Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When we heat an iron bar, it first becomes red, then yellow and finally begins to glow with white light and then blue light. Why ? |
| Answer» SOLUTION :The change in colour is due to increase in the frequency of radiation emitted on heating as RED colour LIES in the lower frequency REGION while BLUE colour lies in the high frequency region. | |
| 2. |
When a greater number of electrons from excited hydrogen atoms reach the ground state, then |
|
Answer» The intensity of SPECTRAL lines in Lyman series increases |
|
| 3. |
Which of the following is a linear molecule? |
|
Answer» `BeCl_2` |
|
| 4. |
Which one of the following processes will produce hard water ? |
|
Answer» SATURATION of WATER with `CaCO_3` |
|
| 5. |
What happens when: |
|
Answer» magnesium is burnt in air `2Mg+O_(2)rarr2MgO`(Magnesium oxide) `3Mg+N_(2)rarrMg_(3)N_(2)` (Magnesium nitride) (ii). When quicklime `(CaO)` is heated with silica, it forms calcium SILICATE. `CaO+SiO_(2)overset(Above 1270 K)(rarr) underset(("Calcium sillicate"))(CaSiO_(3))` (iii). When chlorine reacts with slaked lime, it forms bleaching powder. `underset(("Slaked lime"))(Ca(OH)_(2))+Cl_(2)rarrunderset(("Bleaching powder"))(CaOCl_(2)+H_(2)O)` (iv). When calcium nitrate is heated, it decomposed to give `CaO,NO_(2)` and `O_(2)`. `2Ca(NO_(3))_(2)overset(rarr)2CaCO+4NO_(2)+O_(2)uarr` |
|
| 6. |
What is the hybridisation of carbon in problem |
| Answer» SOLUTION :`H_(3) underset(underset(sp^(3))(UARR))(C ) - underset(underset(sp)(uarr))(C ) -= N` (B) `H_(2) underset(underset(sp^(2))(uarr))(C )= underset(underset(sp)(uarr))(C )= underset(underset(sp^(2))(uarr))(CH_(2))` (c ) `H_(3) underset(underset(sp^(3))(uarr))(C )- underset(underset(sp^(3))(uarr))(CH_(3))` (d) `underset(underset(sp^(2))(uarr))(CH_(2)) = underset(underset(sp^(2))(uarr))(CH) - underset(underset(sp^(3))(uarr))(CH_(2)) - underset(underset(sp^(2))(uarr))(C )-= underset(underset(sp)(uarr))(CH)` (E ) `underset(underset(sp^(3))(uarr))(CH_(3))- underset(underset(sp^(2))(uarr))overset(overset(OH)(|))(C )= O` | |
| 7. |
What is the role of a catalyst in a catalysed reaction |
|
Answer» LOWERS the ACTIVATION energy |
|
| 8. |
Which of the following represents redox reaction ? |
|
Answer» `Cr_(2)O_(7)^(-2) +2OH^(-) rarrCrO_(4)^(2-)+ H_(2)O` b) `overset(+8)underset(_)(S)O_5^(2+)+I^(-)rarrI_2^(0)+overset(+6)(S)ulO_4^(2-)` C) `Ca(OH)_(2)+Cl_(2)^(0) rarr Caoverset(+1)((ClO))_(2)+CaCl_(2)^(-1)` d) `overset(+5 -1)(PCl_5) rarr overset(+3-1)(PCl_3)+Cl_(2)^(0)` |
|
| 9. |
Write the electronic configurations and calculate the bond orders of H_(2)^(+) , H_2 and He_2 Why the bond length in H_(2)^(+) is longer than that in H_2 ? |
|
Answer» Solution :`{: ("Species""""Electronic configuration"""Nb""Na"""Bond order"),(H_(2)^(+)""(SIGMA1S)^(1)""1""0""0.5),(H_(2)""(sigma1s)^(2)""2""0""1),(He_(2)""(sigma1s)^(2)(sigma**1s)^(2)""2""2""0):}` Then bond length in `H_(2)^(+)` is longer than in `H_(2)` because in `H_(2)^(+)` only ONE ELECTRON is present to SHIELD the two nuclei from mutual repulsion. In `H_(2)` there are two electrons to hold the two nuclei, thus the nuclear repulsion is less than that in `H_(2)^(+)`. Hence nuclear separation in `H_(2)^(+)` is more thanin `H_(2)`. |
|
| 10. |
The volume of 0.1 M NaOH will be required to neutralise 100 ml of 0.1 ml H_(3)PO_(4) using methyl red indicator to change the colour from pink (acidic medium) to yellow (basic medium) is 10^(x). What is x? |
|
Answer» `H_(3)PO_(4)iffH^(+)+H_(2)PO_(4)^(-)` `N_(1)V_(1)=N_(2)V_(2)implies0.1xx1xxV_(1)=0.1xx1xx100` `V_(1)=100ml=10^(2)ml` |
|
| 11. |
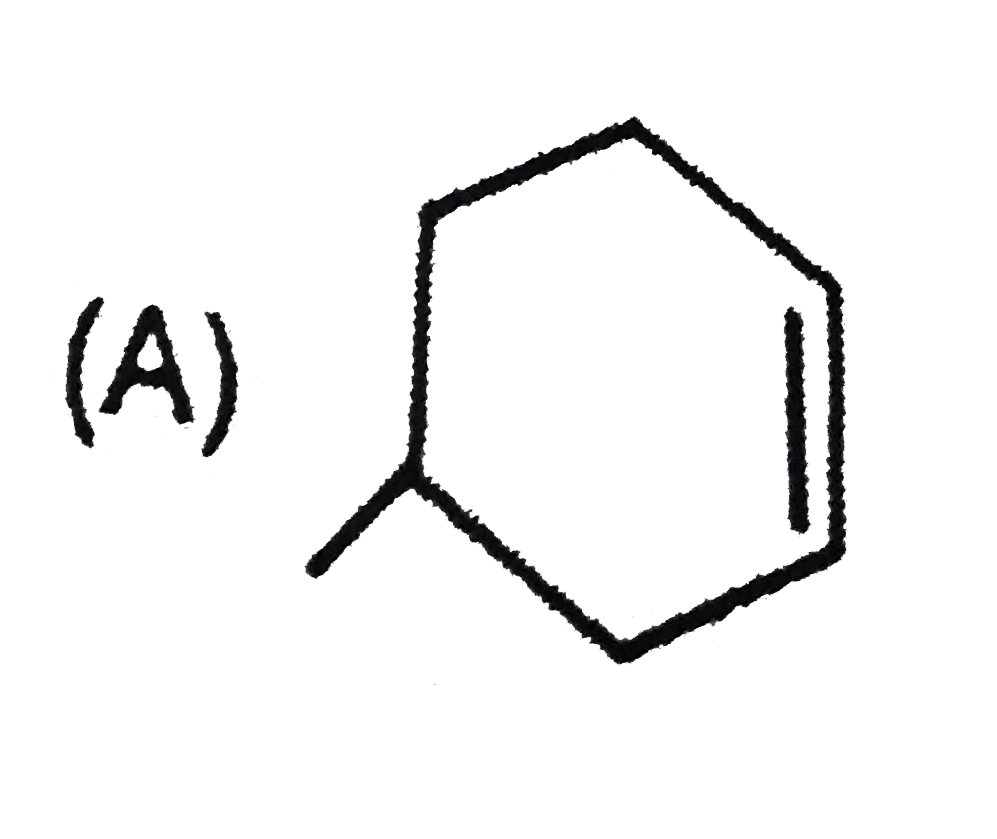
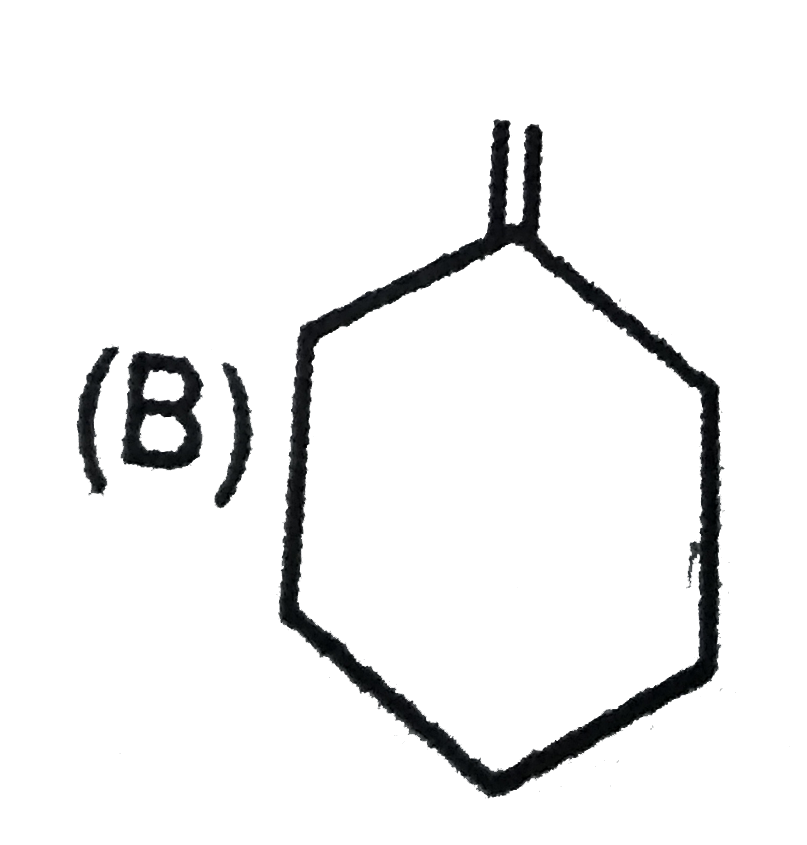
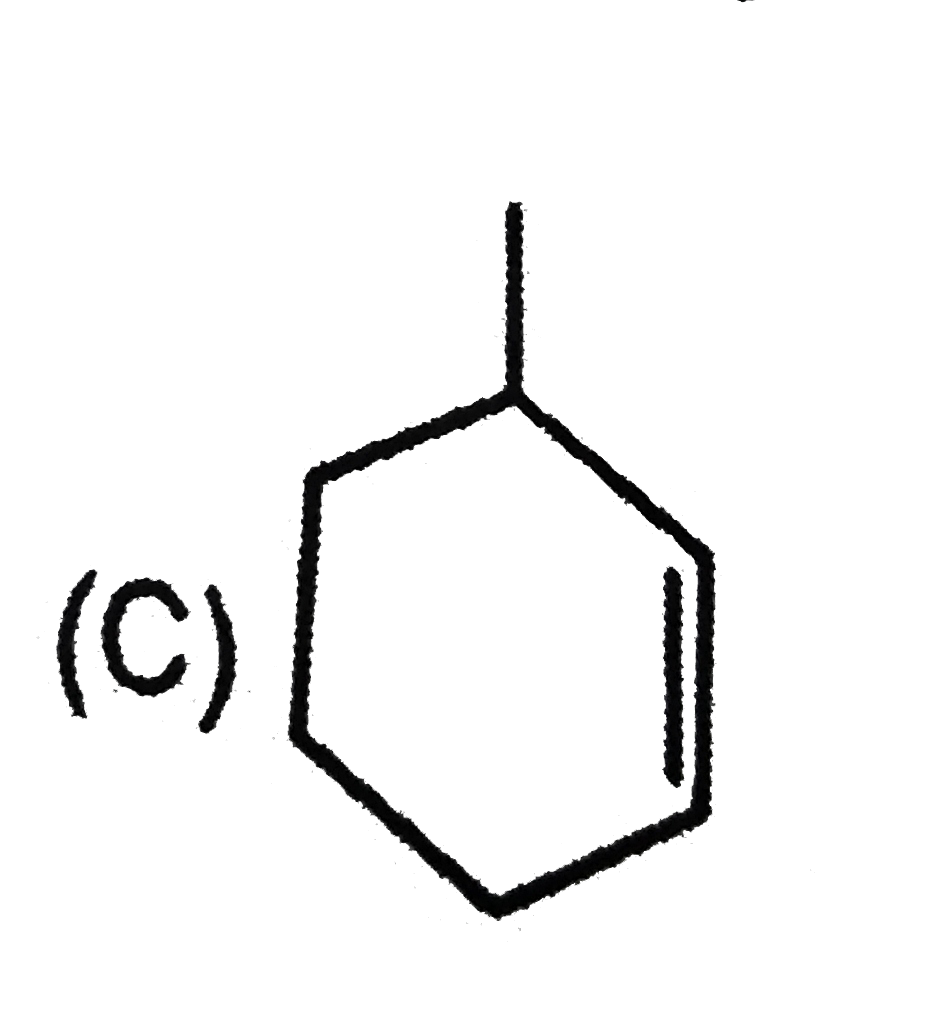
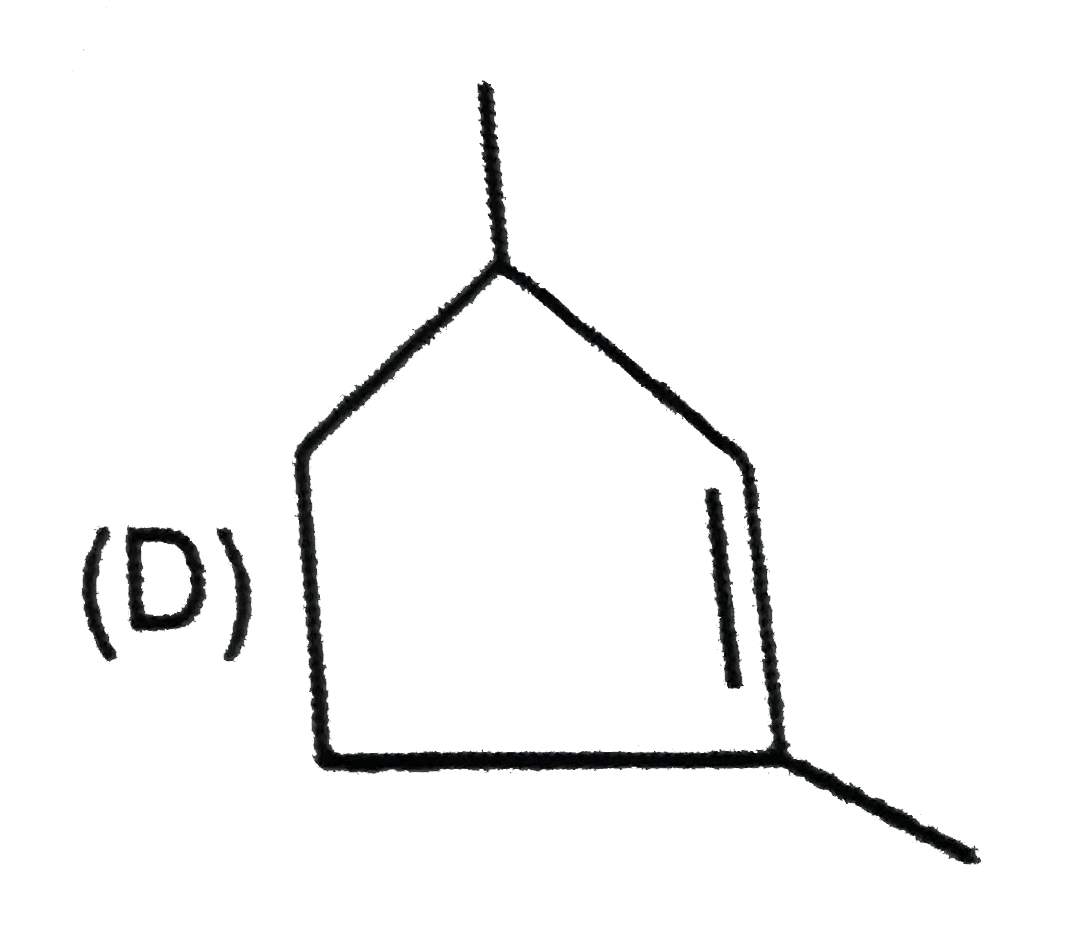
Which of the following systems are aromatic? |
|
Answer»
 (b) has 16(4N) pi-electrons, hence antiaromatic. (c) (azulene) and (d) are aromatic system as they satisfy Huckel rule. |
|
| 12. |
Which of the following have zero dipole moment? |
|
Answer» TRANS-2,3-dichloro-2-butene |
|
| 13. |
What pH do you expect for 10^(-8) M solution of an acid ? |
| Answer» SOLUTION : CLOSE to 7 but `LT 7`. | |
| 14. |
When 100 ML of 1.0 M HCl was mixed 100MI of 1.0 M NaOH in an insulated beakertat constant pressure, a temperature incease of 5.7^(0)C was measured for the beaker and its contents (Expt.1) Because th enthalpy of neutralization of a c 5.7^(0)C strong acid with a strong base is constant (-57.0 kJ moL- I), this experiment could be used to measure the calorimeter constant. In a second experiment (Expt.2) , 100 ML of 2.0 M acetic acid (K_(a) =2.0 xx 10^(-5)) was mixed with 100 ML of 1.0 m NaOH (under identicalconditions to Expt. 1) Where a temperaturerise of 5.0^(0)C was measured. (Consider heat capcity of all solution as 4.2 Jg^(-1) and density of all solution as 1.0 gmL^(-1)) Enthalpy of dissociation (in KJ Mol^(-1)) of acetic acid obtained from Expt, 2 is |
|
Answer» `1.0` |
|
| 15. |
Which set of reagentswould you use to form the following compound? |
|
Answer» `HO-CH_(2)-CH_(2)-OH`and`CH_(2)=O` |
|
| 16. |
What is the name of the reaction 2CH_(3)CH_(2)SH rarr CH_(3)CH_(2)CH_(2)-S-S-CH_(2)CH_(2)CH_(3) Wheter codensation oxdation reduction or polymerization |
| Answer» SOLUTION :This is na exaple of oxidatin reaction since TWO H - ATOMS have been removed | |
| 17. |
When l=0,the number of electrons that can be accommodated in the subshell is……. |
|
Answer» SOLUTION :If l=0,NUMBER of ELECTORNS=(2l+1) `=2(2xx0+1)=2` |
|
| 18. |
When 100 ML of 1.0 M HCl was mixed 100MI of 1.0 M NaOH in an insulated beakertat constant pressure, a temperature incease of 5.7^(0)C was measured for the beaker and its contents (Expt.1) Because th enthalpy of neutralization of a c 5.7^(0)C strong acid with a strong base is constant (-57.0 kJ moL- I), this experiment could be used to measure the calorimeter constant. In a second experiment (Expt.2) , 100 ML of 2.0 M acetic acid (K_(a) =2.0 xx 10^(-5)) was mixed with 100 ML of 1.0 m NaOH (under identicalconditions to Expt. 1) Where a temperaturerise of 5.0^(0)C was measured. (Consider heat capcity of all solution as 4.2 Jg^(-1) and density of all solution as 1.0 gmL^(-1)) The p^(H) of the solution after Expt.2 is |
|
Answer» `2.8` |
|
| 19. |
What is F-centre ? |
| Answer» Solution :The site where ELECTRON is ENTRAPPED in THEANION VACANCY is called F-centre. | |
| 20. |
Which of the following is incorrect for ideal solution ? |
|
Answer» <P>1) `Delta H _(MIX) =0` `therefore` Incorrect is `Delta G _(mix )=0` |
|
| 21. |
Which of the following have equal concentration- |
|
Answer» 200 ML SOLUTION of 20g NaOH |
|
| 22. |
Write the stability order of following resonating structures : |
|
Answer» <P> |
|
| 23. |
Which of the following on treatment with hot concentrated acidified KMnO_(4) will give 2-methylhexane-1,6-dioic acid as the only organic product ? |
|
Answer»
|
|
| 24. |
The volume of nitrogen is measured by which apparatus in Dumas method? |
|
Answer» |
|
| 26. |
The values of K_(p) and K_(p_(2)) for the reaction X hArr Y+Z ...(1) and A hArr 2B...(2) Are in the ratio 9: 1 . It the degree of dissociationof X and A be equal , calculate the ratio of the total prssures of (1) and (2) at equilibrium. |
|
Answer» Solution :Suppose total pressure at EQUILIBRIUM for reactions (1) and (2) are `P_(1)and P_(2)` respectively . Then ` {:(,X,hArr,Y,+,Z),("Intial.",1"MOLE",,0,,0),("At EQM.",1-alpha,,alpha,alpha,"Total"1+alpha):}` ` P_(X) = (1-alpha)/(1+alpha) P_(1), p_(Y) = alpha/(1+alpha) p_(1),p_(Z) = alpha/(1+alpha)P_(1)` ` K_(p_(1)) = ((apha)/(1+alpha) P_(1))^(2)/((1-alpha)/(1+alpha)P_(1))=(alpha^(2)P_(1))/(1- alpha^(2) )CONG alpha^(2) P_(1) ` `{:(,A,hArr,2B,),("Intial",1"mole",,0,),("At eqm.",1-alpha,,2 alpha,"Total"=1=alpha):}` ` p_(A) = (1-alpha)/(1 +alpha)p^(2),p^(B) = (2alpha)/(1 + alpha) P^(2)` ` K_(p_(2)) = ((2alpha)/(1+alpha)P_(2))^(2)/((1-alpha)/(1+alpha)P^(2))= (4 alpha^(2))/(1-alpha^(2)) P_(2) = 4 alpha ^(2) P_(2) ` ` (K_(p_(1)))/(K_(p_(2)))= (alpha^(2) P_(1))/(4alpha^(2)P_(2)) = (P_(1))/(4 P_(2))= 9/1 ("Given") or (P_(1))/(P_(2))= 36 /1 = 36 :1 ` |
|
| 27. |
When light with a wavelength of 400 nm falls on the surface of sodium , electrons with a kinetic energy of1.05 xx 10^(5) " J mol"^(-1) are emitted . What is the minimum energy needed to remove an electron from sodium ? |
|
Answer» |
|
| 28. |
When CH_(2)=CH-COOH is reduced with LiAlH_(4), the compound obtained will be : |
|
Answer» `CH_(3)-CH_(2)-COOH` |
|
| 29. |
When light with a wavelength of 400 nm falls on the surface of sodium , electrons with a kinetic energy of1.05 xx 10^(5) " J mol"^(-1) are emitted . What is the maximum wavelength of light that will cause a photoelectron to be emitted ? |
|
Answer» |
|
| 30. |
What do you observe in the spectrum of NaCl ? |
| Answer» SOLUTION :TWO YELLOW LINE with WAVELENGTHS `5890Å and 5896 Å` | |
| 31. |
What arehorizontal rowsand verticalcolumns of the periodictablecalled ? |
| Answer» SOLUTION :Horizontalrows arecalledperiodswhileverticalcolumnsare calledgroups. | |
| 32. |
What is weak electrolyte ? Give examples. |
|
Answer» Solution :A weak ELECTROLYTE is defined as a substance which dissociates to a SMALL extent in aqueous solution and hence conducts electricity also to a small 1 extent. e.g. `NH_4OH, OH_3COOH` etc. In weak electrolytes, as they are partly ionized, an EQUILIBRIUM is set up between the ions and the UNIONIZED electrolyte. |
|
| 33. |
Which contains both polar and non-polar bonds ? |
|
Answer» ` NH_(4) CL` |
|
| 34. |
Which of the following has least bond enthalpy ? |
|
Answer» `Pb-Pb` |
|
| 35. |
What is the importance of heavy water with regard to nuclear power generation ? |
| Answer» SOLUTION :It is used as a moderator in atomic REACTORS to SLOW down the speed of neutrons produced as a result of fission and thus helps to CONTROL the nuclear reactions. | |
| 36. |
What is Fission of a covalent bond? Explain its type with example and main difference |
|
Answer» Solution :The FISSION of a covalent bond means the REACTION of BREAKING the covalent bond. The covalent bond can get CLEAVED EITHER by: (i) heterolytic cleavage OR (ii) homolytic cleavage 
|
|
| 37. |
The vapour density of undecomposed N_(2)O_(4) is 46. When heated, vapour density decreases to 24.5 due to is dissociation to NO_(2). The % dissociation of N_(2)O_(4) at the final temperature is |
|
Answer» 80 `:. (VD_("innitial"))/(VD_("FINAL")) = (n_("final"))/(n_("innitial"))` `(46)/(25.4) = (25.4)/(46) = (1+alpha)/(1)` `1.8 = 1+alpha` `implies alpha = 0.8`or`80%` |
|
| 38. |
When N_(2) goes to N_(2)^(+), then N-N bond distance ……….and when O_(2) goes to O_(2)^(+) the O-O bod distance……. |
|
Answer» INCREASES, DECREASES |
|
| 39. |
Which of the following compounds cannot exhibit mesomeric effect |
|
Answer» `CH_(3)-CH_(2)=CH-overset(oplus)(C)H-CH_(3)` |
|
| 40. |
Which of the following compounds will not form an anhydride ? |
|
Answer» Propandioic ACID `HOOC - CH_(2) - COOH underset(-2H_(2)O)rarr underset("CARBON suboxide")(O = C = C = C = O)` |
|
| 41. |
What is the melting point of benzene if Delta H_("fusion") = 9.95 kJ//mol and Delta S_("fusion") = 35.7 J//K- mol? |
|
Answer» `278.7^(@)C` `rArr` MP = 278.7 K |
|
| 42. |
What type of addition reactions do alkenes usually undergo ? |
| Answer» SOLUTION :ELECTROPHILIC | |
| 43. |
Thermodynamically most stable allotrope of carbon is |
| Answer» SOLUTION :Graphite is more STABLE than Diamond | |
| 44. |
X, Y and Z elements have 4, 5 and 7 valence electrons . Draw the structure of XH_(4), YH_(5) YH_(3)" and " H - Z. |
Answer» SOLUTION :
|
|
| 45. |
Write redox couples involved in the reaction (a) to (d) given in que. No. 34. Cu+Zn^(+2)toCu^(+2)+Zn Mg+Fe^(+2)toMg^(+2)+Fe Br_(2)+2Cl^(-)toCl_(2)+2Br^(-) Fe+Cd^(+2)toCd+Fe^(+2) |
|
Answer» SOLUTION :(a) `Cu^(+2)//CuandZn^(+2)//Zn` (B) `MG^(+2)//MgandFe^(+2)//Fe` ( c) `Br_(2)//Br^(-)andCl_(2)//CL^(-)` `Fe^(+2)//FeandCd^(+2)//CD` |
|
| 46. |
When KMnO_4 acts as an oxidizing agent and ultimately forms MnO_(4)^(2-), MnO_2, Mn_2O_3 and Mn^(2+). The number of electrons transferred in each case respectively is |
|
Answer» 1,3,4,5 |
|
| 47. |
What is the characteristic of resonance structure? |
|
Answer» Solution :(i) The POSITION of ATOMIC CENTRE is same. (ii) Total electron REMAIN same. (iii) `pi` or p electron migrate and change their position |
|
| 48. |
When CaC_2 is heated in atmospheric nitrogen in an electric furnace the compound formed is |
|
Answer» `CA(CN)_(2)` |
|
| 49. |
Vingegar contains |
|
Answer» `10 - 20%` ACETIC ACID |
|
| 50. |
X' is most inert element and 'Y' is most reactive element in the long form of the periodic table. 'X' and 'Y' are respectively |
| Answer» Answer :B | |