Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is lanthanide contraction? Give one of its consequences. |
|
Answer» |
|
| 2. |
Which one of the following statements is wrong for gases |
|
Answer» GASES do not have a definite shape and VOLUME |
|
| 3. |
The weight of H_2O_2present in 80 ml 10V H_2O_2solution is |
|
Answer» 3.2 G |
|
| 4. |
What is the pH of acidic buffer ? How it is formed ? |
|
Answer» Solution :The pH of acidic buffer is LESS than 7. It is PREPARED by EQUAL MIXTURE of WEAK acid and its salt with strong base. `CH_3COOH+CH_3COONa` pH:4.75 |
|
| 5. |
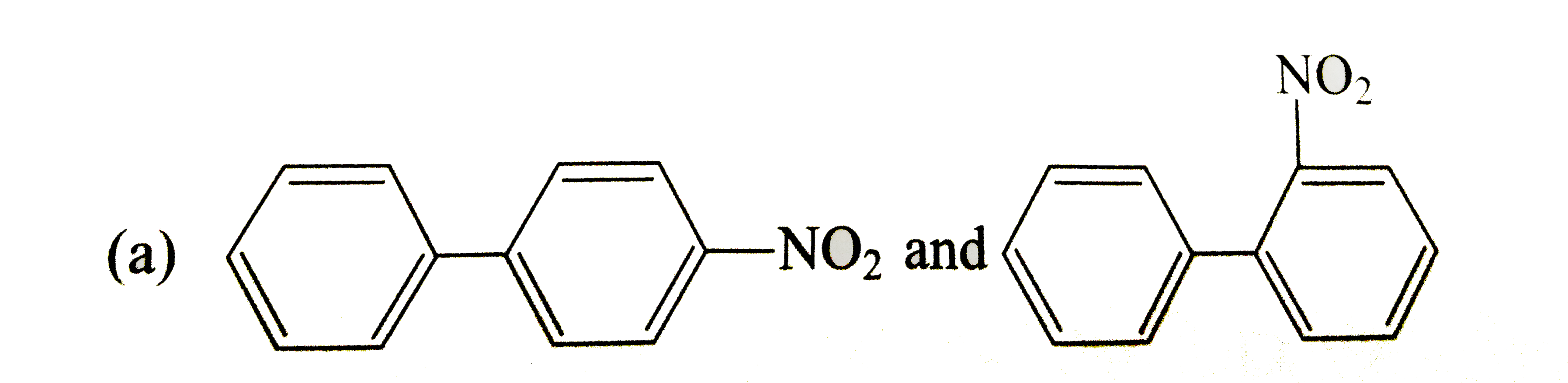
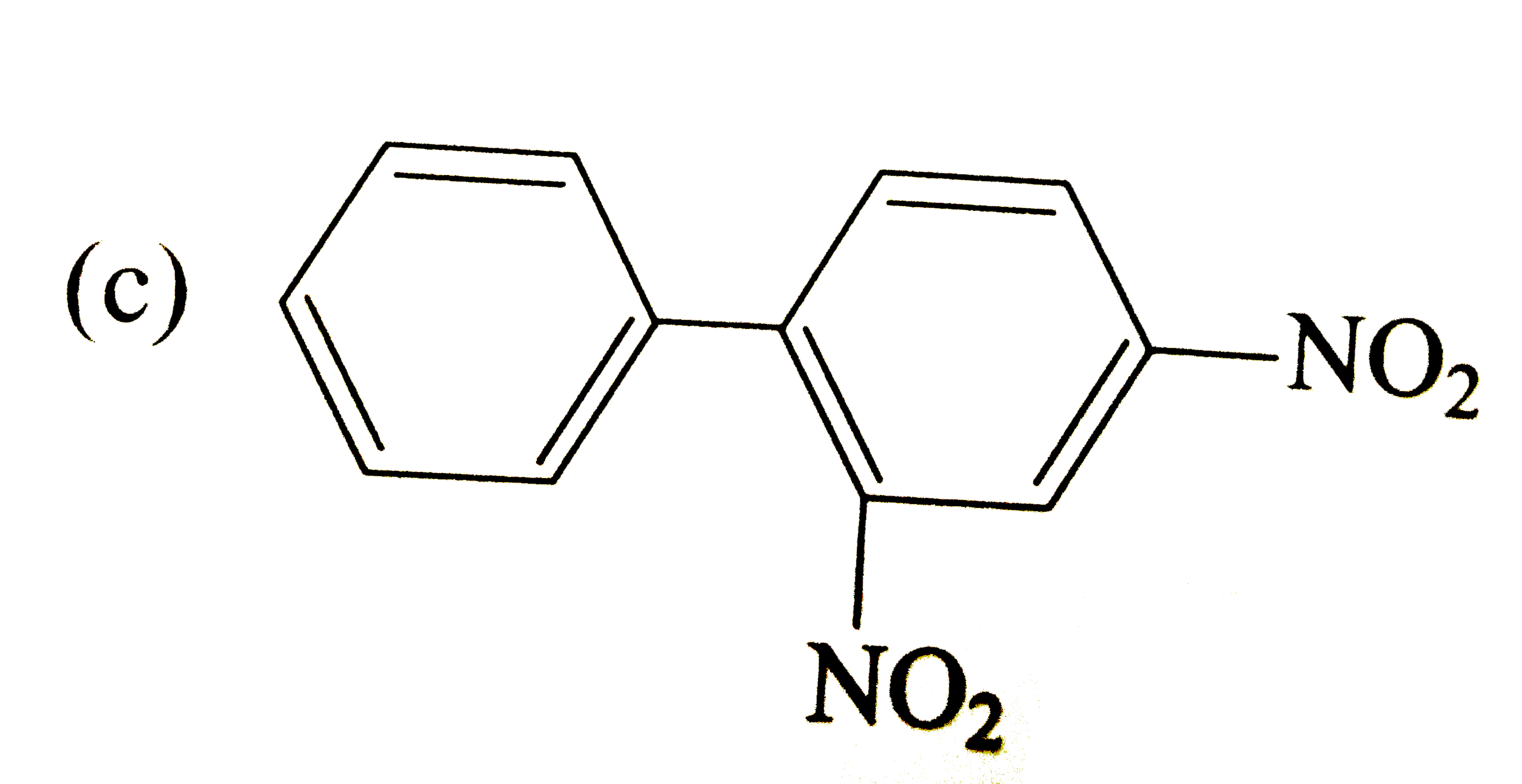
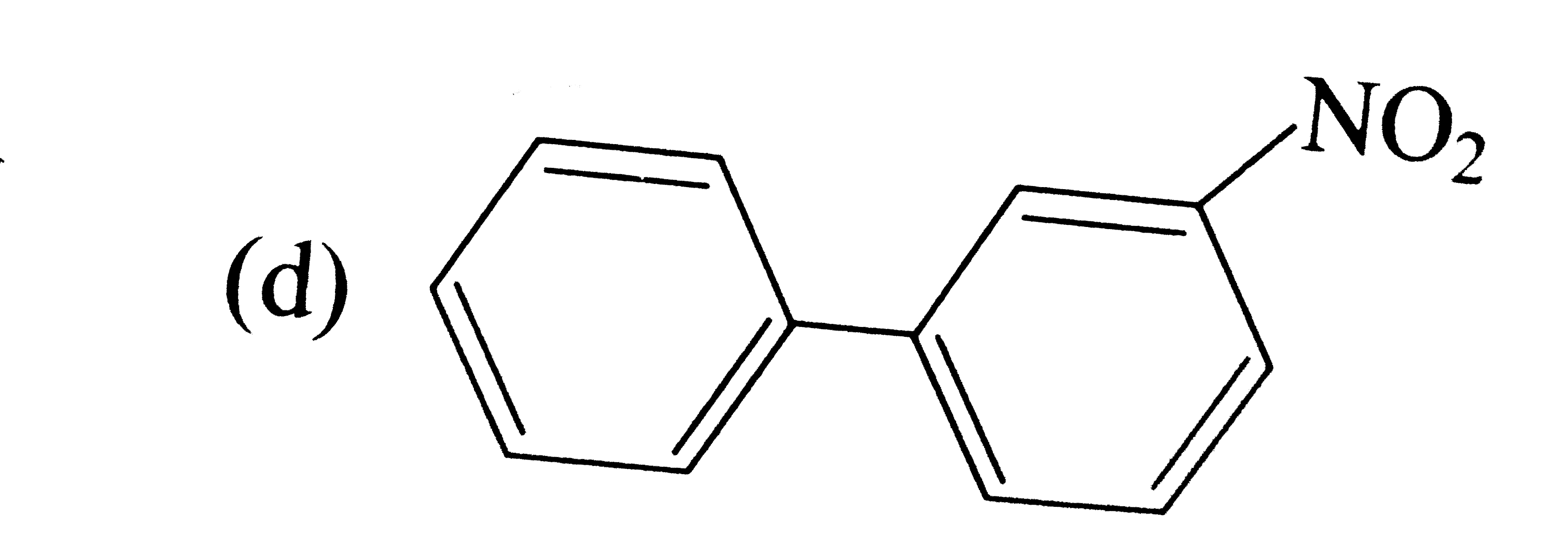
Write the IUPAC names of the following compounds. |
|
Answer» Solution :(i) 2-Phenylethanol, (ii) 1, 1, 1-Trichloro-2, 2-diphenylethane (iii) 2-Phenylethanamine, (iv) 2- (CHLOROMETHYL) chlorobenzene (v) 3, 3-Diphenylbutan-2-ol, (VI) 1, 2-bis (4-chlorophenyl) ETHANE (vii) 2, 4-Dinitrobenzenamine, (VIII) 4-Nitroso-N-dimethylbenzenamine (ix) Benzene 1, 4-dicarboxylic acid, (x) 1-Phenylpropanone. |
|
| 6. |
Vander Waal's equation for one mole of CO_(2)gas at low pressure will be |
|
Answer» <P>`(P + a/(V^2))V = RT` |
|
| 7. |
Which of the following are stored under oil? |
| Answer» Answer :A | |
| 8. |
Which of the following alcohols on oxidation gives a carboxylic acid with lesser number of carbon atoms ? |
|
Answer» `CH_(3)CH_(2)CH_(2)CHO` |
|
| 9. |
Which represents an example of Friedel-Crafts reaction |
|
Answer» `C_6 H_6 + C_2 H_5 CL OVERSET(AlCl_3 ) to C_6H_5 C_2 H_5 + HCl` |
|
| 10. |
Which of the following statements about hydrogen is incorrect ? |
| Answer» Solution :Hydrogen has three ISOTOPES of which TRITIUM is radioactive and RATE. Dihydrogen ACTS as reducing agent. | |
| 11. |
Water add in 1.0 mL 0.1 M HCl solution to give 50 mL. Calculate pH change of solution. |
| Answer» Solution :Initially PH =1 , After dilution pH=2.7 , The change in pH = 1.7 increases. | |
| 12. |
Which of the following is the most powerful oxidising agent? |
|
Answer» `F_(2)` `3S overset(+4)(O_(2))+K_(2)Cr_(2)O_(7)+H_(2)SO_(4) to K_(2)SO_(4) +Cr_(2) (overset(+6)(SO)_(4))_(3)+H_(2)O` Hence, oxidation state of SULPHUR changes from +4 to +6. |
|
| 13. |
Which of the following compounds is oxidised to prepare ethyl methyl ketone ? |
|
Answer» 2-Propanol |
|
| 14. |
Which of the correct IUPAC names of the following compound ? H_3C-CH_2-CH_2-overset(HC(CH_3)_2)overset|"CH"-undersetunderset(H_3C-CH-CH_2CH_3)|"CH"-CH_2-CH_2-CH_2-CH_2-CH_3 |
|
Answer» 5-Butyl-4-isopropyldecane |
|
| 15. |
Which of the following is true about chemisorption? |
|
Answer» It is reversible in nature |
|
| 16. |
The volume strength of 1.54 N H_(2)O_(2) solution is: |
|
Answer» 4.8L `=(1.5"equiv" L^(-1))xx(17"equiv"^(-1))` `underset(68g)(2H_(2)O_(2)) to underset(22.4L)(2H_(2)O+O_(2))` 68g of `H_(2)O_(2)` evolve `O_(2)` under N.T.P conditions=22.4L 25.5g of `H_(2)O_(2)` evolve `O_(2)` under N.T.P conditions `=((22.4L)xx(25.5g))/(68g)=8.4L` |
|
| 17. |
Which of the following statements in not correct regarding preparation of NaOH/ |
|
Answer» NAOH is prepared by ELECTROLYSIS of sodium chloride in castner-kellner cell. |
|
| 18. |
Which of trhe following statements is not correct? |
|
Answer» The first inoisation energies (in ` KJ "mol"^(-1)`) of carbon, silicon , GERMANIUM , tin, and lead are 1086, 786,761,708 and 715 respectively:Down the GROUP , electronegativity dereases from B to T1 in boron FAMILY. |
|
| 19. |
When FeS_2is oxidized with sufficient O_2then its oxidation product is found to be Fe_(2)O_3 and SO_2 if the molecular weight of FeS_(2), Fe_(2) O_3 and SO_2are M. Ma and Mc, then which of the following statements are correct? |
|
Answer» EQUIVALENT wt. of `FeS_2`is M/11 |
|
| 20. |
The vapour density of a gas is 11.2. Calculate the volume occupied by 11.2 g of the gas at NTP. [Hint: mol. Wt =2 xx V.D] |
|
Answer» |
|
| 21. |
Write the coordination number of each ion in the following crystals : (i)NaCl (ii)CsCl (iii)ZnS (iv)Na_2O |
| Answer» Solution :(i)`Na^+`=6,`Cl^-`=6 , (II)`Cs^+`=8, `Cl^-`=8 , (III)`ZN^(2+)`=4 , `S^(2-)` =4 , (iv)`CA^(2+)`=8 , `F^-` =4 , (v)`Na^+` =4 , `O^(2-)`= 8 | |
| 22. |
Which of the following is used in high temperature thermometry ? |
|
Answer» Na |
|
| 23. |
Which of the following statement is wrong for lyophobic sol |
|
Answer» Dispersed PHASE is generally in organic material |
|
| 24. |
When an electron with charge .e. and mass .m. moves with velocity .v. around the nucleus having nuclear charge .Z. in a circular orbit of radius .r., the potential energy of electron is |
|
Answer» `(ZE^(2))/(R)` |
|
| 25. |
The van der Waal equation of gas is (P+ (n^(2)a)/(V^(2))) (V - nb) = nRT |
|
Answer» (i) `CO_(2)` , (ii) `H_(2)` In `CO_(2)`, intermolecular FORCES increases with number of electrons in a molecule. (ii)Size increases in order of `H_(2)LT O_(2)CH_(4)lt CO_(2)` Hence, `H_(2)` will have lowest VALUE of B. |
|
| 26. |
Which of the following is an example of heterogeneous equilibrium ? |
|
Answer» Synthesis of HI Here `CO_(2)` is in gaseous state while `CaCO_3` and CaO are in solid state . |
|
| 27. |
Which of the following species is reduced by N_(2)O_(2) ? |
|
Answer» `[Fe(CN)_(6)]^(4-)` `2|Fe(CN)_(6)|^(3-)+2OH^(-)+H_(2)O_(2)to|Fe(CN)_(6)|^(4-)+2H_(2)O_(2)+O_(2)`. |
|
| 28. |
Which is the pH of 10^(-8) M HCl from 8.0, 7.0, 6.95 ? Why ? |
| Answer» SOLUTION :6.95 because the solution is ACIDIC THEREFORE the VALUE of pH is less than 7.0. | |
| 29. |
The total charge in coulombs required to complete the electrolysis is : |
|
Answer» 24125 MOLES of Na discharged at CATHODE = 1 mol Total charge needed to COMPLETE the electrolysis is `= 2 xx F =xx 96500 C` `= 193000 C`. |
|
| 30. |
Two oxides of a non-metal X contain 50% and 40% of non -metal respectively. If the formula of the first oxide is XO_2, then the formula of second oxide is |
|
Answer» |
|
| 31. |
What is the exothermic reaction? |
|
Answer» |
|
| 32. |
Which of the following oxides is amphoteric ? |
|
Answer» `SnO_(2)` |
|
| 33. |
Write only arrangement of electron and its shape of the following. HgCl_(2) " (ii) " BF_(3) "(iii) " NH_(4)^(+) " (iv) PCl_(5) (v) SF_(6) "(vi) " SO_(2)" (vii)" NH_(3) "(viii)" H_(2) O "(ix)"SF_(4) " (x)" CIF_(3) " (xi)" BrF_(5) " (xii)" XeF_(4) |
| Answer» SOLUTION :Use table Q. No: 52 & 53 | |
| 34. |
What aresuper heavy element? |
| Answer» Solution :ELEMENTWITH `Z GT 100` which havehighdensitiesare called super heavy element. | |
| 35. |
The state of hybridisation of S in SO_(2) in similar to that of C in |
|
Answer» `C_(2)H_(2)` |
|
| 36. |
When is treated with nitrating mixture (HNO_(3)+H_(2)SO_(4)), we get : |
|
Answer»
|
|
| 37. |
What is SI units of viscosity coefficient (eta) ? |
|
Answer» Pascal |
|
| 38. |
Which of the following cannot be produced by acidic dehydration of alcohols? |
|
Answer» Ethers |
|
| 39. |
Which of the following is most reactive towards sulphonation? |
|
Answer» <P>m - XYLENE |
|
| 40. |
Why are lithium salts commonly hydrated and those of the other alkali metal ions usually anhydrous? |
| Answer» SOLUTION :Due to its smallest SIZE`Li^(+)` can polarize WATER molecules easily than the other alkali METAL IONS. | |
| 41. |
Write the resonating structures for the following molecules. CO_(3)^(2-) |
Answer» SOLUTION :
|
|
| 42. |
When a 20 mL of 0.08 M weak base BOH is titrated with 0.08 HCl , the pH of the solution at the end point is 5. What will be the pOH if 10 mL 0.04 M NaOH is added to the resulting solution?[Given:log 2= 0.30 and log 3 = 0. 48 ) |
|
Answer» <P>` 5.40` ` N_1V_1 = N_2V_2 , [BCl ]= (20 XX 0.08)/( 20+20 ) =0.04` ` 5= (1)/(2) [14 -P^(K_b) -log 4 xx 10^(-2) ]rArr P^(K_b) =5.4 ` ` {:(B^(+) +, OH^(-)to, BOH) ,( 1.6 m " mol " , 0.4 m " moles " ,0),(1.2 m " mol " ,-, 0.4 m " mole" ):}` ` P^(OH) =5.4 +log ""(1.2)/(0.4)=5.88` |
|
| 43. |
Which of the following is free radical addition reaction ? |
|
Answer»
|
|
| 44. |
What happens when ferrimagnetic Fe_(3)O_(4) is heated to 850 K and why ? |
| Answer» Solution :Ferrimagnetic `Fe_(3)O_(4)`on heating to 850 K becomes paramagnetic. This is due to GREATER alignment of domains (spins) in one direction on heating . | |
| 45. |
What is the difference between total kinetic energy and translational kinetic energy ? For what type of molecules, the two are equal ? |
| Answer» Solution :Total KINETIC energy is the sum of translational, vibrational and rotational kinetic ENERGIES. The total kinetic energy is equal to the translational kinetic energy for MONOATOMIC gases (He, Ne etc) as they do not POSSES vibrational and rotational motion but have only translational motion. | |
| 46. |
Which of the following salts solution will act as a buffer ? |
|
Answer» `NH_(4)CH_(3)COO(aq.)` Its NEGATIVE ion `(CH_(3)COO^(-))` reacts with `H^(+)` from added acid to from a weak acid `(CH_(3)COOH) :CH_(3)COO^(-)+H^(+)rarr CH_(3)COOH` |
|
| 47. |
What will be the molarity of 200 ml solution of sulphuric acid having pH = 1 ? |
|
Answer» 0.5 M |
|
| 48. |
Which of the following is true about I and II? |
|
Answer» I is ECLIPSED, II is staggered |
|
| 49. |
The solubilty product of the electrolyte of the type A_2B_3 is (s is the solubility in mol/ lit) |
|
Answer» `108 s^(5) ` |
|