Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following metals will give H_(2) on reaction with NaOH? |
|
Answer» Be |
|
| 2. |
Which one of the following react with gringnard reagent followed by hydrolysis will yield primary alcohol? |
|
Answer» `CH_3CHO` |
|
| 4. |
What are all the conditions for aromaticity |
|
Answer» SOLUTION :Huckelproposedthat aromaticitlyfunctionof electromicstructureof anorganiccompoundacompoundmay bearomaticifit obeythe followingrules. (i) Themoleculemusthavea cyclicstructure themoleculemust be co-planar (III) PRESENCE of`(4n-2)` ELECTRONICIN theringwheren is aninteger `(n=0,1,2....)` thisisknownas Huckels rule. |
|
| 5. |
Which of the following compound(s) gives green solution, when treated with K_(2)Cr_(2)O_(7) and dilute H_(2)SO_(4)? |
|
Answer» `KNO_(3)` |
|
| 6. |
Which of the following pairs of atomic numbers represents elements belonging to the same group? |
| Answer» Answer :C | |
| 7. |
Water acts as a powerful ionizing solvent due to its high |
|
Answer» POLAR nature |
|
| 8. |
What is a redox couple |
| Answer» SOLUTION :A redox couple CONSISTS of oxidised and reduced form of the same substance taking part in an oxidation or reduction half reactin for example `Zn^(2+)(AQ)//Zn (s),CI_(2)CI^(-)(aq)`ETC | |
| 9. |
Which d-orbital is involved in dsp^(2)hybridisationwhy ? |
|
Answer» Solution :` d_(Z^(2)- y^(2))` . This is because its four LOBES LIE along the x-axis and y-axis . The two p-0orbitals can COMBINE along these axes. |
|
| 10. |
The volume of perhydrol which on decomposition gives 2 lit ofO_2gas at STP is |
|
Answer» 100ml |
|
| 11. |
Which of the following hydrated chlorides can not be converted into anhydrous chloride only by heating? |
|
Answer» `MgCl_(2).6H_(2)O` `MgCl_(2).6H_(2)OrarrMgO+2HCl+5H_(2)` `2AlCl_(3).6H_(2)OrarrAl_(2)O_(3)+6HCl+9H_(2)O` `SnCl_(2).2H_(2)OrarrSnO+2HCl+H_(2)O` |
|
| 12. |
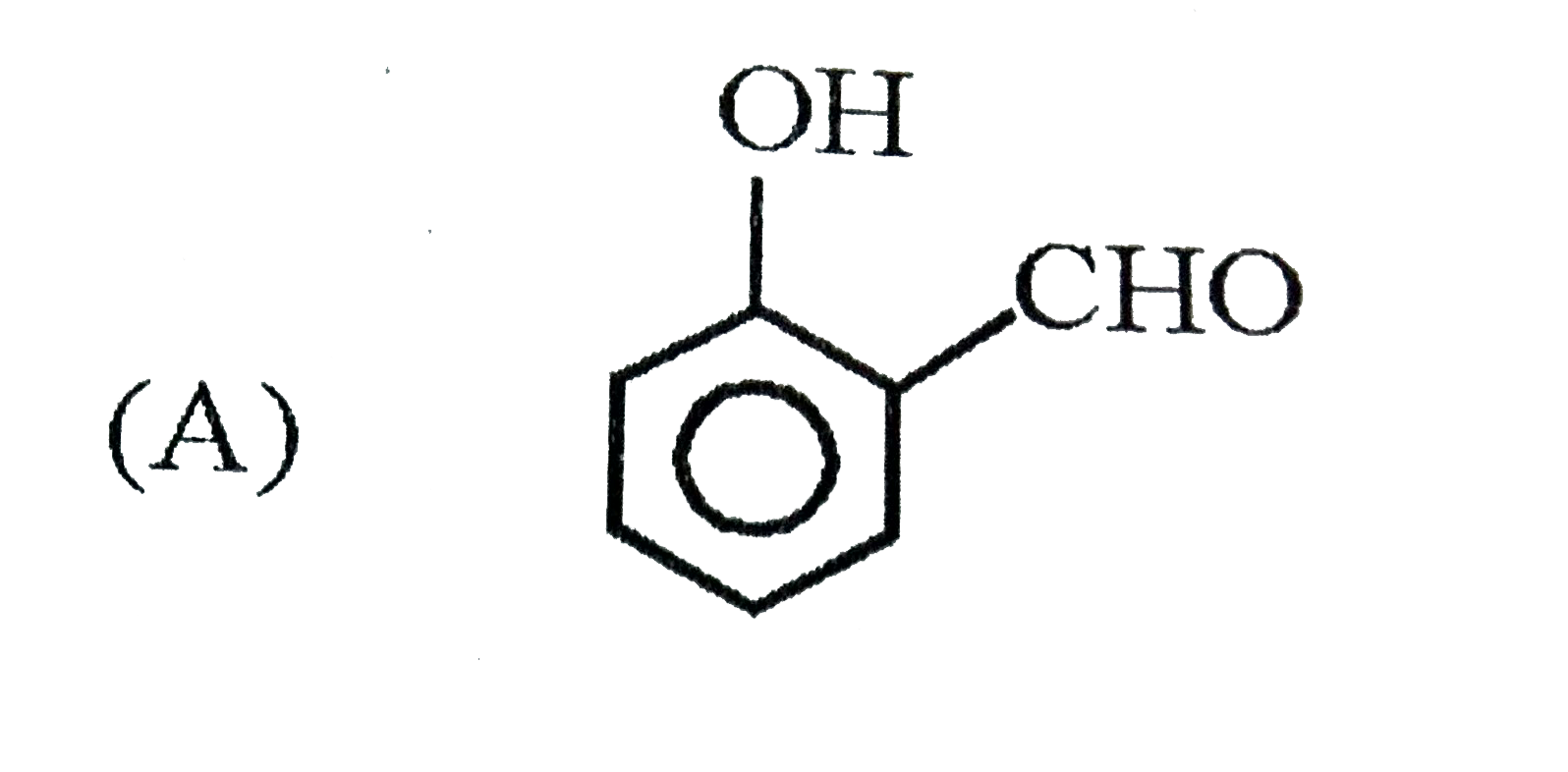
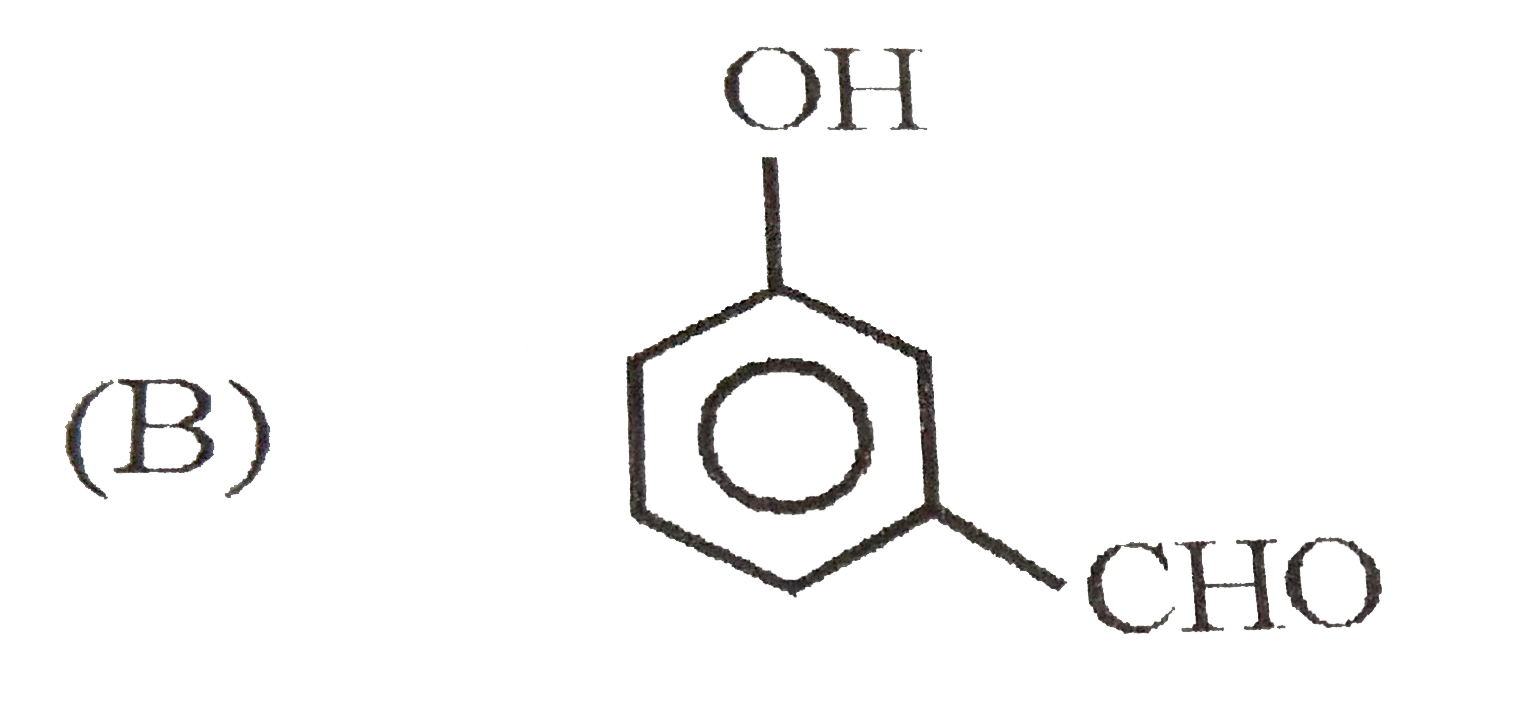
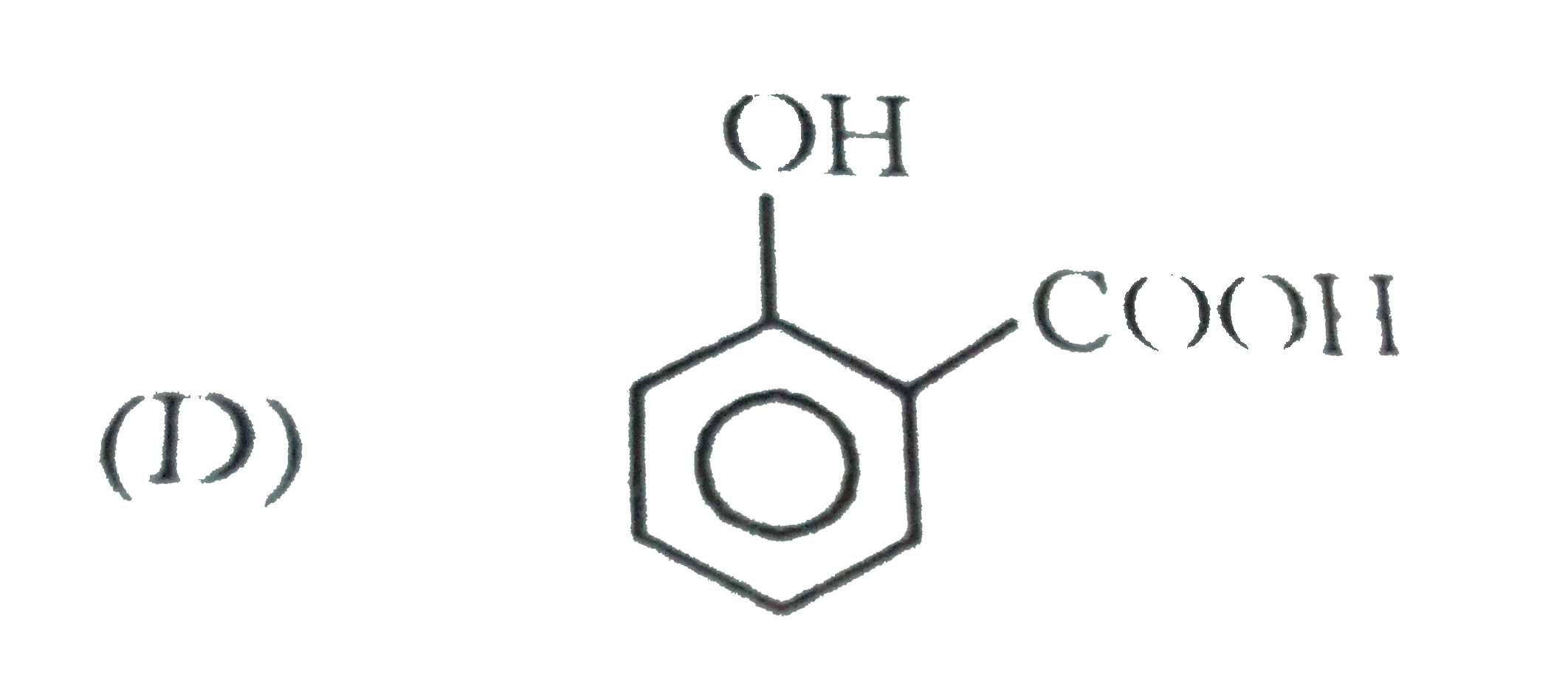
When 1-butyne undergoes oxymercuraction with the help of HgSO_(4)+H_(2)SO_(4), the product(s) formed is/are |
|
Answer» `CH_(3)CH_(2)COOH+HCOOH` 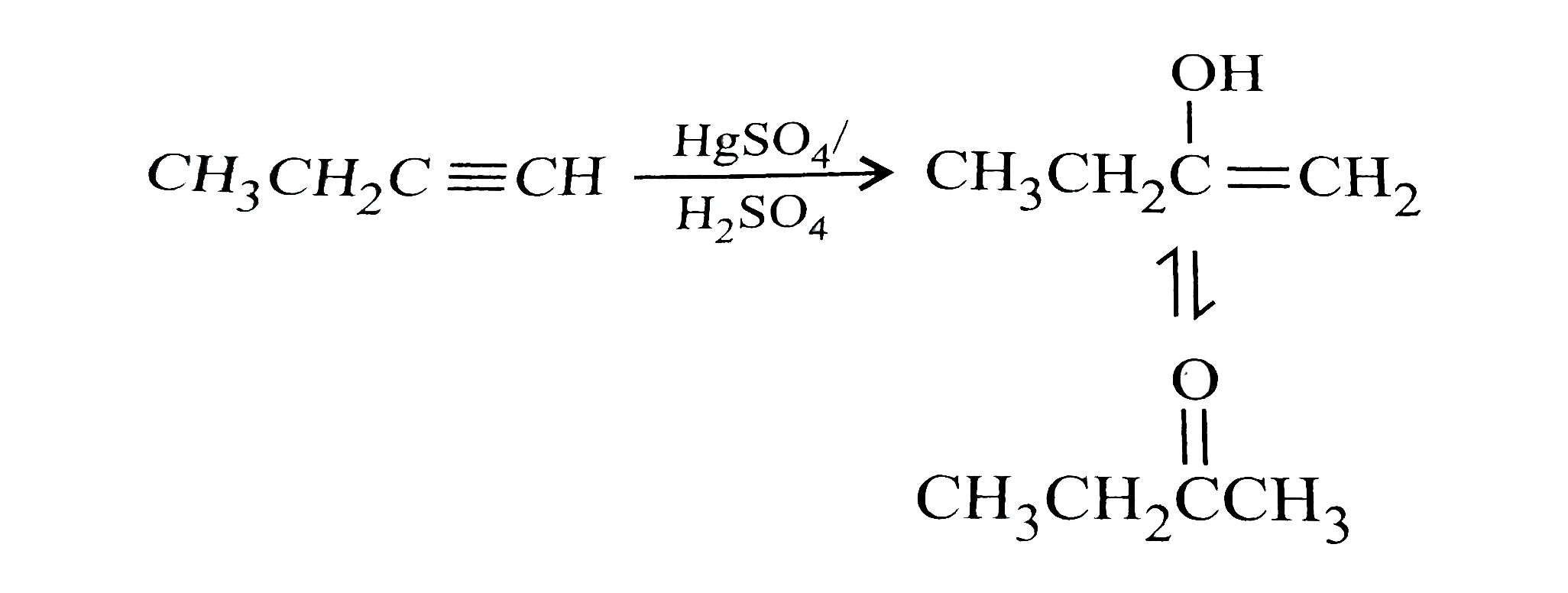
|
|
| 13. |
Which is wrong combination |
|
Answer» <P>`p_(X)-p_(x)` |
|
| 14. |
Which systematics diagram represents the correct chemical relations between sodium and its compound |
|
Answer»
|
|
| 15. |
Which of the following oxidation state do possess by group 14 elements ? |
|
Answer» `+4` |
|
| 16. |
Which of the following statement is /are correct |
|
Answer» Ionic compound are LESS soluble in `D_2O" than "H_2O` b) The temperature of maximum density of `H_2O` is less than that of `D_2O` c) Deuterium compound, can be prepared by exchange reaction where deuterium is EXCHANGES for hydrogen under suitable condiition. |
|
| 17. |
The variation of volume V, with temperature T, keep pressure constant is called the coefficient of thermal expansion ie alpha= (1)/(V) (del V)/(del T)_(P). For an ideal gas alpha is equal to …………. . |
|
Answer» T |
|
| 18. |
Which of the following is non-correct for substitution reaction. |
|
Answer» <P>(II)(iii)(R) |
|
| 19. |
Which of the following is the unit of equilibrium constant of given reaction N_(2(g))+ 3H_(2(g)) hArr 2NH_(3(g)) ? |
|
Answer» MOL `L^(-1)` |
|
| 20. |
When nitrogen and hydrogen combine to form ammonia, the ratio between the volumes of gaseous reactants and products is 1 : 3 : 2. Name the law of chemical combination illustrated here. |
| Answer» Solution :GAY - LUSSAC’ s LAW of Gaseous VOLUMES. | |
| 21. |
What does [H_(9)0_(4)] stands for ? Draw its structure . (b) Can sodium bicarbonate make water hard ? (c) Hard water is softened before make water hard ? (d) What is sequestration ? How is hard water made soft by sequestration ? |
|
Answer» Solution :(a) `H_(9)O_(4)^(+)` stands for hydrated proton . In `[H_(9)O_(4)]^(+) `, `H^(+)` is tetrahedrally surrounded by four water molecules as shown. (b) `NaHCO_(3)` cannot MAKE water hard because soaps themselves are sodium salts of fatty acids which are soluble in water. (c) Hard water on boiling gives precipitates of `MgCO_(3), CaCO_(3) and CaSO_(4)` which form scales in boilers . The formation of these boiler scales causes rapid deterioration of the boiler due to over heating . Further , these boiler scales are non-conducting . Therefore, more fuel is consumed . Therefore, in order to avoid formation of these scales, hard water is softened before using in boilers. (d) Sequestration means to render ineffective . The hardness of water is due to the presence of `Ca^(2+)` and `Mg^(2+)` ions. These ions are rendered ineffective by TREATING them with sodium hexametaphosphate , commercially called calgon. When calgo is added to hard water, the `Ca^(2+) and Mg^(2+)` ions PRESENT in it combine with sodium hexametaphosphate to form soluble complexes of calcium and magnesium salts . These complex calcium and magnesium ions do not form any precipitate with soap and hence READILY produce lather with soap solution. In this way, hard water is softened by sequestration. 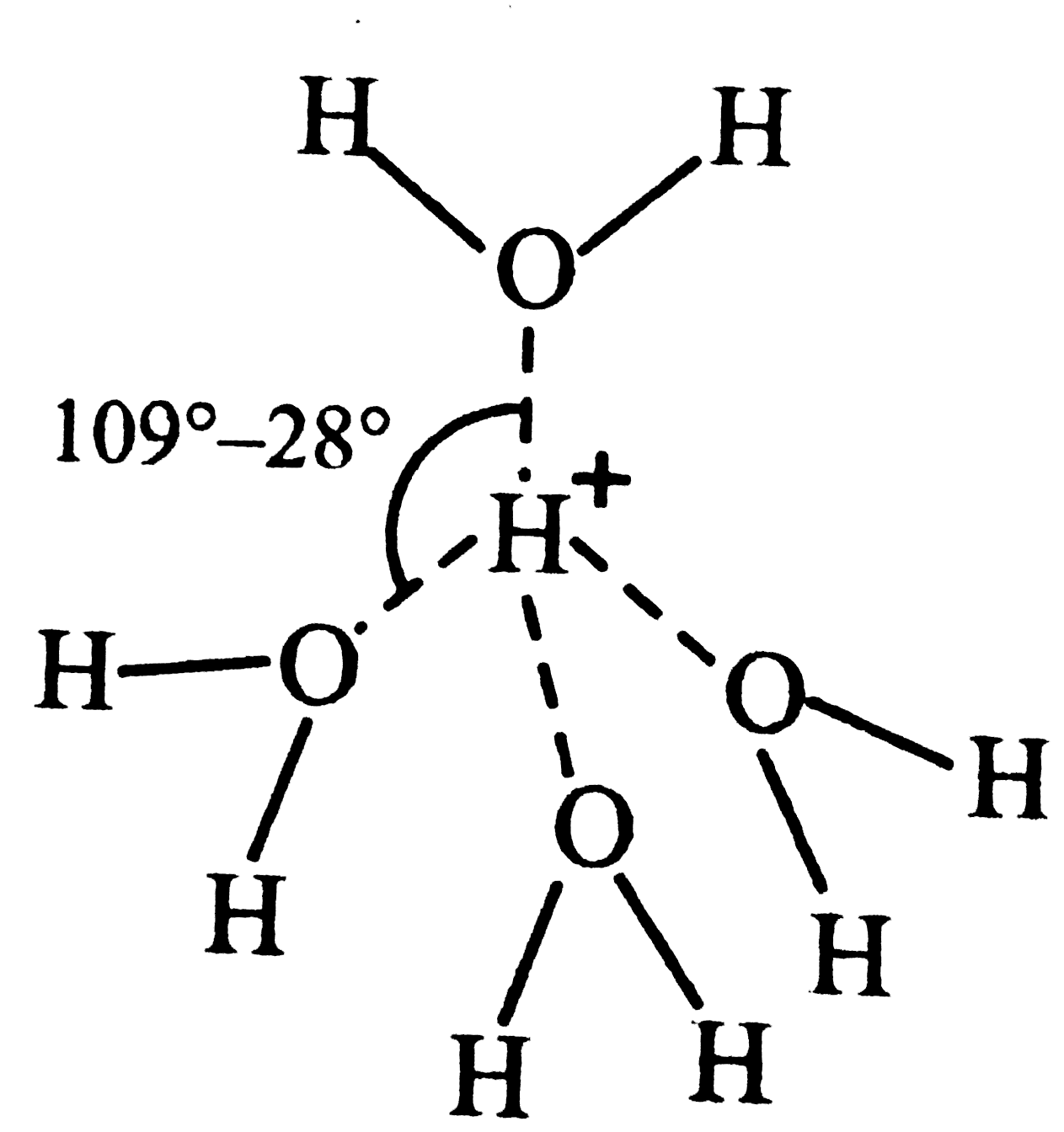
|
|
| 22. |
when electronstrikewith ZnSit shhowsfluorescentwhat isindirector ? |
| Answer» SOLUTION :Electronhasparticlenature . | |
| 23. |
What will be the volume strength of 100mL of KMnO_4 in acidic medium? (Given that 61mL of KMnO_4 reacts completely with 5mL of 1MK_(4) [Fe(CN)_(6))] where it converts into K^(+),Fe^(3+),CO_(3)^(2-) and NO_(3)^(-)) |
|
Answer» 17.31 V |
|
| 24. |
What is the major product obtained when phenol is treated with chloroform and aqueous alkali ? |
|
Answer»
|
|
| 25. |
The units of van der Waal's constant a and b respectively are |
|
Answer» `L^(-2)ATM^(-1)mol^(-1)" and" L mol^(-2)` `therefore`Units of `b=(V)/(n)=(L)/(mol)=mol^(-1)L` |
|
| 26. |
Which of the following substance is used as antiknock compound? |
|
Answer» TEL |
|
| 27. |
The solubility product of CaCl_2 in water is 4.2 xx 10^(-12). The concentration of Ca^(2+) in an aqueous solution of CaCl_2 is ......... M. |
|
Answer» `1.01xx10^(-4)` M |
|
| 28. |
The wave length of light having wave number 4000 cm^(-1) is |
|
Answer» `2.5mum` |
|
| 29. |
The systematic name of the organic compound having the structure |
|
Answer» 4 - ISOPROPYL HEXANE |
|
| 30. |
The sky looks blue due to |
|
Answer» dispersion effect |
|
| 31. |
Which of the following compounds is the most acidic ? |
|
Answer» `HCO_(2)H` |
|
| 32. |
The total number of cyclic isomers possible for the hydrocarbon with molecular formula C_(4)H_(6) is |
|
Answer» |
|
| 33. |
What are the oxidation numbers of 'N' in NH_(4)NO_(3)? |
|
Answer» `+3 -5` |
|
| 34. |
What are thevariousfactorsdue towhichthe ionizationenthalpyof the maingroupelementstendsto decrease down thegroup? |
|
Answer» SOLUTION :Within the maingroupelementsthe ionization enthalpy regularly as we moredown thegroup due to thefollowingtwo FACTORS. (i) Atomic size. On movingdownthe group the atomicsizeincreases graduallydue to theadditionofone newprincipal energyshellat eachsucceedingelement .As aresultthe distanceof thevalenceelectronsfrom thenucleusincreases . Consequently theforceof attraction of thenucleusof the valenceelectronsdecreases and hencethe ionizationenthalpydecreases. (ii) SCREENING effect. Withthe additionof newshells thenumberof innerelectronshellswhichshield the valenceelectronsincreases. In otherwordsthe shieldingeffect of thescreening effectincreases . Asa resultthe force of attractionof thenucleusfor thevalenceelectronsfurtherdecreasesand hence theionizationenthalpydecreases. |
|
| 35. |
What change is observed in real gas than ideal gas in pressure and volume ? |
| Answer» SOLUTION :Change in PRESSURE `= (an^(2))/(V^(2))`, change in VOLUME = NB | |
| 36. |
the vividh bharati station of All india Radio, Delhi , broadcasts on a frequency of 1,368 kHz (kilo hertz). Calculate the wavelength of the electromagnetic radiation emitted by transmitter . Which part of the electromagnetic spectrum does it belong to |
|
Answer» Solution :Wavelength, `lamda = (C)/(v)` Substituting `= c = 3 XX 10^(8) ms^(-1) and v = 1368 kHz` (Given) `= 1368 xx 10^(3) "CYCLES" s^(-1) "" (1 kHz = 10^(3) " cycles " s^(-1))` `= 1368 xx 10^(3) s^(-1)` We have, `lamda = (3 xx 10^(8) ms^(-1))/(1368 xx 10^(3) s^(-1)) = 219.3m` It belongs to the RADIOWAVE region of the electromagnetic spectrum. |
|
| 37. |
Write the lewis dot structure for (i) CO_(2) (ii) CH_(4) |
|
Answer» (II) `H:underset(H)underset(..)overset(H)overset(..)(C):H` |
|
| 38. |
What is valency |
| Answer» Solution :The valence of an element MAY be DEFINED as the combining capacity of element. | |
| 39. |
Turpentine oil can be purified by |
|
Answer» STEAM distillation |
|
| 40. |
The species H_2O, HCO_3^(-) , HSO_4^(-)and NH_3 can act both as Bronsted acids an bases. For each ase give the corresponding conjugate acid and base. |
Answer» SOLUTION :
|
|
| 41. |
What are the common oxidation states exhibited by the elements of group 14 and which state is more stable for Pb ? |
| Answer» SOLUTION :`+4` and +2 , +2 | |
| 42. |
The uncertainty in the position and velocity of a particile are 10^(-10)m and 5.27xx10^(-24)ms^(-1) respectively.Calculate the mass of the particle. |
|
Answer» Solution :Accoding to uncertainty pricople, `/_\x.m/_\v=h/(4PI)or /_\v=h/(4pim/_\x),h=6.626xx10^(-34)KGM^(2)kgm^(2)s^(-1)` `/_\x=10^(-10)m,/_\v=5.27xx10^(-24)ms^(-1)` `m=((6.626xx10^(-34)kgm^(2)s^(-1)))/(4xx3.143xx(10^(-10)m)XX(5.27xx10^(-24)ms^(-1))=0.1kg` |
|
| 43. |
Which of the following statement regarding spectral series is/are correct ? |
|
Answer» The lines in the Balmer series correspond to the electronic TRANSITION from the energy levels HIGHER than N = 1 energy LEVEL |
|
| 44. |
What is the basicity of H_(3)PO_(4) ? |
|
Answer» Solution :The structure of `H_(3)PO_(4)` is tetrahedral. It has three P-OH and one P=O bond as SHOWN below : Since it has three ionizable P-OH bonds, therefore, `H_(3)PO_(4)` is TRIBASIC as shown below : `H_(3)PO_(4)+H_(2)OhArrH_(3)O^(+)+H_(2)PO_(4)^(-),pK_(a_(1))=2.12" at "298K` `H_(2)PO_(4)^(-)+H_(2)OhArrH_(3)O^(+)+HPO_(4)^(2-),pK_(a_(2))=7.2" at "298K` `HPO_(4)^(2-)+H_(2)OhArrH_(3)O^(+)+PO_(4)^(3-),pK_(a_(3))=12.4" at "298K` Since it is tribasic, it forms three series of salts with NaOH, VIZ., `NaH_(2)PO_(4),Na_(2)HPO_(4)andNa_(3)PO_(4)`. 
|
|
| 45. |
Under isothermal conditions, a gas at 300 K expands from 0.1 L to 0.25 L against a constant external pressure of 2 bar.The work done by the gas is [ Given that 1 L bar = 100 J] |
|
Answer» Solution :`w = -p_(ext)(V_(2) - V_(1))` `p_(ext)` = 2 BAR, `V_(1) = 0.1 L, V_(2) = 0.25 L` w = -2(0.25 - 0.1) `= -2 xx 0.15 = -0.30` bar L ` = -0.30 xx 100 = -30` J |
|
| 46. |
The reluctance of valence shell s-electrons pair to get unpaired and to participate in bond formation is called ………………….. . |
| Answer» SOLUTION :INERT PAIR EFFECT | |
| 47. |
Which substance chars when heated with conc. H_2 SO_4? |
|
Answer» carbohydrate |
|
| 48. |
When acetyl chloride reacts with any amine, the reaction is known as : |
|
Answer» saponification |
|
| 49. |
What are the uses of Mendeleev periodic law ? |
| Answer» Solution :CLASSIFICATION of ELEMENTS and predication of NEW elements are the uses of this LAW. | |
| 50. |
Which of the following pair of gases will diffuse at the same rate through a porous plug ? |
| Answer» Answer :A::B::C | |