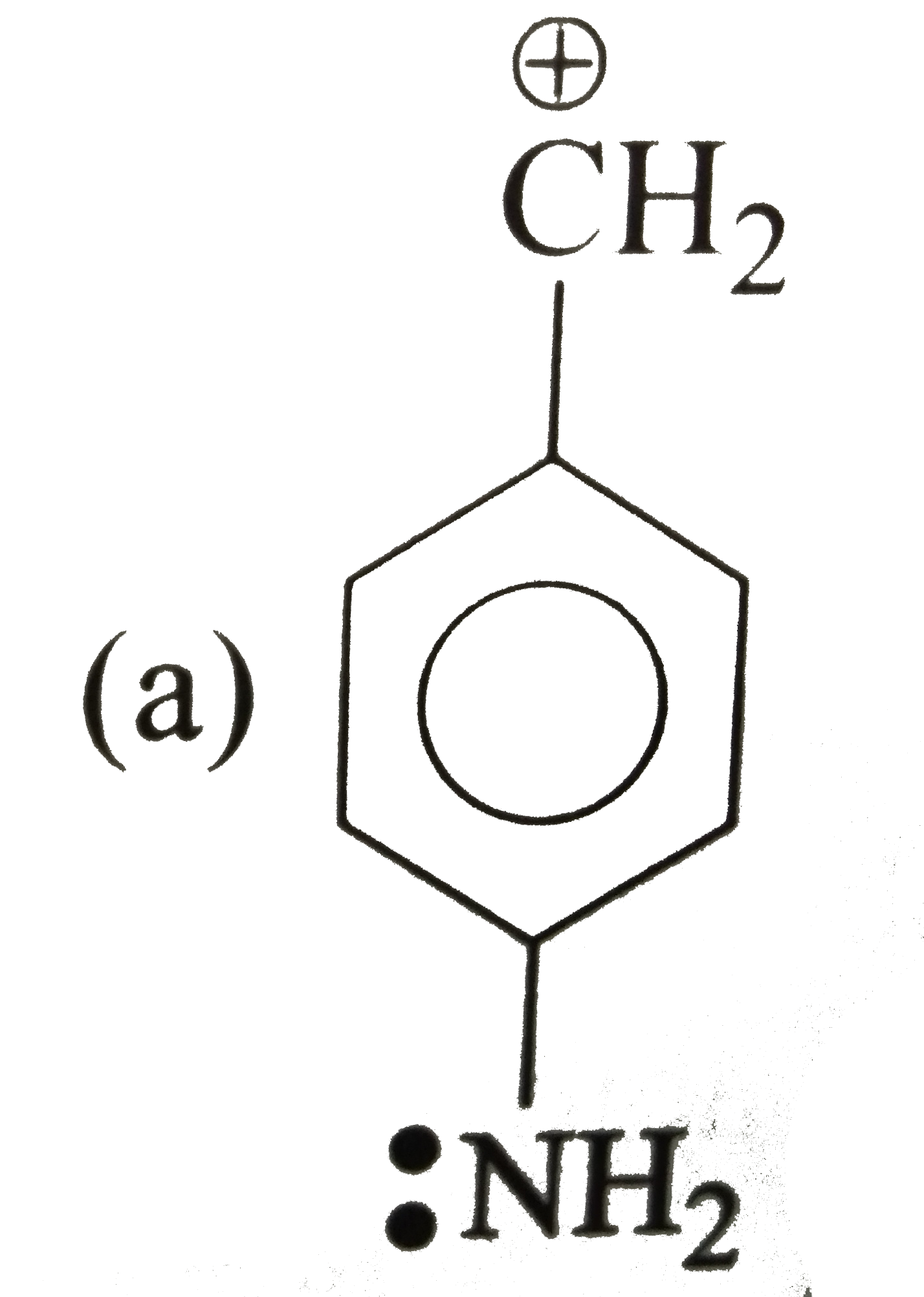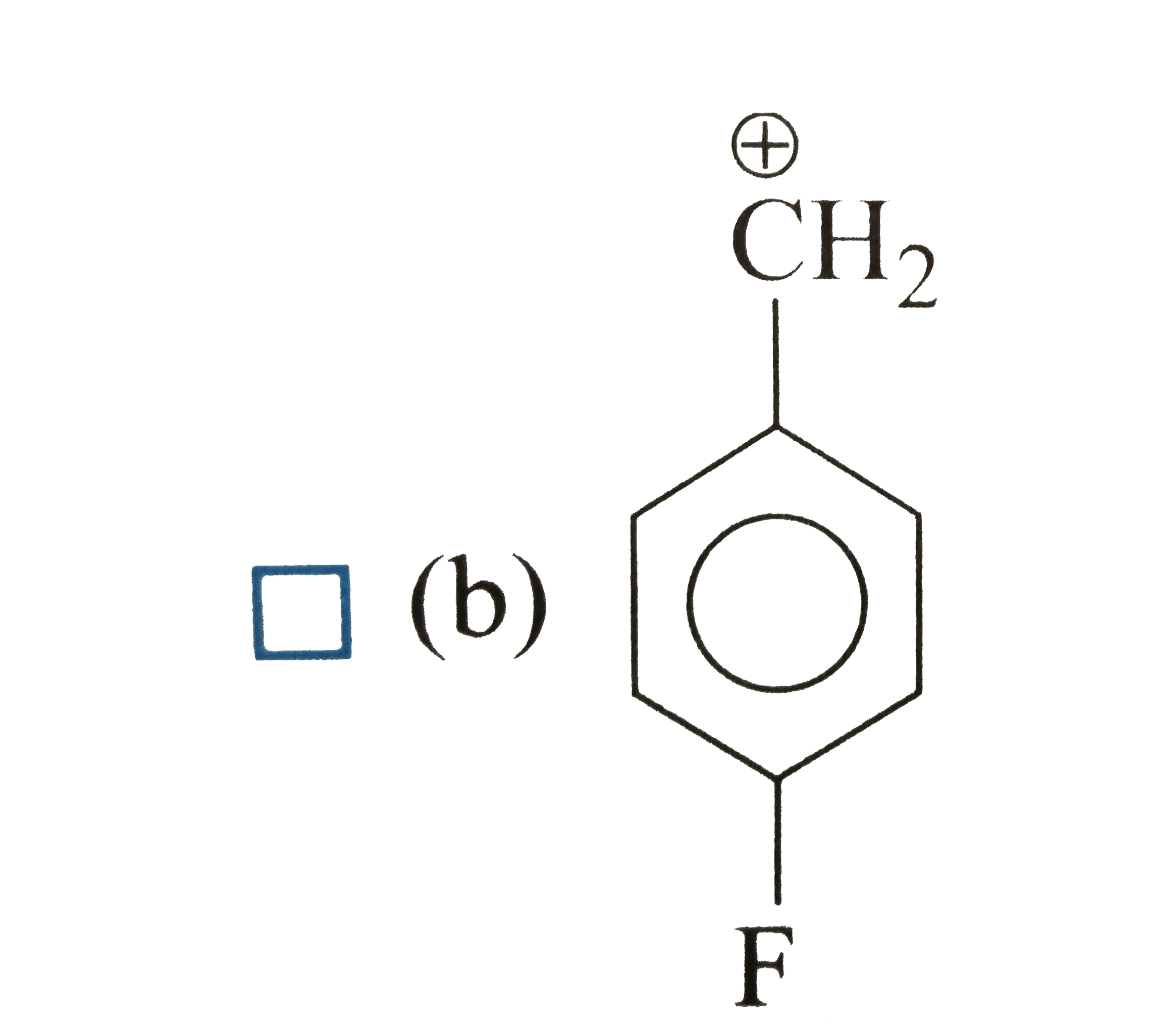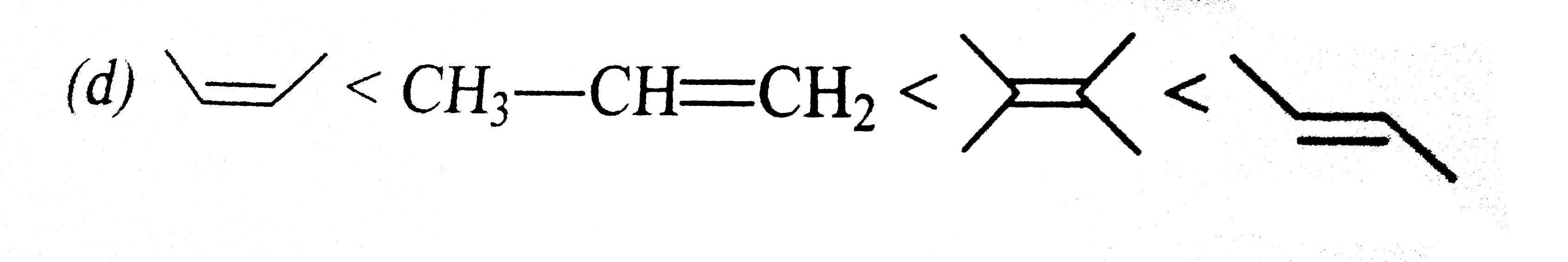Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following statements are correct for alkali metal compounds? I. Superoxides are paramagetic in nature. II. The basic strength of hydroxides increases down the group. III. The conductivity of chlorides in their aqueous solutions decreases down the group. IV. The basic natuer of carbonates in aqueous solution is due to cationic hydrolysis. |
|
Answer» I,II and III |
|
| 2. |
Unknown organic metal oxide (A) heated with carbon will gives compound (B), which on reaction with water gives ethyne. So what are the compound A and B ? |
| Answer» SOLUTION :`underset("LIME")underset((A))(CaO)+3C overset(-CO)RARR underset("Calcium carbie")underset((B))(CaC_(2))underset(-Ca(OH)_(2))overset(+2H_(2)O)rarr underset("Ethyne")(C_(2)H_(2))` | |
| 3. |
Which one is not a pollutant normally ? |
|
Answer» HYDROCARBONS |
|
| 4. |
Which group P-block elements show highest positive oxidation state ? |
|
Answer» GROUP 16 |
|
| 5. |
Which of the following parameter cannot be estimated by using Born-Haber cycle? |
|
Answer» HYDRATION energy |
|
| 7. |
What are (A), (B) and ( C) in the following scheme of reactions ? |
Answer» SOLUTION :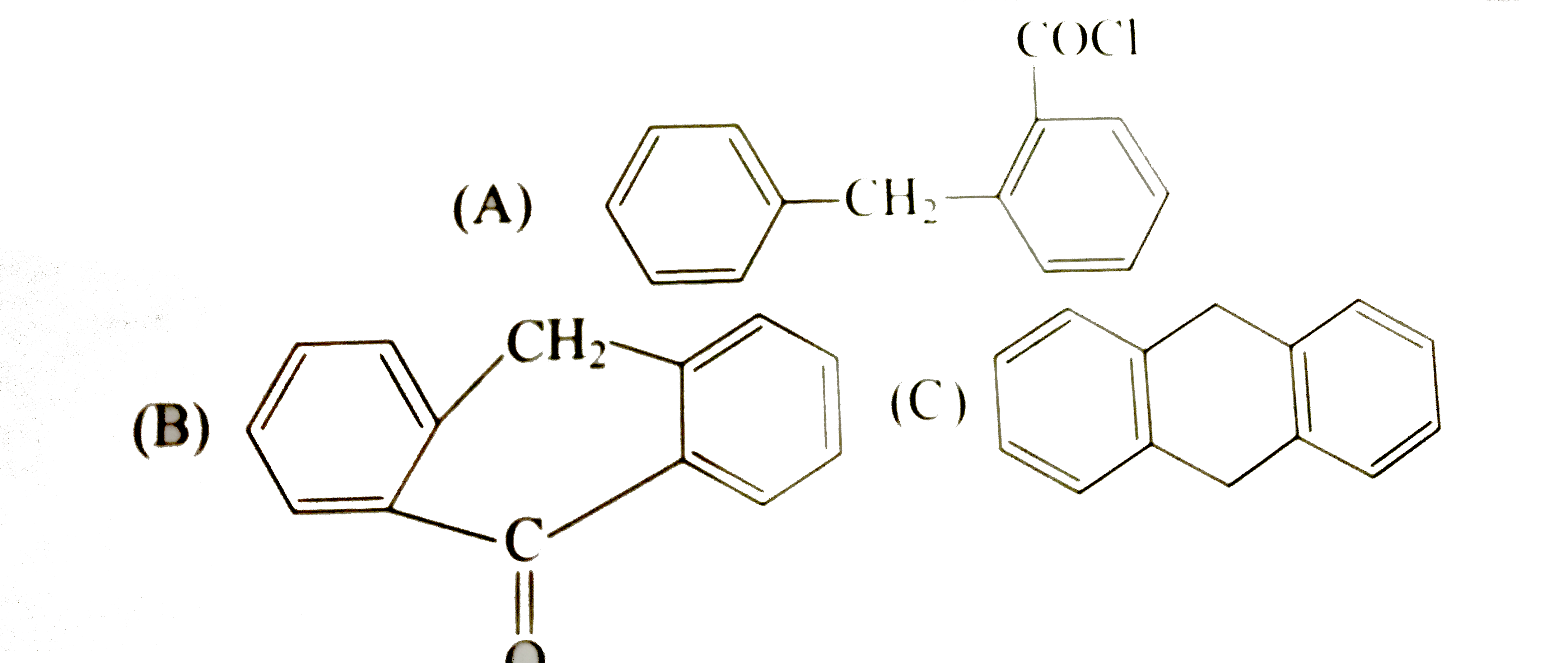
|
|
| 8. |
Tyndall effect would be observed in a |
|
Answer» solution |
|
| 9. |
When benzene sulfonic acid and p-nitrophenol are treated with NaHCO_(3), the gases released respectively are |
|
Answer» `SO_(2), NO_(2)` |
|
| 10. |
The uncertainty in the position and velocity of a particle are 10^(-10)m and 5.25 xx 10^(-24) ms^(-1). What is the mass of the particle ? (h = 6.625 xx 10^(-34) Js) |
|
Answer» SOLUTION :`Deltax=10^(-10)m""Deltav=5.25xx10^(24)ms^(-1)` `m=(6.625xx10^(-34)kgm^(2)s^(-1))/(4xx3.1416xx10^(-10)mxx5.25xx10^(-24)ms^(-1))` `"Mass of a particle" =" 0.1004 KG or 100.4 g "` |
|
| 11. |
What is units of co-efficient of viscosity. |
|
Answer» SOLUTION :Ns `m^(-2)=` Newton SEC / square meter `Pa s^(-1)=` Pascal / sec. CGS = Poise 1 poise `= 1g CM^(-1)s^(-1)=10^(-1)kg m^(-1)s^(-1)` SI unit kg `m^(-1)s^(-1)=10xx` Poise |
|
| 12. |
What orbitals can overlap to form a sigma-bond and which orbitals can do so to form a pi-bond ? |
| Answer» SOLUTION :s-s,s-p,p-p for ` SIGMA `-BOND and only p-p for `PI`-bond. | |
| 13. |
Write the minimum and maximum valencies of elements. |
|
Answer» Solution :The minimum valency EXHIBITED by an ELEMENT is zero. The maximum valency exhibited by OS, Ru or XE in its compounds is 8. |
|
| 14. |
Which among the given molecules are exhibit tautomerism? |
| Answer» Answer :A | |
| 16. |
You are given the following bond energy data E_(H-H) = 104.2 kcal mol ^(-1),E_(F-F) = 36.6kcal mol ^(-1), E_(H-F) = 134.6 kcal mol ^(-1) On the basis of this data, eletronegativity of flurine will be ………. |
|
Answer» reprsented as `chi_(H) and chi_(F)`RESPECTIVELY . APPLING Pauling's formula `|chi_(H)- chi_(F) |=0.208sqrt(Delta) ` `Delta= BE (H-F) - sqrt(BE(H-H)xxBE(F-F))` `= 134.6 - sqrt(104.2xx36.6)` `= 134.6 - 61.8`kcal mol `^(-1)` 72. 8kcal mol `^(-1)` `therefore|chi_(H)- chi_(F) |=0.208sqrt(72.8) = 1.77` As `chi_(F)gt chi_(H),chi_(F)= 1.77 + chi_(H) = 1.77+2.1 = 3.87` |
|
| 17. |
Which of the following concentration terms is/are independent of temperature? |
|
Answer» MOLALITY |
|
| 18. |
What do you mean by torsional strain ? |
|
Answer» Solution :Alkanes can thus have INFINITE number of conformation by rotation around C-C single bonds. However, it may be remembered that rotation around a C-C single BOND is not COMPLETELY free. It is hindered by a small energy barrier of 1-20 kJ `MOL^(-1)` due to weak replusive interaction between the adjacent bonds. Such a TYPE of repulsive interaction is called torsional strain. |
|
| 19. |
Write the equation of Henderson Hassel balch. |
| Answer» Solution :`pH=log K_a (["CONJUGATE BASE" , A^-])/"[WEAK ACID, HA]"` | |
| 21. |
The size of isoelectronic species- F_(-), Ne and Na^(+) is affected by |
|
Answer» nuclear charge (Z) |
|
| 22. |
Which of the following ppt. is soluble in both aqua regia and sodium disulphide (yellow)? |
| Answer» Answer :A::B | |
| 23. |
Which one of the following elements is unable to form MF_6^(3-)ion ? |
|
Answer» In |
|
| 24. |
What is coordination number of Na^(+) and Cl^(-)ion in Nacl ? |
| Answer» SOLUTION :Each has a COORDINATION NUMBER of 6, i.e. each ION is surrounded by SIX oppositely charged ions. | |
| 25. |
Using the Gibb's free energy change,DeltaG^(@) =+63.3kJ for the following reaction Ag_(2)CO_(3)(s)hArr2Ag^(+)(aq)+CO_(3)^(2-)(aq) the K_(sp) of Ag_(2)CO_(3) in water at 25^(@)C is (R=8.314 JK^(-1) "mol"^(-1)) |
|
Answer» `3.2xx20^(-26)` `63.3xx10^(3)J =- 2.303xx8.314xx298 logK_(sp)` `=-5705. 84 log K_(sp)` or log `K_(sp)=(63.3xx10^(3))/(57.5.84)=-11.09` `:. K_(sp)=` ANTILOG `(bar(12).91)` `=8.128xx10^(-12)` |
|
| 26. |
Which ofthe following metal oxides is antiferromagnetic in nature ? |
|
Answer» `MnO_(2)` |
|
| 27. |
which of the following does not reduce Benedict's solution ? |
|
Answer» GLUCOSE |
|
| 28. |
Which of the following is true for spontaneous adsorption of H_2 gas without dissociation on solid surface |
|
Answer» PROCESS is EXOTHERMIC and `DELTAS lt 0` |
|
| 29. |
When a gas is compressed at constant temperature |
|
Answer» the SPEEDS of the MOLECULES increase |
|
| 30. |
Two gases A and B have critical temperatures as 250 and 125 K respectively. Which one of these can be liquefied easily and why? |
| Answer» Solution :GAS a can be EASILY liquefied. Because higher critical TEMPERATURE, greater is the intermolecular force of ATTRACTION. | |
| 31. |
What is the molecular formula of borax ? |
|
Answer» `Na_2B_4O_7 . 4H_2O` |
|
| 32. |
The sequence of ionic mobility in aqueous solution is |
|
Answer» `Rb^(+)gt K^(+) gt Cs^(+) gt Na^(+)` |
|
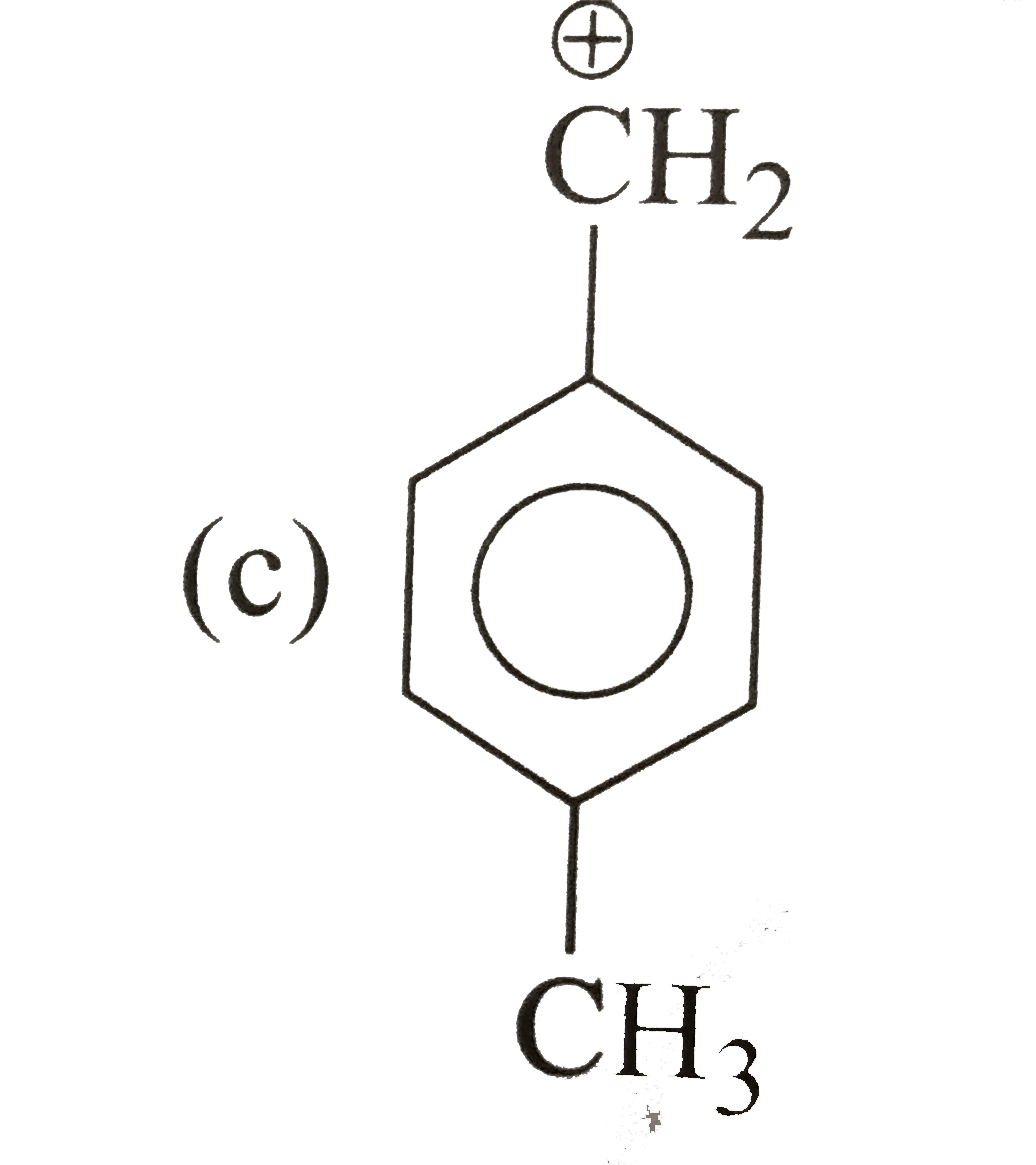
| 33. |
Which of the following is correct order of stability of alkene? |
|
Answer»
|
|
| 34. |
Whatshould be ther ratio of velocities of CH_4 and O_2 moleculesso that they are associated with de Broglie waves of equal wavelength ? |
|
Answer» |
|
| 35. |
The total energy of the electron of H-atom in the second quantum state is -E_(2). The total energy of the He^(+) atom in the third quantum state is: |
|
Answer» `-((3)/(2))E_(2)` `=-(Z^(2))/(n^(2))xx13.6eV` `E_(2)(H)=-(13.6)/(1)eV` `E_(3)(He^(+))=-(13.6xx4)/(9)eV` `(E_(2))/(E_(3))=(9)/(4)"or"E_(3)=(4)/(9)E_(2)`. For negative value of `E_(2),E_(3)` will also be negative. |
|
| 36. |
Which bond angle is high from PH_(3) and PH_(4)^(+)? Why ? |
|
Answer» Solution :In `PH_(4)^(+)` , bond angle is high because it is TETRAHEDRAL and angle is `109.5^(@)` . In `PH_(3) , SP^(3)` hybridization occurs but non- bonding `e^(-)` pair present on P, So due to REPULSION and bond angle is decrease `(93.6^(@))` |
|
| 37. |
What is molecular hydride?Explain with an examples. |
| Answer» | |
| 38. |
Which of the following can shown tautomerism? |
|
Answer» HCN |
|
| 39. |
The tatol number of electron pairs in N_(2) O_(3) is |
Answer»  In each strcuture ,TOTAL number of LONE PAIRS = 8 |
|
| 40. |
which of the following aqueous solution has the highest bolling point? |
|
Answer» `0.1 M KNO_(3)` |
|
| 41. |
Whatwill be thewavelengthof a ballof mass0.1 kgmovingwitha velocityof 10 ms^(-1) |
|
Answer» SOLUTION :According to thede Broglieequation `LAMBDA=(h)/( mv) ` `lambda =(6.626 xx 10^(34) js)/((0.1 kg10 ms^(-1))` where m= 0.1 kg v=10 `ms^(-1)` `h= 6.626xx 10^(34) Js` `=6.626 xx 10^(34) kg ^(-1) m^(-1) s^(2)` but`J= kg m^(2) s^(2)` `=6.626 xx 10^(34) m` |
|
| 42. |
Write the correct order of reducing character of 13th group elements in +3 states. |
| Answer» Solution :Due to inert pair effect, stability of +1 STATE increases and that of +3 state decreases down the group. Order of reducing character in +1 state is : `AL gt B gt GA gt In gt Tl` | |
| 43. |
The threshold frequency V_0 for a metal is 7.0xx10^14 s^1. Calculate the kinetic energy of an electron emitted when radiation of frequency v=1.0xx10^15s^(-1) hits the metal. |
| Answer» Solution :ACCORDING to Einstein’s equation Kinetic energy `=1//2m_eV^2=h(v-v_0)=(6.626xx10^(-34)Js)XX(1.0xx10^15s^-1-7.0xx10^14s^-1)=(6.626xx10^(-34)Js)xx(10.0xx10^14s^-1-7.0xx10^14s^-1)=(6.626xx10^(-34)Js)xx(3.0xx10^14s^-1)=1.988xx10^(-19)J` | |
| 44. |
Whichof thefollowingreactiondo NOToccur? |
|
Answer» `Be + 2HCI toBeCl+H_(2)` |
|
| 45. |
Which one of the following ratio gives the purity of the metal (rho-resistivity (or) specific resistance) |
|
Answer» `(rho_(300^(@)C))/(rho_(4.2^(@)C))` |
|
| 46. |
Which of the following molecule/ion do not have all the identical bond ? |
|
Answer» `SiF_4` |
|
| 47. |
Which of the following has maximum hydrogen bonding? |
|
Answer» Ethyl amine |
|
| 48. |
What type of bond is responsible for the partial compensation of the electron deficiency of a boron halide ? |
|
Answer» <P> SOLUTION :`p PI- p pi` BACK BONDING |
|
| 49. |
Which one of the following two compounds is the stronger acid ? Explain why? |
|
Answer» |
|
| 50. |
Which of the following are correct statement |
|
Answer» Hardy-Schulze RULE is related to coagulation (b) Brownian movement is shown by colloidal solution due to collision between colloidal particles themselves and with that of particles of dispersionmedium. thyndall effect is due to scattering of lightt by colloidal particle. (d) is correct because, higher the gold number, less will be protective power. |
|