Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Write the resonating structures for the following molecules. O_(3) |
Answer» SOLUTION :
|
|
| 2. |
Which of the following is correct regarding the solubility of alkaline earth halides |
|
Answer» `BeCI_(2) gt MgCI_(2) gt CaCI_(2) gt SrCI_(2) gt BaCI_(2)` |
|
| 3. |
Which of the following does not involve oxidation of iorn? |
|
Answer» Formation of `Fe(CO)_(5)` FORM Fe |
|
| 4. |
Which is useful for the separating benzoic acid from methyl benzoate? |
| Answer» Answer :A | |
| 5. |
What is the mass of one molecule of yellow phosphorus in milligrams ? (Atomic mass of P = 30) |
|
Answer» `6.022 xx 10^(23)` MOLECULES of yellow phosphorus have mass `= 120 G = 120 xx 10^(3) MG` 1.0 MOLECULE f yellow phosphorus has mass `= ((120xx10^(3)mg))/(6.022xx10^(23))=1.993xx10^(-23)mg`. |
|
| 6. |
Which solid is much more soluble in 1MHCI than in H_(2)O? |
|
Answer» `CaHPO_(4)` |
|
| 7. |
To 50 mL of 0.5 M H_(2)SO_4 75 mL of 0.25 M H_2SO_4 is added. What is the concentration of the final solution if its volume is 125 mL? |
|
Answer» Solution :No. of MOLES in 0.05 litre of `H_(2)SO_(4) = M XX V` (litre) `=0.5 xx 0.05 = 0.0025` No. of moles in 0.0075 litre of `H_(2)SO_(4)` added `=0.25 xx 0.075` `=0.01875` TOTAL no. of moles in 0.125 litre of `H_(2)SO_(4) = 0.025 + 0.01875` =0.04375 `therefore` MOLARITY of `H_(2)SO_(4) = (0.04375)/(0.0125)= 0.35 M` |
|
| 8. |
Under what condition, the heat evolved or absorbed in a reaction is equal to its free energy change ?. |
|
Answer» Solution :`H_2O_((s)) HARR H_2O_((l)) Delta_"FUS"`H is 6 kJ `"MOL"^(-1)` (a)`DeltaG=DeltaH-TDeltaS` When the reaction is carried out at `0^@`K or `DeltaS=0` `DeltaG=DeltaH` (b)`H_2O_((s)) hArr H_2O_((l))` `Delta_"fus"H ="6 kJ mol"^(-1)` `=6000 "J mol"^(-1)` `Delta_"fus"H ="6 kJ mol"^(-1)` `=6000 "J mol"^(-1)` |
|
| 9. |
The speed of a Helium atom is equal to its root mean square speed at 27^@C. The approximate wavelength of this atom will be equal to : |
|
Answer» `1.37xx10^5` cm/sec C `= lambda = H/(mv)` on SOLVING v = `1.37xx10^5 ` cm/sec |
|
| 10. |
The reagent commonly used todetermine the hardness of water titrametrically is: |
|
Answer» Oxalic acid |
|
| 11. |
What is lattice energy? |
| Answer» Solution :LATTICE energy is DEFINED as the AMOUNT of energy required to completely SEPARATE ONE mole of a solid ionic compound into gaseous constituent. | |
| 12. |
The surface tension of a soap solution is 30xx10^(-3)Nm^(-1). The work done in stretching a bubble of this solution of surface area 5cmxx5cm, to an area of 10cmxx10cm, is |
|
Answer» `4.5xx10^(-4)J` `=2(A_(2)-A_(1))xxS` `=2(10xx10-5xx5)xx10^(-4)m^(2)xx30xx10^(-3)NM^(-1)` `=4.5xx10^(-5)J`. |
|
| 13. |
Under identical experimental conditions, which one of the following pairs of gases will be most easy to separate by diffusion process ? |
|
Answer» `O_(2),N_(2)` |
|
| 14. |
Which of the following compounds will give isocyanide on reaction with CHCl_(3) + KOH ? |
|
Answer»
|
|
| 15. |
Toluene undergoes nitration easily than benzene. Why? |
|
Answer» Solution :TOLUENE undergoes nitration easily than benzene. Why? Ans.(i) Toluene has a METHYL group on the benzene ring which is ELECTRON releasing group and hence activate the benzene ring by pushing the electrons on the benzene ring. (ii) `CH_3` group is ORTHO-para director and ring activator. Therefore in toluene, ortho and para positions are the most reactive towards an electrophile, thus promoting electrophilic substitution reaction. (iii) The methyl group hence makes it around 25 times more reactive than benzene. Therefore it undergoes nitration easily than benzene. 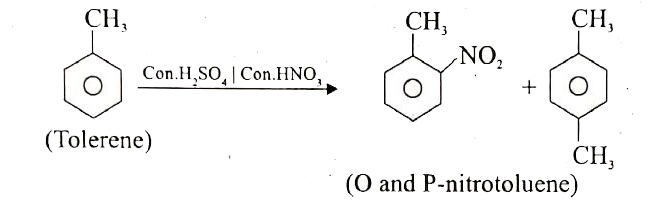
|
|
| 16. |
Which one is main cause for soil pollution ? |
|
Answer» INDISCRIMINATE use of ARTIFICAL FERTILIZERS |
|
| 17. |
Which confomation of ethane has the lowest potential energy |
|
Answer» ECLIPSED |
|
| 18. |
What do you understand by inert pair effect ? |
| Answer» Solution : Inert pair effect: As one MOVES down the group, the tendency of s-block electrons to participate in CHEMICAL bonding decreases. This effect is known as inert pair effect. In case of group 13 elements, the electronic CONFIGURATION is `ns^2 np^1` and their group valency is +3. However, on moving down the group, the +1 oxidation state becomes more stable. This happens because of the poor shielding of the `ns^2` electrons by the d and f-electrons. As a result of the poor shielding, the `ns^2` electrons are held tightly by the NUCLEUS and so, they cannot participate chemical bonding. | |
| 19. |
What is different between elements of first group and other group is s and p-block? Why? |
|
Answer» Solution :Behaviour of first group of elements are different from other elements because their volume are very less and charge or radius are high and more electronegativity. (i)Metallic radius are less : Ex. Metallic radius of first group of elements M/pm : Li (152) , Be(111) , B (88) Metallic radius of second group of elements M/pm : Na (186) , Mg (160) , Al (143) (II) Ionic radius are less : Ionic radius of first group of elements M+/pm : Li (76), Be (31) Ionic radius of second group of elements `M^(+)`/pm : Na (102), Mg (72) Maximum four valence orbitals are form in bond FORMATION `(2s, 2p_(x) , 2p_(y), 2p_(z))`. vailable for bonding, whereas the second member of the groups have nine valence orbitals `(3s , 3p , 3d)`. As a consequence of this, the maximum covalency of the first member of each group is 4 (e.g., boron can only form `[BF_(4)]^(-)` , whereas the other members of the groups can expand their valence shell to accommodate more than four pairs of electrons (e.g. aluminium forms `[AIF_(6)]^(3-)`). Furthermore, the first member of p-block elements displays greater ability to form `p_(pi) - p_(pi)`multiple bonds to itself. e.g. : `C = C, -= C, N = N , N -= N` First member from `p_(pi) - p_(pi)`bond with other elements of second PERIOD. While other elements of group do not form `p_(pi) - p_(pi)` bond. |
|
| 20. |
Which of the following compounds can combine as ligand in complex compound formation ? |
|
Answer» `SnO_2` `Ni+4CO overset"333K-343 K"to [Ni(CO)_4]` |
|
| 21. |
Which one of the following orbitals is spherical in shape? |
|
Answer» 4s |
|
| 22. |
Which one of the following statement about water is false? |
| Answer» Solution :There is extensive INTERMOLECULAR HYDROGEN BONDING in the CONDENSED PHASE instead of intramolecular hydrogen bonding. | |
| 23. |
which one of the following pairs consists of only paramagnetic species ? |
|
Answer» `[O)_(2),NO]` two unpaired ELECTRONS and NO has one unpaired ELECTRON. |
|
| 24. |
What is Pyrolysis? Give example . |
Answer» Solution :The decomposition of higher alkane into a mexture of lower alkanes, ALKENES, ETC by the APPLICATION of heat is CALLED PYROLYSIS or cracking. 
|
|
| 25. |
Two1. 0 gcarbon disks1.00 cm apart have opposite charges of equal magnitude such that there is a1.00 xx 10^(-5) N force between then . Calculate the ration of excess electron between them . Calculate the ration charged disk. |
| Answer» SOLUTION :` 4.2 XX 10^(-14) ,` | |
| 26. |
which one of the particulates is most toxic |
|
Answer» SMOG |
|
| 27. |
The solubility product constant of a metal carbonate MCO_(3) is 2 xx 10^(-12) at 25^(@)C. A solution is 0.1 M in M(NO_(3))_(2) and it is saturated with 0.01 M CO_(3). Also the ionization constant of CO_(2) are : K_(a_(1)) = 4 xx 10^(-7) and K_(a_(2)) = 5 xx 10^(-11) at 25^(@)C. The minimum pH that must be maintained to start any precipitation is |
|
Answer» |
|
| 28. |
Write the formulas for the first five members of each homologous series beginning with the following compound. CH_(3)COCH_(3) |
|
Answer» Solution :`CH_(3)COCH_(3)` `CH_(3)COCH_(2)CH_(2)CH_(3)` `CH_(3)COCH_(2)CH_(2)CH_(2)CH_(3)` `CH_(3)COCH_(2)CH_(2)CH_(2)CH_(3)` `CH_(3)CO(CH_(2))_(4)CH_(3)` |
|
| 29. |
Write the formulas for the first five members of each homologous series beginning with the following compound: H-CH=CH_(2) |
|
Answer» SOLUTION :`H-CH=CH_(2)` `CH_(3)CH=CH_(2)` `CH_(3)CH_(2)CH=CH_(2)` `CH_(3)CH_(2)CH_(2)CH=CH_(2)` `CH_(3)CH_(2)CH_(2)CH_(2)CH=CH_(2)` |
|
| 30. |
Write the uses of equilibrium constant. |
|
Answer» Solution :The equilibrium constant HELPS in predicting the direction in which given REACTION will PROCEED at any stage. For this purpose CALCULATE the reaction quotient Q. The prediction is on the base of value of Q and K. `Qlt K_c`So, forward reaction, `Q gtK_c`. So, reverse reaction, and `Q = K_c` So, reaction mixture is already both equilibrium. Calculate for concentrations equilibrium : For reaction expression of equilibrium reaction and equilibrium constant. Then PUT the equilibrium concentration and equilibrium constant and calculate initial equilibrium concentration. |
|
| 31. |
Using the concept of oxidation number, identify the substances undergoing oxidaton and reduction in the following reactions. Cr_(2)O_(3)+2Al to2Cr+Al_(2)O_(3) |
| Answer» SOLUTION :Al is oxidised `Cr_(2)O_(3)` is REDUCED | |
| 32. |
Using the concept of oxidation number, identify the substances undergoing oxidaton and reduction in the following reactions. C+O_(2)toCO_(2) |
| Answer» SOLUTION :C is oxidised `O_(2)` is REDUCED, | |
| 33. |
Using the concept of oxidation number, identify the substances undergoing oxidaton and reduction in the following reactions. Zn+2HCl toZnCl_(2)+H_(2) |
|
Answer» |
|
| 34. |
Total number of coordinate bonds presentin a molecule of C=uSO_(4) .5 H_(2) O is |
|
Answer» |
|
| 35. |
Which colour vessel is used for storage of biodegradable substance ? |
|
Answer» Yellow |
|
| 36. |
Xunderset("Boil")overset(Dil.H_(2)SO_(4))toYunderset(Delta)overset(Zndust)larrQ where 1mole Y on ozonolysis yields three moles of ethane-1,2-dial, X and Q respectively are |
|
Answer» NAPTHALENE,phenol |
|
| 37. |
Which type of shape have sp, sp^(2) , sp^(3)..... Hybrid orbitals ? |
|
Answer» Solution :All this ORBITALS have same ballon type (+) lobe at ONE site and small lobe at other site. 
|
|
| 38. |
Which of the following has maximum ionization potential ? |
|
Answer» Be |
|
| 39. |
What is meant by electronic configuration?Write the electronic configuration of N(Z=7). |
|
Answer» Solution :The distribution of ELECTRONS into VARIOUS orbitals of an ATOM is CALLED its ELECTRONIC configuration. N(Z=7) 
|
|
| 40. |
The total heat content of a system at constant pressure is |
|
Answer» Enthalpy |
|
| 41. |
Which of the following reactions have same heat of reaction at constant pressure and at constant volume? |
|
Answer» `H_(2(g)) + Cl_(2(g)) hArr 2HCl_((g))` |
|
| 42. |
Which of the following acids is added in the titration of oxalic acid and potassium permanganate ? |
|
Answer» `HNO_(3)` |
|
| 43. |
Which one of the following is correct order of stability of carbanions? |
|
Answer» `""^(-)C(CH_(3))_(3) gt ""^(-)CH(CH_(3))_(2) gt ""^(-)CH_(2)-CH_(3) gt""^(-)CH_(3)` |
|
| 44. |
The second ionization enthalpy of calcium is more than that the first and yet calcium forms CaCl_(2) and not CaCl. Why ? |
| Answer» SOLUTION :The higher ENTHALPY of lattice FORMATION of `Ca^(2+)` IONS more than COMPENSATES the higher second ionization enthalpy of calcium . | |
| 45. |
What volume of N//2 and N//10 HCl should be taken in order to make a 2 litre solution of N//5 strength ? |
|
Answer» 0.5 LITRE `N//2` HCL and 1.5 litre of `N//10` HCl |
|
| 46. |
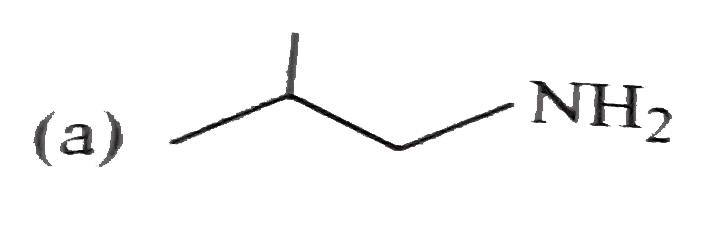
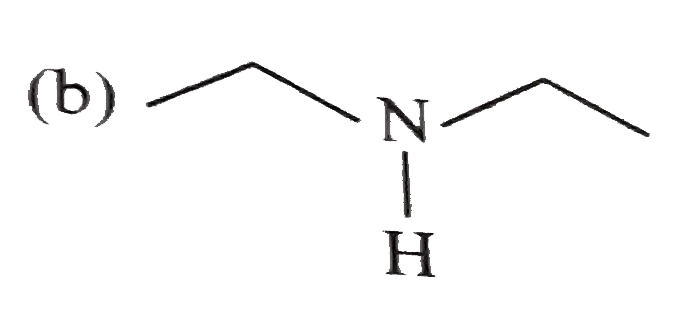
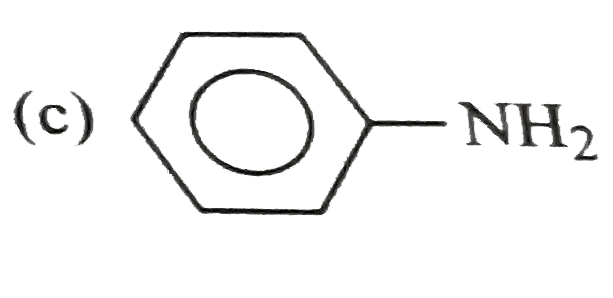
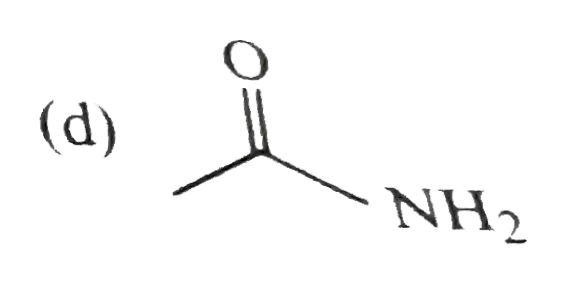
Which one of the following is least basic in character? |
|
Answer»
|
|
| 47. |
What happens when prpene reacts with concentrated H_(2)SO_(4) |
Answer» SOLUTION :Propanereactswith CONC.`H_(2) SO_(4)` toform 2 - proplyhydrogensulphatein accordancewithmarkovnikoffruleFurtherhydrolysisyields2-propanol 
|
|
| 48. |
Which type of intermolecular forces exist among the following molecules ? (i) H_(2)S molecules (ii) H_(2)O molecules (iii) CL_(2) and C Cl_(4) molecules (iv) SiH_(4) molecules (v) Helium atomms (vi) He atoms HCl molecules |
|
Answer» Solution :(i) Dipole-dipole interactions (because `H_(2)S` is POLAR). (II) Hydrogen bonding. (iii) London DISPERSION forces (becausearenon-polar). (iv) London dispersion forces (because `SiH_(4)` is non-polar ) (v) London dispersion forces (because He atoms have SYMMETRICAL ELECTRON clouds). (vi) Dipole-induced dipole forces (because HCl is polar while He atom has symmetrical electron cloud). |
|
| 49. |
Which of the following is not true for (i) Zn + dilute HCl and (ii) Zn + concentrated HCl ? |
| Answer» SOLUTION :ZN + CONCENTRATED HCL is not TRUE. | |