Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following statement is not correct about the characteristics of cathode rays ? |
|
Answer» They start from the CATHODE and move towards the anode |
|
| 2. |
Which one of the following benefication process is used for the mineral . Al_(2)O_(3).2H_(2)O? |
|
Answer» Froth floatation |
|
| 3. |
When does the blue coloured ammonia solution (of alkali metals) changes to bronze colour? |
|
Answer» Solution :(i) Alkali metals dissolve in liquid ammonia to give deep blue solutions that are conducting in nature (ii) The blue colour of the solution is due to the ammoniated electron which absorbs ENERGY in the visible region of light and thus imparts blue colour to the solution. (iii) The solutions are paramagnetic and on standing slowly liberate hydrogen resulting in the formation of amide. (IV) In CONCENTRATED solution, the blue colour change to bronze colour and BECOME diamagnetic. |
|
| 4. |
When freezing of a liquid takes place in a system |
|
Answer» may have `q gt 0 or q LT 0` DEPENDING on the liquid |
|
| 6. |
Which of the following orbitals are degenerate ? 3d_(xy), 4d_(xy) 3d_(z^(2)), 3d_(yz), 4d_(yz), 4d_(z^(2)) |
| Answer» Solution :Degenerate orbitals are the orbitals of the same subshell of the same main shell. HENCE, these are `(3d_(xy), 3d_(Z^(2)), 3d_(YZ)) and (4d_(xy), 4d_(yz), 4d_(z^(2))` | |
| 7. |
Which of the following conditions regarding a chemical process ensures its spontaneity at all temperatures? |
|
Answer» `Delta H GT 0, Delta H lt 0` |
|
| 8. |
Which of the following statements are correct?(i) Helium has the highest first ionisation enthalphy(ii)Chlorine has less electron affinity than fluorine(iii)Ne has more ionisation energy than Boron. (iv)The ionisation energy noble gases of zero |
|
Answer» (i) , (II) and (III) |
|
| 9. |
Which is the correct decreasing order of stability? (i) CH_(3) - overset(+)(C )H- CH_(3) (ii) CH_(3)- overset(+)(C )H- O - CH_(3) (iii) CH_(3)- overset(+)(C )H- CO- CH_(3) |
|
Answer» `(i) LT (ii) lt (iii)` |
|
| 10. |
Which gas is evolved when PbO_(2) is treated with conc. HNO_(3) ? |
|
Answer» `NO_(2)` |
|
| 11. |
Tri alkyl aluminium molecules exist as dimers which contains 3c-2e bonds. The corrdination number of bridged carbon is |
|
Answer» |
|
| 12. |
Which of the following sets of quantum numbers are followed ? |
|
Answer» N = 1,L = 0 and m = 0 .m. cannot be `pm 1 ` for l = 0 and .l. cannot be equal to .2. for n = 2 value of m = -1 _____ 0 _____ + 1 Values of l=0 ,1,2, ….. (n-1) |
|
| 14. |
Which of the following exists in enantiomeric pair |
|
Answer» n-butyl chloride |
|
| 15. |
Which of the following compounds gives benzoic acid on oxidation ? |
|
Answer» Chlorophenol |
|
| 17. |
The three elements A, B and C form three compounds AB, AC and BC. AB contains 75% of A, AC contains 57.14% of C while BC contains 11.11% of B. Prove that these results are in accordance to law of reciprocal proportions. |
|
Answer» Solution :In compound AB, the percentage of A is 75%. This means that 75 g of A combine with 100 - 75 = 25 g of B. THEREFORE, the amount of B that will combine with a fixed amount of A, SAY 100 g, would be `=25/75 xx 100 = 33.33 g` In compound AC, the amount of C is 57.14%. This means that 100 - 57.14 = 42.86 g of A combine with 57.14 g of C. Therefore, the amount of C that will combine with 100 g of A would be: `=(57.14)/(42.96) xx 100 = 133.3 g` `therefore` The RATIO in the amounts of B and C that combine with 100 g of A is: `33.33 : 133.3`, i.e. 1:4 .......(I) In compound BC, the amount of B is 11.11%. This IMPLIES that in this compound, 11.11 g of B combine with 100 - 11.11 = 88.89 g of C. The ratio in these amounts is `11.11 : 88.99,` i.e. 1:8.............(ii) The two ratios (i) and (ii) are related to each other as: `1/4 : 1/8,` i.e. `2:1` which is a simple whole number ratio. Thus, the amounts of B and C in compounds AB and AC bear a simple whole number ratio with the ratio of their amounts in compound BC. Hence, the data are in accordance to the law of reciprocal proportions and prove this law. |
|
| 18. |
The reagents used to convert (E)-2,3-dibromo-2-butene to 2-butyne |
|
Answer» ZN duct/D |
|
| 19. |
The weight percentages of carbon in carbon dioxide and carbon disulphide are respectively 27.27% and 15.79%. What is the composition of oxide of sulphur, if the weight percentage of oxygen is 50? |
|
Answer» Solution :Weight of OXYGEN that combines with 1G of Carbon `=(73.73)/(27.27)=2.67g` Weight of Sulphur that combines with 1g of carbon `=(84.21)/(15.79) =5.33g` Ratio of weights of S and O that can combine with same weight of carbon is 5.33 : 2.67 =2:1 The SIMPLE multiple ratio is 1 : 1 The oxide is sulphur DIOXIDE, `SO_(2)` |
|
| 20. |
When 100 ml of 0.4 M CH_3 COOHis mixed with 100 ml of 0.2 M NaOH , the [H_3O]^(+)in the solution is approximately :[K_a (CH_3COOH)= 1.8 xx 10 ^(-5) ] |
|
Answer» `1.8 xx 10 ^(-4) ` ` =1.8 xx 10 ^(-5)xx ( (100 xx 0.4- 100 xx 0.2))/( (100 xx 0.2 )) =1.8 xx 10 ^(_5)` |
|
| 21. |
Which of following statements is/are correct? |
|
Answer» In Reimer-Tiemann reaction, DICHLOROCARBENE INTERMEDIATE is produced. |
|
| 22. |
Which one of the following pairs of molecules will have permenant dipole moments for both members? |
|
Answer» `SiF_(4)` and `NO_(2)` |
|
| 23. |
Which temperature is used as a stanadard referance on a Pt-Ir bar to define meter ? |
|
Answer» `0 ""^(@)C` |
|
| 24. |
Why are ionic hydrides of only alkali metals and alkaline earth metals are known ? Give two examples. |
| Answer» Solution :Alkali metals and alkaline earth metals are most electropostive due to LOW IONIZATION energy or enthalpy. Therefore, they can form hydride, e.g., NAH, KH and `CaH_(2)` | |
| 25. |
Which would give isobutane on decarboxylation ? |
|
Answer» `(CH_3)_3C-COOH` 
|
|
| 26. |
When 100mL 0.2 MKOH is mixed with 100mL0.2 M HCl in a rigid adiaabatic container ,temperature of solution increase by t_(1)^(@)C while when 300mL0.1 M Koh is mixed with 3000mL 0.1 M HCl then increase in temperature is t_(2)^(@)C then which one is correct? (Assume density as well as specfic heat capacity o final solution are same.) |
|
Answer» `t_(1)=t_(2)` |
|
| 27. |
What is R_(f) ? |
|
Answer» Solution :`R_(f)` means RETARDATION factor. Its value in CHROMATOGRAPHY is ratio of DISTANCE moved by the substance and the solvent from base line. `R_(f) = ("Distance moved by the substance from base line (x)")/("Distance moved by the solvent from base line(y)")` |
|
| 28. |
Total number of electrons present in 1.7 g of ammonia is |
|
Answer» `6.022xx10^(23)` No. of moles of ammonia =`("MASS")/("Molar Mass")` `=(1.7g)/(17gmol^(-1))` 0.1 mol No. of molecules present in 0.1 mol of ammonis `=0.1xx6.022xx10^(23)=6.022xx10^(22)` No. of electrons present in 0.1 mol of ammonialtbrtgt`10xx6.022xx10^(22)-6.022xx10^(23)` |
|
| 29. |
Which is the isotope of dihydrogen ? |
|
Answer» `._1^1H` |
|
| 30. |
Thermal energy dominates over the molecular interaction energy in a liquid. Comment. |
| Answer» Solution :The GIVEN statement is FALSE. Both THERMAL energy and MOLECULAR interaction energy are at equilibrium in at liquid. Molecular association breaks, but they reassociate again. | |
| 31. |
The velocity of electronis 1%of velocityoflight . Calculatedebrobilewavelengthof electron. |
| Answer» SOLUTION :`2.43 XX 10^(10) m` | |
| 32. |
What would have happened if the greenhouse gases were totally missing in the earth's atmosphere ? Discuss. |
|
Answer» Solution :The most dense greenhouse gases on the earth are `CO_2, CH_4, O_3,` CFCs and water VAPOR. These gases are FOUND on the earth.s surface. The absorbed solar energy emits from the atmosphere of the planet and the result is that atmosphere BECOMES warm. Greenhouse gases are IMPORTANT to maintain the earth.s atmosphere. If there is no greenhouse gases, then the earth.s AVERAGE temprature decreases and makes it unfit for living so life on earth will not be possible. |
|
| 33. |
The steam distillation of ……..liquid is done and it is boiled at…….temperature |
| Answer» SOLUTION :ANILINE, below 373K | |
| 34. |
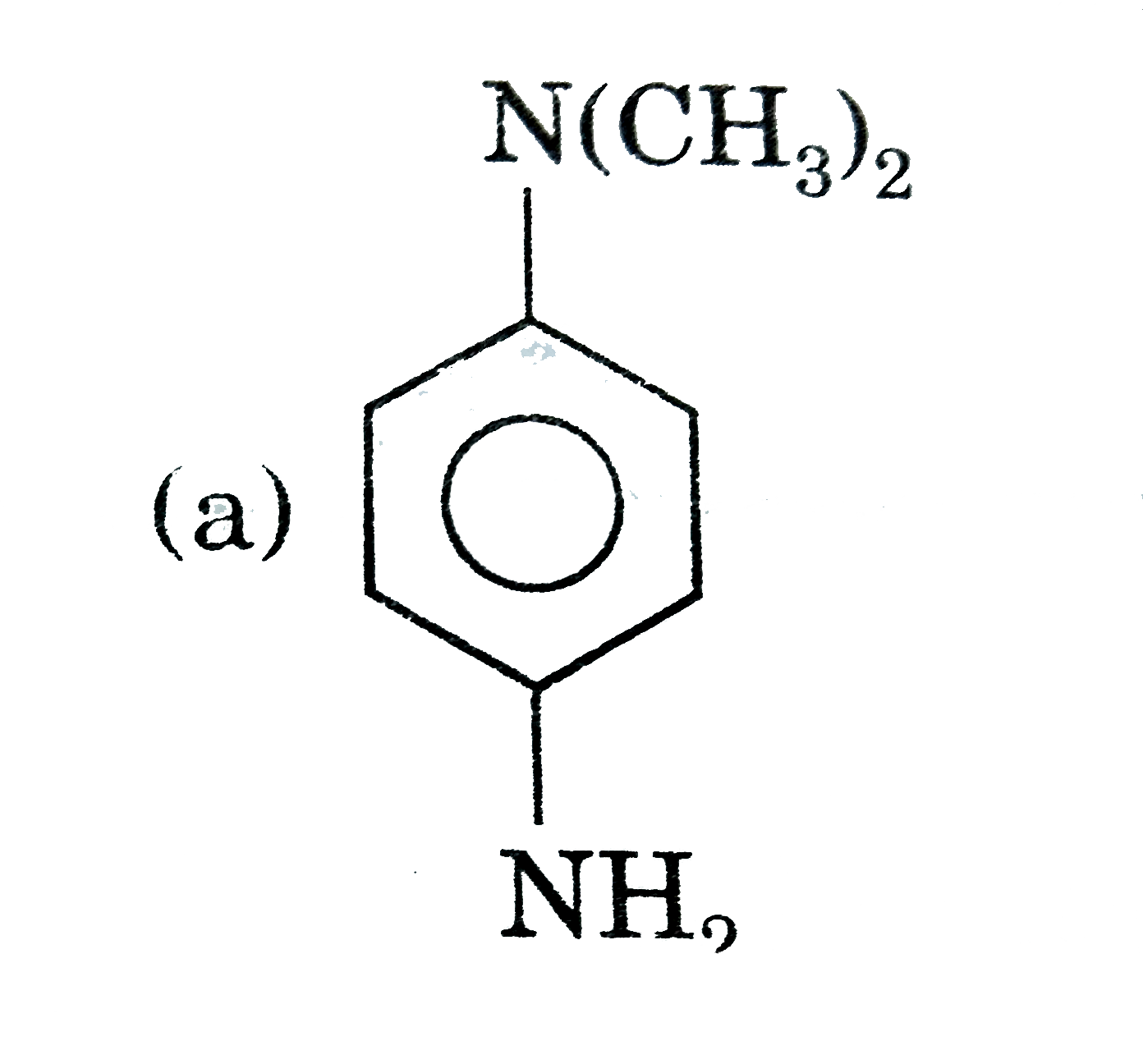
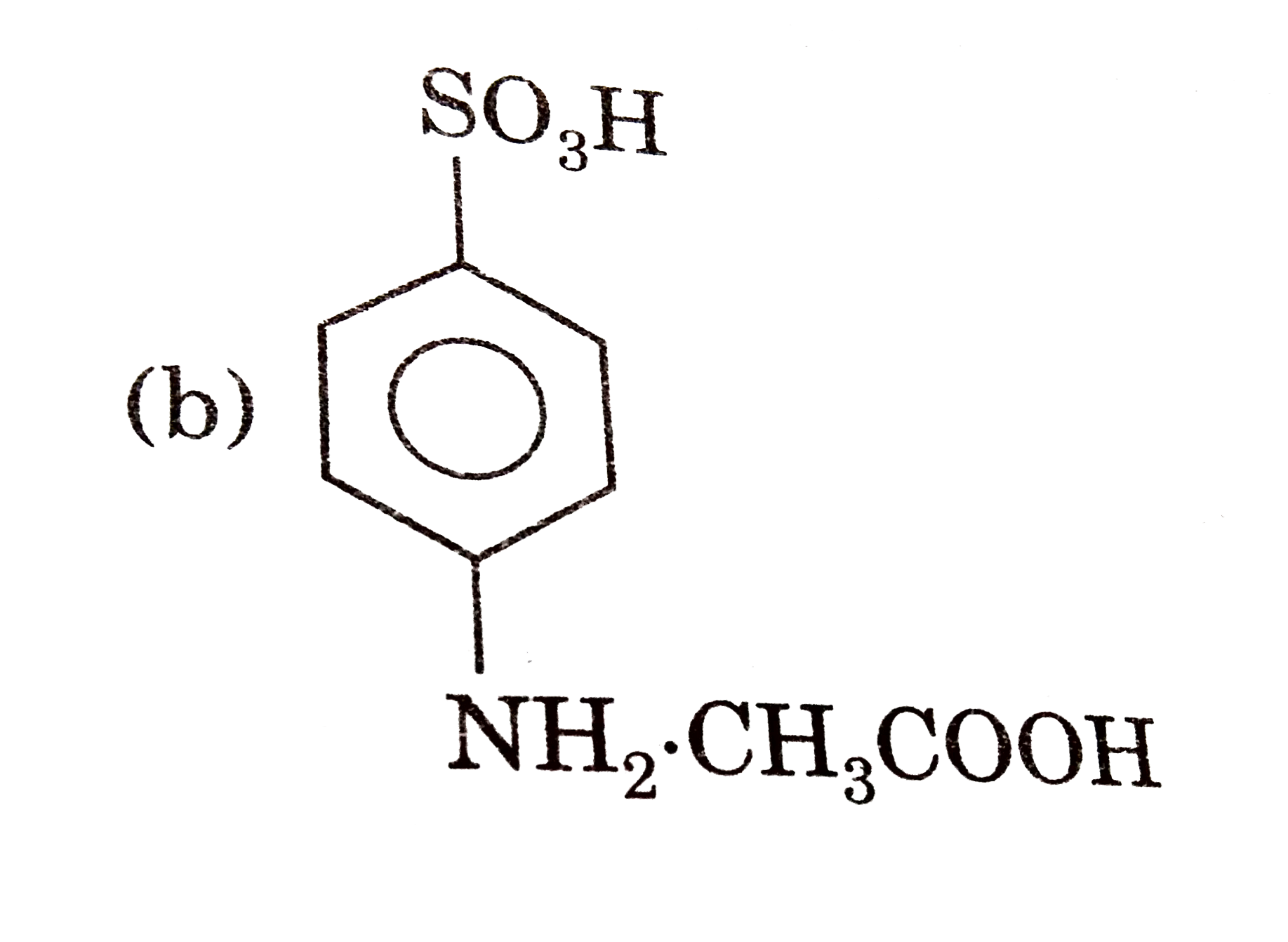
Which of the folloing contains maximum number of lone paris on the central atom ? |
|
Answer» `ClO_(3)^(-)` 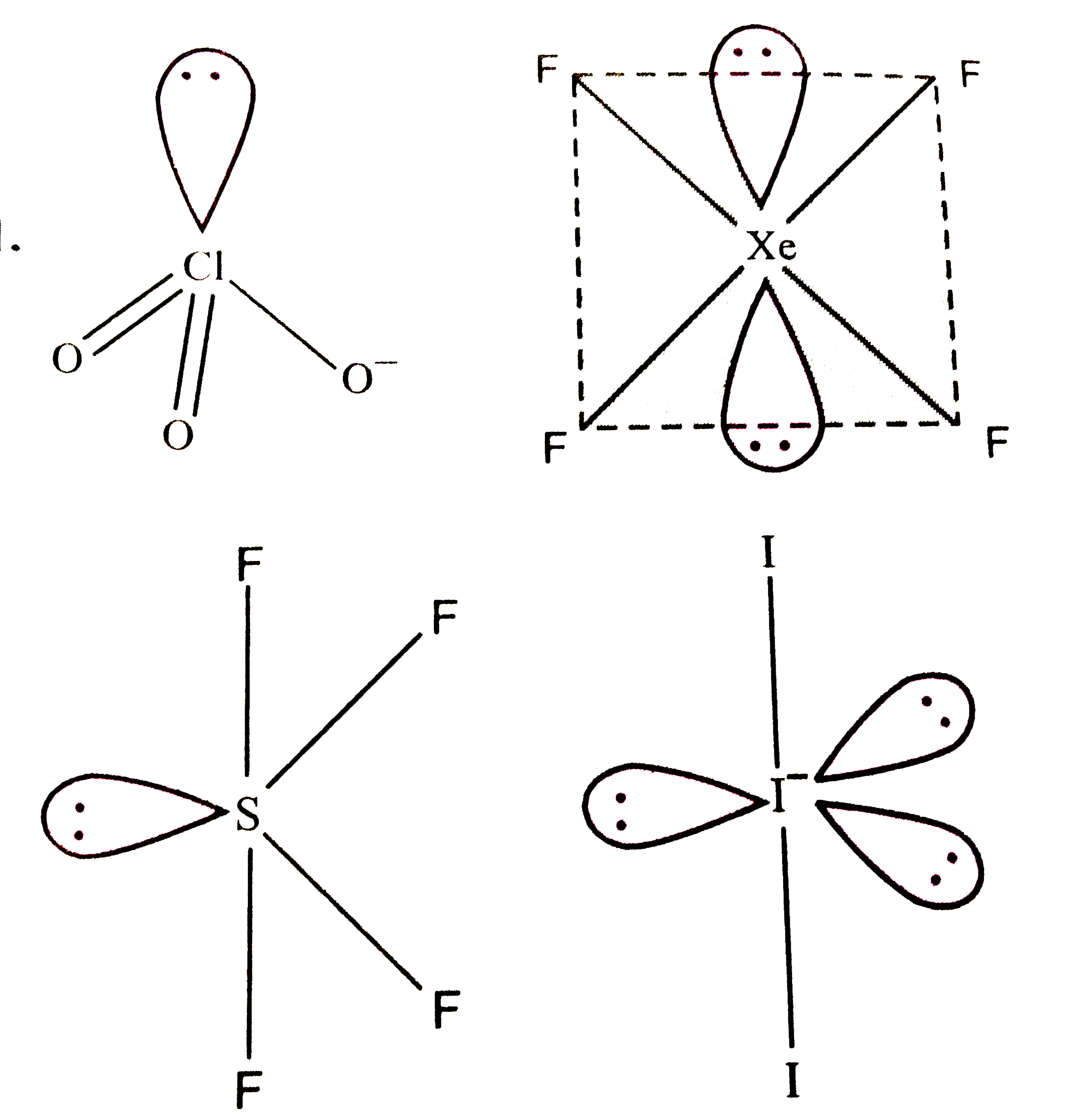
|
|
| 35. |
The tanks used by scuba divers are filled with air diluted with11.7%He , |
|
Answer» ` 56.2% N_2 and 32.1 % O_2 ` |
|
| 36. |
Which of the following compound is not named correctly ? |
|
Answer»
|
|
| 37. |
The reducing agent for conversion of O_(2)NCH_(2)CH_(2)CH=CH_(2) to H_(2)NCH_(2)CH_(2)CH=CH_(2) is : |
|
Answer» `LiAIH_(4)` |
|
| 38. |
Which of the following statement (s) is/are correct |
|
Answer» E2 isa concerted reaction in which bonds break and new bonds form at the same time in a single step |
|
| 39. |
Using the standard electrode potentials given in the table predict if the reaction between the following feasible. (a) Fe^(3+)(aq) andI^(-)(aq) (b) Ag^(+)(aq) andCu(s) (c )Fe^3+ (aq) and Cu(s) Ag(s) and Fe^(3+) (aq) (Br_(2))(aq) and Fe^(2+)(aq) (d) Ag(s) and Fe^(3+)(aq) (e) Br_(2)(aq) and fe^(2+)(aq) |
|
Answer» Solution :A particular REACTION can be feasible if e.m.f of the cell based on the `E^(@)` values is positve. KEEPING this in mind,let us predict the feasibility of the reactions. `(a) I^(-)(aq)+Fe^(3+)(aq)rarrFe^(2+)(aq)+1//2I_(2)(g)` `E^(@)_(cell)=0.77-0.54=0.23 V` `(b) Cu(s)+2AG^(+)(aq)rarrCu^(2+)(aq)rarrCu^(2+)(aq)+2Ag(s)` `E^(@)_(cell)=(0.80-0.34)=0.46 V` `(C )3Cu(s)+2Fe^(3+)(aq)rarr3Cu^(2+)(aq)+2Fe(s)` `E^(@)_(cell)=(0.77-0.34)=-0.43 V` `(d) 3Ag(s)+Fe^(3+)(aq)rarr3Ag(+)(aq)+Fe(s)` `E^(@)_(cell)=(0.77-0.80)=-0.03 V` `(e) Fe^(2)(aq)+Br_(2)(g)rarrFe^(3+)(aq)+2 Br^(-)(aq)` `E^(@)_(cell)=1.08-0.77=0.31 V` |
|
| 40. |
Which of the following relations regarding molecular velocities are true ? a) Most probable velocity = 0.8166 xx RMS velocity b) RMS velocity = 0.9213 xx Average velocity c) Average velocily = sqrt((8RT)/(piM)) d) C_p > bar(C) > C |
| Answer» ANSWER :D | |
| 41. |
Which one of the following alkali metals gives hydrated salts ? (a) Li (b) Na (c) K (d) Cs |
|
Answer» Solution :Smaller the size of an ion, the more highly is it hydrated. Among the given ALKALI metals, Li is the SMALLEST in size. Also, it has the highest charge density and highest polarising power. Hence, it attracts water MOLECULES more strongly than the other alkali metals. As a RESULT, it forms hydrated salts such as `LiCl_(2)`.The other alkali metals are larger than Li and have weaker charge DENSITIES. Hence, they usually do not form hydrated salts |
|
| 42. |
Which of the following reagents (s) is/are required to give water soluble dye stuff, methylene blue in strongly acidic solution with H_(2)S? |
| Answer» ANSWER :A::C | |
| 43. |
When Boyle's law is applicable to all gases reagardless of their chemical identity? |
|
Answer» HIGH PRESSURE |
|
| 44. |
Write the composition of gunpowder. |
| Answer» Solution :Gunpowder is an explosive mixture containing `KNO_3` ALONG with CHARCOAL and SULPHUR. | |
| 45. |
What is the SI unit for mass of substance ? |
|
Answer» MOLE |
|
| 46. |
Which of the substances can be used for drying neutral or basic gases ? |
|
Answer» CALCIUM carbonate |
|
| 47. |
The work function (phi) of some metals is listed below. The number of metals which will show photoelectric effect when light of 300nm wavelength falls on the metal. The Metal is |
|
Answer» `= (hc)/(lambda)n impliesE = (6.6xx10^(-34) x 10^(8))/(300xx0^(-9))` `E` in eV `=(6.6xx10^(-34) xx 3 xx 10^8)/( 300xx10^(-9) xx 1.6xx 10^(-19)) = 4.16 eV` So , number of metals showing photo-electric effects will be (4) i.e., Li, Na , K , MG |
|
| 48. |
The wave number of the radiation whose quantum is 1 erg is |
|
Answer» |
|
| 49. |
Which of the followng product can be obtained on reaction of benzene with chlorine in presence of anhydrous AlCl_(3) ? |
|
Answer» Chlorobenzene (B) Hexachlorocyclohexane `C_(6)H_(6)Cl_(6)` :  (C) Benzene hexachloride `C_(6)Cl_(6)` : 
|
|