Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which is the correct increasing order of acidic strength from the following of Methan, Ammonia, Water and Hydrogen flouride. |
|
Answer» `HF GT gt H_2O gt gt NH_3 gt gt CH_4` `LARR` A ELECTRONEGATIVITY increases `larr` `larr` So, acidic strength increases `larr` Where ,A=F,O,N,C |
|
| 2. |
What other element may be added to silicon to make electrons available for conduction of an electric current ? |
| Answer» SOLUTION :PHOSPHORUS | |
| 3. |
Which of the following is an Arrhenius acid? |
|
Answer» `NH_3` |
|
| 4. |
What is the density of oxygen gas at 227^(@)C and 4 atm pressure (R=0.082 L atom K^(-1)mol^(-1)) |
|
Answer» `3.12g//L` |
|
| 5. |
Whichof the followingis less reactive than benzene towards electrophilic substitutionreactions |
|
Answer» NITROBENZENE |
|
| 6. |
Which of the following is strongest -I group |
|
Answer» `-F` |
|
| 7. |
What is relation between gas constant R and work ? |
|
Answer» Solution :`pV=nRT, R=(dV)/(nT)` `therefore R =("Pressure"xx"Volume")/("Mole number"xx"Temperature")` Where pressure `=("Force")/("Area")xx` Volume `therefore "Pressure"=("force"xx"length"^(3))/("length"^(2))=` force `xx` length `R=("force"xx"length")/("mole number"xx"temperature")` `= ("WORK")/("mole number"xx"temperature")` ..Work required to change in `1^(@)C` temperature is known as gas CONSTANT R... |
|
| 8. |
Which of the following FCC structure contains cations in alternate tetrahedral voids ? |
|
Answer» NaCl |
|
| 9. |
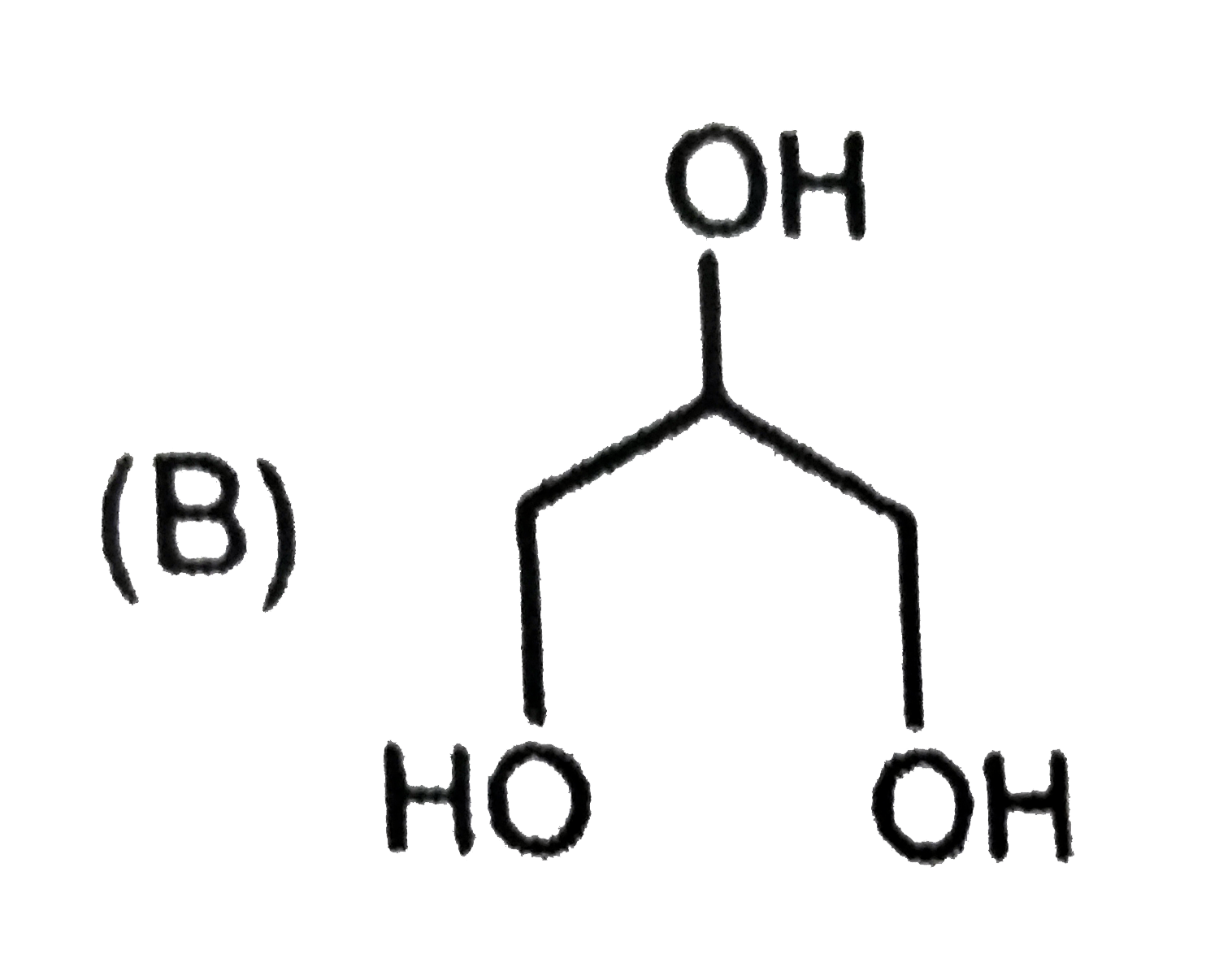
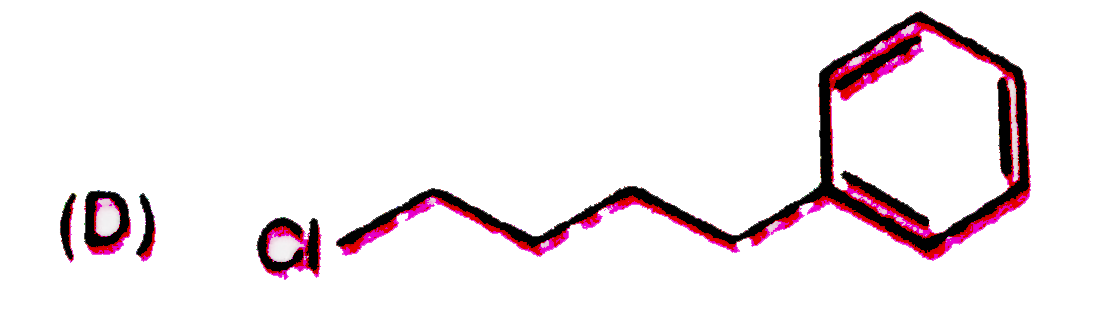
Write structure of the compound 4-tert-butyl-3-iodoheptane and 1,4-dibromo but-2-ene. |
Answer» Solution :(i)  (II) `underset(Br)underset(|)(C)H_(2)-CH=CH-underset(Br)underset(|)(C)H_(2)` |
|
| 10. |
Which one is incorrect name ? |
|
Answer» Propyne |
|
| 11. |
The weight percentage of chloride in a binary halide is 77:45. What is the equivalnet weight of the element forming chloride? |
|
Answer» |
|
| 12. |
Which of the following is/are correct? |
|
Answer» An electron in excited state cannot ABSORB a photon |
|
| 13. |
Which metal does not liberate H_(2) gas from dilute aqueous hydrochloric acid at 298 K? |
|
Answer» Mg |
|
| 14. |
What is similarities in elements for the same groups ? |
|
Answer» Solution :ELEMENTS in the same VERTICAL COLUMN or group have SIMILAR valence shell electronic configurations, the same number of electrons in the outer orbitals and similar properties. e.g. The group 1 elements (alkali metals `ns^(1)` have his valence shell electronic configuration as shown below :  THSU it can be see that the properties of an element have periodic dependence upon its atomic number and not on relative atomic mass . |
|
| 15. |
Which of the following compound is used to increase stability of hydrogen peroxide ? |
|
Answer» SULPHURIC acid |
|
| 17. |
underset(underset("(Several hours)")(100^(@)C))overset(NaOH,H_(2)O)rarr"Product"Product is obtained in the above reaction is : |
| Answer» Answer :A::B::C::D | |
| 18. |
Which of the gas can displace remaining all the gases from the surface of adsorbent ? |
| Answer» ANSWER :A | |
| 19. |
When a substance 'A' reacts with water it produces combustible gas B and a solution of a substance 'C' in water . A has no reaction with the solution of 'C' When another substance 'D' reacts with this solution of 'C' it also produces the same gas 'B' even on reaction with dilute sulphuric acid at room temperature . Here A,B,C and D or respectively. |
|
Answer» `Na, H_(2), NaOH and Zn` |
|
| 20. |
The relative i s ......... proportion of the atoms of tritium and protium. |
|
Answer» `10:10^(17)` `THEREFORE` 1 tritium atoms in (?) and proportionis 99.985 % in HYDROGEN That means99.985 protium in 100 h atom `therefore`protium in (?) H atom thus in 1 h atom `therefore 10^(17)` : 1 is proportion of protium: tritium |
|
| 21. |
The unit cube length forLiCl ( NaCl structure) is5.14 Å . Assuming anion -anion contant, calculate the ionicradius for chloride ion. |
|
Answer» ` BC = sqrt( AB^(2) + AC^(2))= sqrt((2.57)^(2) + (2.57)^(2)) = 3.63` RADIUS of ` CL^(-)` ion ` 1/2 XX 3.63 Å = 1.81 Å` 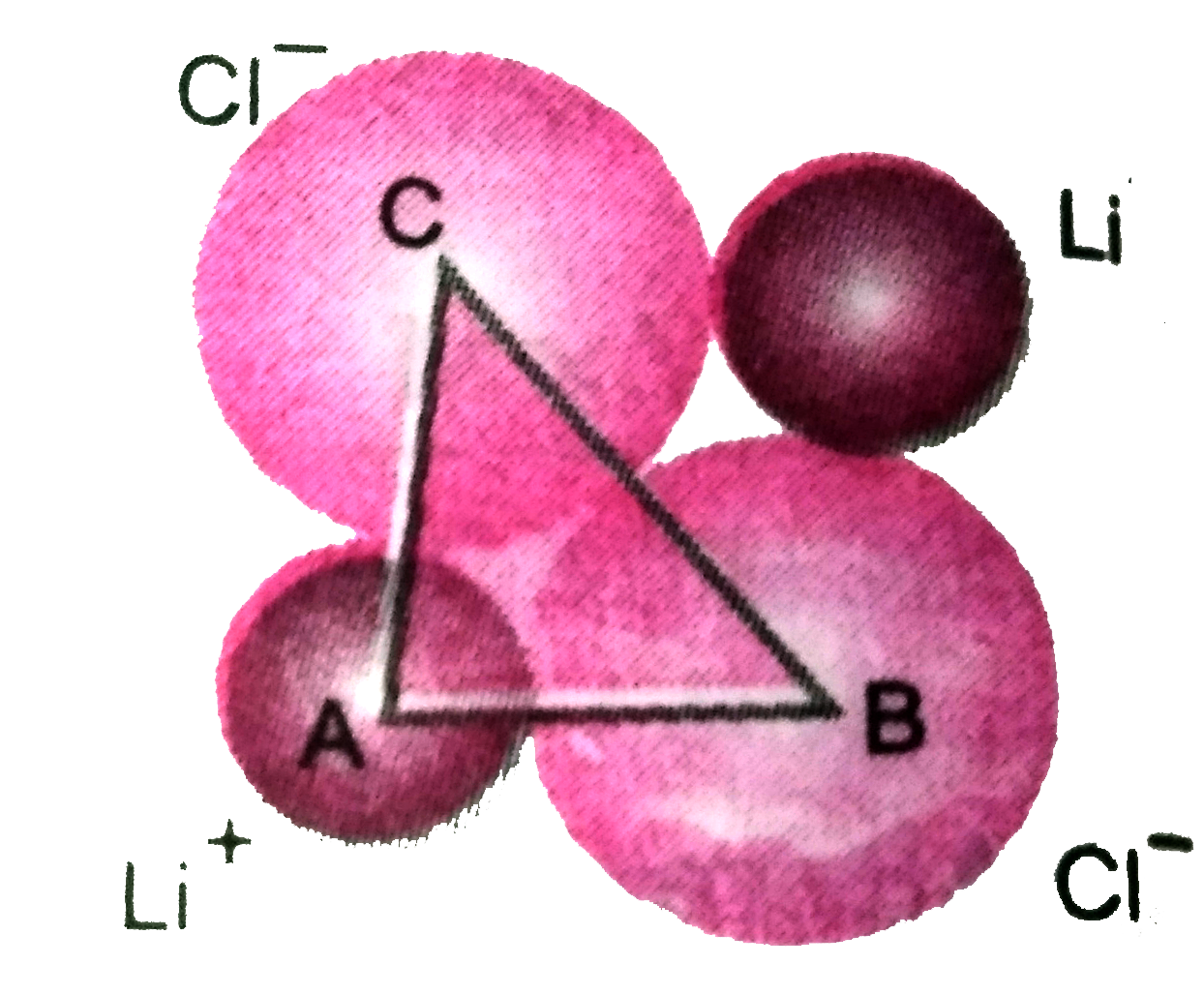
|
|
| 22. |
When sulphur in the form of S_(8) is heated at 900 K , the intial pressure of 1 atm fallsby 29% at equilibrium . Thisis because of conversion of S_(8) "to" S_(2) . Calculate the equilibrium constant for the reaction. |
|
Answer» SOLUTION :` {:(,S_g(g),hArr,4S_(2)(g)), (" Intial",1 atm,,), (" At eqm.",1-0*29 = 0*71 "atm",,4b xx 0*29 "atm" = 1*16 "atm"):} ` ` K_(p) = (p_(s_(2)(g))^(4))/(p_(s_(g(g))))= (1*16 " atm ")^(4)/(0*71 "atm" )=2*55 " atm "^(3)` |
|
| 23. |
Which of the following oxides have rock salt structure with coordination number 6 : 6 ? |
|
Answer» ReO |
|
| 24. |
Time independent schrodinger wave equation is |
|
Answer» `hatH Psi = EPsi` |
|
| 25. |
Which four variables are describe completely the gasous state? |
| Answer» Answer :D | |
| 27. |
Write the drawbacks in Mendeleev's periodic table that led to its modification. |
|
Answer» Solution :Draw backs of Mendeleev.s PERIODIC tables are, (i) Elements having SIMILAR properties are placed in different groups whereas some elements having different properties are placed in the same group. e.g., alkali metals such as Li, Na, K, etc., (IA group) are grouped together with coinage metals such as Cu, Ag, AU (IB group) though their properties are quite different. Chemically similar elements such as Cu (IB group) and Hg (IIB group) have been placed in different groups. (ii) Elements having higher ATOMIC weight are placed before the elements with lower atomic weights in order to maintain the same chemical nature of elements. `""_(18)^(39).9Ar and ""_(19)^(39.1)K, ""_(27)^(58.9) CO and ""_(28)^(58.9)Ni etc` (iii) Isotopes donot find any place in the periodic table. Now, according to Mendeleev.s classification explained, they should be placed at different places in the periodic table. (IV) Position of hydrogen is controversial in the periodic table. (v) Elements of group (VIII) position are not made clear which can be arranged in three triads without any justification. (vi) Even and odd series in IV, V and VI long periods it could not be explain. (vii) Lanthanides and actinides are not discovered later on, the position in the main frame is not be given proper in the periodic table |
|
| 28. |
What does LPG stand for ? Name its important constituents. |
| Answer» Solution :LPG stands for LIQUEFIED petroleum gas . It MAINLY CONTAINS BUTANE and isobutane. | |
| 29. |
Which of the following respresents 2,2,3-Trimethyl hexane? |
|
Answer» `CH_(3)-C(CH_(3))_(2)-CH_(2)-CH_(2)-CH(CH_(3))_(2)` |
|
| 30. |
Thiosulphate reacts differently with iodine and bromine in the reactions given below 2S_(2)O_(3)^(-2)+I_(2)toS_(4)O_(6)^(-2)+2I^(-) 2S_(2)O_(3)^(-2)+2Br_(2)+5H_(2)Oto2SO_(4)^(-2)+2Br^(-)+10H^(+) Which of the following statements justifies the above dual behaviour of thiosulphate ? |
|
Answer» Bromine is stronger oxidants than iodine. |
|
| 31. |
When two reactants A and B are mixed to give products C and D, the reaction quotient , Q, at the intial atages of the reaction |
|
Answer» is ZERO With time, [C] and [D] increase and so does Q. |
|
| 32. |
Which of the following species are hypervalent- |
|
Answer» `ClO_(4)^(-)` |
|
| 33. |
What will be the density of carbon dioxide at 100^@C and 800 mm Hg pressure ? |
|
Answer» In the present case, `P = 800 " mm Hg " = 800/760 = 1.053` atm `T= 100^@C = 100 + 273 = 373 K` `:. ""M =(dRt)/P` `:. "" d = (MP)/(RT)` SUBSTITUTING the values, we have `d= (44 xx 1.053)/(0.0821xx 373) = 1.513g L^(-1)` |
|
| 34. |
Which statement(s) is (are) correct ? |
|
Answer» A SIGMA bond is stronger than `pi`-bond |
|
| 35. |
Which of the following shows highest oxidation number in combined state |
| Answer» Solution :`OVERSET(+8)(OsO_4),overset(+8)(RuSO_4)` | |
| 36. |
What is order of first, second, third ionisation enthalpy of any elements ? |
|
Answer» SOLUTION :For any elements… FIRST ionisation enthalpy `(Delta_(i)H(1)) lt` Second ionisation enthalpy `(Delta_(i)H(2)) lt` THIRD ionisation enthalpy `(Delta _(i)H(3)) lt `…….. |
|
| 37. |
Which species doesnot exist |
|
Answer» `[BF_(6)]^(3-)` |
|
| 38. |
Which of the following classes of compound is unreactive toward sulphuric acid? |
|
Answer» Alkanes |
|
| 39. |
What are ambident nucleophiles ? Explain with an example . |
|
Answer» Solution :Nucleophiles which can attack two different SITES are called AMBIDENT nucleophiles . For example , cyanide group is a resonance hybrid of two contributing structures and therefore it can ACT as nucleophile in two different ways : `""^(@)C = N hArr : C - N^(@)` It can attack through CARBON to FORM cyanides and through nitrogen to form isocyanides or carbylamines |
|
| 40. |
Which of the polymer (s) in Example is // are optically active or chiral ? |
Answer» Solution :In Example , `(i) P.M.M.A.``(II)``P.A.N.`,`(iv)` Styron, and `(ix)``P.P.` are optically active or have a CHIRAL centre, for example, polypropene monomer part ofpolypropylene is representes as `:` 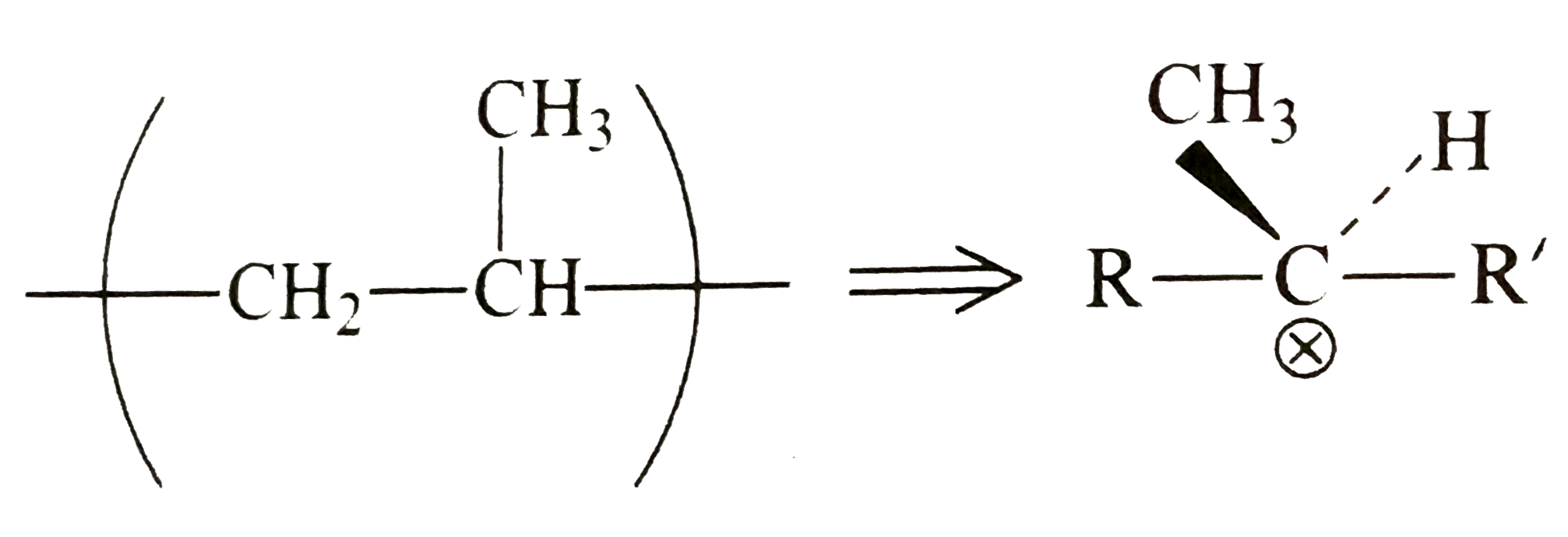 Each `C` ATOM in the monomer is chiral and millions of stereoisomers can be FORMED depending on the size of the POLYMER. |
|
| 41. |
Which of the following is correct for SF_(4)? |
|
Answer» It has a see-saw shape |
|
| 42. |
Which reaction(s) reflect amphoteric nature of BeO? |
|
Answer» `BeO+C+Cl_(2)OVERSET(600-800K)hArrBeCl_(2)+CO` |
|
| 43. |
The solubility of AgCl in water is ........than that in NaCl solution. |
|
Answer» |
|
| 44. |
Which of the two ions from the list given below that have the geometry that is explained by the same hybridization of orbitals , NO_(2)^(-), NO_(3)^(-), NH_(2)^(-), NH_(4)^(+), SCN^(-) ? |
|
Answer» `NO_(4)^(+) and NH_(2)^(-)` `NO_(2)^(-) to SP^(2), NO_(3)^(-) to sp^(2)` ` NH_(2)^(-) to sp^(3), NH_(4)^(+) to sp^(3)`, ` SCN^(-) to sp ` |
|
| 45. |
Which of the following is true about fullerenes? |
|
Answer» It CONTAIN twelve six-MEMBERED rings and twenty five membered rings |
|
| 46. |
Which of the following statements is true about Ca(OH)_2 ? |
|
Answer» It is used in the PREPARATION of bleaching powder |
|
| 47. |
Which of the following is not the correct relationship |
|
Answer» II & IV are metamer |
|
| 48. |
Which of the following molecules is most suitable to disperse benzene in water ? |
|
Answer»
|
|
| 49. |
Which substance has fruity sweet smell ? |
|
Answer» Pent-1-ene |
|
| 50. |
What efforts are being made by the government to save the Taj Mahal ? |
|
Answer» Solution :The government of India announced an action plan NAMED .Taj Trapezium. in 1995 to prevent the Taj Mahal. This plan aims at clearing the air of the NEARED city or towns of Taj Mahal. Under this plan more than 2000 polluting industries dying inside the Trapezium would switch over to the use of natural GAS or liquefied petroleum gas instead of coal or oil. A new natural gas pipeline would bring more than half a million cubic metres of natural gas a DAY to this area. People living in the city will also be encouraged to use liquefied petroleum gas in PLACE of coal, kerosene or firewood. Vehicles plying on highways in the vicinity of Taj would be encouraged to use low sulphur content diesel. |
|