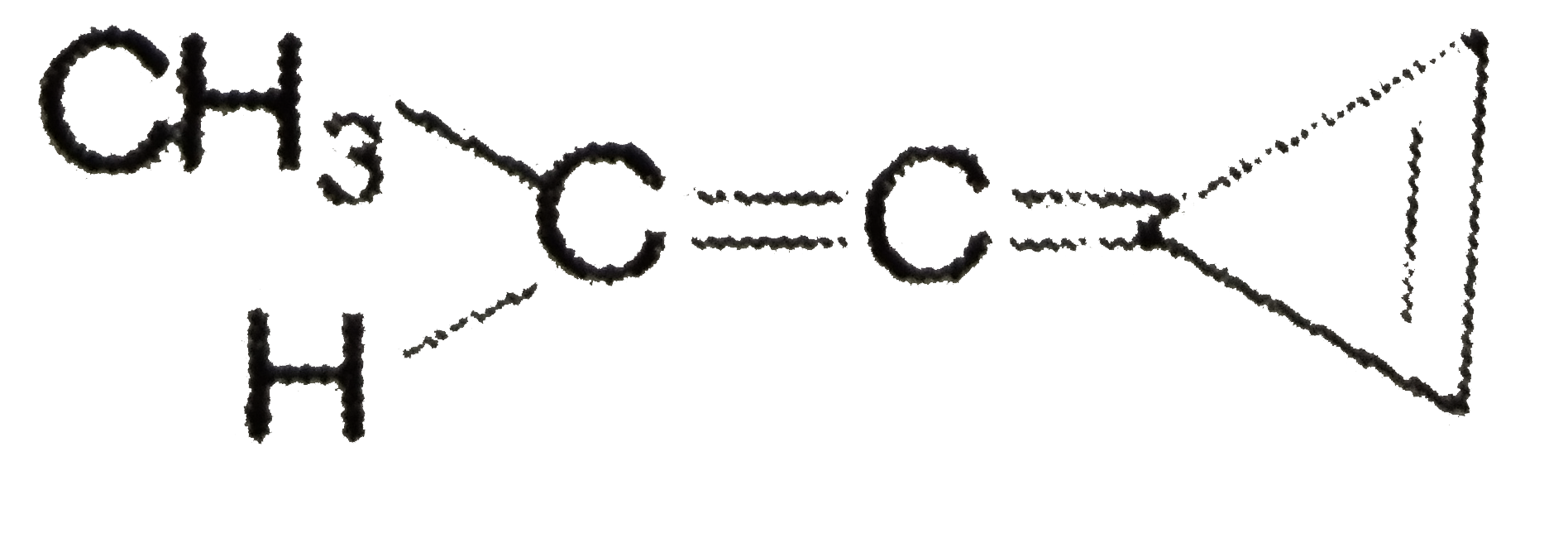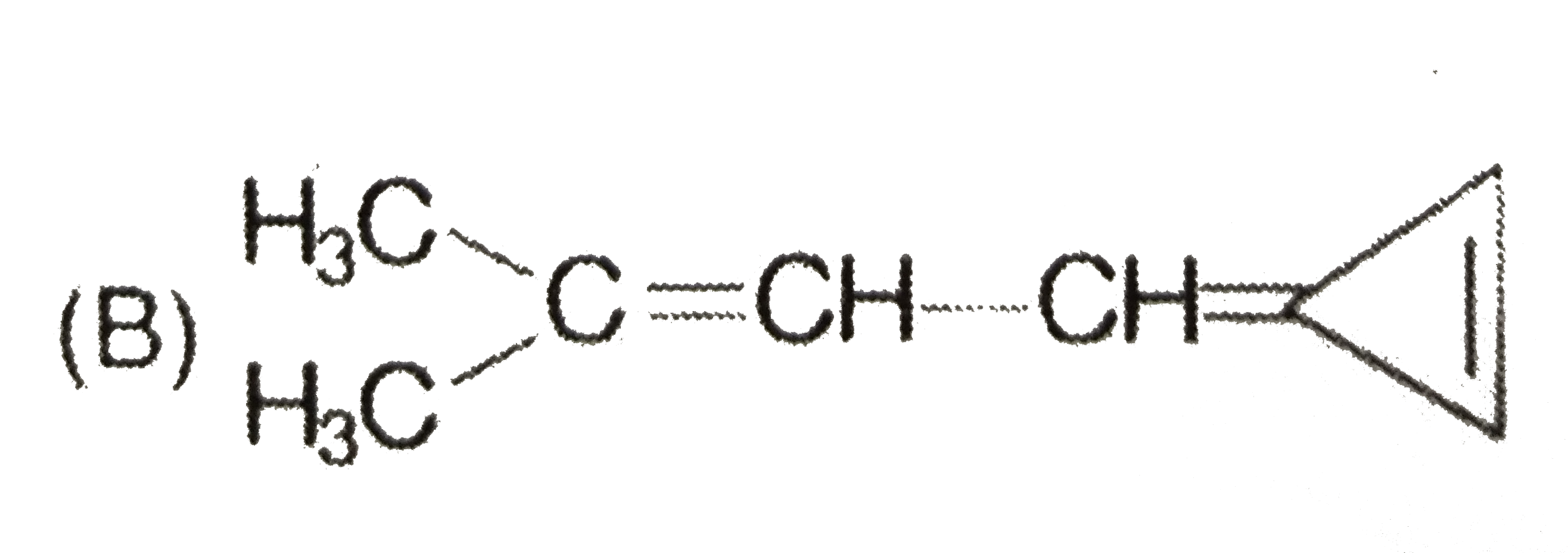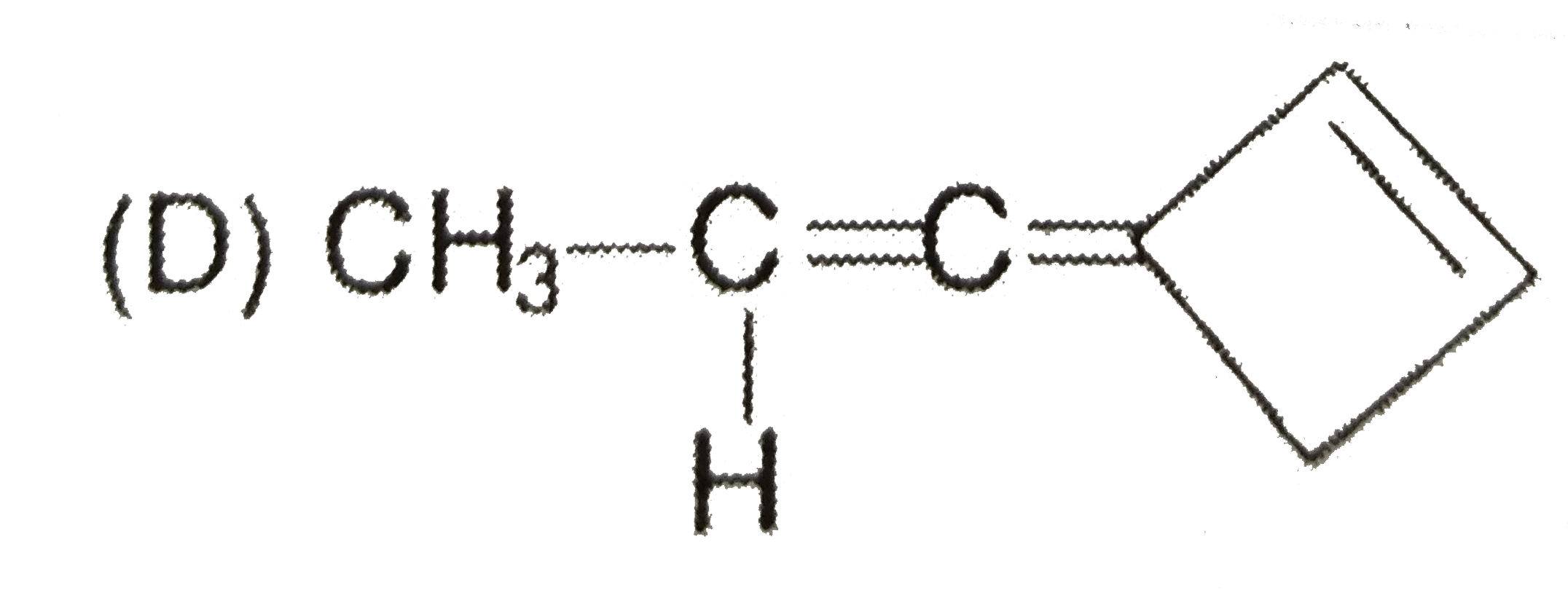Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
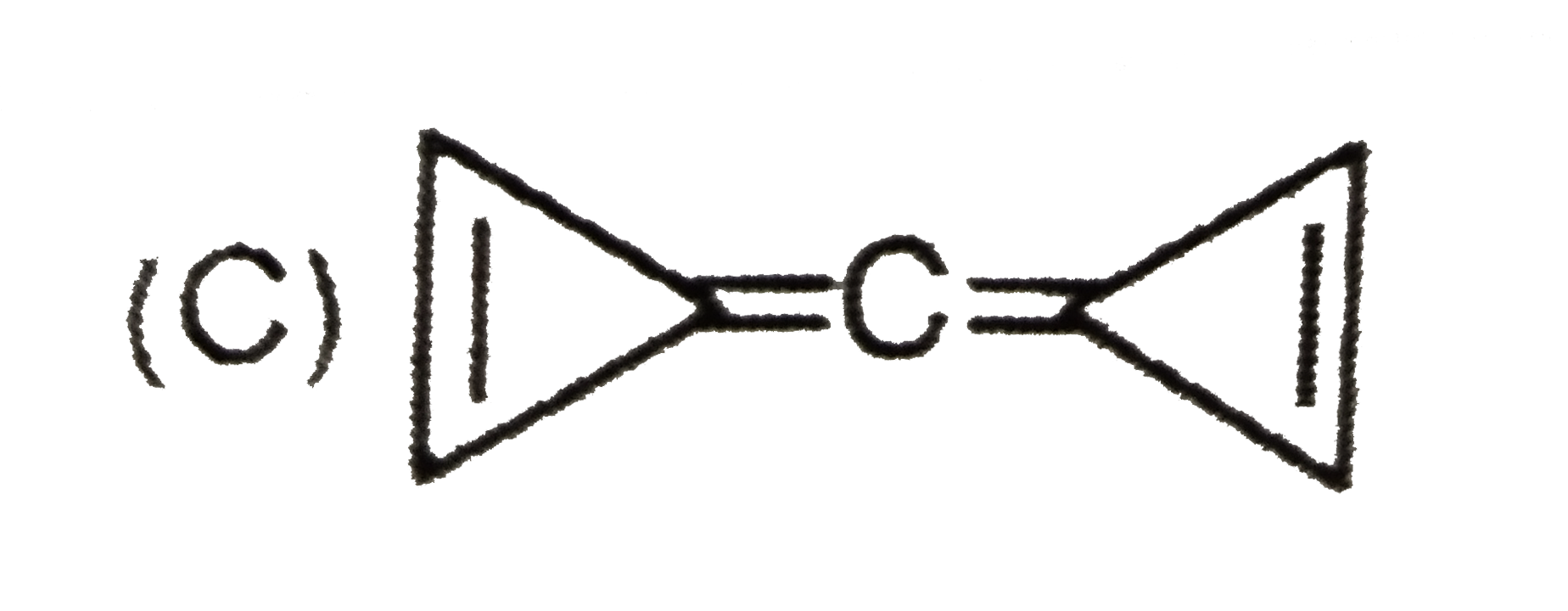
Which of the following compounds have the same type of hybridised carbon atoms? |
|
Answer» `CH_(2) = CH - CH= CH_(2)` |
|
| 2. |
Write the application of chromatography |
| Answer» Solution :(i) To PURIFY the compounds. (II) To separate mixture into their components (iii) To TEST the purify of compounds. | |
| 3. |
What is the empirical formula of Kaolin the percentage composition of which is as under : Al_(2)O_(3) = 39.5 %, SiO_(2)=46.6 %,H_(2)O=13.9 % (Atomic masses : Al = 27, Si= 28,O = 16,H=1) Hint : Radical mass of Al_(2)O_(3) = 102,SiO_(2) = 60,H_(2)O=18. |
|
Answer» `{:("Radical","Percentage","Radical mass","Radical ratio","SIMPLEST ratio","Simplest whole no. ratio"),("Al"_(2)"O"_(3),39.5,102,(39.5)/(102)=0.38,(0.38)/(0.38)=1.0,1),("SiO"_(2),46.6,60,(46.6)/(60)=0.78,(0.78)/(0.38)=2.05,2),("H"_(2)"O",13.9,18,(13.9)/(18)=0.77,(0.77)/(0.38)=2.03,2):}` Empirical formula of Kaolin `= Al_(2)O_(3).2SiO_(2).2H_(2)O`. |
|
| 4. |
Which of the following orders of properties is /are correct? |
|
Answer» `i)"Meltimng point of halides : "MF_(2)gtMCl_(2)gtMBr_(2)gtMI_(2)` |
|
| 5. |
Which will not result in a reaction? |
|
Answer» `C_6H_5OH + Br_2` |
|
| 6. |
Which of the following reactions give meso product? |
|
Answer»
B) CIS alkene `UNDERSET(OH^(-)) OVERSET("cold" KMnO_4)to` cis hydrogenation `to 1 ` cis addition is anti additionracemic mixture (CAR) cis syn addition-meso C) C) Trans alkene- epoxide formation with peroxidesepoxide on hydrolysis gives trans diols so anti addition Trans-anti addition (ATM) gives meso D) Cyclo hexen is cis alkene anti addition racemic so (D) is not the CORRECT answer |
|
| 7. |
Whichof the followingpairsof elements would you expectto havelowerfirst ionizationenthalpy ? Explain (i)CI or F (ii) CI or S(iii) K or Kr(iv) Kr or Xe |
|
Answer» (ii)Amongs andCIS haslower `Delta_(i) H_(1)` due tobiggersize andlowernuclearcharge (iii)K haslower `Delta_(i) H_(1)`than Ar hasconfiguration . (iv)Xe becauseof itsbigger sizeand stronger shieldingeffecthas lower `Delta_(i) H_(1)` than Kr. |
|
| 8. |
Which substance is used to stop radioactive rays ? |
|
Answer» SiC |
|
| 9. |
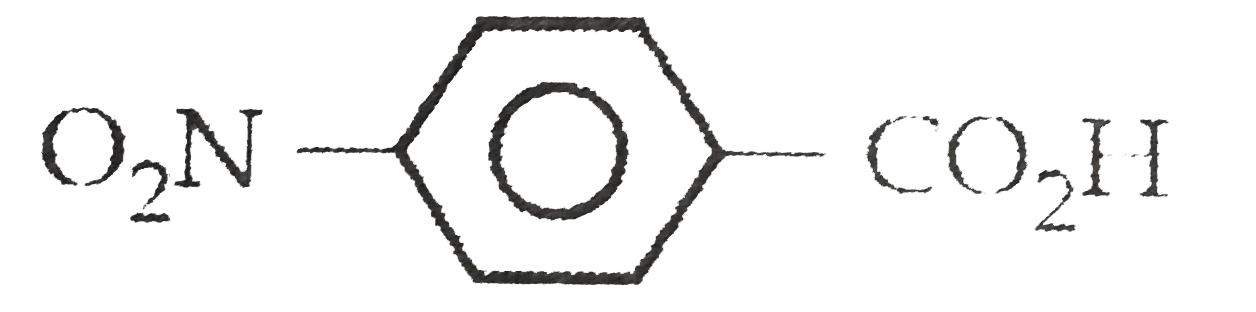
when benzyl chloride is boiled with aqueous KOH solution and the solution is acidified by a few drops of dilute HNO_(3) followed by AgNO_(3) solution, a white precipitate is formed. However, no such precipitate is noticed when the reaction is carried with chlorobenzene. explain. |
Answer» Solution :In case of `C_(6)H_(5)CH_(2)Cl`, the benzyl carbocation `(C_(6)H_(5) OVERSET(o+)(C)H_(2))` FORMED by releasing `Cl^(-)` ion is resonance stabilised. THEREFORE, the `C - Cl` bond can cleave to form `KCl`. HOWEVER, the phenyl carbocation `(C_(6)overset(o+)(H)_(5))` expected to be formed in case of chlorobenzene does not show any resonance. therefore, `C - Cl` bond does not cleave in case of chlorobenzene and no white precipitate is formed in this case. 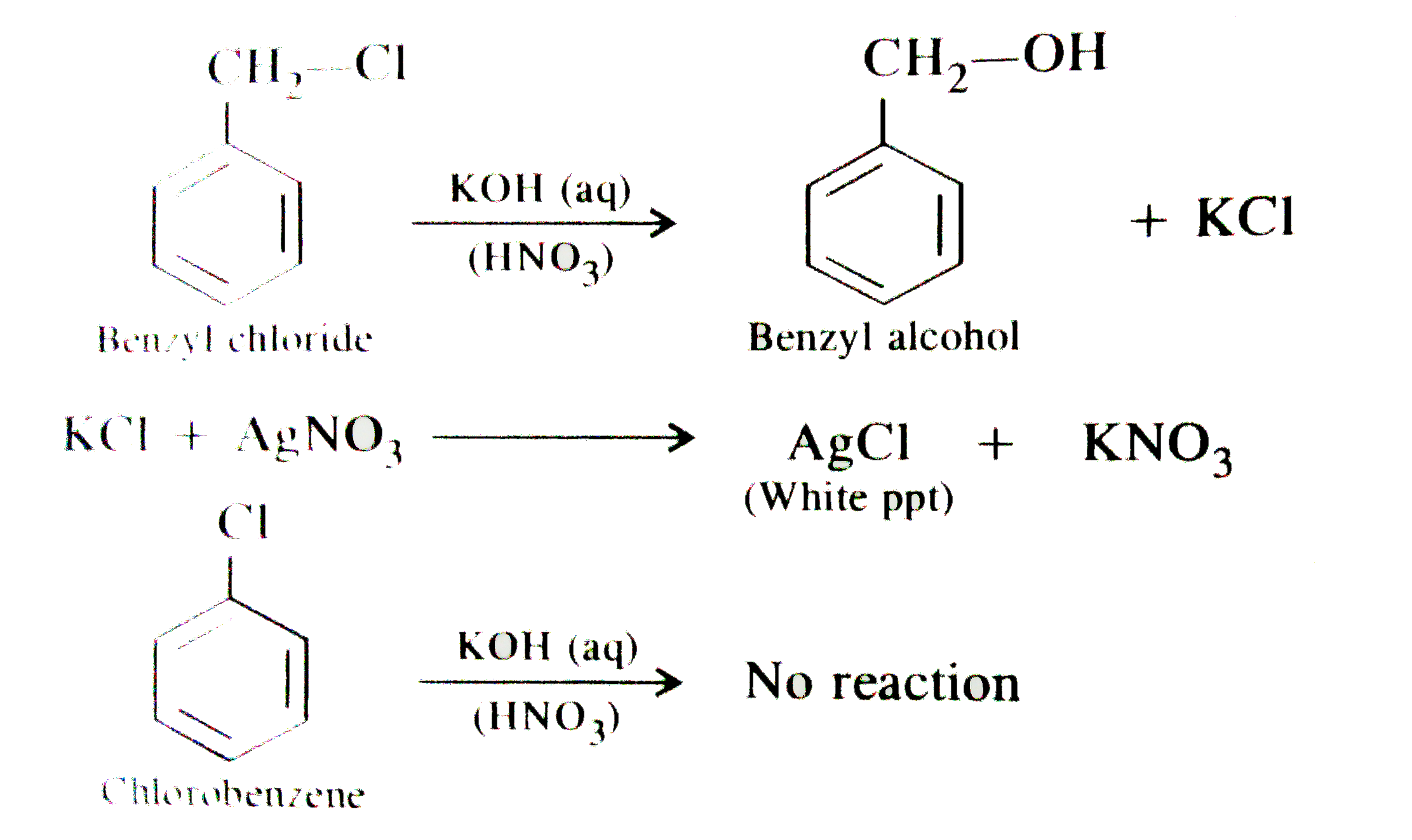
|
|
| 10. |
Which cycloalkane is almost considered to be a planar molecule |
|
Answer» CYCLOHEXANE |
|
| 11. |
(X) overset(O_(3)//Zn)underset(H_(2)O)toCO_(2)+CH_(3)-underset(O)underset(||)(C)-H+OHC -underset(O)underset(||)(C)-CHO X is |
|
Answer»
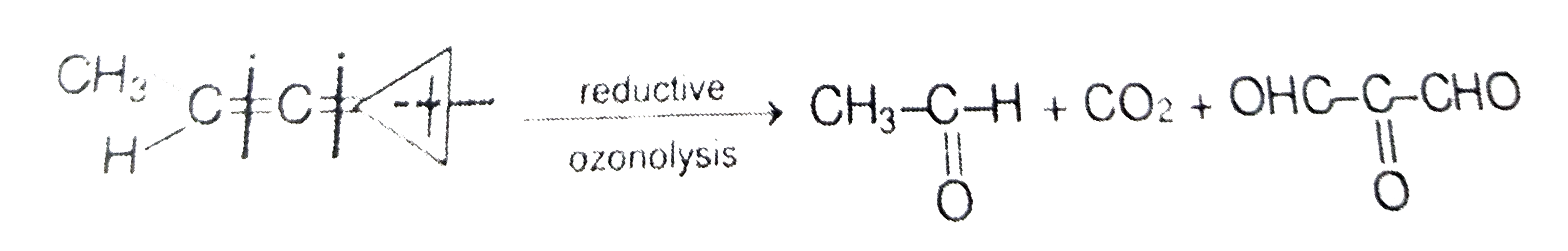
|
|
| 12. |
When benzene reacts with excess of chlorine in the presence of anhy. AlCl_3, the product(s) formed |
|
Answer»
|
|
| 13. |
Which of the following reagents will be able to distinguish between 1-butyne and 2-butyne ? |
|
Answer» `NaNH_2` |
|
| 14. |
Write the Lewis structure of hydrogen peroxide. |
Answer» SOLUTION :The LEWIS STRUCTURE of `H_2O_2` is 
|
|
| 15. |
Under the same conditions how many mL of 1M KOH and 0.5M H_(2)SO_(4) solutions, respctively when mixed for a total volume of 100mL produce the highest rise in temperature |
|
Answer» `67:33` |
|
| 16. |
Which of the following molecular type will have seesaw shape ? |
|
Answer» `AB_(2)E_(2)` |
|
| 18. |
When is a precipitate formed when solutions of BaCl_(2) and Na_(2)SO_(4) are mixed ? |
| Answer» SOLUTION :A PRECIPITATE is formed when in the final solution after mixing, the ionic product `[Ba^(2+)][SO_(4)^(2-)] GT K_(SP)` for `BaSO_(4)`. | |
| 19. |
What is shape of the orbital with (i) n = 2 and I = 0. |
| Answer» SOLUTION :`N = 2 and I = 0`. The ORBITAL is 2s. Its shape is symmetrically spherical. | |
| 20. |
Which of the following metals is most commonly used in photochemical cells? |
|
Answer» Lithium |
|
| 21. |
Which is the correct ascending order for the acidic strength of methane, ammonia, water and hydrogen fluoride ? |
|
Answer» `HF GT gt H_2O gt gt NH_3 gt gt CH_4` |
|
| 22. |
Which of the following statements is correct about the reaction given below ? 4 Fe_((s)) +3O_(2(g)) rarr 2Fe_(2)O_(3(g)) |
|
Answer» Total mass of iron and OXYGEN in reactants `=` total mass of iron and oxygen in product THEREFORE it FOLLOWS law of conservation of mass. The total mass of products `=` Total mass of reactants The Amount of `Fe_(2)O_(3)` is decided by limiting reagent. |
|
| 23. |
What is loam soil ? |
| Answer» Solution :The SOIL containing 34% air, 66% water ALONG with HUMUS is called loam soil It is best for the crops | |
| 24. |
The species : H_(2)O,HCO_(3)^(-),HSO_(4)^(-) and NH_(3) can act both as Bronsted acids andbases. For each case give the corresponding conjugate acid and base. |
|
Answer» Solution :CONJUGATE acids : `H_(3)O^(+), H_(2)CO_(3), H_(2)SO_(4), NH_(4)^(+)`. Conjugate bases : `OH^(-) , CO_(3)^(2-), SO_(4)^(2-), NH_(2)^(-)`. |
|
| 25. |
Which one of the following is an example of homogeneous catalysis? |
|
Answer» HABER process of synthesis of ammonia |
|
| 26. |
The salt of an alkali metal gives violet colour in the flame test . Its aqueous solution gives a white precipitate with barium chloride in hydrochloric acid medium . The salt is |
|
Answer» `K_(2)SO_(4)` |
|
| 27. |
Which of the following sulphate salt have least solubility in water? |
|
Answer» `MgSO_(4)` |
|
| 28. |
Which of the following is classified as Z and trans-isomer? |
|
Answer»
|
|
| 29. |
Which one of the following is in solid state at room temperature ? |
|
Answer» BROMINE |
|
| 30. |
Discuss the trend of the following (i) Thermal stability of carbonates of Group 2 elements. (ii) The solubility and the nature of oxides of Group 2 elements. |
|
Answer» `BaCO_3 GT SrCO_3 gt CaCO_3 gt MgCO_3` |
|
| 31. |
What is meant by isolated system? Give example. |
|
Answer» Solution :(i)A system which can exchange neither matter nor energy with its surroundings is CALLED an isolated system. (ii) Here boundary is sealed and INSULATED. (III) Hot WATER contained in a thermos FLASK, is an example for an isolated system. |
|
| 32. |
Which one of the following compounds is most acidic? |
| Answer» Solution : Inductive effect and resonance. | |
| 33. |
Write Lewis symbols for the followint atoms and ions: S and S^(2-): Al and Al^(3+), H and H^(-) |
Answer» SOLUTION :
|
|
| 34. |
We are consuminga lot of electrical energy, solar energy etc. Do you, therefore, conclude that energy of the universe is continuously decreasing ?Explain. Which other thermodynamic quantity is continously increasing or decreasing ? |
| Answer» Solution :No, energy of the universe remains constant( LAW of CONSERVATION of energy ) . ENTROPY of the universe iscontinuously increasing. | |
| 35. |
Which of the following ions can cause coagulation of proteins? |
|
Answer» `Ag^(+)` |
|
| 36. |
Very pure N_(2) can be obtained by |
|
Answer» thermal DECOMPOSITION of ammonium dichromate `underset("Sod.azide")(2NaN_(3))to2Na+N_(2)` |
|
| 38. |
The substance not likely to contain CaCO_(3) is |
|
Answer» DOLOMITE |
|
| 39. |
When the bond not form between two atom as per VB theory ? Give e.g. |
|
Answer» Solution :If the atomic orbitals are not overlap an internuclear AXES than bond will not from. s - p or `p_(x) - p_(y)` ZERO OVERLAPPING 
|
|
| 40. |
Whichof thethefollowingis anexampleofeliminationreaction ? |
|
Answer» CHLORINATIONOF METHANE |
|
| 41. |
One mole of an ideal monoatomic gas at temperature T and volume 1L expands to 2L against a constant external pressure of one atm under adiabatic conditions, then final temperature of gas will be: |
|
Answer» <P>`(T)/(2^(2//3))` `Delta E= - P_("EXT") Delta V` `nC_(V) Delta T = -P_("ext") (V_(2) - V_(1))` ` 1 xx (3)/(2) R xx (T_(2) - T_(1)) = -1(2-1) = -1` `T_(2) = T- (2)/(3R)` |
|
| 42. |
Which of the following reagents is useful to separate benzoic acid from phenol ? |
|
Answer» DILUTE HCl |
|
| 43. |
Which of the following statements are correct about CO_(3)^(2-) ? |
|
Answer» Thehybridisation of central atomis `sp^(3)` Resonance structure has both C-O bond of equallength . Hence , (b) is wrong. formal charge on each O-atom = `(" TOTAL charge ")/(" No, of O-atoms " ) = (-2)/(3) = -0.67 ` units All C-O bond lengths are equal as mentioned above . |
|
| 44. |
Using VSEPR thory, drawthe molecular structures of OSF_(4) and XeF_(4)indicating the lacation of lone pair(s) of electrons and hybridisation of central atoms . |
Answer» SOLUTION :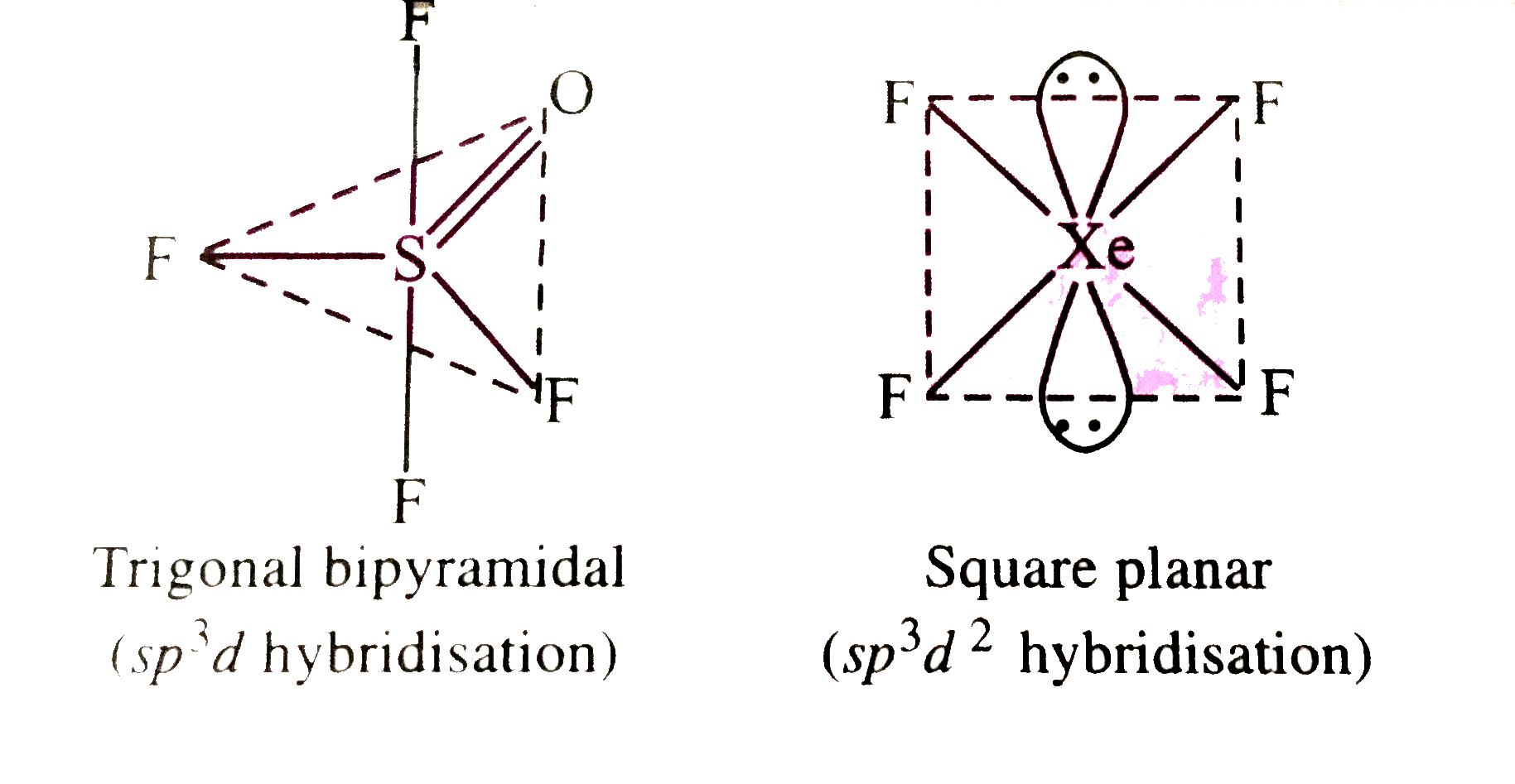
|
|
| 45. |
Write the expression for Delta U under an adiabatic process. |
| Answer» SOLUTION :`D U = Q + W, DU = W`. | |
| 46. |
The volume (in mL) of 0.1 M AgNO_(3) required to completely precipitat the chloride ions present in 30 mL of 0.01 M of [Cr(H_(2)O)_(5)Cl]Cl_(2), as silver chloride is close to : |
|
Answer» `(M_(1)V_(1))/(n_(1))=(M_(2)V_(2))/(n_(2))` `(0.1xxV)/(2)=(0.01xx30)/(1)` V=6 mL |
|
| 47. |
What could be the harmful effects of improper management of industrial and domestic solid waste in a city? |
| Answer» Solution : If domestic waste in a city is not PROPERLY managed, it may find its way into sewers or may be eaten up by the CATTLE. The non-biodegradable waste like POLYTHENE bags, metal scrap etc. choke the sewers. The polythene bags if swallowed by the cattle can result into their death. Similarly, if industrial waste is not properly managed, it will cause pollution of the AIR, SOIL and water | |
| 48. |
What is diagonal relationship? |
| Answer» SOLUTION :lithium and beryllium is more SIMILAR with the second element of the following group i.e., MAGNESIUM and aluminium, RESPECTIVELY. This sort of similarity is commonly referred to as diagonal relationship in the PERIODIC properties. | |
| 49. |
Explain amphoteric nature of water when it react with NH_3 and H_2S ? |
| Answer» | |
| 50. |
What is meant by EIA? |
|
Answer» Solution :In this system, HYDROGEN ATOM OSCILLATES between TWO directly linked POLYVALENT atoms. e.g., 
|
|