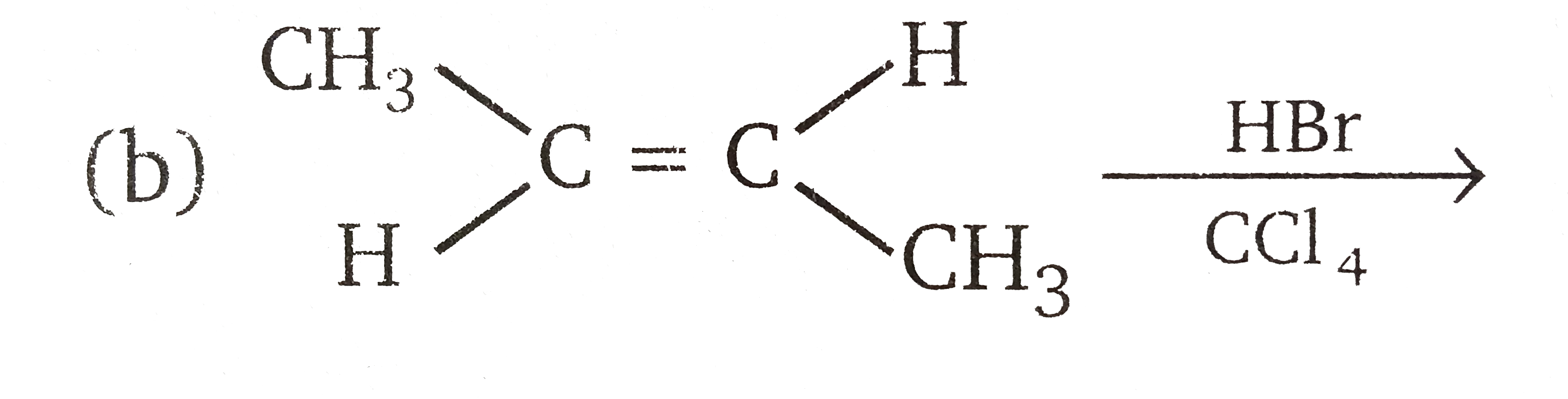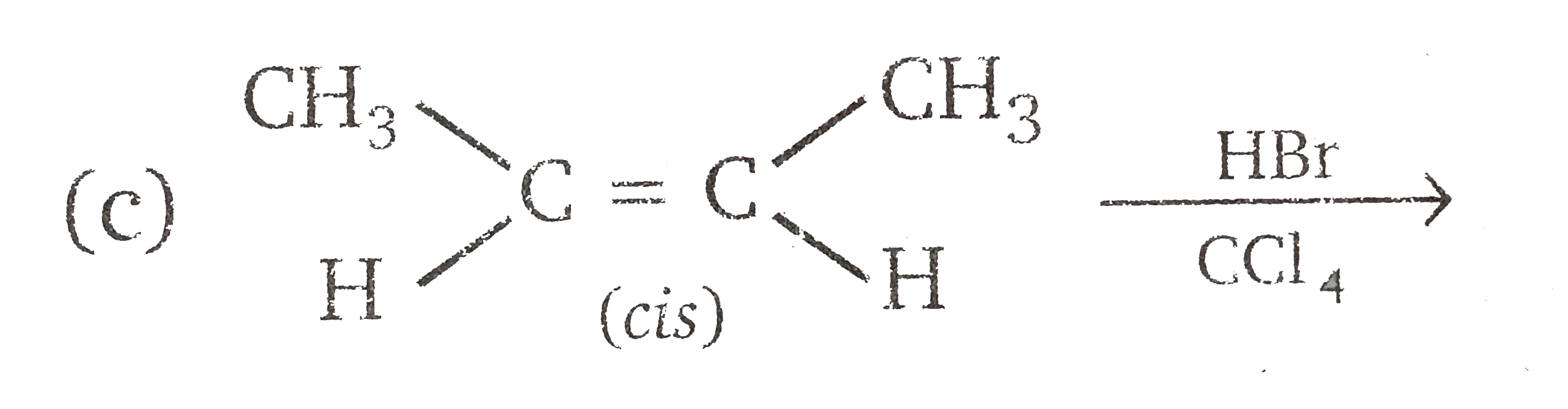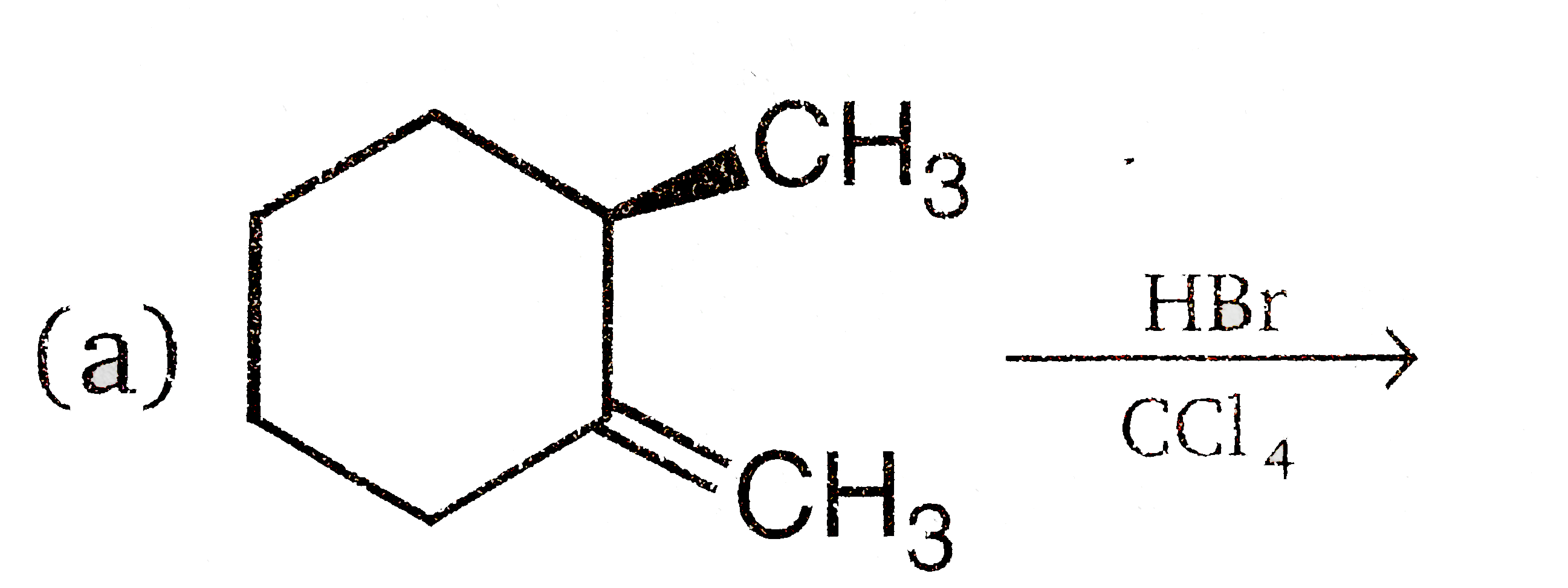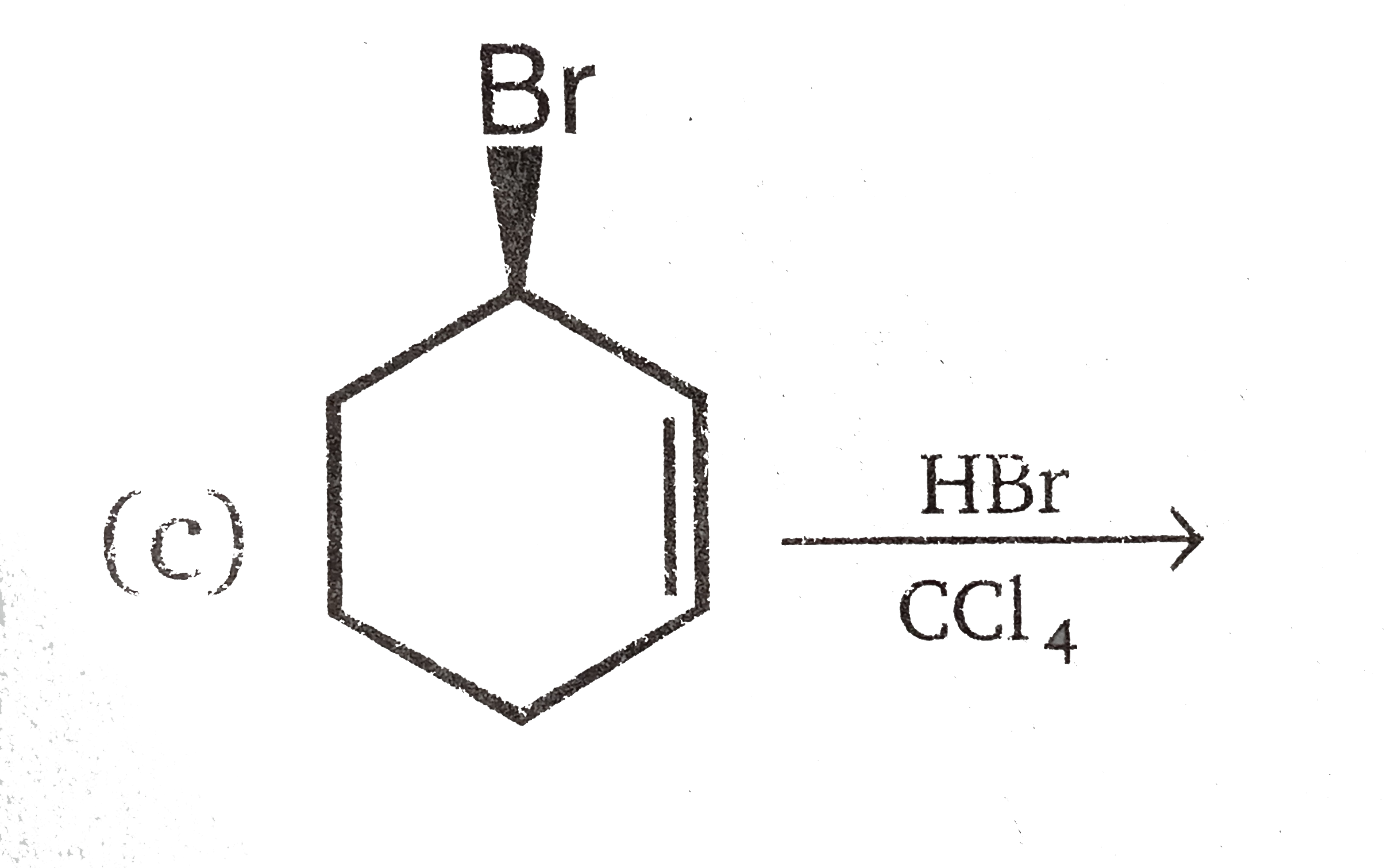Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What happens when ehtanol is heated with cone H_(2)SO_(4)? Give equation. |
| Answer» Solution :`underset("ETHANOL")(CH_(3)CH_(2)OH)overset(CONOH_(2)SO_(4))(to)underset("ETHENE")(CH_(2))=CH_(2)+H_(2)O` | |
| 2. |
Which one of the following has highest bond order ? N_(2) , N_(2)^(+) " or " N_(2)^(-) |
|
Answer» SOLUTION :`N_(2) ` (14 electrons) BOND order = 3 Bond order ` = (N_(b)- N_(a))/2 = (10 - 4)/2 = 3` `N_(2)^(+)` ( 13 electrons) Bond order= `2.5` Bond order ` = (N_(b) - N_(a))/2 = (9-4)/ 2 = 2.5` `N_(2)^(-) ` ( 15 electrons) Bond order ` = 2.5` Bond order ` = (N_(b) - N_(a))/2 = (10 - 5)/2 = 2.5`. So , `N_(2)` has the highest bond order . |
|
| 3. |
What happens when a jar full of H_2gas is placed inverted over a jar containing Cl_2 gas? |
| Answer» SOLUTION :The two gases diffuse into each other and the MIXTURE of gases in the two JARS ACQUIRE the colour of chlorine. | |
| 4. |
Which of the following statements is not a postulate of kinetic molecular theory of gases? |
|
Answer» K.E. is dependent on temperature |
|
| 5. |
Which molecule has the longest carbon chain? |
|
Answer» Isopentane |
|
| 6. |
Which of the following compounds has the lowest melting point ? |
|
Answer» `CaF_(2)` |
|
| 8. |
What are quantum numbers ? |
|
Answer» Solution :(i) The electron in an atom can be characterized by a set of four quantum numbers,namely PRINCIPAL quantum number (n) , AZIMUTHALQUANTUM number (I),magnetic quantum number (m) and SPIN quantum number (s). (ii) When Schrodinger EQUATION is solved for a wave function`Psi` ,the solution contains the FIRST three quantu numbers n,l and m. (iii) The fourth quantum number arises due to the spinning of the electron about its own axis. |
|
| 9. |
Write the chemical equations for combustion of propane. |
|
Answer» Solution :PROPANE BURNS in excess of oxygen to FORM water and carbon dioxide. `underset("Propane")(CH_(3)CH_(2)CH_(3))+50_(2)rarr3CO_(2)+4H_(2)O` |
|
| 10. |
Which of the following statements are correct about the ionic solids KI and CaO- |
|
Answer» lattice enthalpy of Ca0 is greater than that of KI |
|
| 11. |
Which of the following represents calcium chlorite ? |
|
Answer» `CaClO_2` |
|
| 12. |
What types of bonds are present in NH_4 CI? Write its structure. |
|
Answer» IONIC |
|
| 13. |
Which of the following group -14 elements is a radioactive element? |
|
Answer» Flerovium |
|
| 14. |
Which one of the following does not exhibit hyperconjugation ? |
|
Answer» Ethanal |
|
| 15. |
Which of the following alkaline earth metal sulphate has higher hydration enthalpy than lattice enthalpy |
|
Answer» `CaSO_(4)` |
|
| 17. |
What is understood by hydrogenation ? |
|
Answer» Solution :Hydrogenation means addition of `H_(2)` in PRESENCE of a catalyst to multiple bonds to form SATURATED compounds . For EXAMPLE, `UNDERSET("Ethane")(CH_(2)=CH_(2)+H_(2)) underset(473 K)overset(Ni or Pt or Pb)to underset("Ethane")(CH_(3)-CH_(3))` |
|
| 18. |
Which of the following is not a valid resonating structure ? |
|
Answer»
|
|
| 19. |
What are the possible types of electrons movement ? Represent then by clearly indicating the electrons shift . |
|
Answer» SOLUTION :There are THREE types of electron movement viz., 1. lone pair BECOMES a bonding pair . 2. bonding pair becomes a lone pair 3. a BOND breaks and becomes another bond .  
|
|
| 20. |
Which of the following statements is correct for hydrogen ? |
|
Answer» Hydrogen has same ionization POTENTIAL as alkali METALS |
|
| 21. |
Which causes water pollution? |
|
Answer» JET PLANES |
|
| 22. |
Which of the following ester on reaction with methyl magnesium chloride produces a secondary alcohol ? |
|
Answer» Ethyl acetate |
|
| 23. |
What is the pH of 0.01M glycine solution ? For glycine K_(a_(1))=4.5xx10^(-3) and K_(a_(2))=1.7xx10^(-10) at 398 K |
|
Answer» Solution :Glycine `H_(3)N^(+)CH_(2)CO O^(-)` is more acidic than basic. Instead of `K_(b)` value, the SECOND given `K_(a)` value is corresponding to the `K_(b)` value `(.:' K_(a)xxK_(b)=10^(-14))`.Hence, OVERALL IONIZATION constant, `K_(a_(1))xxK_(a_(2))=4.5xx10^(-3)xx1.7xx10^(-10)` `=7.65xx10^(-13)` `[H^(+)]=sqrt(KC)=sqrt(7.65xx10^(-13)xx0.01)` `=sqrt(0.765xx10^(-14))=0.87xx10^(-7)M` `pH = - log (0.87xx10^(-7))=7-0.93=6.07` |
|
| 24. |
When the temperature is increased, surface tension of water |
|
Answer» INCREASES |
|
| 25. |
What is water gas?How is itperpared ? |
|
Answer» SOLUTION :An equimolarmixture of `CO` with `H_(2)` is called water gas.It is obtainedby passing steam over red not COKE. `C(s) + H_(2)O (G) overset(1273 K)rarr CO(g) + H_(2)(g)`. |
|
| 26. |
Two moles of a perfect gas undergo the following processes : (a) a reversible isobaric expansion from ( 1.0atm, 20.0 L)to ( 1.0 atm,40.0 L) (b) a reversible isochoric change of state from ( 1.0 atm,40.0L ) to (0.5 atml, 40.0L) (c ) a reversible isothermal compression from ( 0.5 atm, 40.0 L) to ( 1.0 atm, 20.0L) (i) Sketch with labels each of the processes on the sameP-Vdiagram. (ii) Calculate the total work (w ) and the total heat change ( q) involved in the above processes (iii) What will be the valeu of DeltaU , DeltaH and DeltaS for the overall process? |
Answer» Solution :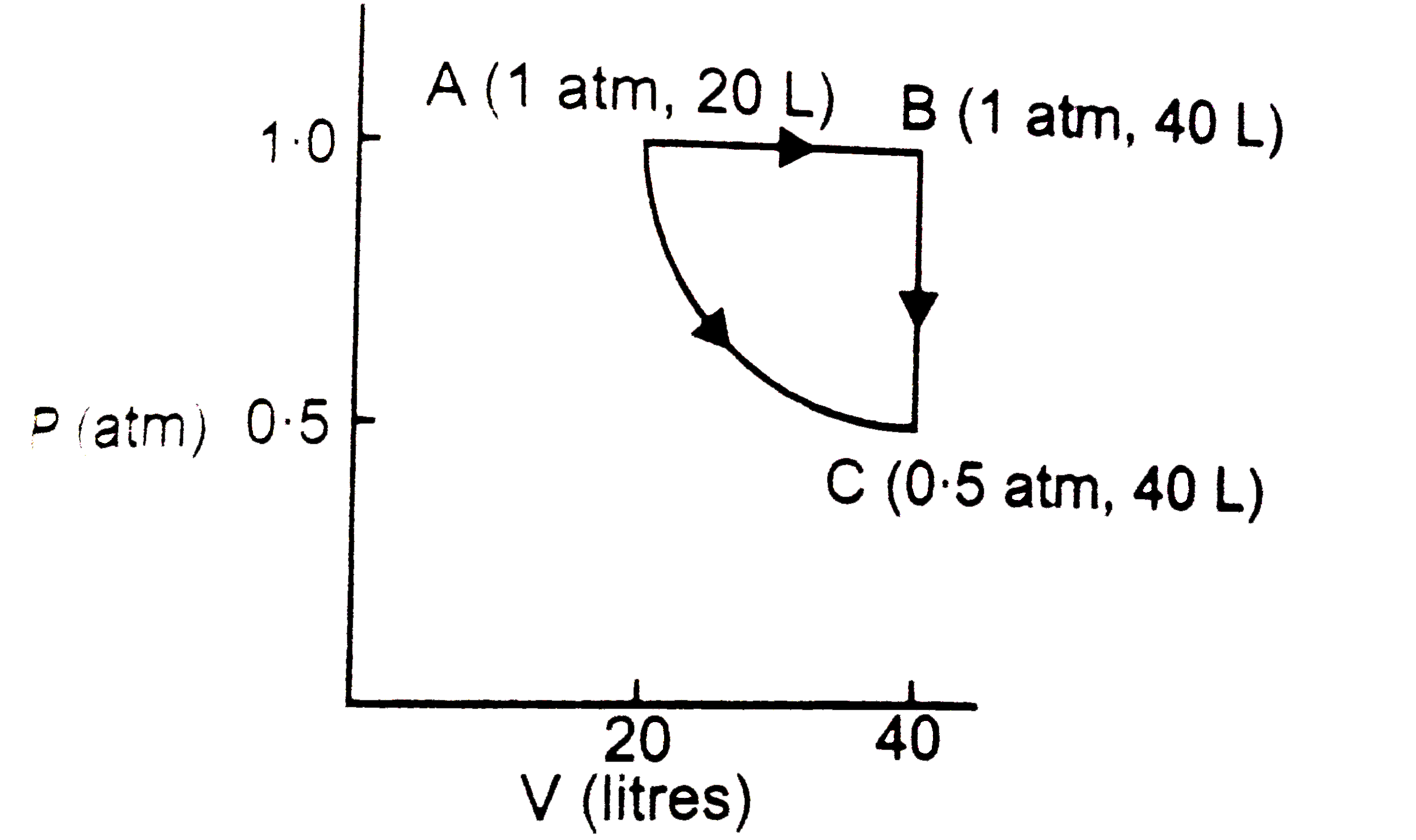 (ii)TOTAL work `(w) = w_(1)+w_(2)+w_(3) =- P DeltaV+ 0 + 2.303 nRT log .(V_(1))/(V_(2))` `= -1 xx 20+2.303 xx 2 xx 0.082 xx 121.95 log 2 (T =(PV)/(nR) = ( 1 xx 20)/( 2 xx 0.082) = 121.95 K) ` `= -20+ 13.86 = - 6.13L atm=-6.13 xx 101.3 J = - 621.0 J ` (In a cyclic process,HEAT abosorbedis completely converted into work) (iii) As U, H and S arestate FUNCTIONS and the system has returned to its original state,`DeltaU =0, DeltaH=0,` and`Delta S=0`. |
|
| 27. |
Which one of the following is crystalline form of carbon ? |
|
Answer» COAL |
|
| 28. |
Which of the following has largest size ? |
|
Answer» Al |
|
| 29. |
What is the name for the following chemical reaction? CO(g) + H_2O(g)to CO_2(g) + H_2(g) |
|
Answer» |
|
| 30. |
Write the expressions for K_(c)andK_(p) for the reaction, 2A+3Bunderset(V_(b))overset(V_(f))(hArr)4C+5D |
|
Answer» <P> SOLUTION :`K_(c)=([C]^(4)[D]^(5))/([A]^(2)[B]^(3))andK_(p)=(P_(C)^(4)P_(D)^(5))/(P_(A)^(2)P_(B)^(3))` |
|
| 31. |
What is electronegativity ? Explain periodicity in periodic table. |
|
Answer» Solution :Electronegativity: "CAPACITY to attract electron of element in chemical compound is known as electronegativity." It is not a measurable QUANTITY. Linus Pauling, an American scientist in 1922 assigned arbitrarily a value of `4.0` to fluorine. Periodicity : When we GO left to RIGHT in periodic table electronegativity increases. Ex. Second Period  When we go top to bottom in periodic table electronegativity decreases.  Electronegativity of noble gas is almost zero. Electronegativity Values (on Pauling scale) Across the Periods  Explanation : In period attraction of nucleus and electron increases so atomic radius decreases so electronegativity increases. When we go to top to bottom in group, atomic radius increases with electronegativity decreases. So tendency of electronegativity is same as Ionisation enthalpy OPPOSITE as atomic radius. |
|
| 32. |
The weight of methane which occupies the same volume at STP as 7.5 gm of ethane is __________g. |
|
Answer» 0.25 moles methane `(CH_(4))=4G` |
|
| 33. |
What is exchange energy?How it is related with stability of atoms?Explain with suitable example. |
|
Answer» Solution :(1)If TWO or more electrons with the same spin are present in DEGENERATE ORBITALS,there is a possibility for exchanging their positions.During echange process,the energy is released and the released energy is called exchange energy. (2)If more number of exchanges are possible ,more exchange energy is released.More number of echanges are possible only in the case of half filled and fully filled configurations. (3)For example.in chromium,the electronic configuration is `[Ar]3d^(5)s^(1)`.The 3d orbital is half filled and there are ten possible exchanges.  (4) On the other hand only six exchanges are possible for `[Ar]3d^(4)4s^(2)` configuration. (5)Hence,exchanges energy for the half filled configuration is more .This increases the STABILITY of half filled 3d orbitals. |
|
| 34. |
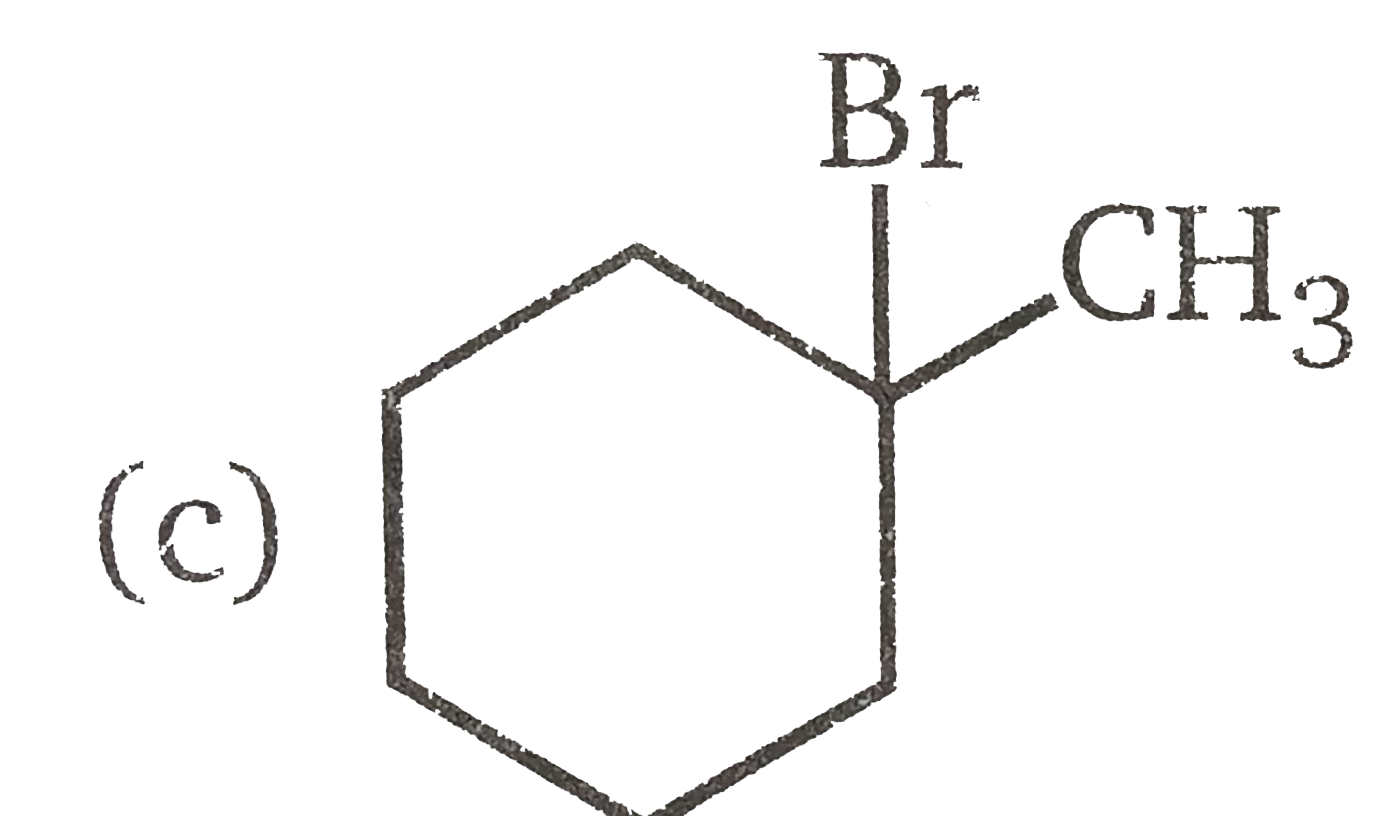
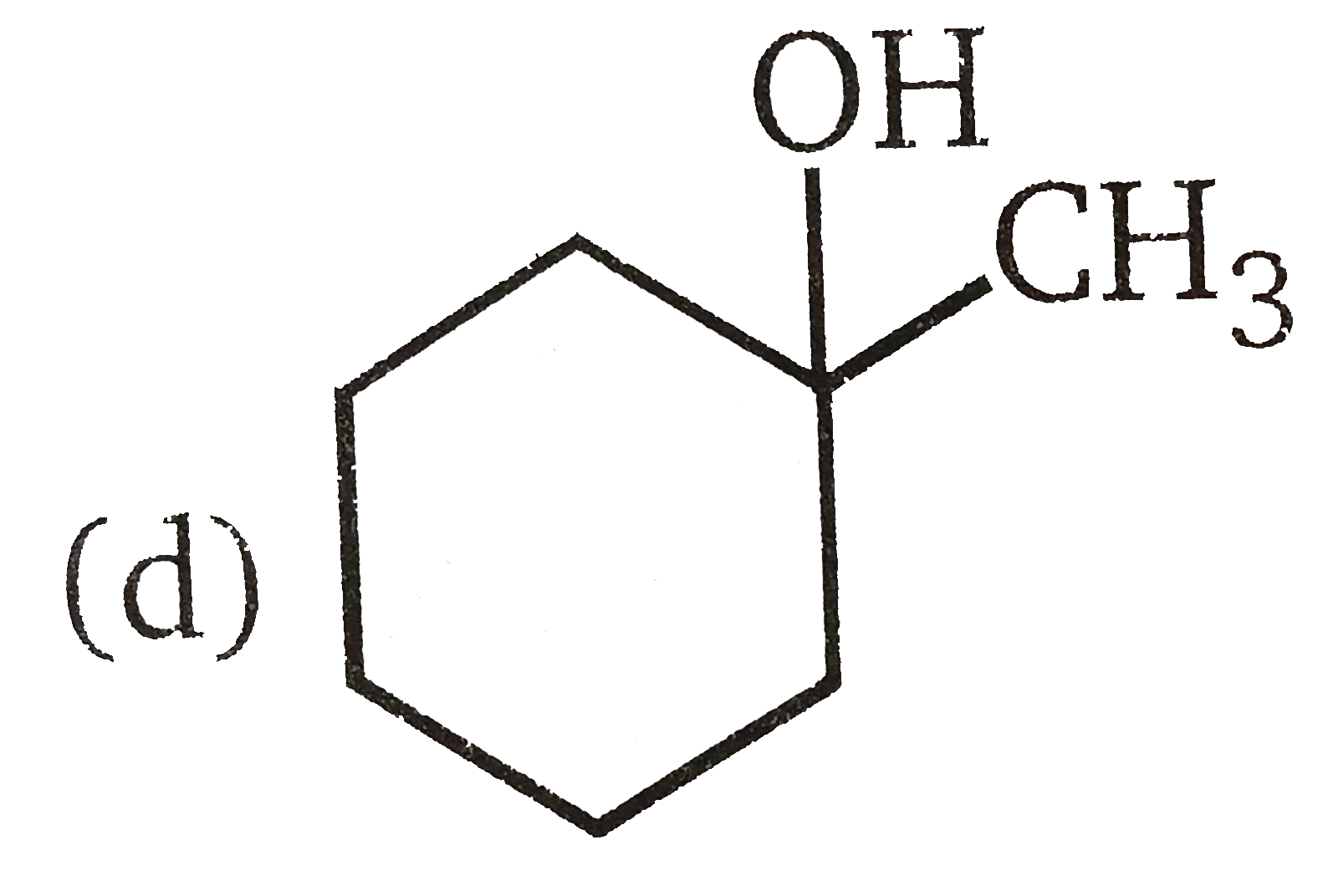
Vladimir Markovnikov rule: Alkenes undergo electrophilic addition reactions. It is triggered by the acid acting as a electrophile toward pi-electrons of the double bond. Markovnikov's rule states that when an unsymmetrically substituted alkene reacts with a hydrogen halide, the hydrogen atom adds to the carbon that has the greater number of hydrogen, e.g., In which of the following reactions, product is racemic mixture? |
|
Answer» `CH_(3)-CH_(2)-CH=CH_(2)overset(HBR)UNDERSET(C Cl_(4)) to ` 
|
|
| 35. |
Vladimir Markovnikov rule: Alkenes undergo electrophilic addition reactions. It is triggered by the acid acting as a electrophile toward pi-electrons of the double bond. Markovnikov's rule states that when an unsymmetrically substituted alkene reacts with a hydrogen halide, the hydrogen atom adds to the carbon that has the greater number of hydrogen, e.g., Identifythe major products r_(1), r_(2) and r_(3) in the given reactions |
|
Answer»

|
|
| 36. |
Vladimir Markovnikov rule: Alkenes undergo electrophilic addition reactions. It is triggered by the acid acting as a electrophile toward pi-electrons of the double bond. Markovnikov's rule states that when an unsymmetrically substituted alkene reacts with a hydrogen halide, the hydrogen atom adds to the carbon that has the greater number of hydrogen, e.g., In which of the following reaction, diastereomers will be formed? |
|
Answer»
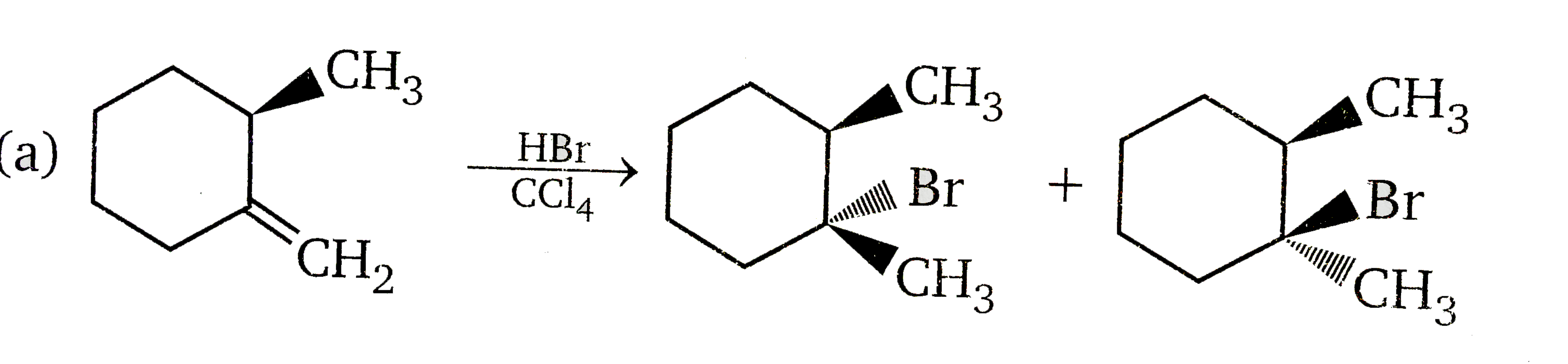  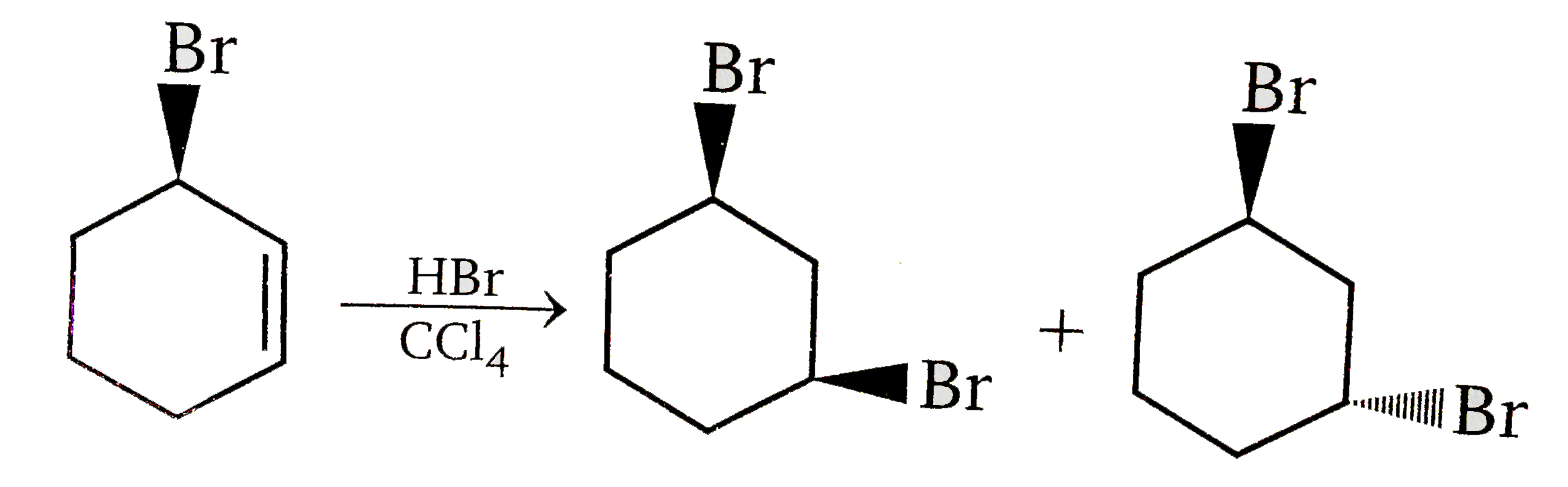
|
|
| 37. |
Whichof the followingcompoundcannot be prepared by kolbe electrolytic method |
|
Answer» `CH_(3)-CH_(3)` |
|
| 38. |
When a mixture of ammonium chloride and potassium dichromate was heated, a stable colourless gass (A) was evolved which did not support combustion, but magnesium continued to burn in it. The gas (A) reacted with calcium carbide in an electric furnace a solid (B) which was slowly hydrolysed by water forming an insoluble substance (C) and the solution of a substance (D) which turned Nessler's reagent brown. Identify substances (A) to (E) and explain all the reactions involved. |
|
Answer» Solution :(i) `2NH_(4)Cl+K_(2)Cr_(2)O_(7)tounderset("Amm.dichromate")((NH_(4))_(2)Cr_(2)O_(7)+2KCl)` (ii) `(NH_(4))_(2)Cr_(2)O_(7)overset("Heat")tounderset((A))(N_(2))+Cr_(2)O_(3)+4H_(2)O` (iii) `3Mg+N_(2)overset(Delta)tounderset("Magnesium nitride")(Mg_(3)N_(2)` (iv) `underset("Calcium carbide")(CaC_(2))+N_(2)overset("Heat")tounderset("Cal.cyanamide (B)")(CaCN_(2))+C` (v) `underset((B))(CaCN_(2))+3H_(2)Otounderset("Cal.carbonate (C)")(CaCO_(3))+underset("Ammonia (D)")(2NH_(3))` (VI) For reaction with NESSLER's reagent, |
|
| 39. |
Write the names of isotopes of hydrogen. What is the mass ratio of these isotopes? |
| Answer» SOLUTION :Protium`("_1^1H)`, Deuterium `("_1^2H)`, and Tritium `("_1^3H)` having the MASS RATIO 1:2:3 | |
| 40. |
The role of using mercury as cathode and anode at the bottom of the tank in the Castner-Kellner cell is |
|
Answer» MERCURY is non TOXIC |
|
| 41. |
When sodium is dissolved in liquid ammonia , a solution of deep blue colour is obtained . The colour of the solution is due to |
|
Answer» ammoniated ELECTRON |
|
| 42. |
Which of the following defects is also known as dislocation defect ? |
|
Answer» frenkel DEFECT |
|
| 43. |
Which of the following statements is incorrect in relation to ionization enthalpy ? |
|
Answer» IONIZATION enthalpy increases for each successive electron |
|
| 44. |
What would be the pH of a solution obtained by mixing 100 ml of 0.1 N HCl and 9.9 ml of 1.0 N NaOH solution ? |
|
Answer» Solution :100 ml of 0.1 N HCl = `100xx0.1 ` milli eq. = 10 milli eq. 9.9 ml of 1 N NaOH `=9.9xx1 ` milli eq. = 9.9 milli eq. `:.` HCl LEFT unneutralized `=10-9.9= 0.1` milli eq. Volume of solution = 100 + 9.9 = 109.9 ml `:.` Normality pf HCl in RESULTING solution `= (0.1)/(109.9)~~(0.1)/(110)=0.09xx10^(-4)N = 9.09 XX 10^(-4)M` As HCl completely ionizes as `HCl + H_(2)O RARR H_(3)O^(+) + Cl^(-)` `:. [H_(3)O^(+)]=[HCl]=9.09xx10^(-4)M` `pH = - LOG [H_(3)O^(+)]=- log (9.09xx10^(-4))=- [ log 9.09 + log 10^(-4)]= - [0.9546-4]` `=3.0454 = 3.05` |
|
| 45. |
The standard hydrogen electrode potential is zero, because |
|
Answer» there is no potential DIFFERNCE between the electrode and the solution |
|
| 47. |
What are the limitations of Bohr's atom model? |
|
Answer» Solution :(i)The Boh.s atom model is applicable only to species having one electron such as hydrogen,`Li^(2+)`ETC and not applicale to multi-electron atoms. (ii)It was unable to explain the SPLITTING of spectral lines in the presence of magnetic field(ZEEMAN effect) or an electric field(Stark effect). (iii)Bohr.s theory was unable to explain why the electron is restricted to revolve around the nucleus in a fixed orbit in which the ANGULAR momentum of the electron is equal to `nh/(2pi)` |
|
| 48. |
Urea was prepared first time in the laboratory by heating |
|
Answer» AMMONIUM cyanate |
|
| 49. |
Unit of K_pfor NH_4COONH_(2(s)) harr 2NH_(3(g))+ CO_(2(g)) is |
|
Answer» No units |
|
| 50. |
Which of the following is incorrect: |
|
Answer»
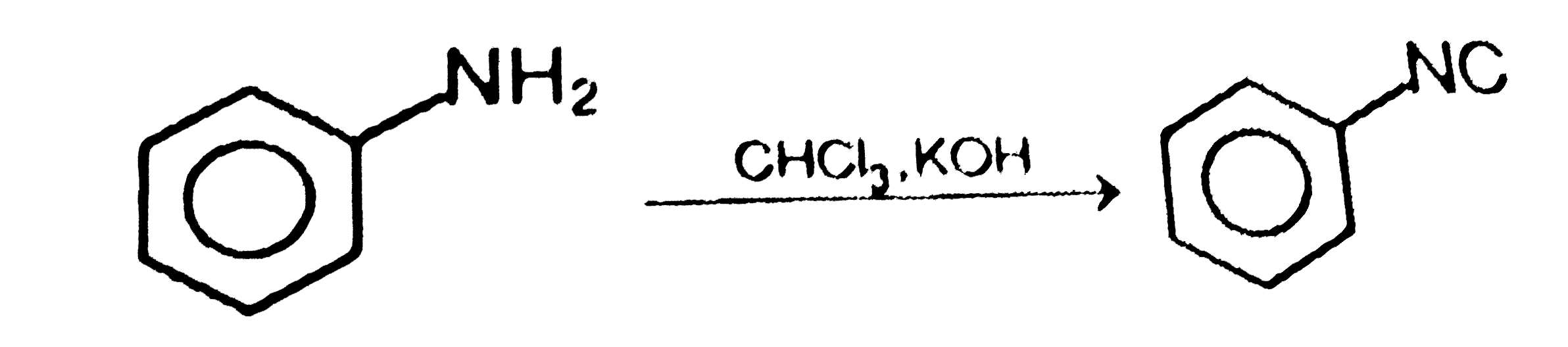
|
|