Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
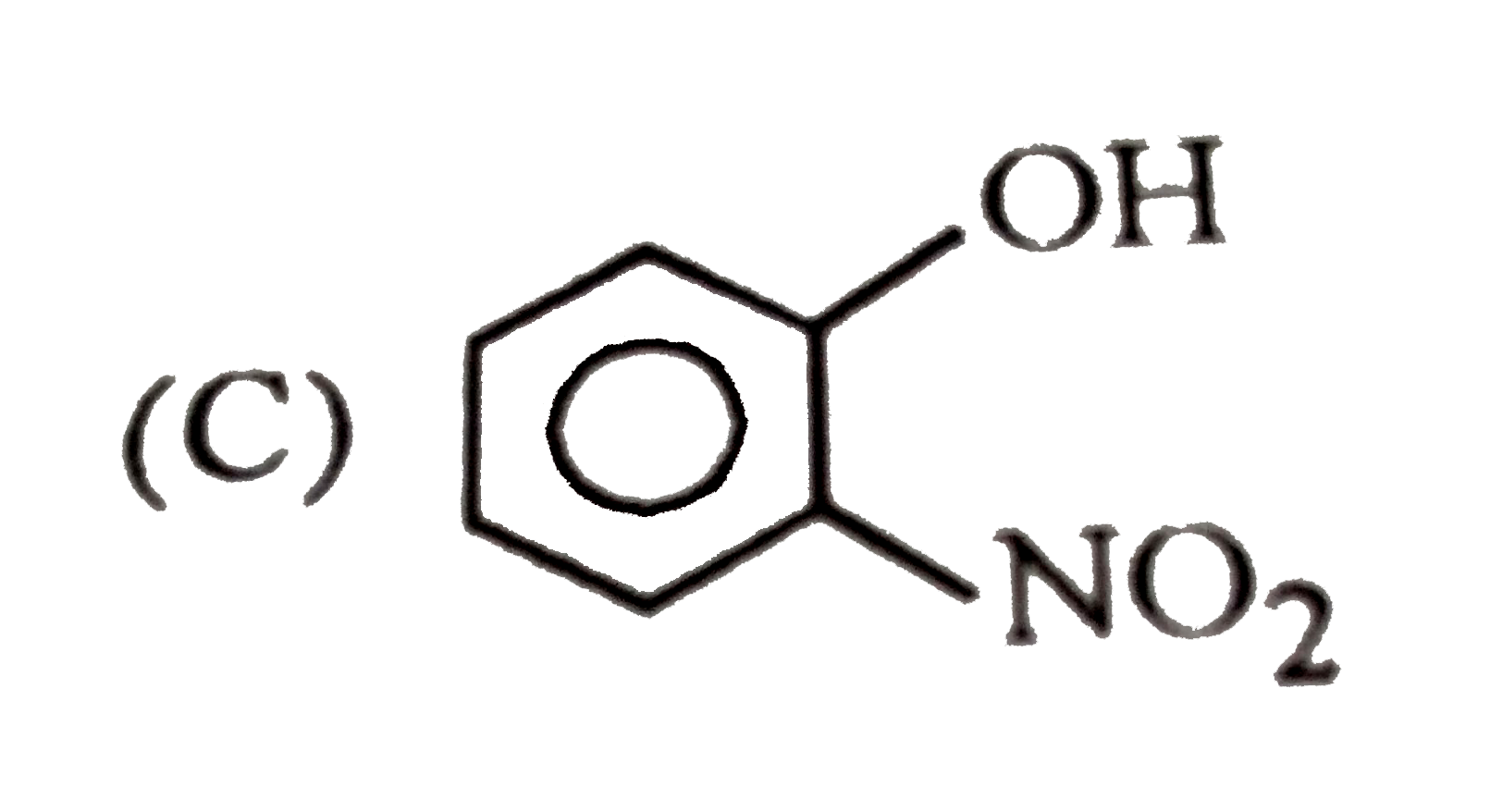
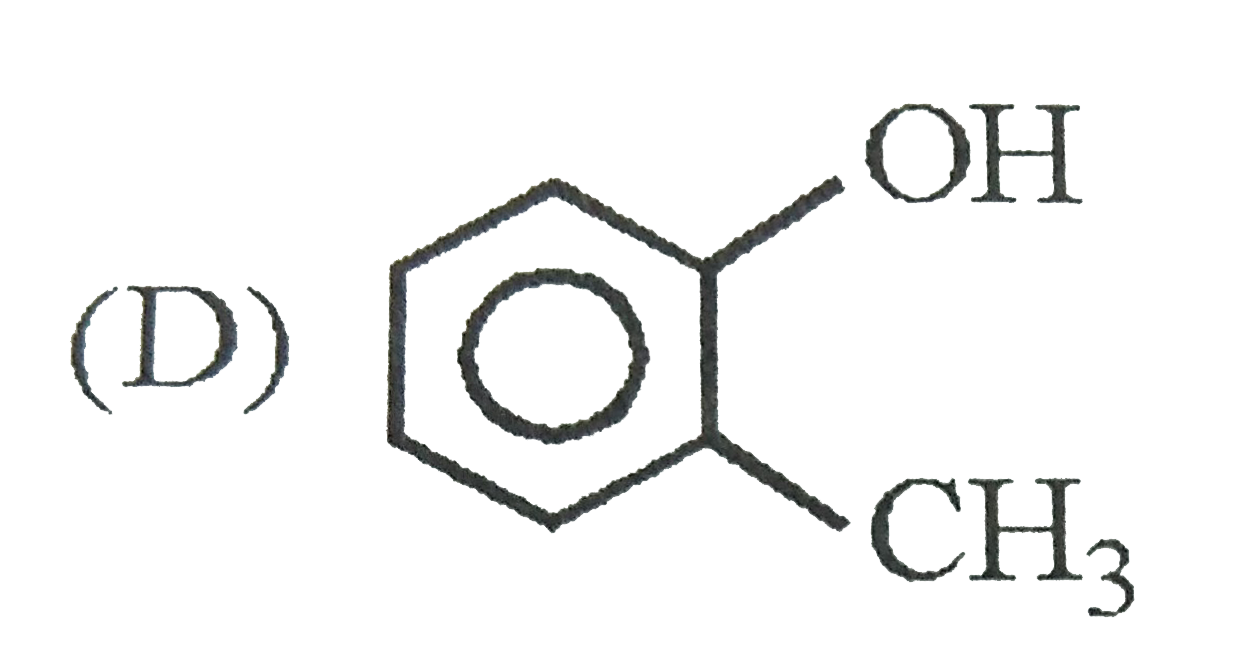
| 1. |
Which one of the following has largest size? |
|
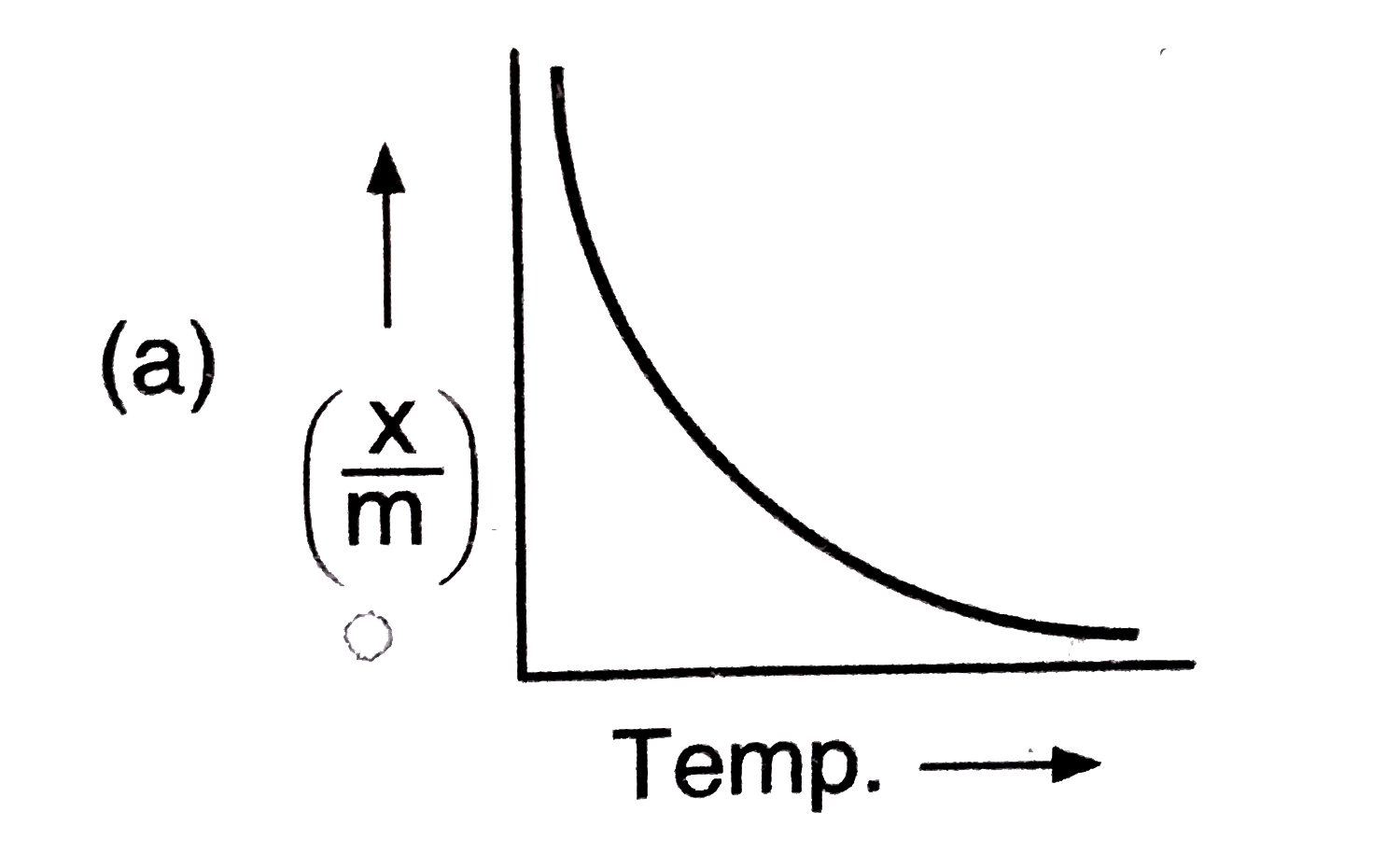
Answer» `NA` |
|
| 2. |
Which of the following specieswill have the largest and smallest size Mg,Mg^(2+),Al,Al^(3+)?Give reason. |
|
Answer» Solution : The largest SPECIES is Mg The smallest species is `Al^(3+)`Reason : Atomic RADII DECREASES across a period. Cations are smaller thantheir parent atoms. Among the ISOELECTRONIC ions , the greater the charge on the cation smaller will be the radius. |
|
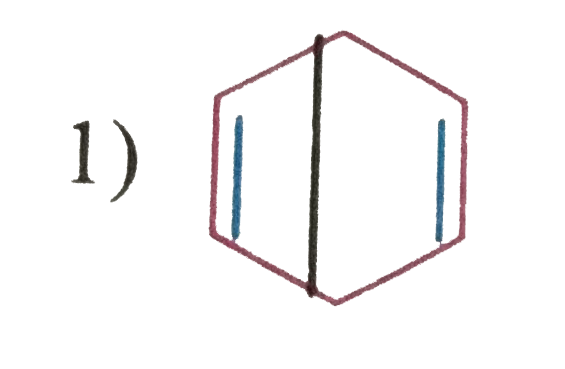
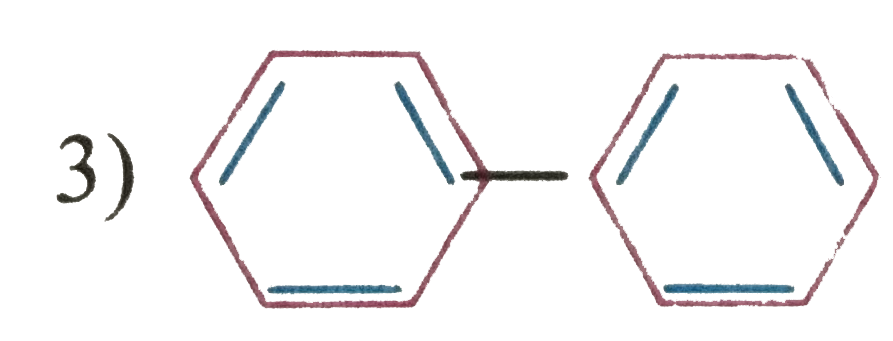
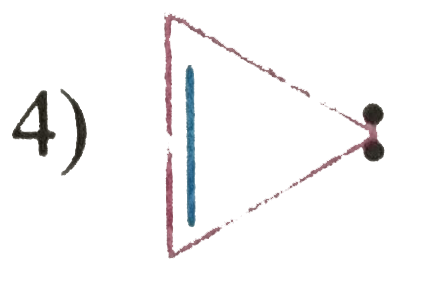
| 3. |
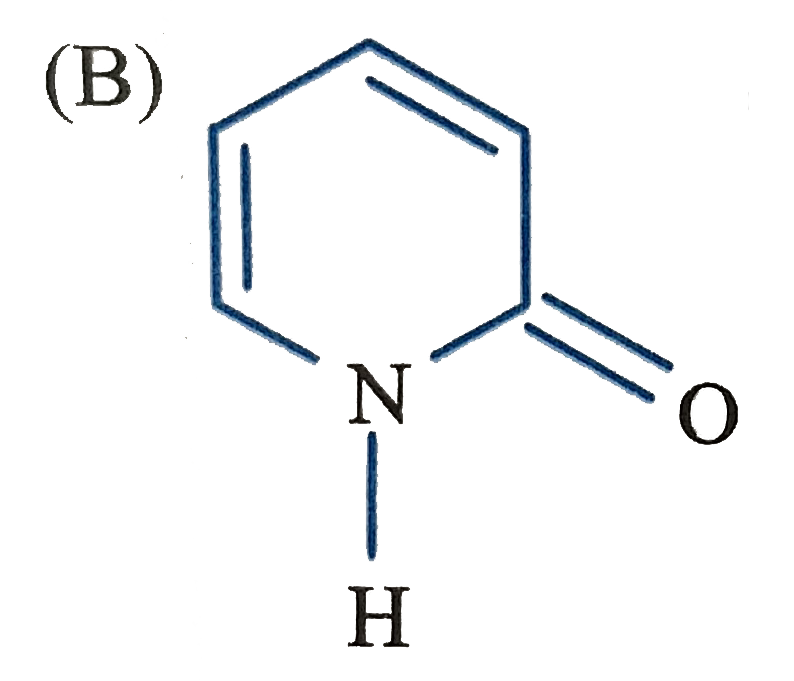
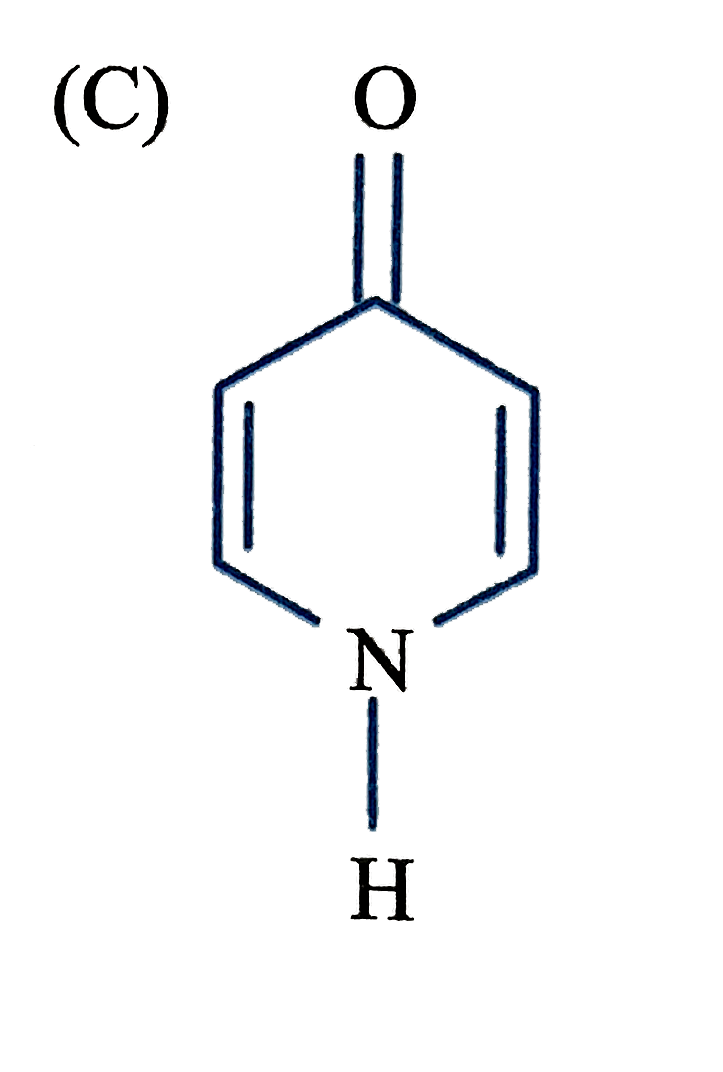
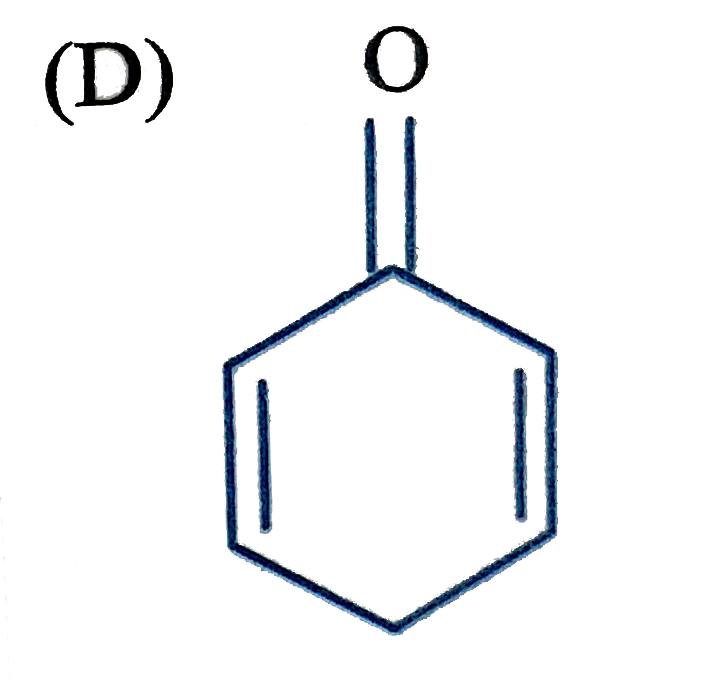
Which of the following structure will not have 4pi electrons |
|
Answer»
|
|
| 4. |
when benzyyl choride is treemrnt with ethanlolic KCN, the major product formed is : |
|
Answer» Benzyrhtyl ehter |
|
| 5. |
Which of the following does not depict properties of fullerenes? |
|
Answer» Fullerenes are MADE by heating graphite |
|
| 6. |
What will be the work done on an ideal gas enclosed in a cyliner, when it is compressed by a constant external pressure, p_("ext") in a single step as shown in figure ? Explain graphically. |
|
Answer» Solution :Suppose total volume of the gas is `V_(i)` and pressure of the gas inside CYLINDER is p. After compression by constant external pressure, `(p_("ext"))` in a single step, final volume of the gas becomes `V_(f)`. Then volume change, `Delta v = (V_(f) - V_(i))` If W is the work done on the system by movement of the piston, then `W = P_("ext") (-Delta V)` `W = - p_("ext") (V_(f) - V_(i))` This can be calculated from `p - V` GRAPH as shown in the figure. Work done is equal to the shaded are `ABV_(f)V_(i)` 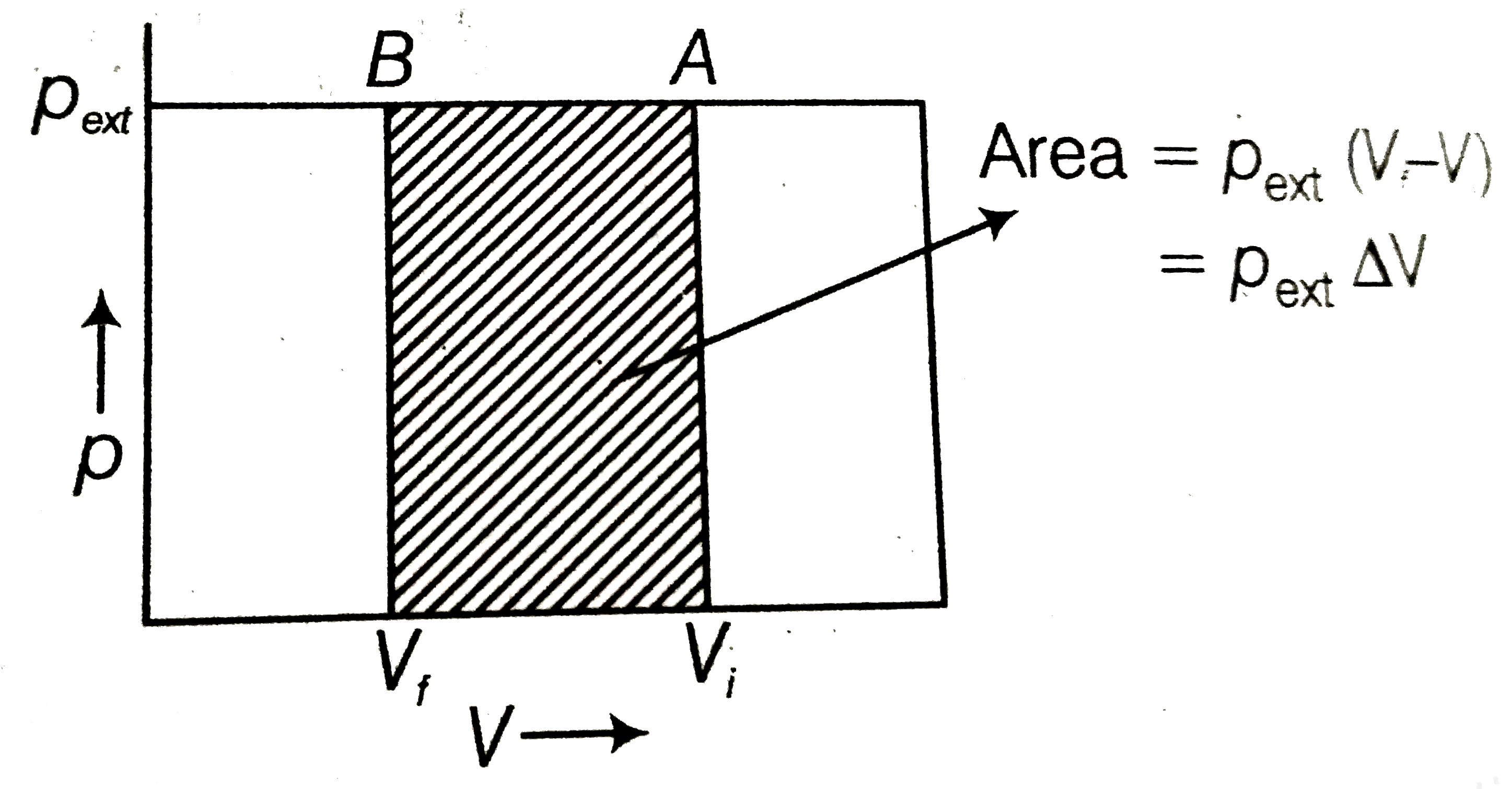 The negative sign in this expression is required to obtain CONVENTIONAL sign for W which will be POSITIVE. Because in case of compression work is done on the system, so `Delta V` will be negative |
|
| 7. |
When ethyl bromide is heated with sodium in dry ether solvent, the alkane obtained is |
|
Answer» Butane |
|
| 8. |
What do you understand by stoichiometric coefficients in a chemical equation ? |
|
Answer» SOLUTION :The co-effecients of REACTANTS and PRODUCTS INVOLVED in a chemical equation represent by the balanced from are known as stoichiometric co-efficients. Eg: `N_(2)(g)+3H_(2)(g) rarr 2NH_(3)(g)` The stoichiometric co-efficients are 1, 3 and 2 respectively. |
|
| 9. |
Which of the combination of reaction result in formation of an alkene. |
|
Answer» (I) (IV) Q |
|
| 10. |
Which can not exhibit tautomerism here |
|
Answer»
|
|
| 11. |
Which of the following orders for electron affinity is/are correct? (a)S gt O lt Se ,(b)Cl gt F , (c) S gt O , (d) O gt S, (e)N gt P , (f) C gt N |
|
Answer» a,B |
|
| 12. |
When pressure of 0.09 mole will become half then calculate volume at 300 K ? |
| Answer» Solution :ACCORDING to Boyle.s law if PRESSURE doubled then VOLUME BECOMES half (As temperature and volume CONSTANT). | |
| 13. |
Whichamong thefollowingspecieshas thesamenumber of theelectronsin itsoutermostas well aspenultimate shell ? |
|
Answer» `MG^(2+)` |
|
| 14. |
When does the carbon in an alkene molecule acquires a positive charges ? Explain . |
|
Answer» Solution :When an ELECTROPHILE such as ` H^(+)`APPROACHES an alkene molecule , the ` pi` ELECTRONS are instantaneously shifted to the electrophile and a new bond is FORMED between carbon and hydrogen . This makes the other carbon electron decient and hence it acquires a POSITIVE charges . ` (##SUR_CHE_XI_V02_C12_E02_058_S01.png" width="80%"> |
|
| 15. |
What are the harmful effects of chemical water pollutants? |
|
Answer» Solution :(i) Cadmium and mercury can cause kidney damage. (II) Lead poisoning can lead to SEVERE damage of kidneys, liver and brain. It also affects the central NERVOUS system. (III) Polychlorinated biphenyl causes skin diseases and are CARCINOGENIC in nature |
|
| 16. |
Towards electrophilic reagents |
|
Answer» ethene is more reactive then ethyne |
|
| 17. |
Write the formula of the compound of iodine which is obtained when conc. HNO_(3) oxidises I_(2). |
| Answer» Solution :`{:(2HNO_(3)toH_(2)O+2NO_(2)+[O]xx5),(I_(2)+5[O]=I_(2)O_(5)),(I_(2)O_(5)+H_(2)Oto2HIO_(3)):}/{:I_(2)+10HNO_(3)tounderset("IODIC ACID")(2HIO_(3))+10NO_(2)+4H_(2)O:}` | |
| 18. |
Which one of the following is a metal? |
|
Answer» N |
|
| 20. |
What is system ? |
| Answer» Solution :A very SMALL part of UNIVERSE in which experiments are done and OBSERVATIONS are made, this small part of universe is called SYSTEM. | |
| 21. |
When KMnO_(4) acts as an oxidizing agent and ultimately forms MnO_(4)^(-2),MnO_(2),Mn_(2)O_(3)andMn^(+2), then number of electrons transfered in each case respectively is _____ |
|
Answer» 4, 3, 1, 5 |
|
| 22. |
UV radiations bring about |
| Answer» Solution :U.V radiation brings about skin cancer. | |
| 23. |
Which of the following set of quantum number represent the 19th electron in chromium (Z = 24 for Cr) |
|
Answer» 4, 0, 0, `(1)/(2)` 19th electron enters 4s because 4s is filled earlier than 3d. For `4s, n = 4, L = 0, m = 0, s = + (1)/(2) or - (1)/(2)` |
|
| 24. |
Which is the following shows maximum -I effect? |
|
Answer» `-CH_(3)` |
|
| 25. |
Which one of the compound behaves as an electrophile as well as nucleophile. |
| Answer» Answer :A | |
| 26. |
Though Cl has nearly same electronegativeity as N, yet there is no H-bonding is HCl . Why ? |
| Answer» SOLUTION :CHLORINE ATOM has a LARGE SIZE . | |
| 27. |
The unit of 'b' in Vander Waals equation of state. |
|
Answer» |
|
| 28. |
What is water pollution ? Give a detailed account on causes and harmful effects of water pollution. |
|
Answer» Solution :Water pollution is defined as "The addition of foreign substances or factors like heat which degrades the quality of water, so that it becomes health hazard or unfit to use." (i) The water pollutants originate from both natural and human activities. The source of water pollution is classified as Point and Non-point source. (ii)Easily identified source of place of pollution is called as point source. Example `:` municipal and industrial discharge pipes. Non-point source cannot be identified easily. Example`:` agricultural runoff, mining wastes, ACID rain, and storm -water drainage and construction sediments.  ( a) Causes of water pollution `:` (i) Microbiological ( Pathogens ) `:` Disease causing microorganisms like bacteria, viruses and protozoa are most serious water pollutants. They come from domestic sewage and animal excreta. Fish and shellfish can BECOME contaminated and people who eat them can become ill. Some serious diseases like polia and CHOLERA are water borne diseases. Human excreta contain bacteria such as Escherichia coli and Streptococcus faecalis which cause gastrointestinal diseases. (ii) Organic wastes `:` Organic matter such as leaves, grass, trash etc can also pollute water. Water pollution is caused by excessive phytoplankton growth within water.Microorganisms present in water decompose these organic matter and consume dissolved oxygen in water. (iii)Eutrophication `:` Eutrophication is a process by which water bodies receive excess nutrients that stimulates excessive plant growth ( algae, other plant weeds ) . This enhanced plant growth in water bodies is called as algae bloom. The growth of algae in extreme abundance covers the water surface and reduces the oxygen concentration in water. Thus,bloom-infested. (b) Biochemical oxygen demand (BOD) `:` (i)The total amount of oxygen in MILLIGRAMS consumed by microorganisms in decomposing the waste in one litre of water at `20^(@)C` for a period of 5 days is called biochemical oxygen demand ( BOD) and its value is expressed in ppm. < br> ( c ) Chemical wastes `:` (i) A whole variety of chemicals from indusctries, such as metals and solvents are poisonous to fish and other aquatic life. (ii) Some toxic pesticides can accumulate in fish and shell fish and poison the people who eat them. Detergents and oils float and spoil the water bodies. Acids from mine drainage and salts from various sources can also contaminate water sources. Harmful effects of chemical water pollutants `:` (i) Cadmium and mercury can cause kidney damage. (ii) Lead poisoning can leads to the severe damage of kidneys, liver, brain etc. it also affects central nervous system. (iii) Polychlorinated biphenyls ( PCBs) causes skin diseases and are carcinogenic in nature. |
|
| 29. |
When a brown compound of Mn (A) is treated with HCl, it gives a gas (B). The gas (B) taken in excess reacts with NH_(3) to give an explosive compound (C). The compouns A, B and are |
|
Answer» `A=MnO_(2),B=Cl_(2),C=NCl_(3)` `NH_(3)+3underset((B))(Cl_(2))tounderset((C))NCl_(3)+3HCL` |
|
| 30. |
Which of the following undergoes reduction with hydrogen peroxide in an alkaline medium ? |
| Answer» SOLUTION :`HOCl to Cl_2` | |
| 31. |
Which of the following compounds will most readily be dehydrated to give alkene under acidic condition ? |
|
Answer» 4-Hydroxypentan-2-one |
|
| 32. |
Which of the following hydrocarbon is most difficult to prepare by Wurtz's reaction? |
|
Answer»
|
|
| 34. |
What would be the expected product of the reaction of propyne with Br_2//H_2O if the mechanism of this reaction is analogous to that of of propene ? |
|
Answer» 2-Bromopropenol 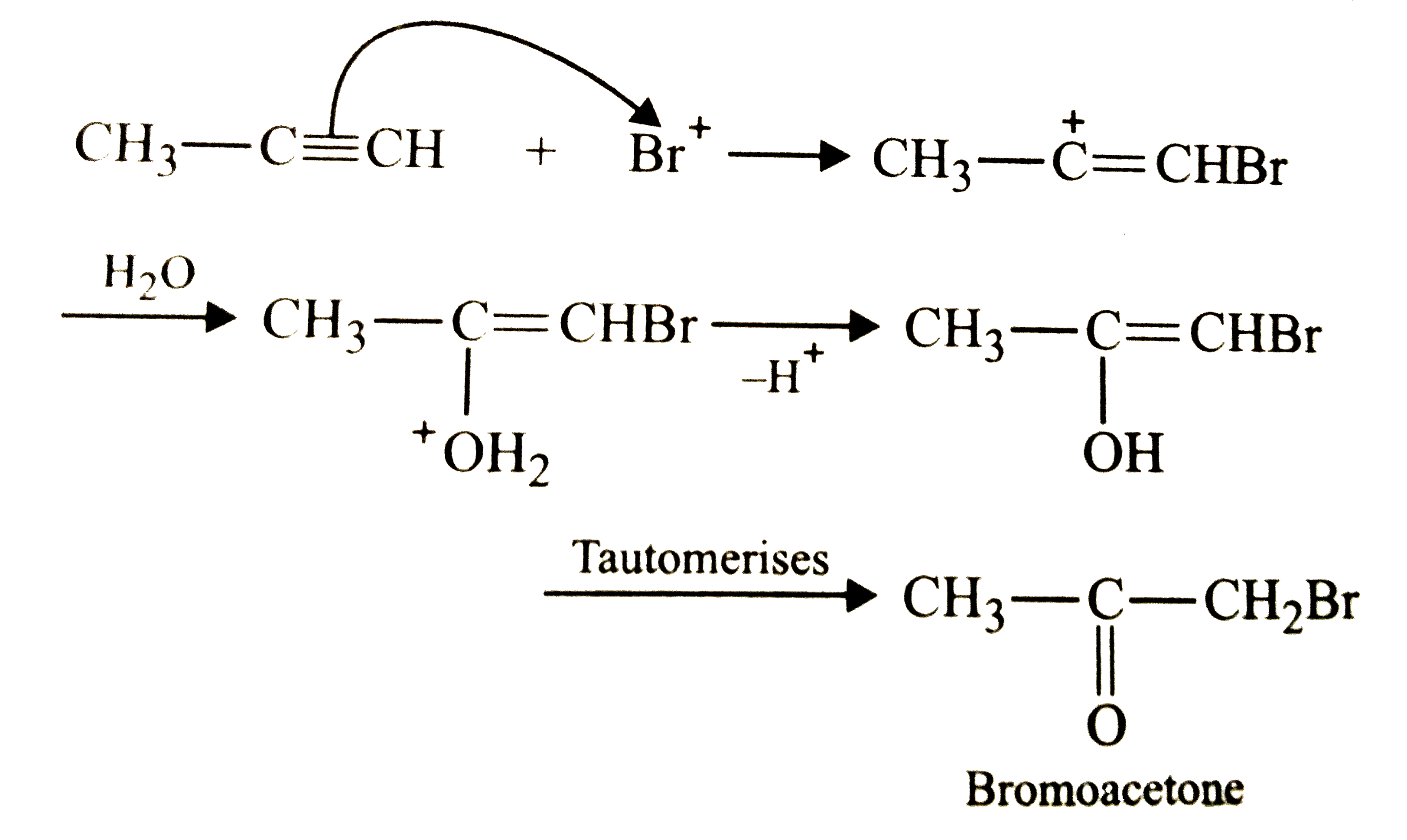
|
|
| 35. |
What is fuming nitric acid? What colour is it? |
| Answer» Solution :Concentrated nitric ACID in which oxides of NITROGEN `(i.e., NO, NO_(2),N_(2)O_(3), etc)` are DISSOLVED is called fuming nitric acid. Its colour is yellow. | |
| 36. |
Write the formulae of borax and boric acid. |
|
Answer» `Na_2 B_4 O_7 * 4H_2 O` |
|
| 37. |
Total number of species which exhibit mesomersim (Conjugation) in the following species are |
Answer» 
|
|
| 38. |
Which of the following reaction will be least forward ?(i)N_(2(g)) +O_(2(g)) hArr 2NO_((g)) , K_c=4.8xx10^(-31) at 298 K (ii)Fe_((aq))^(3+) +SCN_((aq))^(-) hArr [Fe(SCN)]^(2-) , K_c= 140 at 298 K (iii)H_(2(g)) + Cl_(2(g)) hArr 2HCl_((g)), K_c=5xx10^31 at 298 K |
| Answer» SOLUTION :(i)`K_c` of this REACTION is much less so it is so FAR of COMPLETION. | |
| 39. |
What is retrograde solubility ? |
| Answer» Solution :Gypsum is a soft mineral and it is less soluble in water as the TEMPERATURE in known as RETROGRADE SOLUBILITY, which is a DISTINGUISHING characteristic of gypsum | |
| 40. |
What do you understand by bond pairs and lone pairs of electrons? Illustrate by giving one example of each type. |
|
Answer» Solution :The SHARED pair of electrons PRESENT between TWO covalently bonded atoms are called bond pairs, while the electron pairs on the atoms that do not TAKE part in bonding are called lone pairs. Example: 
|
|
| 41. |
Value of Delta G is -ve for ice to water system at .......... temperature. |
|
Answer» `260` K |
|
| 42. |
Which one of the following compounds is most acidic ? |
|
Answer» `Cl-CH_(2)-CH_(2)-OH` |
|
| 43. |
What is dead burnt plaster? |
| Answer» Solution :Above 393K, no water of CRYSTALLIZATION is l eft and anhydrous calcium sulphate, `CaSO_(4)`is formed. Thhis is KNOWN as .dead BURNT plaster.. | |
| 44. |
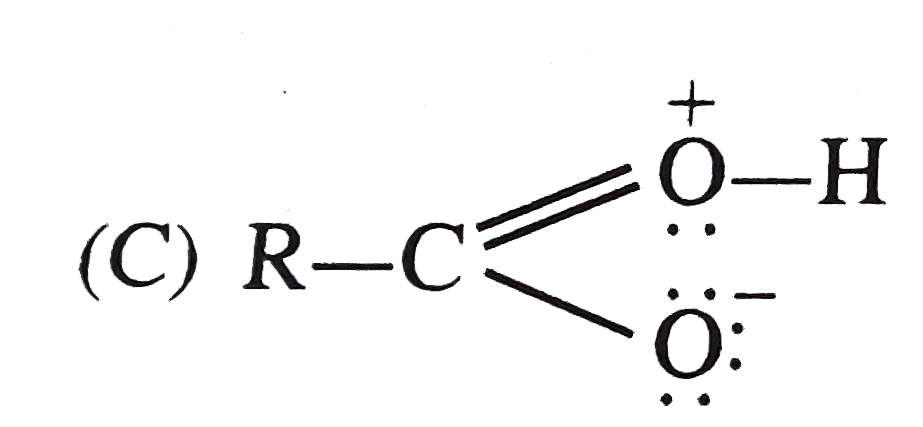
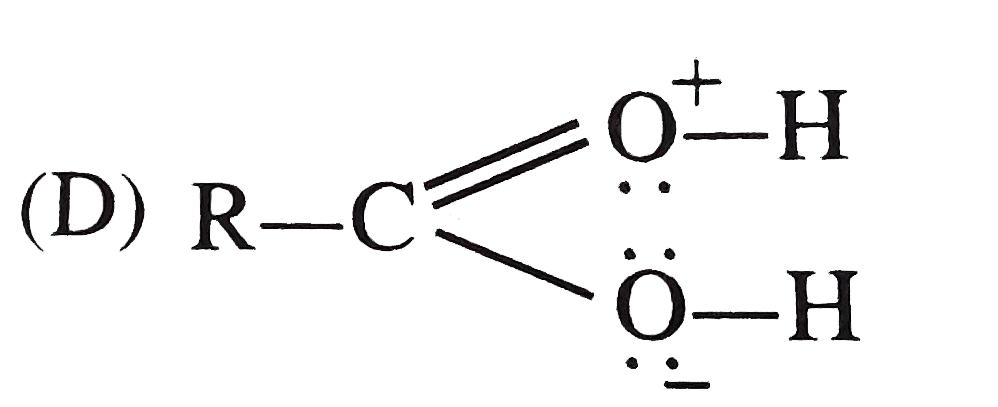
The resonating structure whose contribution result in the acidic nature of carboxylic acid |
|
Answer»
|
|
| 45. |
Which of the following will be produced when ethanoic acid and diazomethane are allowed to react ? |
|
Answer» Ethyl acetate |
|
| 46. |
When is the entropy of a perfectly crystalline solid zero? |
| Answer» SOLUTION :At ABSOLUTE ZERO of TEMPERATURE ( OK). | |
| 47. |
When IM H_2SO_3 is completely neutralised by sodium hydroxide, the heat liberated is 114.64 KJ. Enthalpy of neutralisation in KJ is? |
|
Answer» `+ 114.64` |
|
| 48. |
Why B-Xbond distance in BX_(3) is shorter than theoretically expected value ? |
| Answer» Solution :This is due to`ppi-ppi`BACK bondingof the FULLY filled p-orbital of halogen(X) into the empty p-orbital of boron. As a result, B-X bondhas some double bondcharacter and hence B-X bond is SHORTER tha expected. | |
| 49. |
What iscoreelectrons andvalenceelectron ? |
|
Answer» Solution :Careelectron: The ELECTRONIN the`He^(2) NE^(10)Ar^(18)` Valenceelectrons : Aftercompletelyfilledshellselectronicshellwith thehighestprincipalquantumnumberare calledvalenceelectron of 3sand 4of 3pare valenceelectron. Example, `S^(16)(Ne)3s^(2)3P^(4) .` in thistwo electronof 3s and 4 of3pare valenceelectron. |
|
| 50. |
Write two resonating structures of N_(2) Othat satisfy the octet rule . |
Answer» SOLUTION :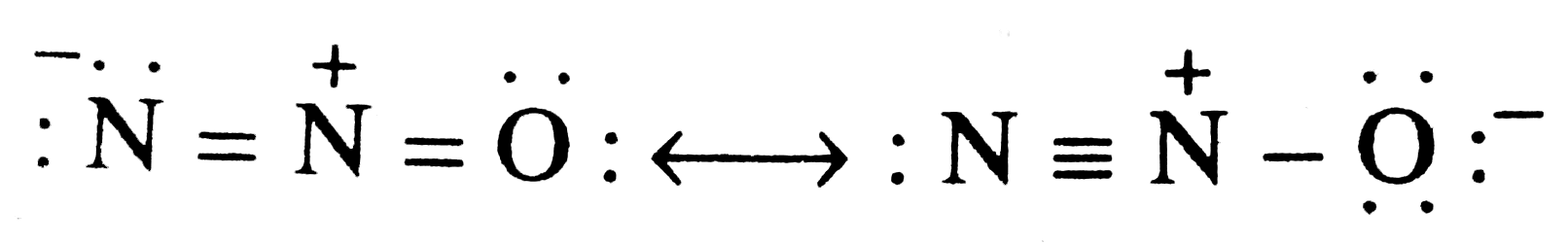
|
|