Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is added as stabilizer to aqueous solution of H_2O_2 ? |
|
Answer» PHOSPHORIC acid |
|
| 2. |
What is the coordination number ( C.N.)of an atom in (i)primitive cubic unit cell (ii) body- centred cubic unit cell (iii)face-centredcubic unit cell. |
|
Answer» Solution : (i) In a primitive cubic unit cell, each ATOM has 6 EQUALLY spaced NEAREST nighbours.Hence, C.N. = 6. (ii)In BCC,an atom at the body centre is surrounded by 8 nearest neighbours situted at the corners of the cube. Hence , C.N. = 8 ( REFER to fig 1.12) (iii) In FCC, an atom is surrounded by 12 nearest neighbours ( 6 in its own, plane, 3 above and 3 below ). Hence C.N. =12 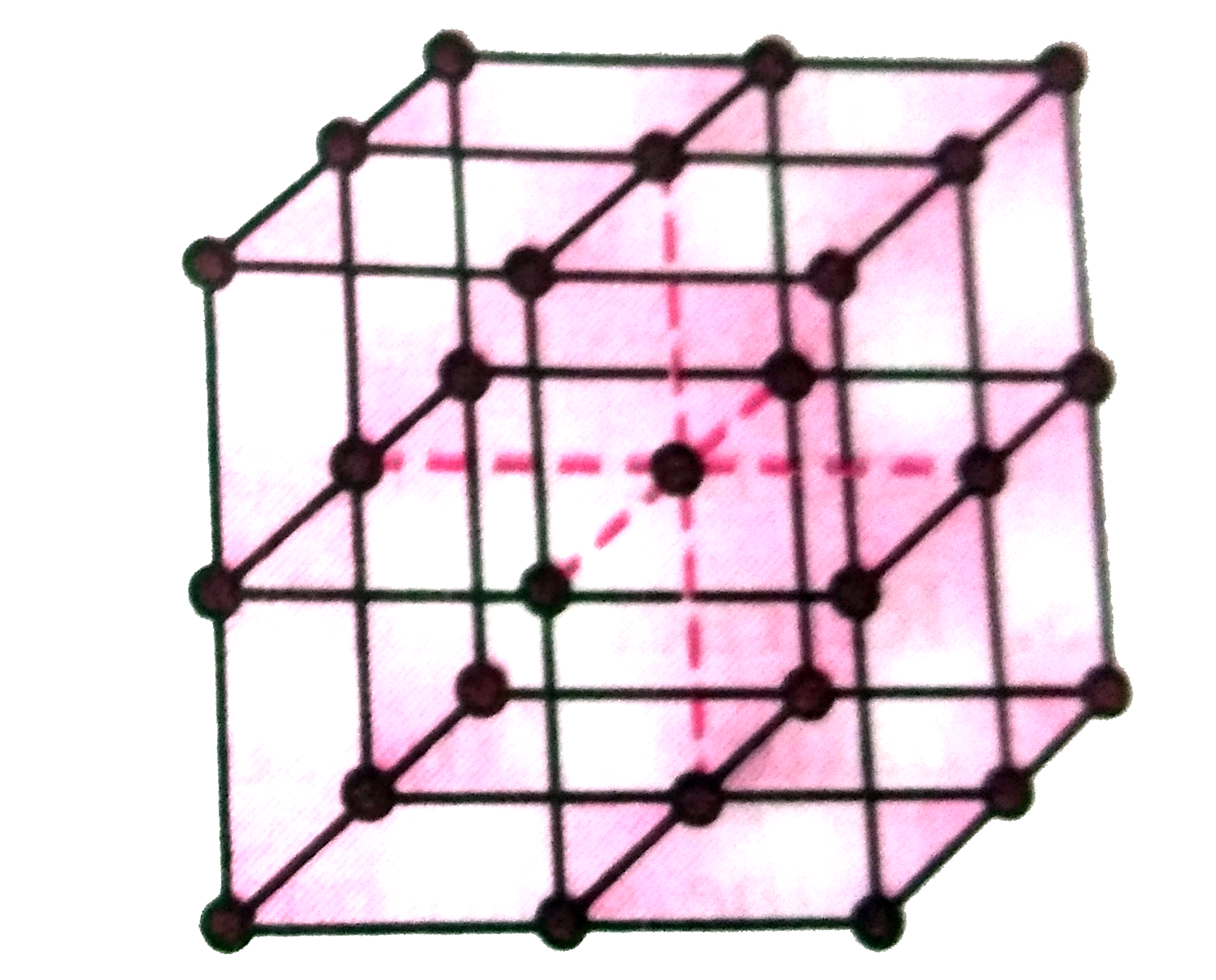
|
|
| 4. |
When one mole of A is reacted completely in an ice calorimeter at 0^(@)C and at 1 atm, it is found that the volume of equlibrium mixture of ice and water decreased by 0.25 Ml. The Delta _(r)H_(273) "for the reaction" 2Ararr B +2C is : (the densities of water and icfe at 0^(@) and 1 atm are 1.00 and 0.96 gm//cm^(3), respectively and latent hete of fusion of ice at 0^(@) C and 1 atm is 80cal//gm) [Use:1ifDelta_(r)H_(273 is+ ve and 2if Delta _(r)H_(273 is -ve and answer as a abc or 2abc, where abc is magnitude ofDelta _(r)H_273] |
|
Answer» |
|
| 5. |
The volume of gases H_(2),CH_(4),CO_(2) and NH_(3) absorbed by 1 gm of activated charcoal at 298 K are in the order: |
|
Answer» `H_(2) gt CH_(4) gt CO_(2) gt NH_(3)` `NH_(3) gt CO_(2) gt CH_(4) gt H_(2)` |
|
| 6. |
What combination of acyl chloride or acid anhydride and arene are choosen to prepare the following compounds ? |
|
Answer» |
|
| 7. |
Which of the following compounds is not formed in the pyrolysis of Hexane |
|
Answer» `C_(6)H_(12)` |
|
| 8. |
Which one is the correct order of acidity? |
|
Answer» `CH_2 = CH_2 GT CH_3 - CH = CH_2 gt CH_3 - C -= CH gt CH -= CH` |
|
| 9. |
Which of the following have identical bond order ? |
|
Answer» `CN^(-)` `CN^(-)`(NUMBER of electrons= 6 + 7 + 1 = 14) `NO^(+)`(number of electrons= 7 + 8 - 1 = 14 ) `O_(2)^(-) `(number of electrons= 8 + 8 + 1 = 17) `O_(2)^(2-)` (number of electrons = 8 + 8 + 2 = 18 ) so, `CN^(-) and NO^(+)` because of the presence of same number of electrons, have same bond order. |
|
| 10. |
Which of the following pairs of compounds are not postional isomers ? |
|
Answer»
|
|
| 11. |
What will be the total no. of isomers of an acylic saturated hydrocarbon having molecular mass 72g/mol ? |
|
Answer» 3 |
|
| 12. |
Which one of the following is the correrct statement ? |
|
Answer» `B_(2)H_(6).2 NH_(3)` is KNOWN as 'inorganic benzen' |
|
| 13. |
Which of the following reaction occurs during Lewis acid-base reaction ? |
|
Answer» EXCHANGE of PROTON |
|
| 14. |
Which one of the following statements is correct? |
|
Answer» ionic RADIUS of `Fe^(3+)` is greater than `Fe^(2+)` |
|
| 16. |
Which of the following will have nearly equal pH ? |
|
Answer» 100 ml 0.1 M HCl mixed with 50 ml water or `[HCl]=(1)/(15) M :. [H^(+)]=(1)/(15)M` (b) 50 ml of 0.1 M `H_(2)SO_(4) = 100 ml "of ? " MH_(2)SO_(4)` or `[H_(2)SO_(4)]=(1)/(20)M :. [H^(+)]=(1)/(10)M` (c) 50 ml of 0.1 `M H_(2)SO_(4) = 150 ml "of ?" M H_(2)SO_(4)` or `[H_(2)SO_(4)]=(1)/(30)M :. [H^(+)]=(1)/(15)M` (d) 50 ml of 0.1 M HCl = 100 ml of ? M HCl or `[HCl]=(1)/(20)M :. [H^(+)]=(1)/(20)M` (a) and (c) have same `[H^(+)]` and HENCE same pH. |
|
| 17. |
Which among the following compound will give maximum enol content in solution : |
|
Answer» `C_(6)H_(6)-overset(O)overset(||)C-CH_(2)-overset(O)overset(||)C-CH_(3)` |
|
| 18. |
what is the usual definition of entropy ? What is the unit of entropy ? |
|
Answer» Solution :(i) Entropy is a thermodynamic state function that is a measure of the RANDOMNESS or disorderliness of the system . (ii) For a reversible CHANGE TAKING PLACE at a constant temperature (T), the change in entropy (`DeltaS`) of the system is equal to heat energy absorbed or evolved (q) by the system divided by the constant temperature (T) `DeltaS_"SYS"=q_"rev"/T` (iii) SIunit of entropy is `JK^(-1)` |
|
| 19. |
Which of the following reactions involve both oxidation and reduction ? |
|
Answer» `NaBr+HClrarrNaCl + HBr` |
|
| 20. |
Which of the two structures (A) and (B) given below is more stabilised by resonance ? Explain. underset((A))(CH_(3)COOH) and underset((B))(CH_(3)COoverset(Θ)(O)) |
Answer» Solution :Resonance STRUCTURES of compound (A) and (B) are given below : 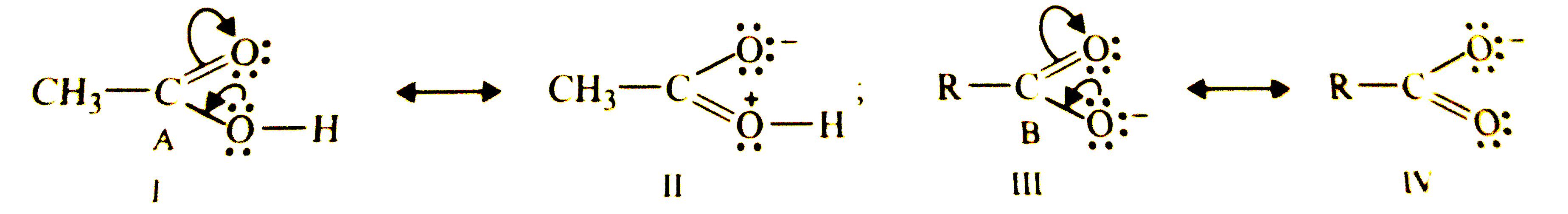 Resonance structure (II) carries separation of +ve and -ve charges and hence it is less stable than structure (I). AS a result, contribution of structure (II) towards resonance HYBRID of compound A is less than that of structure (I). In CONTRAST, both the resonance of compound (B), i.e., III and IV are of equal energy and hence contribute EQUALLY towards the resonance hybrid of structure (B). THUS, structure (B) is more stable than structure (A). |
|
| 21. |
Which is not a property of H_2O_2 |
|
Answer» Conc. `H_2O_2` SOLUTION is acidic in nature |
|
| 22. |
The standard free energy change DeltaG^(@) is related to equilibrium constant K_(p) as |
|
Answer» <P>`K_(p) = e^(-DELTAG^(@))/(RT)` |
|
| 23. |
Which one are not correct about CH_(2)="CC"l_(2)? |
|
Answer» Both carbon are in +2 OXIDATION state |
|
| 24. |
Which of the following is acidic and why : SiO_(2) , AI_(2) , PbO_(2), SiO_(2)? |
| Answer» SOLUTION :`SiO_(3)` is an ACIDIC oxide because Si is a non - METAL. | |
| 25. |
Which is a Lewis acid of the following ? |
|
Answer» `NH_3` |
|
| 26. |
Which of the statement is wrong among the following:- |
|
Answer» Haber's PROCESS of `NH_(3)` REQUIRES IRON as catalyst |
|
| 28. |
The transport of oxygen by hemoglobin in our body as an illustration for a ___________ change. |
|
Answer» REVERSIBLE |
|
| 29. |
When heated in air , the alkali metals form various oxides . Mention the oxides formed by Li , Na and K . |
|
Answer» Solution :The reactivity of ALKALI metals towards oxygen increases down the group as the atomic size increases . Thus , Li forms only lithium oxide `(Li_(2)O)` , sodium forms mainly sodium peroxide `(Na_(2)O_(2))` along with a small amount of sodium oxide while potassium forms only potassium superoxide `(KO_(2))` `4 Li + O_(2) overset(Delta) (to) 2 Li_(2) O , 2 Na + O_(2) overset(Delta)(to) underset("(Major)") (Na_(2)O_(2)) + underset("(Minor)") (Na_(2)O)` `K + O_(2) overset(Delta)(to) KO_(2)` This is because of the following two reasons : (i) As the size of the METAL cations increases , the POSITIVE field around it becomes weaker and weaker therebypermitting the intially formed oxide `(O^(2-))` ion to combine with another oxygen atom to form FIRST peroxide ion `(O_(2)^(-))` and then superoxide `(O_(2)^(-))` ion . `underset("Oxide") (O^(2-)) overset(1//2 O_(2)) (to) underset("Peroxide")(to) (O_(2)^(2-)) overset(O_(2))(to) underset("Superoxide")(2O_(2)^(-))` (II) Since larger cations stabilize larger anions due to higher lattice energies , therefore , the stability increases from oxide `to` peroxide`to` superoxide as the size of the metal cation increases down the group and the size of the anion increases from oxide `to` peroxide `to` superoxide . |
|
| 30. |
What will be the unit of R if the unit of pressure and volume are dyne cm^(-2) and cm^(3) respectively ? |
|
Answer» BAR. lit `k^(-1)MOL^(-1)` |
|
| 31. |
Westrosol overset(90% H_(2)SO_(4))rarr X overset(NH_(3))rarr Y Here, Y is |
|
Answer» ACRYLIC acid `H_(2) underset(Cl)underset(|)(C) - COOH underset(_HCl)overset(NH_(3))rarr H_(2)N - underset("Glycine")(CH_(2)) - COOH` |
|
| 32. |
The value of compression factor , Z for critical constant is |
|
Answer» `(1)/(2)` `(p_(c)V_(c))/(RT_(c))=3/8` |
|
| 33. |
Which of the following compounds will give racemic mixture on nucleophilie substitution by OH ion? (i) CH_3 - underset(C_2H_5)underset(|)(CH) - CH_2Br(ii) H_3C - underset(Br) underset(|)overset(CH_3)overset(|)C-C_2H_6(iii) CH_3 - underset(Cl)underset(|)overset(H)overset(|)C-C_2H_5 |
|
Answer» (i) |
|
| 34. |
What is hydrolith ? How is it prepared ? |
| Answer» Solution :`CA + H_(2) OVERSET(DELTA)to CaH_(2)` (HYDROLITH). | |
| 35. |
What would happen to a reversible dissociation reaction at equilibrium when an inert gas is added while the pressure remains unchanged ? |
|
Answer» LESS of the PRODUCT will be formed |
|
| 36. |
When atomic orbitals overlap head-on , the bond formed is ………..Whereas when they overlap laterally , the bond formed is ………. |
| Answer» SOLUTION :` sigma-bond,pi-bond` | |
| 37. |
What is the correct order of reactivity of the following alkylhalide towards Friedel - Craft's alkylation with benzene in the presence of AlCl_3 ? |
|
Answer» `CH_3 - F GT CH_3 - CL gt CH_3 - Br gt CH_3 -I` `CH_3F gt CH_3Cl gt CH_3 - Br gt CH_3 - I` |
|
| 38. |
What is the other name for photochemical smog? |
|
Answer» LONDON smog |
|
| 39. |
Which of the following is observed in ethylene nw lecule? |
|
Answer» ELECTROMERIC EFFECT |
|
| 40. |
What is the oxidation state of K in KO_(2) ? |
| Answer» Solution :The superoxide species is REPRESENTED as `O_(2)^(-)`, since the compound is neutral, therefore, the oxidation state of POTASSIUM is +1. | |
| 41. |
The volume occupied by O_2gas at STP which is obtained by the decomposition of 17g of H_2O_2is |
|
Answer» 5.6 LIT |
|
| 42. |
Which of the following functional groups passes on electron towards the phenyl ring ? |
|
Answer» `-SO_(3)H` |
|
| 43. |
The value of n in the following equation if balanced Cr_(2)O_(7)^(3+)+14H^(+)+nFe^(2+) to 2Cr^(3+)+nFe^(3+)+7H_(2)O |
|
Answer» 2 |
|
| 44. |
Write Lewis structure of O_(2)^(-)ion and find out oxidation state of each oxygen atom? What is the average oxidation state of oxygen in this ion? |
| Answer» Solution :The Lewis STRUCTURE of `O_(2)^(-)` is`:underset(..)overset(..)O-overset(..)O overset("ϴ"):`. Oxygen atom having NOCHARGE has 6 electrons so its oxidation number is zero, but the other oxygen atom CARRYING -1 charge has 7 electronsso its oxidation number is -1. Hence, the average oxidation state of oxygen is `-(1)/(2)`. | |
| 45. |
Write the bond line structure of 2-bromobutane. |
Answer» SOLUTION :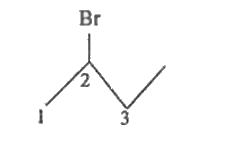
|
|
| 46. |
Under what conditions do we use fractional distillation ? |
| Answer» SOLUTION :When the volatile MISCIBLE liquids differ in the b.p by less than `25^(@)C`. | |
| 47. |
Which of the following effects can explain the stability of tertiary butyl carbonium ion? |
|
Answer» Electromeric |
|
| 48. |
What is important of troposphere in our life ? |
| Answer» Solution :TROPOSPHERE which is HELD to the surface of the earth by gravitational force. The thin layer of atmosphere is VITAL to our LIFE. It shields us from harmful radiations. | |
| 49. |
When 0.684 g of sucrose (Molar mass =342) is burnt in a constant pressure calorimeter, the temperature rises by 5.6 K./ If enthalpy of combustion of sucrose is -5600 Kj/mole then heat capacity of the calorimeter system is : |
|
Answer» 200J/K |
|



