Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When a metal of group-1 was dissolved in liquid ammonia, the following observations were obtained: (a) Blue solution was obtained initially. (b) On concentrating the solution, blue colour changed to bronze colour. How do you account for the blue colour of the solution ? Give the name of the product formed on keeping the solution for some time. |
|
Answer» Solution :(a) Alkali metals dissolve in liquid AMMONIA and give BLUE solution because of ammoniated electrons. These electrons ABSORB energy in the visible region of light and IMPART blue colour to the solution. `M+(x+y) NH_(3) to [M(NH_(3))_(x)]^(+) +[(NH_(3))_(y)]^(-)e` (b) In concentrated solution, the blue colour changes to bronze colour due to the formation of clusters of metal ion, on standing, blue solution liberate H, gas with the formation of amide. `M^(+)+e^(-) +NH_(3) to underset("(amide)")(MNH_(2))+(1)/(2) H_(2)` |
|
| 2. |
When P reacts with caustic soda. The products are PH_(3) and NaH_(2)PO_(2). This reaction is an example of |
|
Answer» OXIDATION reaction |
|
| 3. |
Which of the following compounds shall not produced propene by reaction with HBr followed by elimination of direct only elimination reaction ? |
|
Answer» `H_(2)C=C=O`<BR>`H_(3)C-underset(H_(2))underset(|)OVERSET(H_(2))overset(|)(C)-CH_(2)Br`  `CH_(2)=C=Ounderset("Elimination")overset(HBR)rarr H_(3)C-CH=CH_(2)` `CH_(2)=C=O overset(HBr)rarr H_(2)C=overset(Br)overset(|)(C)-OH hArr H_(3)C-overset(O)overset(||)(C)-Br` `CH_(3)-CH_(2)-CH_(2)-Br overset("Elimination")rarr CH_(3)-CH=CH_(2)` |
|
| 4. |
Which of the following sets of quantumnumber is correct for an electrons in 4f orbital ? |
|
Answer» N = 4,L = 3, m= + 4, s = +1/2 |
|
| 5. |
Which information obtained by electronic conflguratJon of Molecule In MO ? |
|
Answer» Solution :Stability of molecules : If `N_(b) gt N_(z) `, means a stable molecule while `N_(b) lt N_(a)` , means an unstable molecule so, stability `prop ` bond order. Bond order : `(1)/(2) (N_(b) - N_(a))` Nature of bond : Integral bond order values of 1, 2 and 3 correspond to single, double or triple bonds respectively. Bond length : The bond length decrease of bond order increase Magnetic nature: If all the MOLECULAR orbitals in a molecule are doubly OCCUPIED, the substance is DIAMAGNETIC (repelled by magnetic field). If ONE order more molecular orbitals are singly occupied it is PARAMAGNETIC (attracted) by magnetic field), e.g. `O_(2)`molecule. Where, `N_(a)` = Number of electrons in antibonding orbitals. `N_(b)` = Number of electrons in bonding orbitals. BO Bond order |
|
| 6. |
What happens when BF_3 is reacted with ammonia ? |
Answer» Solution :`BF_3`(a Lewis ACID) reacts with `NH_3` (a Lewis base) to FORM an adduct. This RESULTS in a complete OCTET around B in `BF_3` 
|
|
| 7. |
What do youmean by significant figures ? |
|
Answer» |
|
| 8. |
The solubility product of AgCl is 4.0xx10^(-10) at 298 K. The solubility of AgCl in 0.04 m CaCl_(2) will be |
|
Answer» `2.0xx10^(-5) m` `[Ag^(+)]= x "mol" L^(-1), [Cl^(-1)]=(0.04xx2)+x ~~0.8 m` `:. x (0.08 ) 4xx10^(-10) "or" x = 5.0 xx 10^(-9) m` |
|
| 9. |
The solubility of A_(2)X_(3) is y mol dm^(-3) . Its solubility product is |
|
Answer» `6 y^(4)` `K_(ap)=(2Y)^(2)xx(3y)^(3)=108y^(5)`. |
|
| 10. |
Which of the following statement(s) regarding friedal craft alkylation is/are correct? |
|
Answer» The action of `Me_3C . CH_2 CL// AlCl_3` on benzen yields almost wholly the REARRANGED product Ph. `Cme_2. CH_2 Me ` |
|
| 11. |
Whichion has 18electron in its outermostorbit ? |
|
Answer» `Cu^(+)` |
|
| 12. |
What type of metals are used in photoelectric cell ? Give one example. |
|
Answer» |
|
| 13. |
What happens when chloroform is treated with heavy water in presence of an alkali ? |
|
Answer» Solution :Due to greater electronegativity , the three Cl atoms in `CHCl_(3)`, decrease the electron density in C-H bond. In other words, H in `CHCl_(3)` is slightly acidic and THUS can be easily abstracted by a strong base such as `OH^(-)` to form `"CC"l_(3)^(-)` which then reacts with `D_(2)O` to formdeuterochloroform `(CDCl_(3))`, `H-"CCl"_(3) underset(-H_(2)O)overset(OH^(-))to overset(-): "CC"l_(3)overset(D_(2)O)to CDCl_(3)+ DOH` |
|
| 14. |
Why cathode rays are produced only when the pressure of the gas inside the discharge tube is very low ? |
| Answer» Solution :At HIGHER PRESSURE (e.g., at 1 atomospheric pressure), no electric current flows through the TUBE because gases are POOR conductor of ELECTRICITY. | |
| 15. |
Whate are polar and None Polar convalent bond explain with example. |
|
Answer» Solution :Polar covalent molecules : In DIATOMIC covalent molecule, the shared PAIR of electrons is displaced towards more electronegative ATOM present is called polar covalent molecule. EXAMPLE HCI Chlorine pulls the shared electrons from hydrogen towards itself. Hence it is polar molecule. `H_(2)O` Oxygen pulls the shared pair of electron from two hydrogen atom surrounding. It hence it is a polar molecule. Non Pollar covalent BOND : In diatomic covalent molecules. the shapred pair of electrons is equally distributed between the atoms in the molecule. Example `H_(2), O_(2), N_(2), O_(3)` etc. |
|
| 16. |
Which one of the following equations does not correctlyrepresents the first law of thermodynamics for the given process ? |
|
Answer» isothermal PROCESS`:q= - w` Forisothermal process, `DeltaE = 0`. Hence,`q= - w` For cyclic process,`DeltaHE =0`. Hence, `q= -w` For isochoric process , `DeltaV =0`. Hence,`DeltaE =q``( w= P DeltaV= 0)` For adiabaticprocess, `q=0`. Hence, `DeltaE= w`. For expansion into vacuum , `w=0` . Hence, `DeltaE=q`. Thus, only(d) is incorret. |
|
| 17. |
Which one of the following conversions involves change in both hybridisation and shape ? |
|
Answer» `CH_(4)to C_(2)H_(6)` |
|
| 18. |
Which one of the following is true about metallic character when we move from left to right in a period and top to bottom in a group? |
|
Answer» DECREASES in a PERIOD and INCREASES along the GROUP |
|
| 20. |
When Cl_(2) water is added to an aqueous solution of potassium halide in presence of chloroform, a violet colour is obtained upon shaking. This confirms the presence of |
|
Answer» iodide |
|
| 21. |
Which substance serves as a reducing agent in the following reactions ? 14H^(+) +Cr_(2)O_(7)^(2-) +3Ni rarr 2Cr^(3+) + 7H_(2)O+3Ni^(2+), |
|
Answer» `H_2O` |
|
| 22. |
Which of the following transition involves maximum amount of energy? |
|
Answer» `M_((G))^(-) to M_((g))` |
|
| 23. |
Which among the following are expected to form hydrates? |
|
Answer» `MgCl_(2)` |
|
| 24. |
Write structural formulae for compounds names as (a)1-bromoheptane (b) 5-bromoheptanoic acid |
|
Answer» SOLUTION :(a) `underset("1-bromoheptane")(overset(1)(C )H_(2)Br- overset(2)(C )H_(2)-overset(3)(C )H_(2)- overset(4)(C )H_(2)- overset(5)(C )H_(2)- overset(6)(C )H_(2)- overset(7)(C )H_(3))` (b) `underset("5-bromoheptanoic ACID")(overset(7)(C )H_(3)- overset(6)(C )H_(2)- underset(underset(Br)(|))overset(5)(C )H- overset(4)(C )H_(2) - overset(3)(C )H_(2) - overset(2)(C )H_(2)- underset(underset(O)(||))overset(1)(C )-OH)` |
|
| 25. |
What is the value of Planck's constant |
|
Answer» `6.626xx10^(-27)JS` |
|
| 26. |
Which are natural sinks for overset(.)(C ) lO racdical in other parts of stratosphere? |
|
Answer» `SO_(2)` and `NO_(2)` |
|
| 27. |
Whichelectronicconfigurationwill showthehighestfirstionizationpotential ? |
|
Answer» `1S^(2) 2s^(2)2P^(1)` |
|
| 28. |
What is the formula of hypo? |
| Answer» SOLUTION :`Na_2S_2O_3 . 5H_2O` | |
| 29. |
Which one of the following constitutes a group of the isoelectronic species? |
|
Answer» `CN^(-1), N_(2), O_(2)^(-2), O_(2)^(-2)` |
|
| 30. |
what is the corrdination number of (i) Sodium in sodium oxide ( Na_(2)O) ?(ii)oxide ion in sodim oxide (Na_(2)O) ? (iii) Calcium in calcium fluoride(CaF_(2))? (iv) Zinc in zinc blende (ZnS) ? |
| Answer» Solution :(i) 4 (II) 8 (III)8 (iv)4(v) 4. | |
| 31. |
Water transported throughlead pipes becomes poisonous due to the formation of |
|
Answer» `PB(OH)_(2)` |
|
| 32. |
What type of magnetism is shown by a substance if magnetic moments of domains are arranged in same direction |
| Answer» SOLUTION :FERROMAGNETIC SUBSTANCE | |
| 33. |
(v) P+HNO_(3) to H_3PO_4+NO_2+H_2O |
|
Answer» <P> Solution : Equalise the increase / DECREASE in O N by multiplying P SPECIES by `+1` and N species by `+5`. |
|
| 34. |
Write the value of n,l and m for electron present in 2P_(z) orbital. |
|
Answer» |
|
| 35. |
Which compound is used for themanufacture of chemicals like sodium perborate and per-carbonate which are used in high quality detrgents. |
|
Answer» `H_(2)S` |
|
| 36. |
What are surfactants chemical used in synthetic detergents ? |
| Answer» SOLUTION :ALKYL BENZENE SULPHONATES | |
| 37. |
What happens to the pH of acetic acid when aqueous sodium acetate is added to it ? Explain. |
| Answer» SOLUTION :PH INCREASES | |
| 38. |
What do you mean by silicones ? |
| Answer» | |
| 39. |
Which of the following compound(s) is/are more reactive than benzene towards nitration? |
|
Answer»

|
|
| 40. |
Vapour density of the equilibrium mixture of NO_(2) and N_(2)O_(4) is found to be 40 for the equilibrium N_(2)O_(4) harr 2NO_(2). From this information answer the following Abnormal molecular weight is |
|
Answer» 80 |
|
| 41. |
Vapour density of the equilibrium mixture of NO_(2) and N_(2)O_(4) is found to be 40 for the equilibrium N_(2)O_(4) harr 2NO_(2). From this information answer the following Percentage of NO_(2) in the mixture is |
|
Answer» SOLUTION :`underset(1-alpha)(N_(2)O_(4)) harr underset(2alpha)(2NO_(2))` `%=(2alpha)/(1+alpha)=(2 XX 0.15)/(1.15)=0.26` |
|
| 42. |
Vapour density of the equilibrium mixture of NO_(2) and N_(2)O_(4) is found to be 40 for the equilibrium N_(2)O_(4) harr 2NO_(2). From this information answer the following Degree of dissociation of N_(2)O_(4) is |
|
Answer» 0.18 `(46)/(40)=1+alpha, alpha=(6)/(40)=0.15` |
|
| 43. |
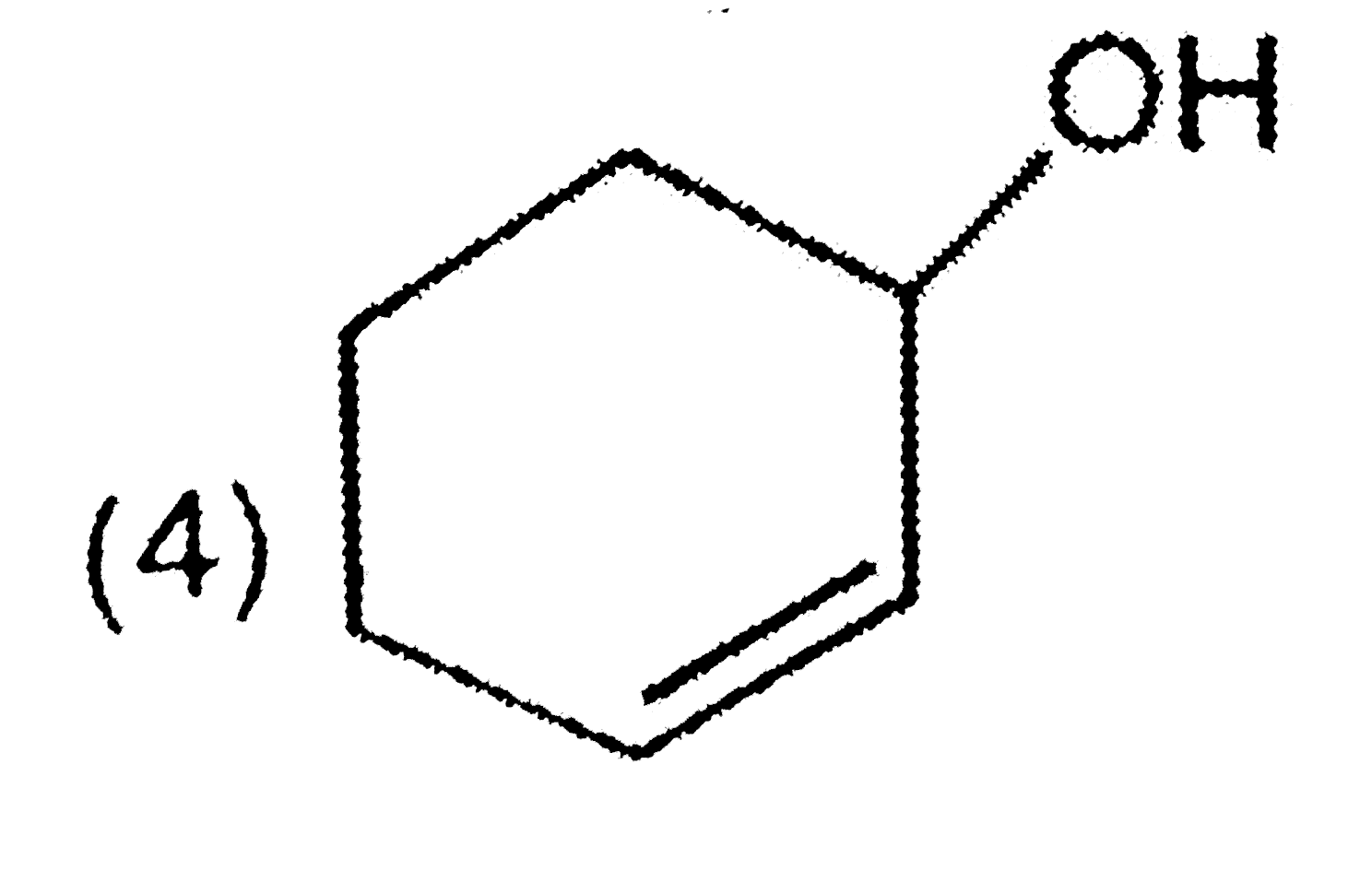
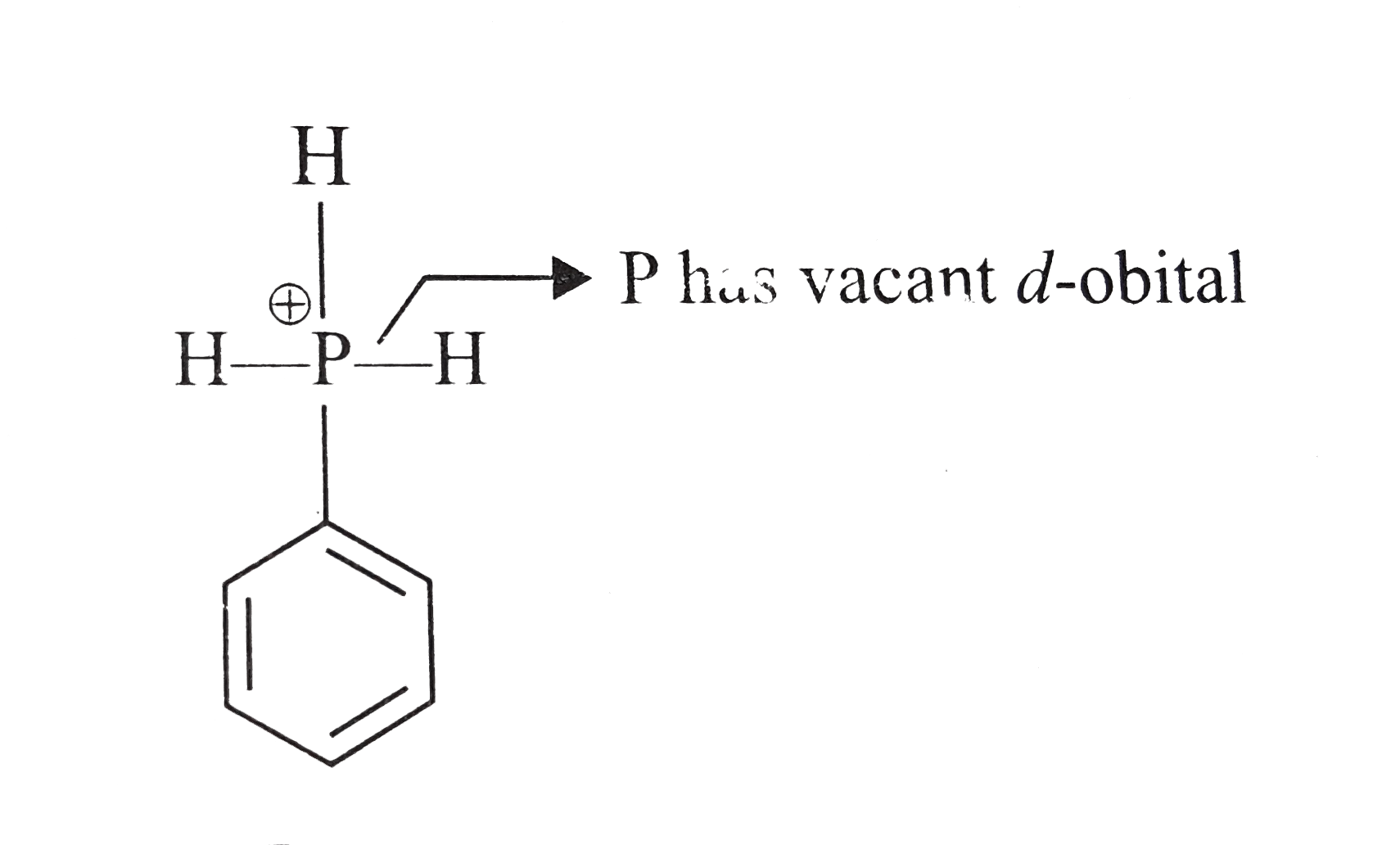
Which of the following compounds will exhibit d-orbital resonance ? |
|
Answer»
 . .
|
|
| 44. |
Which of the following is the correct major product for given reaction ? |
|
Answer»
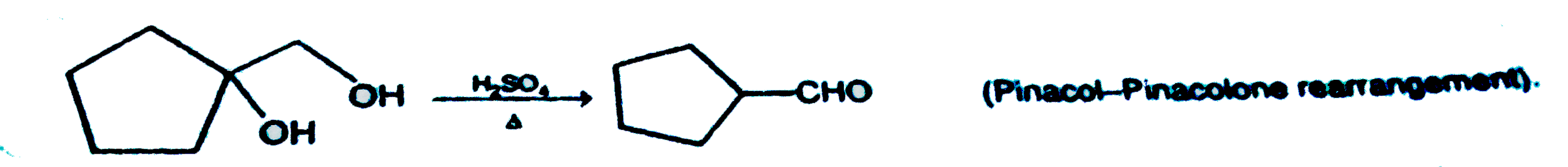
|
|
| 45. |
What is the effect of catalyst on equilibrium constant ? |
|
Answer» Increase |
|
| 46. |
Which of the following has the largest de - Broglie wavelength given that all have equal velocity ? |
|
Answer» `CO_(2)MOLECULE` for same velocity `lambda ALPHA 1//m` Electron has the LEAST MASS, so its wavelength is maximum. |
|
| 47. |
Which of the following is correct with respect to -I effect of the substituents? (R = alkyl) |
|
Answer» `-NR_(2) GT - OR gt - F` `-NH_(2) lt -OR lt -F` |
|
| 48. |
Which of the following reagents can provide distinction between acetic and bezoic acid ? |
|
Answer» Na |
|
| 49. |
Which of the following represents the correct IUPAC name for the compounds concerned? (a) 2, 2-Dimethylpentane OR 2-Dimethylpentane (b) 2, 4, 7-Trimethyloctane OR 2, 5,7-Trimethyloctane (c ) 2-Chloro-4-methylpentane OR 4-Chloro-2-methylpentane (d) But 3-yn-1-ol OR But -4-ol-1-yne |
|
Answer» Solution :(a) 2, 2-Dimethylpentane is correct NAME because, for two alkyl groups on the same carbon its LOCANT is REPEATED twice. (b) 2, 4, 7-Trimethyllocatane is correct name. Here, 2, 4, 7-locant set is lower than 2, 5,7 (c ) 2-Chloro-4-methylpentane is correct name. Because here function group is chloro so, lowest no. is give to chloro (d) But-3-yn-1-ol is correct name. Here, In compound two functional group `-C -= C- and -OH` present. In which `-OH` is a principal functional group so, it CARRY lower locant |
|
| 50. |
Which of the following elements is extracted commercially by the electrolysis of an aqueous solution of its compound ? |
|
Answer» Cl `Al_2O_3 HARR underset"cathode"(Al^(3+)) + underset"anode"(Al_3^(3-))` `Al^(3+) + 3e TOAL` (at cathode) `4AlO_3^(3-) to 2Al_2O_3 + 3O_2 + 12e` (at anode) Aluminium of 99.8% purity is obtained fromthis process. |
|