Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 2. |
When PbS is oxidised by H_(2)O_(2) the increase in oxidation state of sulphur is |
|
Answer» 8 |
|
| 3. |
The temperature above which formation of micelles takes place is known as |
|
Answer» Critical TEMPERATURE |
|
| 4. |
Velocity of the electron in the 1st Bohr orbit |
|
Answer» `2.18xx10^(8)cm//sec` |
|
| 5. |
Write IUPAC name of Parachloro toluene. |
|
Answer» 1-chloro-2-methyl benzene 
|
|
| 6. |
Write the important principles of VSEPR theory to predict the shape of molecules. |
|
Answer» Solution :(i) The shape of a molecule depends upon the no. of electron pairs AROUND the CENTRAL atom. (ii) There is a repulsive force between the electron pairs, which TEND to repel one another. (iii) The electron pairs in SPACE tend to occupy such POSITIONS that they are at maximum distance, so that the repulsive force will be minimum. (iv) A multiple bond is treated as if it is a single bond and the remaining electron pairs which constitute the bond may be regarded as single super pair. |
|
| 7. |
Which of the following is used in Friedel-Craft acylation reaction |
|
Answer» `CH_3COOCH_3` 
|
|
| 8. |
Which of the following is not considered to be a pollutant ? |
|
Answer» `NO_2` |
|
| 9. |
Which of the following physical properties is used to determine the molecular mass of a polymer solution? |
|
Answer» Relative LOWERING of VAPOUR pressure |
|
| 10. |
What is the effect of higher concentration of NO_2 ? |
|
Answer» DAMAGE the leaves of PLANT |
|
| 11. |
Which one of the following sets of units represents the smallest and the largest amount of energy respectively ? |
|
Answer» J and ERG `1eV = 1.6 XX 10^(-19) J ,1 cal = 4.186 J` ` 1 erg = 10^(-7) J , 1 `lit atm` =101.3 J` |
|
| 13. |
Which of the following can decolourless bromine wate solution? |
|
Answer»
|
|
| 15. |
Which of the following fectures are not shown by quartz glass ? |
|
Answer» This is a crystalline solid. |
|
| 16. |
Write the product of following reactions. |
|
Answer» |
|
| 17. |
Write formulas for the following compounds : Iron (III) sulphate |
| Answer» SOLUTION :`Fe_(2)(SO_(4))_(3)` | |
| 18. |
Whichonedecomposesonheating |
|
Answer» `MgCO_(3)` |
|
| 19. |
Where does the new attractive and repulsive force arise in the hydrogen molecule ? |
|
Answer» Solution :Thenew ATTRACTIVE forces ( green ARROWS ) arise between (i) Nucleus of `H_(a)` and valence electron of `H_(B)` (ii) Nucleus of `H_(b)` and thevalence electron of `H_(a)`. The NEW repulsive forces ( red arrows ) arise between (i) The nucleus of `H_(a)" and " H_(b)` (ii) Valence electrons of `H_(a) " and " H_(b)` . |
|
| 20. |
Water is present in [Cr(H_2O)_6]^(3+).3Cl^(-) in the form of _________. |
|
Answer» |
|
| 21. |
The specific heat at constant volume for a gas 0.075cal//g and at constant pressure is 0.125cal//g Calculate : (i) The molecular weight of gas, (ii) Atomicity of gas, (iii) No. ofatoms of gas in its 1 mole. |
|
Answer» `(iii) 6.023xx10^(23)at OMS.` |
|
| 22. |
Thermal decomposition of ammonium dichromate gives |
|
Answer» `N_(2),H_(2)OandCr_(2)O_(3)` |
|
| 23. |
Write the products of following reactions |
|
Answer» |
|
| 24. |
Which of the following statements about a compound is incorrect? |
|
Answer» A molecule of a COMPOUND has ATOMS of different elements |
|
| 25. |
The thermochemical equations for solid and liquid rocket fuels are given below : (i) 2AI(s) 3/2O_(2)(g) to AI_(2)O_(3)(s), DeltaH = -1667.8 kJ (ii)H_(2)(g) + 1/2O_(2)(g) to H_(2)O(l), DeltaH = -285.9 kJ (a) If equal masses of aluminium and hydrogen are used, which is better rocket fuel? (b) Determine DeltaH for the reaction, AI_(2)O_(3)(s) to 2AI(s) + 3/2 O_(2)(g). |
|
Answer» Solution :According to reaction (i) `2AI (s) + 3/2O_(2)(g) to AI_(2)O_(3)(s)DeltaH = -1667.8 kJ` Heat evolved in the combustion of `(2 XX 27) 54` g ofAI = 1667.8 kJ. According to reaction(ii) `H_(2)(g) + 1/2O_(2)(g) to H_(2)O(l) DeltaH = -285.9 kJ` Heat evolved in the combustion of 2 g of hydrogen = 285.9 k.J MAKIN equal masses, (a)Heat evolved in the combustion of 54 g of hydrogen ` = (285.9 xx 54)/(2) = 7719.3 kJ`. `therefore` Hydrogen is a better rocket fuel because heat evolvedfrom 54 g of`H_(2)` is more than that `DeltaH` for the reaction: `AI_(2)O_(3)(s) to 2AI(s) + 3/2 O_(2)(g) DeltaH = `? This is the reverse of equation (i). Therefore, `DeltaH` for this reaction is `DeltaH = + 1667.8 kJ`. |
|
| 27. |
Which of the following is correct about a solution showing positive deviation? |
|
Answer» VAPOUR PRESSURE observed will be the less than that calculated from Raoult's LAW |
|
| 28. |
Which is a good fuel CH_(4) or C_(2)H_(6) ? Thestandard enthalpy of formation ofCH_(4),C_(2)H_(6),CO_(2) and H_(2)O are -74.8, -84.6, -393.5 and -286kJ mol^(-1) respectively . |
|
Answer» |
|
| 29. |
Which of thefollowingprocessesinvolves absorption ofenergy ? |
|
Answer» `C1 + E^(-)to C1^(-)` |
|
| 30. |
What are semi - metal ? Give example. |
|
Answer» SOLUTION :Some ELEMENTS in the periodic table show properties that are characteristic of both METALS and non-metals. These elements are called semi-metals or metalloids. Example: SILICON, germanium, arsenic, antimony and TELLURIUM. |
|
| 31. |
Which of the following does have minimum mass in grams ? (i) 1 gram atom of oxygen (ii) 4.2 gram molecules of H_2 (iii) 2 gram molecules of helium (iv) 1.6 gram atoms of chlorine |
|
Answer» |
|
| 32. |
Three pairs of compounds aregiven below, identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. (i) TlCl_(3).TlCl (ii) AlCl_(3).AlCl (iii) InCl_(3).InCl |
|
Answer» SOLUTION :(a) TlCl more stable than `TlCl_(3)` due to inert pair EFFECT. `TlCl_(3)` is less stable and covalent in nature but TlCl is more stable and ionic is nature (B) Due to absence of d-orbitals, Al does not show inert pair effect. Hence, its most stable oxidation state is +3. Thus, `AlCl_(3)` is much more stable then AlCl. Further, in the solid or the vapour state `AlCl_(3)` covalent in nature but in aqueous solutions, it ionises to form A `Al^(3+)(aq) and Cl^(-)(aq)` ions (c) Due to inert pair effect, INDIUM exists in both +1 and +3 oxidation states out of which +3 oxidation state is more stable is more stable than +1 oxidation state. In other words, `InCl_(3)` is more stable than InCl. Being unstable, In Cl undergoes disproportionation REACTION `3InCl(aq)rarr2ln(s)+ln^(3+)(aq)+3Cl^(-)(aq)`. |
|
| 33. |
The valencyshell configuration of IV A element is |
|
Answer» `ns^(2)np^(1)` |
|
| 34. |
Which of the following is correct about the following compound |
|
Answer» All the `C-C` BOND length are same |
|
| 35. |
Which one is the preferred structure of CO_(2)? |
|
Answer» |
|
| 36. |
Two gases have densities 0.388 g L^(-1) and 0.097 g L^(-1). What is the rate of diffusion of the first gas, if the second gas diffuses at the rate 10 cc s^(-1) under similar conditions ? |
| Answer» SOLUTION :5 CC `s^(-1)` | |
| 37. |
What is meant by nodal surface? |
|
Answer» SOLUTION :i.The region where there is probability density function reduces to zero is called NODAL SURFACE or a RADIAL node. ii.For ns orbital (n-1)nodes are FOUND in it. |
|
| 38. |
What is the shape of pi-electron cloud in ethene and benzene ? |
Answer» SOLUTION :In benzene, `pi`-ELECTRON cloud ARRANGE symmetrically above an beaneath all the SIX carbon ATOMS and form hexagonal shape.  In ethyne, `pi`-electron cloud arrange in cylinderical manner between two symmetrical carbon of ethyne. 
|
|
| 39. |
Write the structures of following alkanes. 5-(2-Ethyl butyl)-3, 3-dimethyldecane |
| Answer» Solution :`CH_(3)-CH_(2)-CH_(2)-CH_(2)-UNDERSET(C_(2)H_(5))underset(|)underset(H_(3)C-C-CH_(3))underset(|)underset(CH_(2))underset(|)(CH)-CH_(2)-OVERSET(CH_(2)-CH_(3))overset(|)(C )H-CH_(2)-CH_(3)` | |
| 41. |
What is meant by boiling point and condensation point of the liquid? |
| Answer» SOLUTION :The TEMPERATURE at which the liquid and vapour phases are at equilibrium is called the BOILING point and CONDENSATION point of the liquid. | |
| 42. |
What volumes of 10N NaOH and 2N NaOH should be mixed to obtain 4L of 5N |
|
Answer» |
|
| 43. |
What is K_(c) for the follwoing reaction in state of equilibrium ? 2SO_(2)(g)+O_(2)(g) hArr 2SO_(3)(g) Given [SO_2] = 0.6M,[O_2]=0.82 M, and [SO_3] =1.90 M |
|
Answer» Solution :`2SO_(2)(g) + O_(2)(g) hArr 2SO_(3)(g)` `K_(c) =([SO_3]^2)/([SO_2]^2[O_2]) = ((1.9 M )XX( 1.9 M))/((0.6 M)xx(0.6 M) xx (0.82 M))` `= 12.229 M^(-1) =12.229 MOL^(-1)` |
|
| 44. |
Which of the following statement is correct in relation to the hydrogen atom : |
|
Answer» 3s-orbital is LOWER in ENERGY than 3P-orbital |
|
| 45. |
When 200 ml 0.2M NaCl and 200 ml 0.4M BaCl_(2) and 100 ml 0.2 M KCl are mixed. {:("Column-I","Column-II"),("A) molarity of "Na^(+)" ion","P) 0.04 M"),("B) molarity of "Ba^(+2)" ion","Q) 0.08 M"),("C) molarity of "Cl^(-)" ion","R) 0.16 M"),("D) molarity of "K^(+)" ion","S) 0.44 M"):} |
|
Answer» B) `[Ba^(+2)]=(200xx0.4)/(500)=0.16M` C) `[Cl^(-)]=(200xx0.2+200xx0.4xx2+100xx0.2)/(500)` = `0.44M` D) `[K^(+)]=(100xx0.2)/(500)=0.04M` |
|
| 46. |
Write the reversible reaction taking place betweenferric ions and thiocyanate ions and write the colour of each reactant and product. |
| Answer» Solution :`{:(Fe^(3)(aq),+,SCN^(-)(aq),hArr,[Fe(SCN)]^(2+)),("(YELLOW)",,"(COLOURLESS)",,"(Reddish brown)"):}` | |
| 47. |
When H_(2)S is passed through Hg_(2)^(2+), we get : |
|
Answer» `HGS` |
|
| 48. |
What should be the ratio of concentration of acetic acid to sodium acetate while preparing an acid buffer mixture with pH = 5.7? (K_(a) for acetic acid is 1.8xx10^(-5)) |
|
Answer» Solution :`pK_(a)=-logK_(a)` = `-log1.8xx10^(-5)` = +4.6447 `pH=pK_(a)+"log"(["Salt"])/(["Acid"])` `5.7=4.7447+"log"(["Salt"])/(["Acid"])` `"log"(["Salt"])/(["Acid"])=5.7-4.7447=0.9553` `(["Salt"])/(["Acid"])="ANTI"log0.9553=9.02~~9` `therefore` Ratio of concentration of acid : salt = 1 : 9 |
|
| 49. |
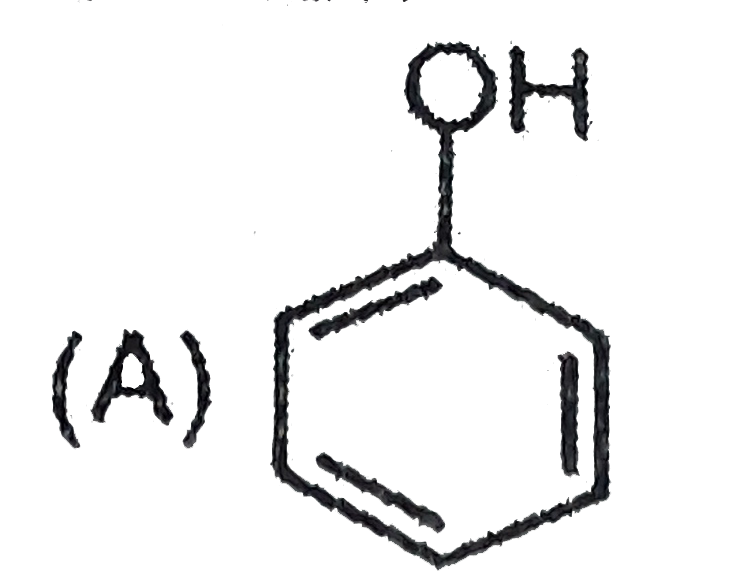
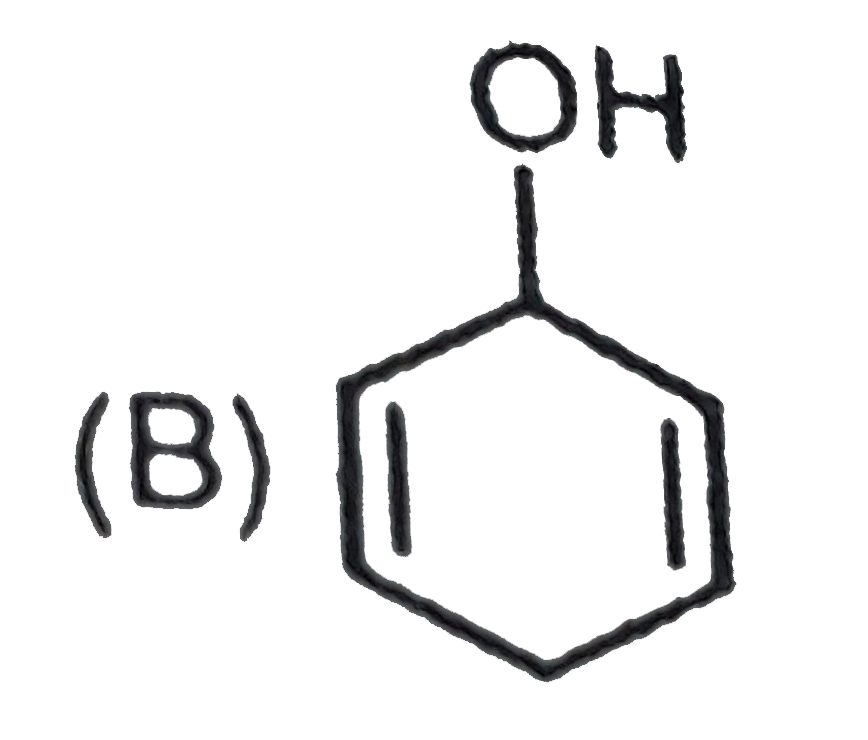
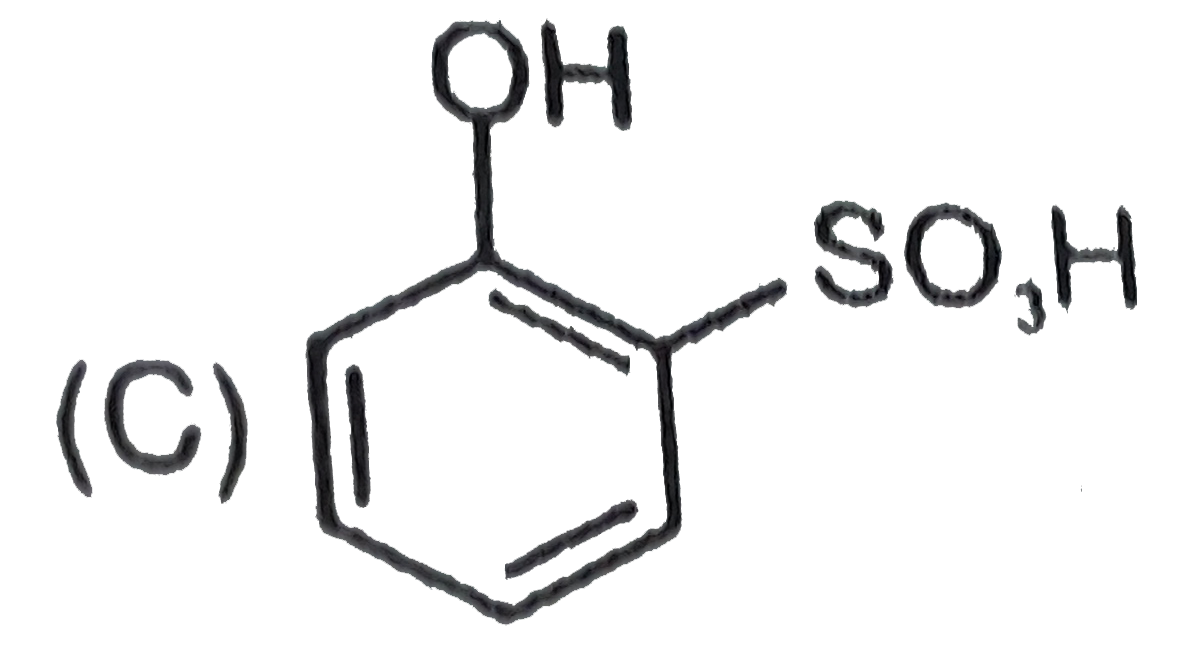
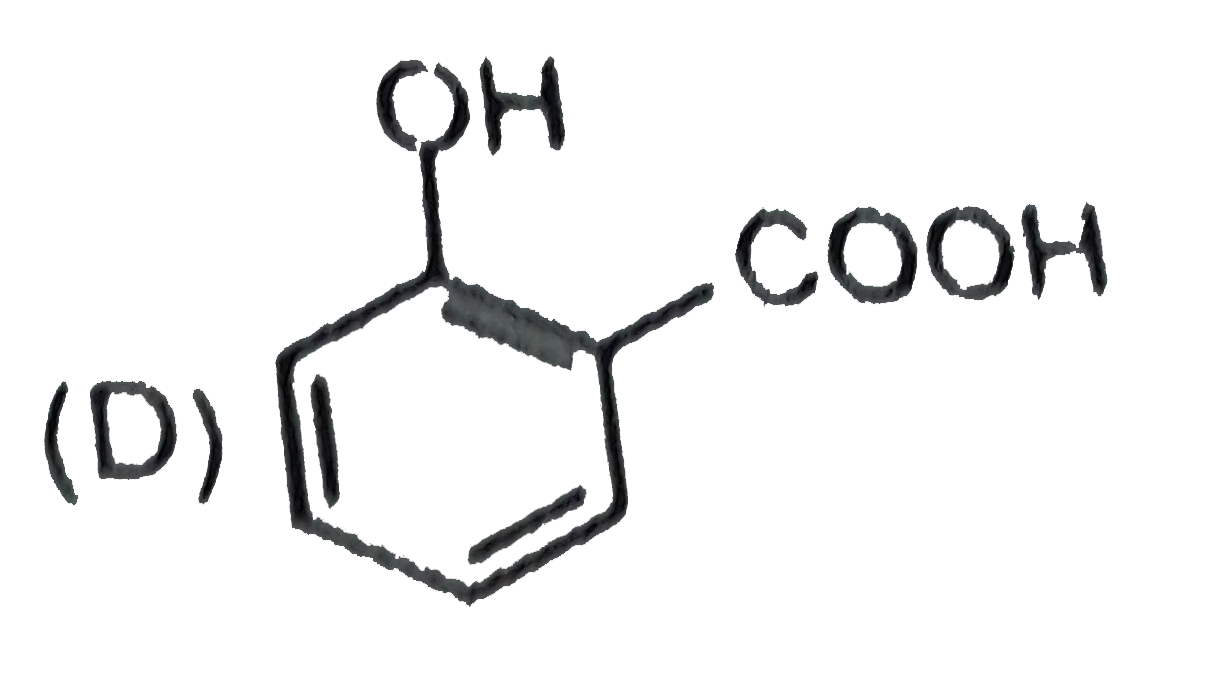
Which one of the following compounds has the most acidic nature ? |
|
Answer»
|
|