Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which one of the following conditions will favour maximum formation of the product in the reaction,A_2(g) + B_2(g) iffX_2(g), Delta_r H = -X kJ |
|
Answer» High TEMPERATURE and low pressure |
|
| 2. |
Which of the following easily reacts with water producing hydrogen? |
|
Answer» `PH_(3)` |
|
| 3. |
Which of the following sets of atomic numbers is of the alkali metals ? |
|
Answer» `2,10,18` |
|
| 4. |
What are the value of n, l and m for 2p_(x) and 3p_(z) orbitals ? |
|
Answer» SOLUTION :For `2p_(X), n = 2, l = 1, m = +1` For `3p_(z), n = 3, l = 1, m = 0` |
|
| 5. |
Which one of the following reactions does not involve either oxidation or reduction |
|
Answer» `VO_(2)^(+) to V_(2)O_(3)` |
|
| 6. |
What happens when aniline is treated with methyl chloride in presence of AlCl_3 ? |
|
Answer» It under GOES friedel craft reaction to form a mixture of P - methyl and O-methyl aniline. |
|
| 7. |
Why at low pressures, the real gas behaves as ideal gas? |
|
Answer» Solution :At LOW PRESSURES, when volume is large, both correction factors in the van der Waals. equations may be neglected. The equation `(P+(a)/(V^2)) (V - b) = RT,` reduces to PV =RT, ideal gas law. This is nothing but representing the LIMITING behaviour of gases at infinite small pressures. |
|
| 8. |
Which of the following options represents the correct bond order : |
|
Answer» `O_(2)^(-) GT O_(2) gt O_(2)^(+)` |
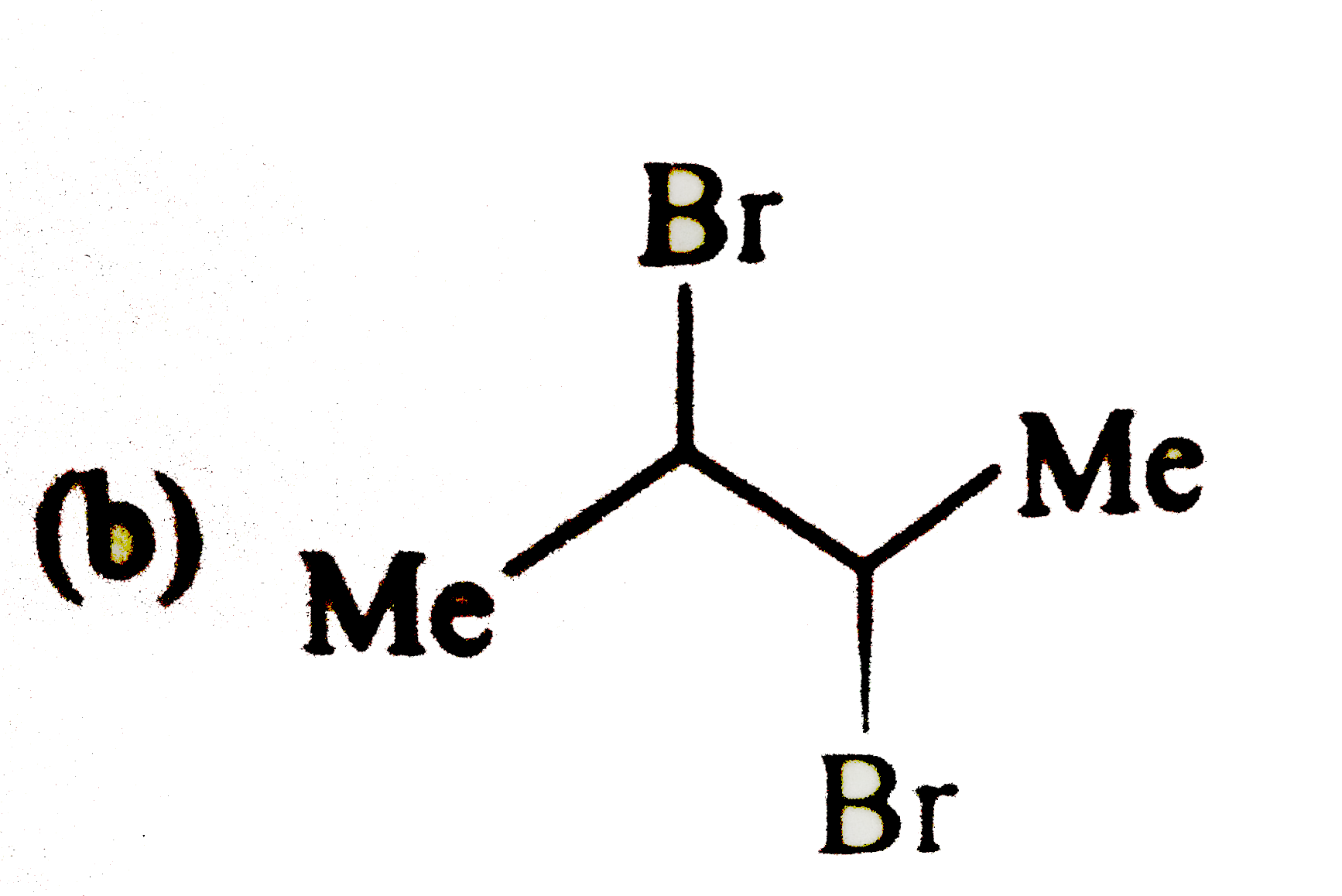
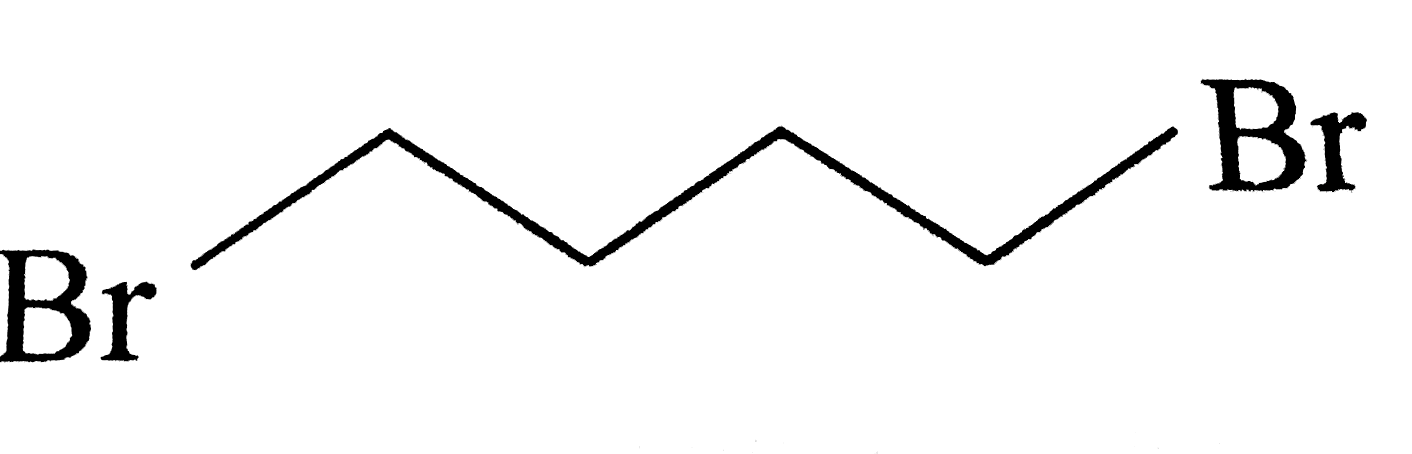
|
| 9. |
Using the data given below, calculate the value of equilibrium constant for the reaction. underset("Acetylene")(3HC-=CH(g)) hArr underset("Benzene")(C_(6)H_(6)(g)) at 298 K , assuming ideal gas behaviour. Delta_(f)G^(@)for HC-= CH(g) = 2.09 xx 10^(5) J mol^(-1),Delta_(f)G^(@)forC_(6)H_(6)(g) =1.24xx 10^(5) J mol^(-1) R = 8.314 J K^(-1) mol^(-1) Based on your calculated value, comment whether this process can be recommended as a practical method for making benzene. |
|
Answer» `Delta_(r)G^(@) = - 2.303 RT LOG K `. Calculate K `DELTA _(r)G^(@) ` is negative and K is very high . Hence, the PROCESS can OCCUR easily. |
|
| 10. |
Use of Al for domestic purpose is reduced now. Why ? |
| Answer» SOLUTION :Aluminium metal and COMPOUNDS of aluminium are TOXIC. Hence the utility is now REDUCED. | |
| 11. |
Which of the following presents the correct order of the acidity in the given compounds ? |
|
Answer» `FCH_(2)COOH gt ClCH_(2)COOH gt BrCH_(2)COOH gt CH_(3)COOH` |
|
| 12. |
When Q_(sp) is used ? |
| Answer» Solution :When in representation of `K_(sp)` ONE or more species concentration is not equal to concentration of `K_(sp)` than `Q_(sp)`USED. | |
| 13. |
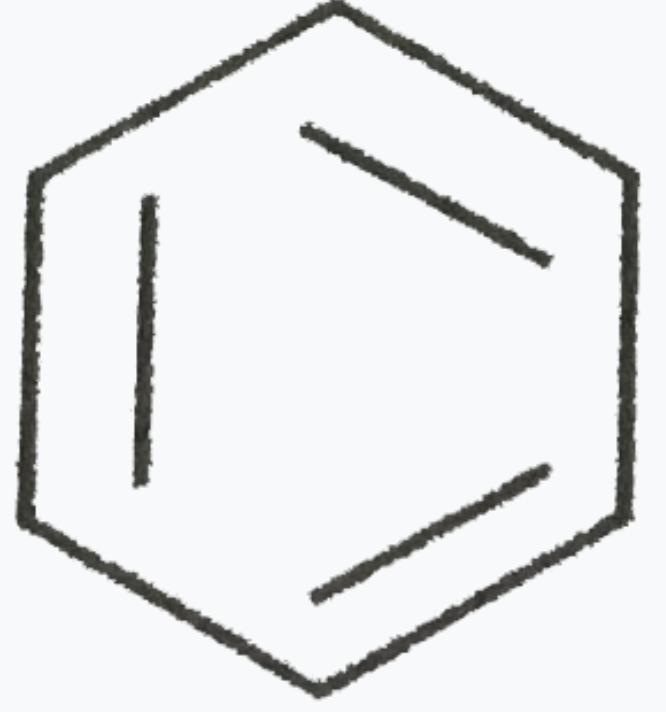
Which one of the following is non -aromatic ? |
|
Answer»
|
|
| 14. |
What is polarisation? |
| Answer» SOLUTION :The ABILITY ofa cation to POLARISE an anion is CALLED its polarising ability and the tendency of the anion to get polarised is called its polarisability. This plenomenon is known as POLARISATION. | |
| 15. |
What is oxidation number of Cl in bleacing powder ? |
|
Answer» Solution :Molecular formula : `CaOCl_(2),Ca^(+2)(OCl^(-))Cl^(-)` `THEREFORE` Oxidation NUMBER of Cl in `OCl^(-)=+1` and oxidation number of SECOND Cl = -1 `therefore` Average oxidation number is ZERO. |
|
| 16. |
Which of the following is wrong ? |
|
Answer» (1) Degree of DISSOCIATIONS of a weakelectrolyte increase with dilution. |
|
| 17. |
Which of the following is an intermediate in the reaction of benzene with CH_3Cl and AlCl_3 ? |
|
Answer»
|
|
| 18. |
When R-C-=CH is treated with cuprous in ammonical medium, product formed is |
|
Answer» `R-C-=C-CU` |
|
| 19. |
Which of the following sequences contain atomic numbers of only representative elements ? |
|
Answer» 3,33,53,87 With reference to this division, elements listed under option (A) and option (D) are all representative elements. |
|
| 20. |
What is the energy of state in a triply ionized beryllium (Be^(++)) whose radius is equal to that of ground state of hydrogen ? |
|
Answer» `= 0.529 Å implies0.529 = 0.529 xx (n^2)/(4) impliesn=2 ` |
|
| 21. |
Which of the followinghydrogen halide is most volatile ? |
|
Answer» HF has least BOILING POINT , viz , HCl . |
|
| 22. |
What is the thermodynamic product obtained from the addition of 1 mole of HBr to 1,3-butadiene? |
|
Answer» 1-bromo-2-butene |
|
| 23. |
The wave function for an orbital is H-atom is given as psi=(sqrt(2)/(81sqrt(pi)))((1)/(a_(0)))^((3)/(2))(6-(r )/(a_(0)))((r )/(a_(0)))e^((r )/(3a_(0))). sinthetasinphi The orbital is : |
| Answer» Answer :A::B | |
| 24. |
Write the molecular formula of iron (III) hexacyanoferrate (II). |
| Answer» Solution :`Fe_(4)[Fe(CN)_(6)]_(3)`. | |
| 25. |
What would have happened if the greenhouse gases were totally missing in the earth's atmosphere ? Discuss |
| Answer» Solution :The solar energy RADIATED back from the earth surface is absorbed by the greenhouse gases (i.e. , `CO_2, CH_4 , O_3 , CFC's`and water vapour ) present near the earth's surface Thus, they heat up the atmosphere near the earth's surface and KEEP it WARM As a result, they keep the temperature of the earth constant and HELP in the growth of PLANTS and existence of life on the earth. If there were no greenhouse gases, there would have no vegetation and life on the earth | |
| 26. |
The total energy of electron in an atom is a combination of potential energy and kinetic energy. If total energy is -E for an electron in an atom, then its K.E. and P.E. respectively are |
|
Answer» |
|
| 27. |
Which of the following compound(s) has /have percentage of carbon same as that in ethylene (C_2H_4) . |
|
Answer» BENZENE |
|
| 28. |
What is Gay - Lussac.s formula ? |
|
Answer» `p PROP T` |
|
| 29. |
Which alkali metal carbonate decomposes on heating to liberate CO_2? |
| Answer» SOLUTION :`Li_2CO_3` | |
| 30. |
Volume of 0.1 M K_(2)Cr_(2)O_(7) required to oxidise 35 mL of 0.5 M FeSO_(4) solution is : |
|
Answer» 29.2 mL |
|
| 32. |
Which of the following act as a stabiliser for the storage of H_(2)O_(2)? |
|
Answer» Solution :`H_(2)O_(2)` decomposes slowly on exposure to light. `2H_(2)O_(2(l))to 2H_(2)O_((l))+O_(2(g))` In the presence of metal surfaces or traces of alkali (present in glass containers), the above REACTION is catalysed. It is, therefore, stored in wax-lined glass or plastic vessels in dark. Urea can be added as a stabiliser. It is kept away from dust because dus can induce explosive decomposition of the compound. |
|
| 33. |
The state of equilibrium is in a dynamic balance between forward and backward reaction. This balance can be disturbed by changing concentration, temperature or pressure. If done so a cartain net change occurs in the system. The direction of change can be predicted with the help of Le Chatelier principle. It states that when a system in equilibrium is disturbed by a change in concentration of temperature, a 'net' change occurs in it in a direction that tends to decrease the disturbing factor. Consider the following exothermic heterogenous equilibrium, M_(2)O(s)+2HNO_(3)(aq)hArr2MNO_(3)(aq)+H_(2)O(l) withK_(C)=3 at 300K. Select the incorrect option. |
|
Answer» Addition of `H_(2)O(l)` to above EQUILIBRIUM has no EFFECT on equilibrium COMPOSITION `(%)` of `HNO_(3)` and `MNO_(3)`. |
|
| 34. |
The state of equilibrium is in a dynamic balance between forward and backward reaction. This balance can be disturbed by changing concentration, temperature or pressure. If done so a cartain net change occurs in the system. The direction of change can be predicted with the help of Le Chatelier principle. It states that when a system in equilibrium is disturbed by a change in concentration of temperature, a 'net' change occurs in it in a direction that tends to decrease the disturbing factor. For the equilibrium, underset(("yellow"))(Fe^(3+)(aq))+SCN^(-)(aq)hArrunderset(("deep red"))[[Fe(SCN)^(2+)](aq) Select the correct option. |
|
Answer» Addition of `H_(2)C_(2)O_(4)` which FORMS `[Fe(C_(2)O_(4))_(3)]^(3-)` DEEPENS red colour. |
|
| 35. |
Which colour is imparted when Lithium, sodium, potassium and Rubidiumk undergoes flame test? |
|
Answer» Solution :Lithium `to` CRIMSON RED Sodium `to` yellow or golden yellow Potassium `to` Pale Violet Rubidium `to` REDDISH violet |
|
| 36. |
Van der Waal's forces are attraction inter molecular forces. Write the names of any two types of van der Waal's forces. |
| Answer» SOLUTION :Dipole-dipole FORCES, DISPERSION forces. | |
| 37. |
Which of the following is the most stable intermdiate formed during the acid catalysed dehydration of neopentyl alcohol |
|
Answer» `(CH_(3))_(3)Coverset(o+)(C)H_(2)` `(CH_(3))_(3)C CH_(2)OH underset(-H_(2)O)overset(H^(+))rarr (CH_(3))_(3)C CH^(o+) overset("Methyl shift")rarr underset(3^(@)-"carbocation")((CH_(3))_(3)overset(o+)(C)CH_(2)CH_(3))` |
|
| 38. |
Which sequence of reaction produce |
|
Answer» `CH_3-C -= C CH_3 underset("Lindlar's catalyst")overset(D_2)to` |
|
| 39. |
What are the products in the following sequence of reaction Compound (B) is: |
|
Answer»
|
|
| 40. |
When conc. H_(2)SO_(4) was treated with K_(4) [Fe(CN)_(6)], CO has was evolved. By mistake, somebody used dilute H_(2)SO_(4) instead of conc, H_(2)SO_(4), then the gas evolved was |
|
Answer» `CO` `K_(4)[Fe(CN)_(6)]+underset("(dil.)")(3H_(2)SO_(4)) rarr 2K_(2)SO_(4)+FeSO_(4)+6HCN uarr` |
|
| 41. |
Which is the correct increasing order of boiling points of the following compounds ? 1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene |
|
Answer» `"BROMOBENZENE" LT "1-Bromobutane" lt "1-Bromopropane" lt "1-Bromoethane"` |
|
| 42. |
Which of the following is not an application of adsorption phenomenon? |
|
Answer» PURIFICATION of muddy water |
|
| 43. |
Which of the following has highest hydration energy |
|
Answer» `MgCl_(2)` |
|
| 44. |
What happens when (i) Sodium metal is dropped in water?(ii) Sodium metal is heated in free supply of air? (iii) Sodium peroxide dissolves in water? |
|
Answer» Solution :`2NA + 2H_(2)O to 2NaOH + H_(2)` (ii) `2Na +O_(2) to2Na_(2)OH_(2)` (iii) `Na_(2)O_(2) + 2H_(2)O to 2NaOH + H_(2)O_(2)` |
|
| 45. |
The temperature at which second virial coefficient of a real gas is zero is called |
|
Answer» critical temperature ` PV=A+(B)/(V)+…` At Boyle temperature, the gas SHOWS IDEAL behaviour, i.e., PV=RT which is so only when A=RT and B=0. |
|
| 46. |
Which of the following is an amphoteric hydroxide ? |
|
Answer» `Be(OH)_(2)` `Be(OH)_(2) +2HCl to BeCl_(2) +2H_(2)O` `Be(OH)_(2) +2NAOH to Na_(2)[Be(OH)_(4)]` |
|
| 47. |
Two minerals that contain Cu are CuFeS_(2) and Cu_(2)S. What mass of Cu_(2)S would contain the same mass of Cu as is contained in 125 lb of CuFeS_(2)? |
|
Answer» |
|
| 48. |
Which of the following gases would you expect to deviate from ideal behaviur under conditaions of low temperature F_(2),Cl_(2)orBr_(2) ? Explain. |
| Answer» Solution :The condition for a GAS to deviate from ideal behavior is high pressure and low temperature. As both conditions greater the DEVIATION from the ideal behaviour. `F_(2),Cl_(2)` and ` Br_(2)` are all non polar molecules. But the heaviestmolecule will have stronger DISPERSION FORCE. Dispersion force increases with molar maas, so `Br_(2)` would deviate most. | |
| 49. |
What is the entropy change for conversion of one gram of ice to water at 273K and one atmospheric pressure ( Delta _(fus) H = 6.025 kJ mol^(-1)) ? |
|
Answer» |
|
| 50. |
The stability of 2,3-dimethyl-but-2-ene is more than 2-butene. This can be explained in terms of |
|
Answer» Resonance |
|