x)^(2) 2p_(y)^(2) 2p_(Z)^(1)`. Hence, for the UNPAIRED `2p_(z)^(1)` ELECTRON, `N = 2, l = 1` | |
| 19. |
Which compound are taken in Kjeldabl's flask? |
| Answer» SOLUTION :The compound which should be ESTIMATED + conc. `H_(2)SO_(4) + CuSO_(4)` | |
| 20. |
Which of the following is oxidized by H_(2)O_(2) in the alkaline medium |
| Answer» Answer :D | |
| 21. |
What is the purpose of levelling ? |
| Answer» SOLUTION :To RECORD VOLUME under ATMOSPHERIC PRESSURE. | |
| 22. |
Wrlle Lewis structure of Nitrite ion. NO_(2)^(-1) |
|
Answer» Solution :(i) Total ELECTRON : N ( Z = 7) : [He] `2s^(2) 2p^(3)` = 5 valence electron O (Z = 8) : [He] `2s^(2) 2p^(4)` = (6) 2 valences electron = 6 `XX` = 12 (Two oxygen) (ii) Structural PRESENTATION : Electronegativity of N is less than O So N is central atom so the arrangement will be.  (iii) To get octet on N and O double bond is required between N and O.  In this WAY N has one double bond and one single bond. One .O. has one double bond and second .O. has single bond. |
|
| 23. |
Which of the following resonating structures is not correct for carbon dioxide- |
|
Answer» `:OVERSET(..)(O)=C=overset(..)(O):` |
|
| 24. |
Which of the following are correct? |
|
Answer» Ordinary water is electrolyzed more rapidly than `D_2O` B) Reaction between `H_2 and Cl_2` is much faster than `D_2 and Cl_2` d) Bond dissociation energy for `D_2` is greater than `H_2` |
|
| 25. |
When lactic acid is subjected to oxidation with Fenton's reagent, the main product formed is |
|
Answer» ACETIC acid |
|
| 26. |
Which compound is formed by heating with nitric acid in estimation of phosphorus in Carious method? |
|
Answer» |
|
| 27. |
Which of the following order(s) is correct |
| Answer» Answer :D | |
| 28. |
Which one of the following solutions are added to Lassaigne's extract to identify halogens? |
|
Answer» ACETIC acid + Lead acetate |
|
| 29. |
Write the molecular orbital electronic configuration for carbon monoxide molecule. Calculate its bond order and comment on its magnetic property. |
|
Answer» SOLUTION :`sigma1s^(2) sigma^(**) 1s^(2) sigma 2s^(2) sigma^(**) 2 s^(2) pi 2 px^(2)=pi 2 py^(2)` bond ORDER `=(N_(B)-N_(A))/2=1/2(8-4)=2` Since there are no UNPAIRED electron it is diamagnetic in nature. |
|
| 30. |
What is chain isomerism? Explain by giving example |
|
Answer» Solution :When two or more compounds have similar molecular FORMULA but different CARBON skeletons, these are referred to as chain isomers and the PHENOMENON is termed as chain isomerism. e.g., `C_(5)H_(12)` (PENTANE) represents three compounds. (i) `CH_(3)CH_(2)CH_(2)CH_(2)CH_(3)`: Pentane (ii) `CH_(3)-underset(underset(CH_(3))(|))(CH)-CH_(2)CH_(3)`: Isopentane (2-methylbutane) (iii)  : Neopentane (2, 2-dimethyl propane) : Neopentane (2, 2-dimethyl propane)
|
|
| 31. |
The total number of structural isomers possible for hydrocarbon C_(5)H_(10) is: |
|
Answer» 9  
|
|
| 32. |
Which of the following statement is/are wrong about pericyclic reaction? |
|
Answer» If the value of l = 0, the electron distribution is SPHERICAL |
|
| 33. |
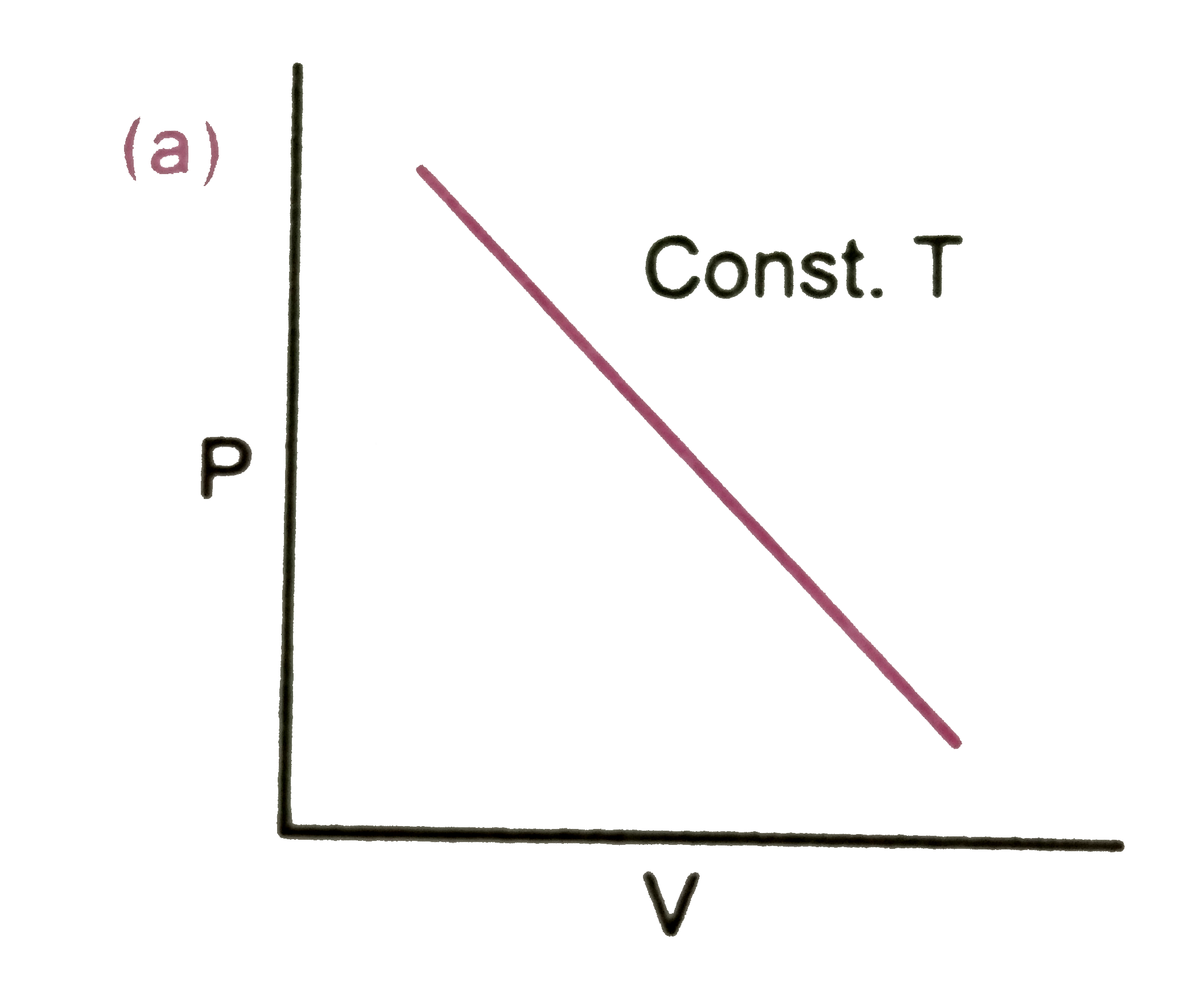
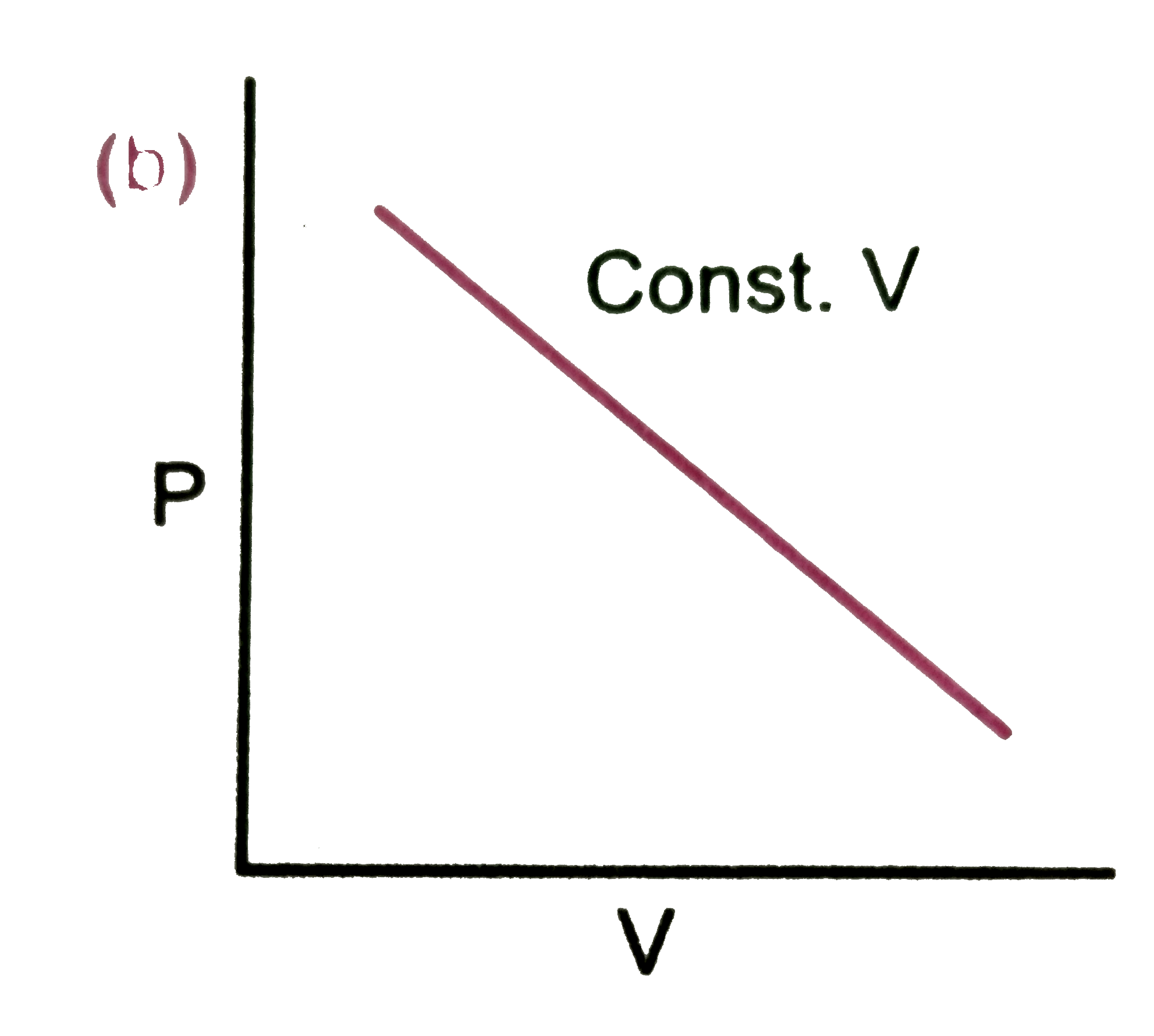
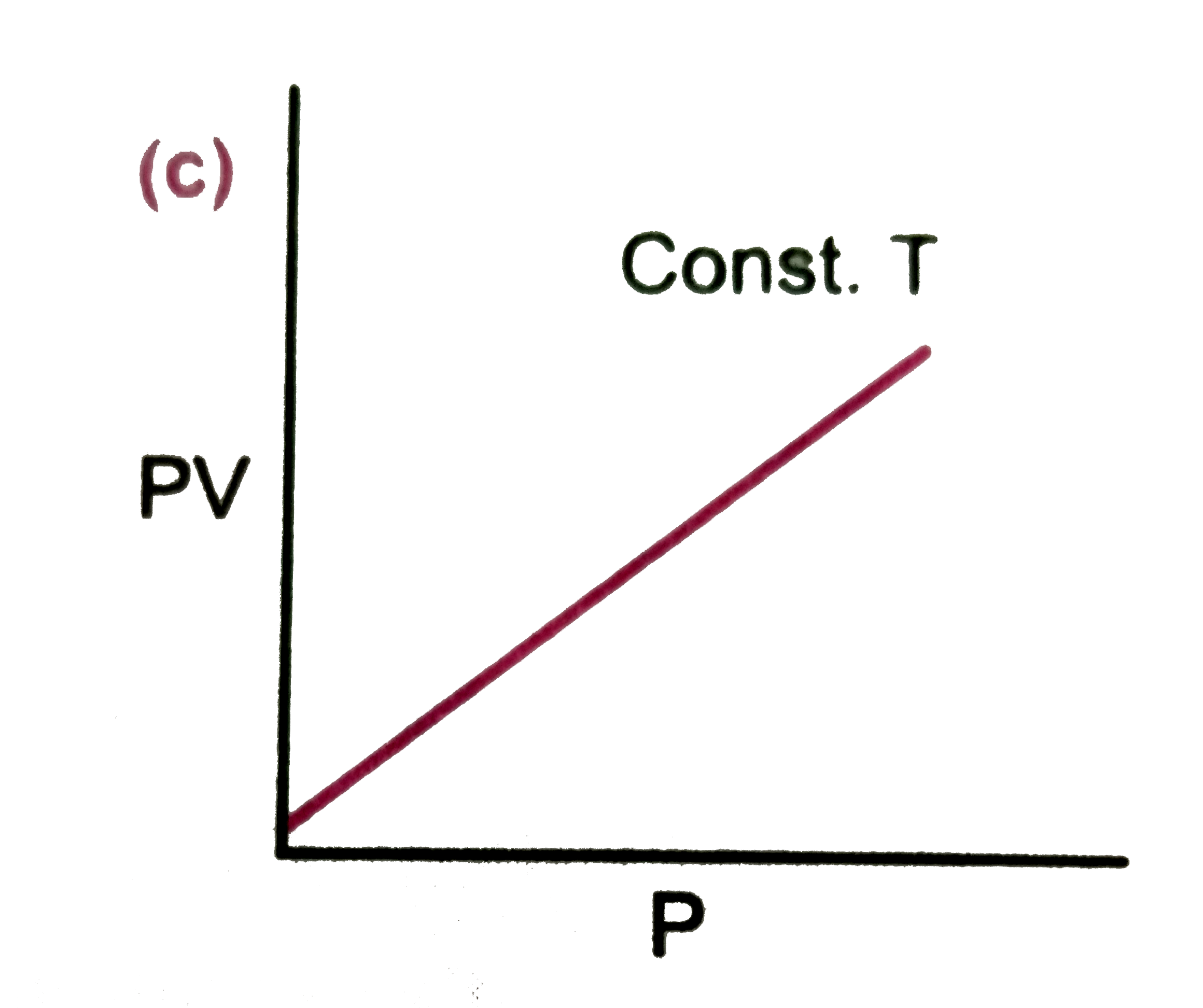
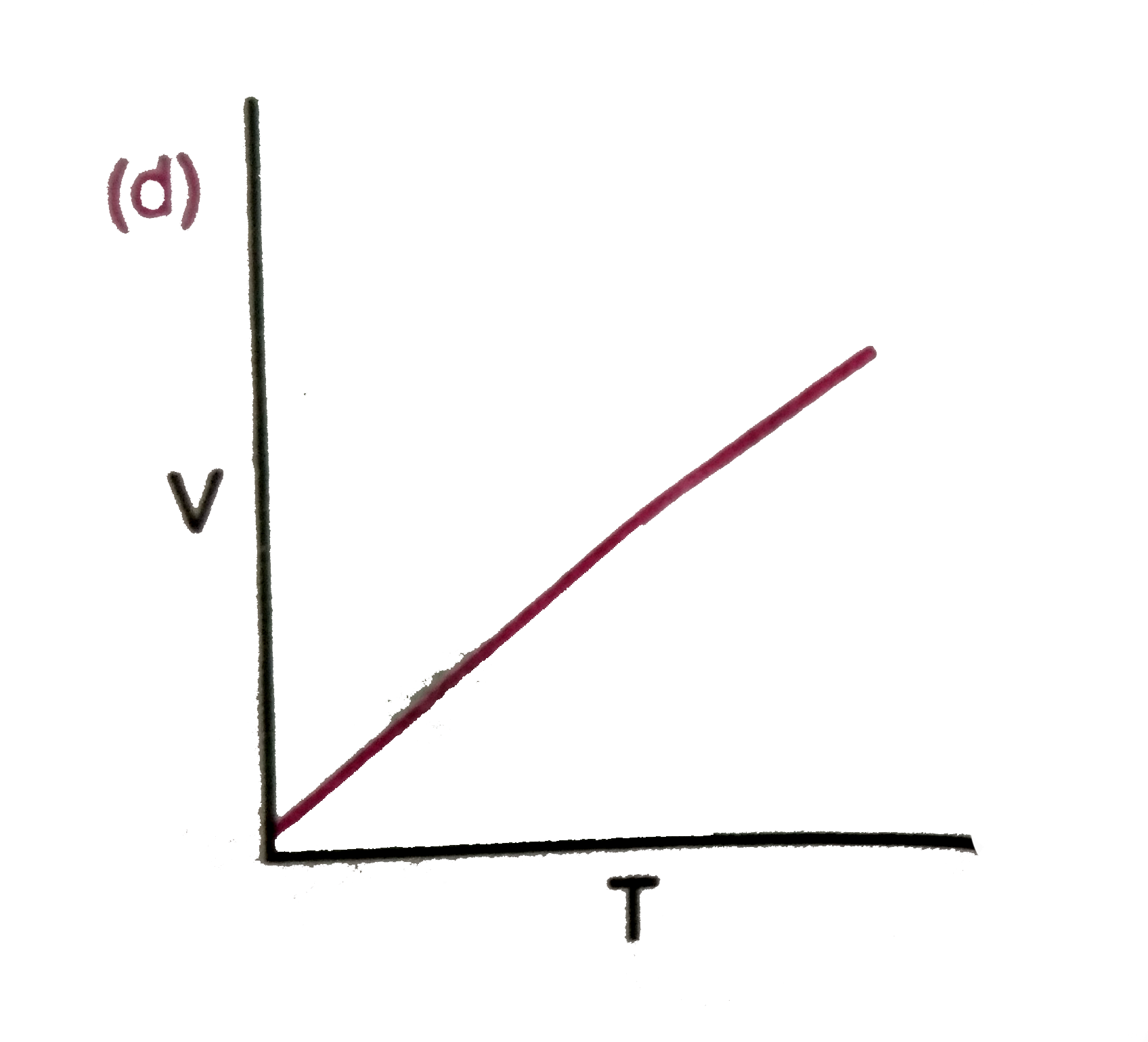
Which of the following diagram correctly describes the behaviour of a fixed mass of an ideal gas ? (T is measured in K) |
|
Answer»
|
|
| 35. |
What is the effect of temperature on ionic product of water and why ? |
| Answer» Solution :Ionic PRODUCT of water `(K_(W))` INCREASES with INCREASE of temperature because `K_(w)=[H_(3)O^(+)][OH^(-)] and ` dissociation of `H_(2)O` to give `H_(3)O^(+)` and `OH^(-)` increases with increase of temperature. | |
| 36. |
Which of the followingN^(3-),O^(2-),F^(-) is largest in size? |
| Answer» Answer :a | |
| 37. |
Which of the following statements is/are wrong |
|
Answer» The GAS displaced in VICTOR Meyer.s method is AIR |
|
| 38. |
Which of the following is dihydric alcohol? |
|
Answer» Glycerol |
|
| 39. |
When FeCl_(3) is added to the sodium extract of an organic compound containing both N and S, the red coloured precipitate is formed due to the formation of |
|
Answer» `[Fe(CN)_(6)]^(3-)` `NaCNS+FeCl_(3)RARR UNDERSET("Red coloured complex")([(Fe(SCN)]Cl_(2))DARR)+NaCl` |
|
| 40. |
the energy of photon is equal to the kineticenergy of a porton,If lambda_1 is the De-Broglie wavelengthof a proton,lambda_2 the wavelength associated with the proton and if the energt of the protonis E, then (lamda_1//lambda_2) is proportional to |
|
Answer» `lambda_1+ lambda_2` = `lambda_3` `1/(lambda_1) + 1/(lambda_2) = 1/(lambda_3)` |
|
| 42. |
When mixture of CuCO_(3).Cu(OH)_(2)(s) and Pb(NO_(3))_(2)(s) is added in dil. HCI solution: |
|
Answer» suspension of `CuCO_(3).CU(OH)_(2)(s)` in colourless SOLUTION is precipitated |
|
| 43. |
What happens when steam is passed over red hot iron? |
|
Answer» Solution :When steam is passed over RED hot IRON, iron oxide will be formed with the RELEASE of hydrogen. `3Fe_((s))+4H_(2)O_((L))toFeO_(4(s))+4H_(2(g))` |
|
| 44. |
What is meant by ionic product of water (K_(w))? |
|
Answer» SOLUTION :It is product of CONCENTRATION of `[H_(3)O^(+)]and[OH^(-)]` at a specific TEMPERATURE. `K_(W)=[H_(3)O^(+)][OH^(-)]=1.0xx10^(-14)` at 298 K. |
|
| 45. |
White fumes appear around the bottle of anhydrous aluminium chloride. Give reason. |
| Answer» SOLUTION :ANHYDROUS aluminium chloride is PARTIALLY hydrolysed with atmospheric moisture to LIBERATE HCL gas. Moist HCl appears white in colour. | |
| 46. |
Using the following Latimer diagram for bromine, pH=0, BrO_(4)^(-) overset(1.82 V)rarr BrO_(3)^(-)overset(1.50 V)rarr HBrO overset(1.595 V) rarr Br_(2) overset(1.06552 V) rarr Br^(-) the species undergoing disproportionation is |
|
Answer» `BrO_(4)^(-)` |
|
| 47. |
Using "Le" Chateller's principle, predict the effect of (i) decreasing the temperature and (ii) increasing the pressure on each of the following equilibria: (A) N_(2)(g)+3H_(2)(g)hArr2NH_(3)(g)+"Heat" (B) N_(2)(g)+O_(2)(g)hArr2NO(g)+"Heat" (C) H_(2)O(g)+"Heat"hArrH_(2)(g)+(1)/(2)O_(2)(g) (D) 2CO(g)+O_(2)(g)hArr2CO_(2)(g)+"Heat" |
|
Answer» |
|
| 48. |
Which of the following statements is/are wrong? |
|
Answer» Sulphur is estimated by Carius method as `BaSO_4` |
|
| 49. |
What is used to represent the C= C in ball and stick model? |
| Answer» Solution :In this model, SPRINGS is USED in PLACE of stick | |