Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The solubility of Ba_3(AsO_4)_2(formula weight=690 )is 6.9 xx 10^(-2)g//100 ml. what is the K_(sp)? |
|
Answer» `1.08 xx 10^(-11)` ` =(6.9xx10 ^(-2))/( 690 )mol //100 ML ` ` = 10 ^(-4)" mole " //100ml ` `therefore 100ml -10 ^(-4)"mole " ` ` 100 ml -10 ^(-4)" mole " ` `rArr 10 ^(-3)" moles" rArr K_(Sp) =108 S^(5) ` |
|
| 2. |
Which of the following hydrocarbons is not formed when Wurtzreaction takes place between ethyl iodide and propyl iodide |
|
Answer» BUTANE |
|
| 3. |
Which of the followin compound(s) on treatment with HCl forms a stable salt which can be separated and analyzed using AgNO_(3). |
|
Answer»
|
|
| 4. |
Which compound gives beta-elimination reactions ? What are their main product ? |
| Answer» SOLUTION :(i) ALKYL HALIDE (II) alcohol compounds gives `beta` -ELIMINATION reaction. Alkene is the main product of this reaction. | |
| 5. |
To prepare a pure sample of n-hexane, using sodium metal as one of the reactant, the other reactant(s) will be |
|
Answer» ETHYL CHLORIDE and n-butyl chloride |
|
| 7. |
what type of substances would make better peremanent magnets, ferromagnetic or ferrimagnetic ? |
| Answer» SOLUTION :Ferromganetic SUBSTANCES would make better PERMANENT MAGNETS. | |
| 8. |
When lithium is irradiated with light, one finds a stopping potential of 1.83V for lambda = 3000A^@ and 0.80V for lambda = 4000A^@. With the known charge on the electron, the work function of lithium is |
|
Answer» 4.6eV `therefore W_0 = 2.3 eV` |
|
| 9. |
The RMS velocity of a gas at 0°C is 2m/s. The RMS velocity of the same gas at 819°C |
|
Answer» `1 m//s` |
|
| 10. |
Which of the following is not a homgeneous mixture. |
|
Answer» AIR |
|
| 11. |
The weakest base among the following is |
|
Answer» `C_(6)H_(5)SO_(3)^(-)` |
|
| 12. |
Which method is used to make water germ free effectively and rapidly? |
| Answer» Solution :Passing Ozone | |
| 13. |
The specific heat of a liquid was measured by placing 100 g of the liquid in a calorimeter. The liquid was heate by an electric immersion coil. The heat capacity of the determined to be 31.4 J//K. With the 100g sample placed in the calorimeter, a current of 0.5 ampere was passed through the immersion coil for exactely 3 minutes. the voltage across the terminal of coil was measure to be 1.50 V. the temperature of the sample rose by 0.8^(@)C. find the specific capacity of liquid. |
|
Answer» |
|
| 14. |
Write the structures of the following : (i) But-2-en-1-ol (ii) 2-Aminoethan-1-ol (iii) 2, 4-Dimethylhexan-3-one (iv) 1, 3-Diaminopropane (v) 4-Ethyl-2, 4-dimethylhexane (vi) 5-(1, 2-Dimethylpropyl) nonane. |
|
Answer» SOLUTION :(i) `CH_(3)-CH=CH-CH_(2)-OH` (ii) `HO-CH_(2)-CH_(2)-NH_(2)` (iii) `{:(""O),("||"),(CH_(3)-CH-C-CH-CH_(2)-CH_(3)),("|""|"),(""CH_(3)""CH_(3)):}` (iv) `H_(2)N-CH_(2)-CH_(2)-CH_(2)-NH_(2)` (v) `{:(""CH_(3)),("|"),(CH_(3)-CH-CH_(2)-C-CH_(2)-CH_(3)),("|""|"),(""CH_(3)""C_(2)H_(5)):}` (vi) `{:(CH_(3)-CH_(2)-CH_(2)-CH_(2)-CH-CH_(2)-CH_(2)-CH_(2)-CH_(3)),("|"),(""CH-CH_(3)),("|"),(""CH-CH_(3)),("|"),(""CH_(3)):}` |
|
| 15. |
What are the anomalies of the long form of periodic table? |
|
Answer» Solution :The long form of periodic table need clarification about the following: (i) Position of hydrogen is not defined till now. (II) Lanthanides and actinides STILL find PLACE in the BOTTOM of the table. |
|
| 16. |
Which of the following test can be used for identification of 1^(@) amine ? |
|
Answer» Carbylamine test |
|
| 17. |
Which of the following is not a similarly of beryllium with aluminium? |
|
Answer» It BECOMES passive when TREATED with conc. `HNO_(3)`. |
|
| 19. |
Which property of liquid is responsible shape of liquid drops ? |
| Answer» SOLUTION :SURFACE TENSION and VISCOISTY. | |
| 20. |
Which of the following alkali metals when burnt in air forms a mixture of oxide as well as nitride? |
|
Answer» K `4Li+O_(2) OVERSET(Delta)to 2Li_(2)O,6Li+N_(2) overset(Delta)to 2Li_(3)N` |
|
| 21. |
Which is the incorrect statement ? |
|
Answer» `FeO_0.98` has non-stoichiometric metal deficiency DEFECT |
|
| 22. |
Whichtwo elements of the followingbelong to thesameperiod ? A1 , Si , Baand O. |
| Answer» SOLUTION :A1 and SI. | |
| 23. |
The velocity of a gas molecule present in a cubic vessel in X-direction is 1000 cm / sec. The side of that cubic vessel is 10 cm. The number of bombardments made by the molecule on one face in X-direction in one second is |
| Answer» ANSWER :D | |
| 24. |
When does Z = 1 ? |
|
Answer» SOLUTION :`Z=(pV)/(NRT)` If `Z =1to pV = nRT` If GAS is IDEAL then Z = 1. |
|
| 25. |
Which one known as ODS ? |
| Answer» Solution :Both (A) and (B) | |
| 26. |
The two elements which react with air readily are |
| Answer» Solution :Al and Tl REACT READILY with AIR. | |
| 27. |
Zeolite used to soften hardness of water is hydrated: |
|
Answer» POTASSIUM ALUMINIUM borate |
|
| 28. |
Which of the following does not show geometrical isomerism ? |
|
Answer» 1,2 - dichloro - 1 - PENTENE |
|
| 29. |
Which graph of Moseley suggests that properties of elements depend upon atomic number? |
|
Answer» `v^(2) to Z ` As a result periodic law of Mendleev was corrected by putting the word atomic number. instead of .atomic weight. "The physical and CHEMICAL PROPERTIES of elements are periodic function of their atomic numbers" is modern periodic law. |
|
| 30. |
What is the coordination number of (i)sodium in sodium oxide (Na_2O) ? (ii)oxide ion in sodium oxide (Na_2O) ? (iii)calcium in calcium fluoride (CaF_2) ? (iv)zinc in zinc blende (ZnS) ? |
| Answer» SOLUTION :(i)4 , (ii)8 , (iii)4 , (IV)4 | |
| 31. |
Thermodynics is no concerned about "…............". |
|
Answer» ENERGY changes involved in a CHEMICAL REACTION. |
|
| 32. |
Which of the folowing represent Z or trans? |
|
Answer»
|
|
| 33. |
The strenth of 10 volume of H_(2)O_(2) solution is : |
|
Answer» 10 `underset(68 g)(2H_(2)O_(2))to 2H_(2)O_underset(22.4 " L at STP")(O_(2))` `because 22.4 L O_(2)` is OBTAINED at STP from `68 g H_(2)O_(2)` `therefore L O_(2)` is obtained at STP from `(68)/(22.4)xx10 g H_(2)O_(2)` =30.36 g Thus, strength of `H_(2)O_(2)=30.36 g//L`. |
|
| 34. |
What is the coordination number in a square close packed structure in two dimensions ? |
|
Answer» 2 |
|
| 35. |
Which one of the following can not act as nucleophile? |
|
Answer» `CH_(3)OH` |
|
| 36. |
Which of the following reaction produce dihydrogen from synthesis gas? |
|
Answer» `CH_(4)(g)+H_(2)O(g) UNDERSET(NI)overset(1270K)(rarr)CO(g)+3H_(2)(g)` |
|
| 37. |
When Friedel-Crafts alkylation of benzene is carried out with n-propyl bromide, the major product is |
|
Answer» n-propylbenzene 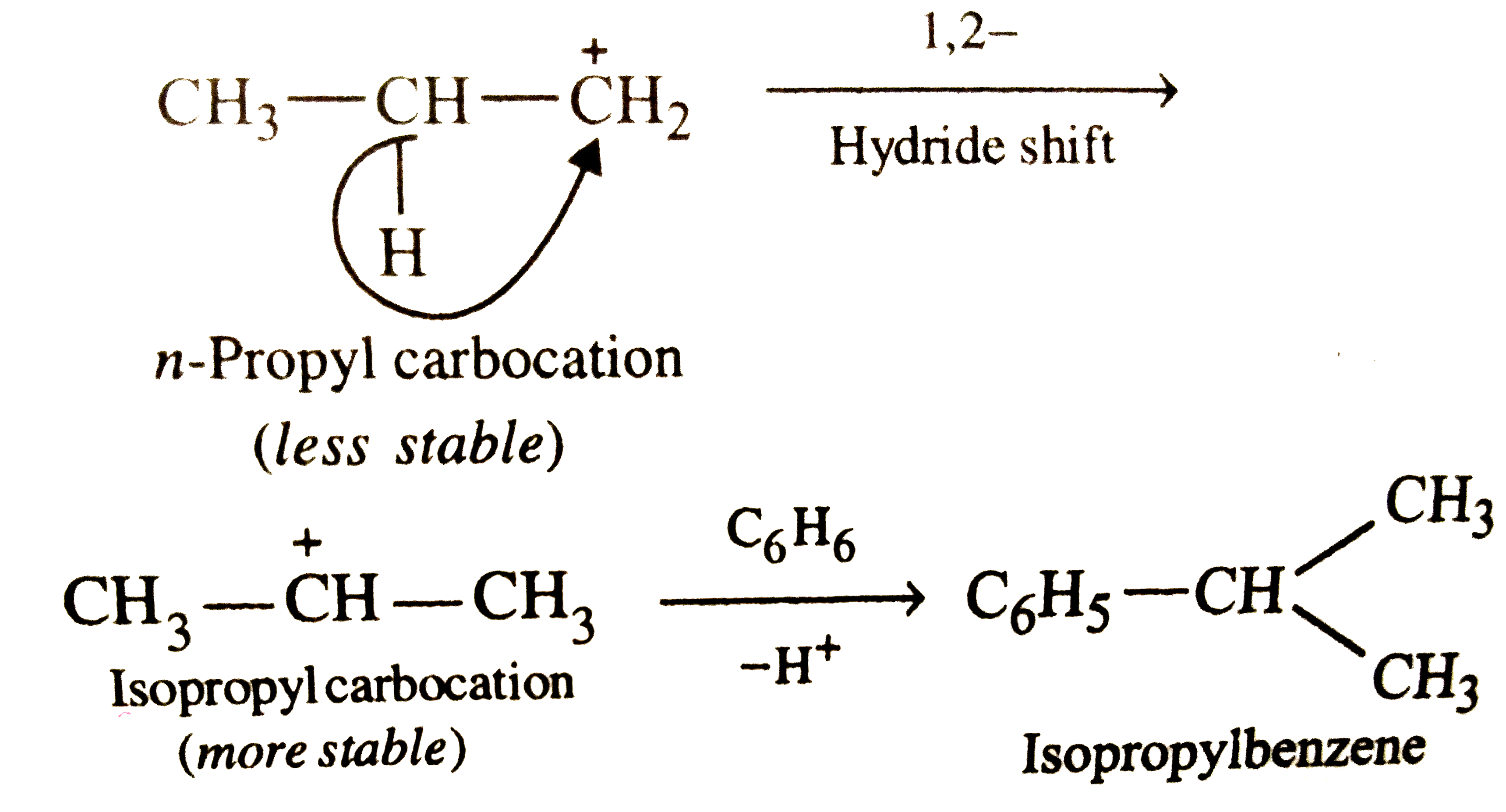
|
|
| 38. |
Which of the following solvents are aprotic? |
|
Answer» 1,2,3 |
|
| 39. |
Which of the following statement is/are correct? |
|
Answer» The conjugate acid of `NH_(2^((-)))` is `NH_(3)` `K_(a)` values decrease successively since it is difficult to remove `H^(o+)` ion from an anion thatn a netural compound. Similarly, it is difficult to remove `H^(o+)` ion from an dianion than an anion. HENCE `K_(a_(1)) gt K_(a_(2)) gt K_(a_(3))` B. Statement B is incorrect. `H_(3)PO_(4) overset(K_(a_(1)))hArrH^(o+)+H_(2)PO_(4^(T))` `H_(2)PO_(4^(T))overset(K_(a_(2)))hArrH^(o+) + HPO_(4^(2-))` `pH(H_(2)PO_(4^(T))) = (pK_(a_(1)) + pK_(a_(2)))/(2)` This is valid only when `H_(3)PO_(4)` during titrating is completely CONVERTED to `H_(3)PO_(4)^(T)`. `HPO_(4^(2-)) overset(K_(a_(2)))hArrH^(o+) + PO_(4^(3-))` `pH(HPO_(4)^(2-))= (pKa_(2) + pKa_(3))/(2)` This is again valid when starting with `H_(2)PO_(4)^(2-)` and is completely converted to `HPO_(4)^(2-)` C. Statement C is correct From the `K_(a)` values it is evident that both `H_(3)PO_(4)` and `H_(2)PO_(4^(T))` are more acidic than `HPO_(4^(2-))` Statement D is incorrect. Both `H_(2)PO_(4^(T))` and `HPO_(4^(2-))` are amphoprotic anion in the solution. `H_(3)PO_(4)overset(+H^(o+))larrH_(2)PO_(4^(T))overset(-H^(o+))rarrHPO_(4)^(2-)+H^(o+)` `H_(2)PO_(4^(T)) overset(+H^(o+))larrHPO_(4)^(2-)overset(-H^(o+))rarrPO_(4^(3-))+H^(o+)` |
|
| 40. |
What are inner transition elements ? |
| Answer» Solution :The ELEMENTS in which extra electron enters (n-2) f- orbitals are called f- BLOCK elements . Theseelements form a transition SERIES within the transition elements . So these elements are called as inner transition elements. | |
| 41. |
Total number of nodes (planar and spherical) present in the 5f-orbital is |
|
Answer» |
|
| 42. |
Which is the radioactive element in group 1 element? What is its half-life period? |
| Answer» Solution :Francium is highly RADIOACTIVE ELEMENT in GROUP. It has half-life of only 21 MINUTES. | |
| 43. |
What happens when p-xylene is treated with concentrated sulphuric acid and the resultant product is fused with KOH and finally dilute acid is added? |
Answer» SOLUTION :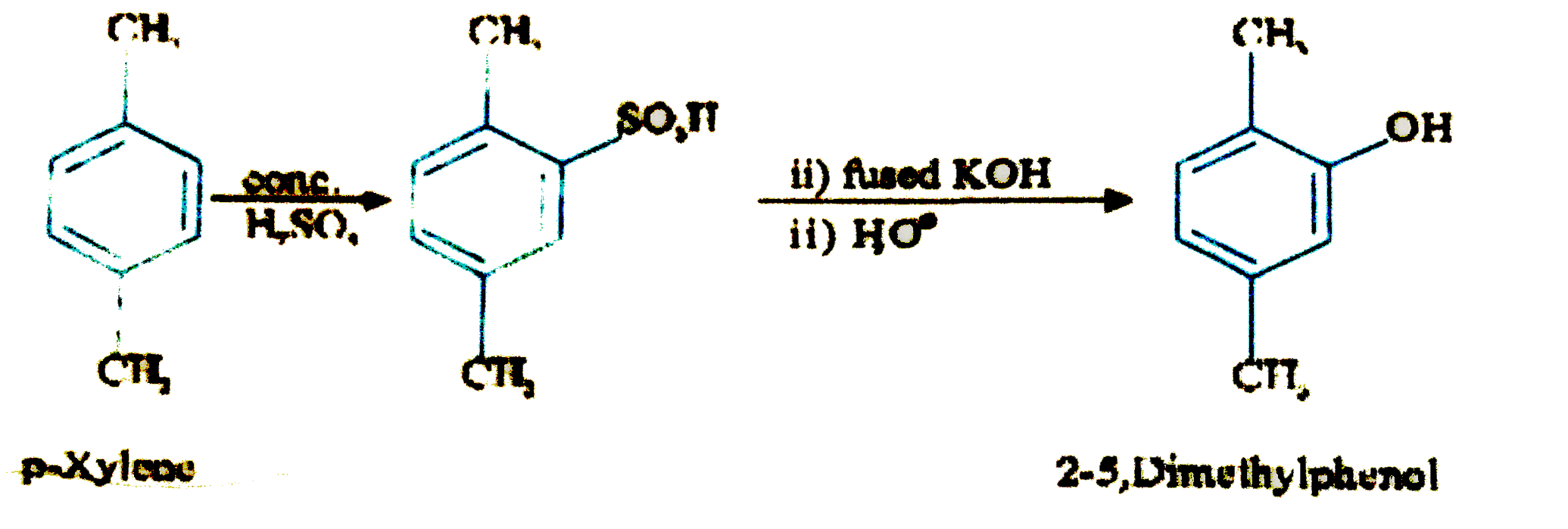 . .
|
|
| 44. |
When phenyl magensium bromide reacts with tert butanol, which of the following is formed? |
|
Answer» tert-Butyl methyl ether |
|
| 45. |
The relative reactivities of acyl compounds towards nucleophilic substitution are in the order of |
|
Answer» ACID ANHYDRIDE `gt` Amide `gt` ESTER `gt` Acyl CHLORIDE |
|
| 46. |
The weakest acid among the following |
|
Answer» `CH_(3)COOH` |
|
| 47. |
Write the chemical equations when sodium fusion extract is prepared from an organic compound containing nitrogen and sulphur. |
|
Answer» `2Na+StoNa_(2)S` |
|
| 48. |
The R_(f) values of A and B in a mixture determined by TLC in a solvent mixture are 0.65 and 0.42 respectively. If the mixture is separated by column chromatography using the same solvent mixture as a mobile phase, which of the two components, A or B, will elute first? Explain. |
| Answer» Solution :Since the `R_(F)` value of A is 0.65, therefore, it is less strongly adsorbed as compared to component B with `R_(f)` value of 0.42. Therefore, on extraction of the COLUMN, A will elute first. | |
| 49. |
The stoichiometric coefficients do not represent |
|
Answer» RELATIVE NUMBER of molecules of the rectants and products |
|