Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which type of Van der Waals attractive forces exists in a vessel filled with N_(2) molecules ? |
|
Answer» DISPERSION FORCES and DIPOLE - Dipole Forces |
|
| 2. |
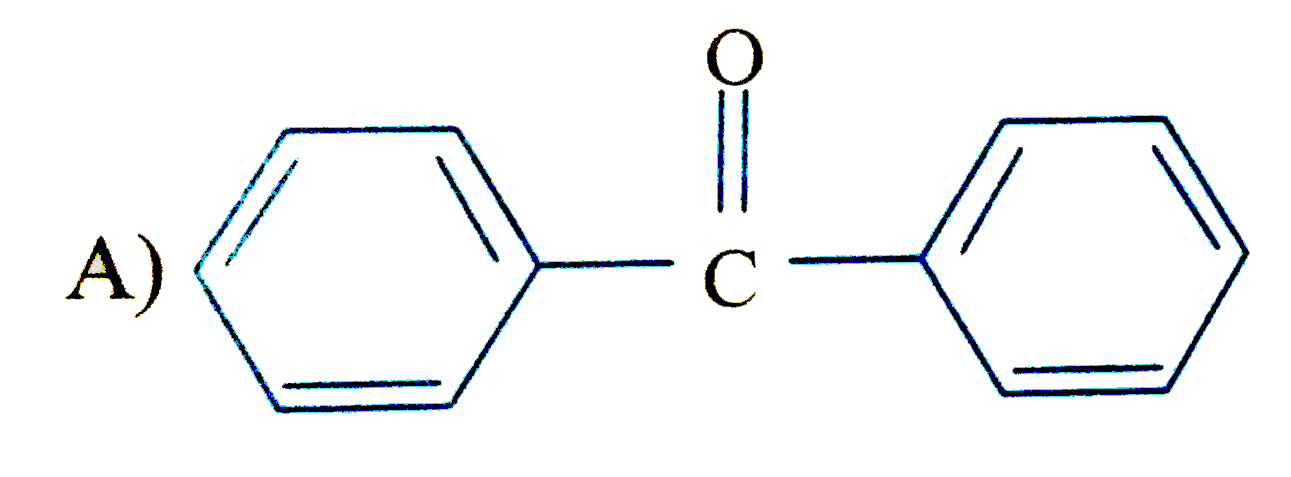
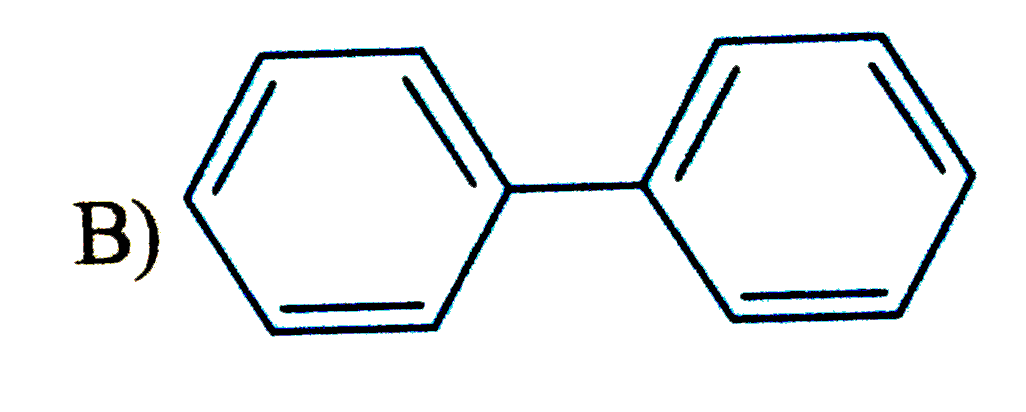
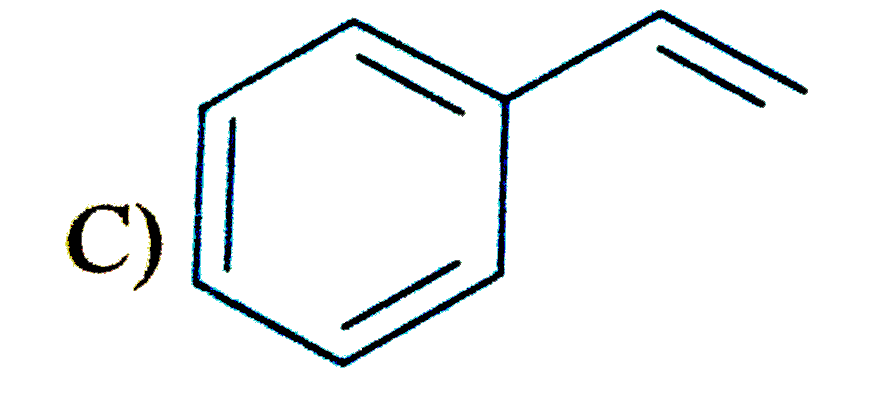
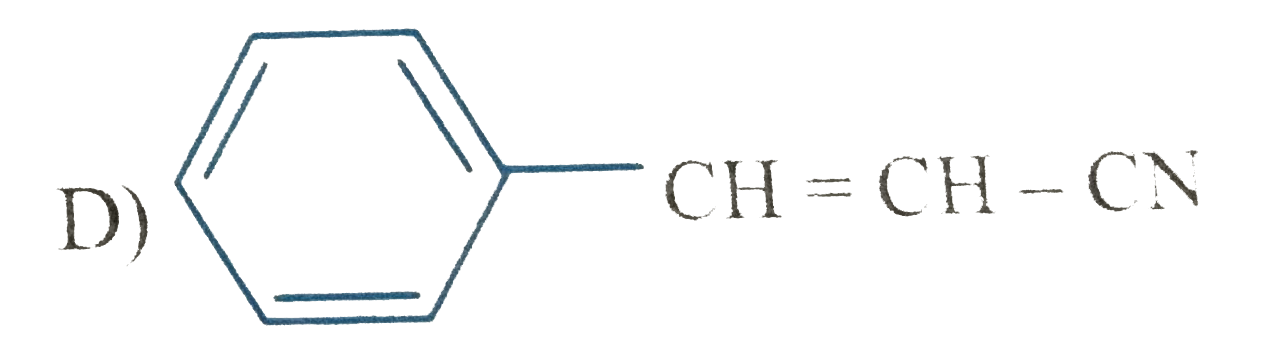
Which of the following compound is expected to undergo fastest electrophilic aromatic substitution than unsubstituted benzene |
|
Answer»
|
|
| 3. |
The solubility product of AgCl at a particular temperature is 1.08xx10^(-10)mol^(2)dm^(-6). Calculate its solubility in 0.01 M HCl. |
|
Answer» Solution :In the presence of a common ion with the concentration of c MOL `DM^(-3)`, the solubility S of the sparingly soluble salt is generally given by `(K_(sp))/c`. `AgClhArrunderset(S)(AG^(+))+underset(S)(Cl^(-))` `underset(0.01M)(HCl)tounderset(0.01M)(H^(+))+underset(0.01M)(Cl^(-))` Solubility of AGCL, `K_(sp)=[Ag^(+)][Cl^(-)]` `1.08xx10^(-10)=(S)(S+0.01)` Assuming S to be small when compared to 0.01 `S+0.01~~0.01` `therefore1.08xx10^(-10)=(S)(0.01)` Solubility (S) = `(1.08xx10^(-10))/0.01=1.8xx10^(-8)"mol "dm^(-3)`. |
|
| 4. |
What is the minimum volume of water required to dissolve 1 g of calcium sulphate at 298 K. K_(sp) for CaSO_(4) is 9.0xx10^(-6). |
|
Answer» 2.45 L `K_(sp)=[Ca^(2+)][SO_(4)^(2-)]` Suppose thesolubility of `CaSO_(4) = s "mol" L^(-1)` `K_(sp)=sxxs=s^(2)` `9.0xx10^(-6) = s^(2) or s=3xx10^(-3) "mol" L^(-1)` Molar MASS of `CaSO_(4) = 40 + 32 + 64 = 136 g "mol" ^(-1)` `:. 1g CaSO_(4) = (1)/(136) ` mol `3xx10^(-3)` mol of `CaSO_(4)` require minimum WATER to dissolve = 1 L `:. (1)/(136) ` molof `CaSO_(4)` will require water `=(1)/(3xx10^(-3))xx(1)/(136) ~=2.45 L` |
|
| 5. |
Which of the following is the correct form ula for one m ole of p h o to n energy ? |
|
Answer» `(Nh)/(LAMBDAC)` |
|
| 6. |
Which components are present in troposphere? |
| Answer» SOLUTION :TROPOSPHERE is made of turbulent, DUSTY zone containing AIR, much water vapour and clouds. | |
| 7. |
What is the atomic number of the element, having maximum number of unpaired 2p electrons? To which group does it belong? |
|
Answer» |
|
| 8. |
What is the change ininternal energy of a system, if 10 J of heat is supplied to it and 15 J of work is done by it? |
|
Answer» SOLUTION :SINCE it is a CLOSED SYSTEM. `Deltau=q-w` `Deltau=10J-15J` `Deltau=-5J` |
|
| 9. |
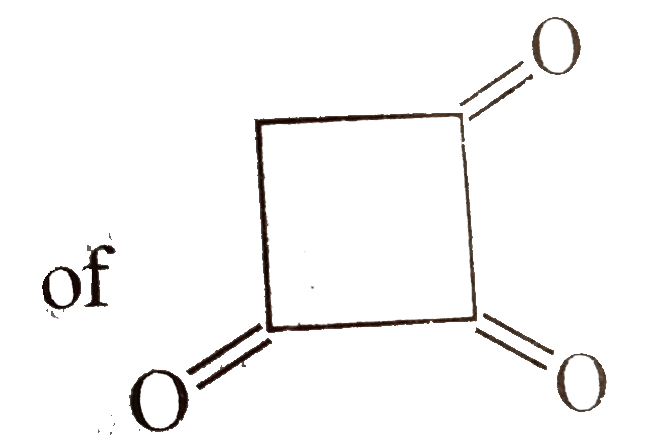
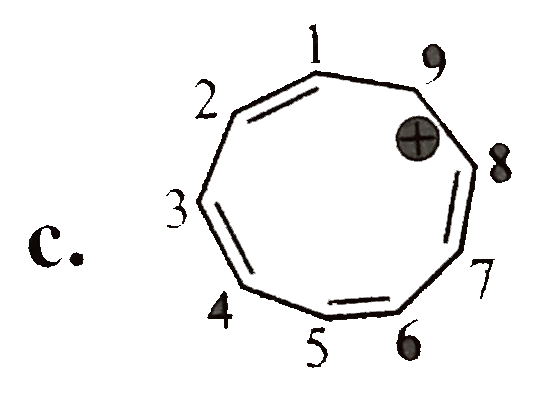
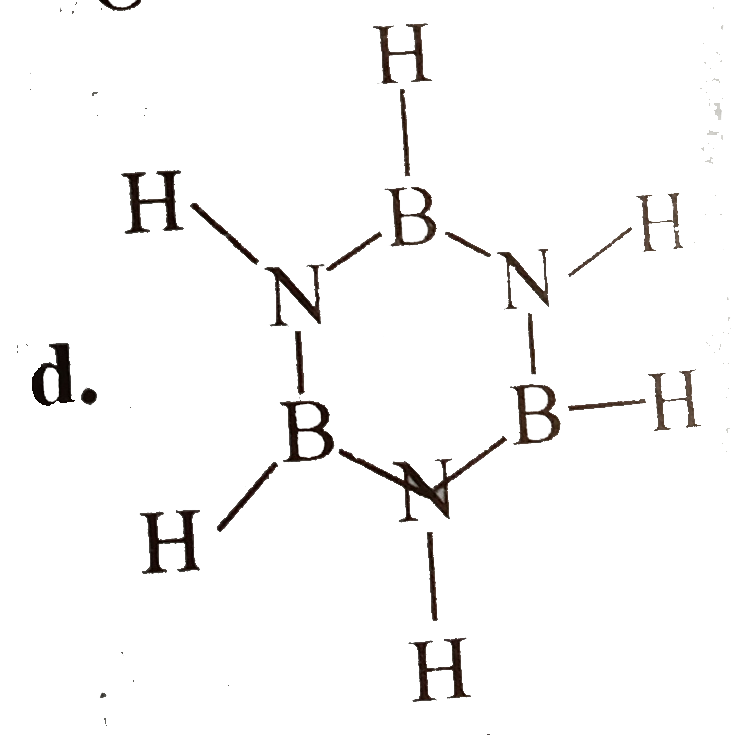
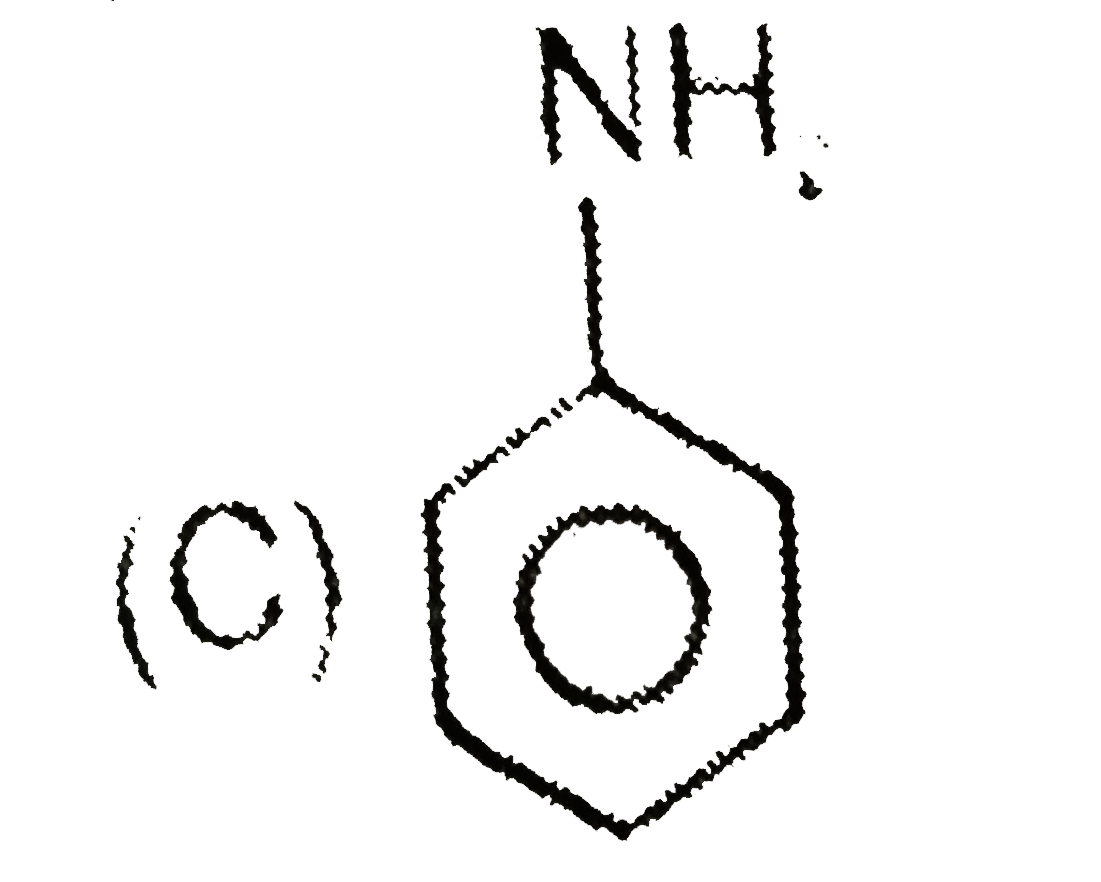
Which of the following is a non-aromatic compound ? |
|
Answer»
(B) is AROMATIC. (C) is anti-aromatic (d) is aromatic. |
|
| 10. |
Using Stock notation, represent the following compounds : HAuCl_(4), TI_(2)O, FeO, Fe_(2)O_(3), CuI, CuO, MnO and MnO_(2). |
| Answer» Solution :The OXIDATION number of the metallic ELEMENT Au in `HAuCl_(4)` is +3. Hence, the compound is REPRESENT as `HAU(III) Cl_(4).` | |
| 11. |
The threshold wavelength for the ejection of electron from metal is 330 nm, then work function for the photoelectric emission is |
|
Answer» `6XX10^(-12)` J `W=(hc)/lambda_(0)=(6.62xx10^(-34)xx3xx10^(8))/(330xx10^(-9))=6xx10^(-19)` J |
|
| 12. |
X and Y are: |
|
Answer» |
|
| 13. |
The virial equation for 1mole of a real gas is written as : PV=RT [1+(A)/(V)+(B)/(V^(2))+(C)/(V^(3))+ ........ To higher power of n] Where A,B, and C are known as virial coefficients . If Vander wall's equation is written in virial form, then what will be value of B : |
|
Answer» `a-(B)/(RT)` |
|
| 14. |
Which of the following is a compound? |
|
Answer» `CO_2` |
|
| 15. |
Write structureal formula of (a) Neopentane (b) Chlorobenzene (c) Propane. |
Answer» Solution :(a) `{:(""CH_(3)),("|"),(CH_(3)-C-CH_(3)),("|"),(""CH_(3)):}` (b) 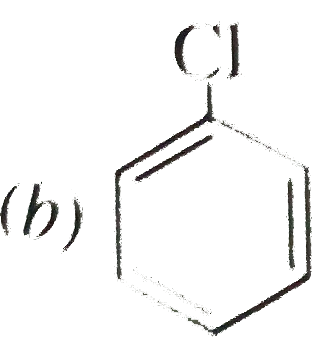 (c) `CH_(3)-CH_(2)-CH_(3)` (c) `CH_(3)-CH_(2)-CH_(3)`
|
|
| 16. |
Total number of lonw pairs and bond pairs of electrons present around xenon in XeF_(4) is |
Answer»  THUS , there are 4 BOND pairs and 2 LONE pairs presnet around XE. |
|
| 17. |
The solubility product of AgClin water is 1.5xx10^(-10). Calculate its solubility in 0.01 M NaCl aqueous solution . |
|
Answer» SOLUTION :As NaCl dissociates completely, therefore, in 0.01 M NaCl solution, `[Cl^(-)] = 0.01M` If SOLUBILITY of AgCl in 0.01 M NaCl solution is s MOL `L^(-1)`, then from AgCl that dissolves, `[Ag^(+)]=[Cl^(-)]=s ` mol `L^(-1) :. ` Total `[Cl^(-)]=0.01 + s ~= 0.01M` `K_(SP) ` for AgCl `=[Ag^(+)] [ Cl^(-)] = s xx 0.01 = 0.001 s :. 0.01 s = 1.5 xx 10^(-5) xx 10^(-10) or s= 1.5 xx 10^(-8) M` |
|
| 19. |
There are two constitutional isomers with the formula C_(2)H_(6)O . Write structural formulas of these isomers . |
Answer» Solution :If we RECALL that carbon form four covalent bonds oxygen can form TWO , and hydrogen only one , we can ARRIVE at the following constitutionalisomers .  It should be noted that these two isomers are clearly different in their physical PROPERTIES .At room temperature and 1 atm pressure ,dimethyl ether is a gas . Ethanol is a liquid . |
|
| 20. |
The organic substance which contains both the sp and sp^(3) hydridised carbon atoms is |
|
Answer» n-Butane |
|
| 21. |
What are X and Y respectively in the following reaction?Z-pro duct overset(Y)larr 2-butyn eoverset(X)toE - pro duct |
|
Answer» `Na//NH_(3(LIQ)) and Pd// BaSO_4 + H_2` 
|
|
| 22. |
Which of the following options does not represent ground state electronic configuration of an atom ? |
|
Answer» `Is^(2) 2s^(2) 2p^(6) 3s^(2) 3P^(6) 3d^(8) 4s^(2)` |
|
| 23. |
Which of the following statement is/are not correct? |
|
Answer» I.P.increases down the group |
|
| 24. |
Which of the following alloy is needed to make PbEt_(4) ? |
|
Answer» `Mg-Pb` |
|
| 25. |
Which one of the following ore dies not contain magnesium ? |
|
Answer» CARNALLITE |
|
| 26. |
The solubility of sulphates of alkaline earth metals in water shows the order |
|
Answer» `Be gt CA gt MG gt BA gt SR` |
|
| 27. |
Which of the following angle corresponds to sphydridisation ? |
|
Answer» `90^(@)` with each other. |
|
| 28. |
The wave number of the spectral line in the emission spectrum of hydrogen will be equal to 8/9 times the Rydberg's constant if the electron jumps from |
|
Answer» `N = 3 " to " n = 1` `(8)/(9) R_(H) = R_(H) ((1)/(n_(1)^(2)) - (1)/(n_(2)^(2))) or (1)/(n_(1)^(2)) - (1)/(n_(2)^(2)) = (8)/(9)` This is so only when `n_(1) = 1, n_(2) = 3` |
|
| 29. |
What is molar enthalpy of vaporization ? |
| Answer» SOLUTION :Amount of heat REQUIRED to vaporize ONE MOLE of a liquid at CONSTANT temperature and under standard pressure (1 bar) is called molar enthalpy of vaporization. | |
| 30. |
The substances used as an antichlor in bleaching is |
|
Answer» `Na_(2) S_(2)O_(3)` |
|
| 31. |
Whichtypeof spectraobtainedif electrontransists fromM - orbitto L-orbit ? |
|
Answer» ABSORPTION |
|
| 32. |
What is strong electrolyte ? Give examples. |
| Answer» Solution :A strong ELECTROLYTE is DEFINED as a substance which dissociates almost completely into ions in AQUEOUS solution and hence is a very good conductor of electricity. e.g. NaOH, KOH, HCI, `H_2SO_4`, NACL, `KNO_3`, etc. | |
| 33. |
What is co-ordinate covalent bond ? |
Answer» Solution :The type of covalent BOND in which electron pair require for bond formation is denoted by only one ATOM form the two atom join together. e.g. : In CO triple bond`(C epuiv O) ` EXIST in which one is co-ordinate covalent bond Both electrons are of oxygen in tripal bond between C and O. |
|
| 34. |
Which of the following will not give positive test with CHCl_(3)//KOH. |
|
Answer» `CH_(3)-CH_(2)-NH-CH_(3)` |
|
| 35. |
Write down the outermost electronc configuration of alkali metals. How will you justify their placement in group 1 of the periodic table? |
Answer»  Hence, placement of all the elements in group 1 of the periodic table because of similarity in electronic configuration and all the elements have similar properties. |
|
| 36. |
Which of the following is most effective in causing the coagulation of ferric hydroxide sol? |
| Answer» Answer :D | |
| 37. |
Which of the following statements is incorrect |
|
Answer» `Li^(+)` has minimum degree of hydration |
|
| 38. |
What is the product when C_(5)H_(5)CH_(2)NH_(2) reacts with HNO_(2)? |
| Answer» SOLUTION :`C_(6)H_(5)CH_(2)NH_(2) overset(HNO_(2))to C_(6)H_(5)CH_(2)OH + N_(2) uarr` | |
| 39. |
the solution of natural rubber in benzene is an example of : |
|
Answer» lyophobic acid |
|
| 40. |
Trimethylamine molecule is ............ in shape, while the shape of trisilylamine is ……………. . |
| Answer» SOLUTION :PYRAMIDAL , TRIGONAL PLANAR | |
| 41. |
whichof thefollowingpair(s) represents (s)the isoelerontsspecies ? |
|
Answer» `S^(2-) andSc^(3+)` |
|
| 42. |
What is |W|^(2)? What isindicate ? |
| Answer» SOLUTION :It givesprobabilityto findelectrons . It doeshavephysicalmeaningprobabilityto findelectron `ALPHA |W|^(2)` | |
| 43. |
Which one of the following is a correct set ? |
|
Answer» `H_2 O , sp^3`ANGULAR |
|
| 44. |
Which is true statement? |
|
Answer» the layer of ICE on the surface of river in the winter acts as a thermal insulator between the WATER below and the air above |
|
| 45. |
Which statement is wrong for sulphur ? |
|
Answer» `S_2` is a paramagnetic |
|
| 46. |
When a copper wire is placed in a solution of AgNO_(3), the solution acquires blue colour. This is due to |
|
Answer» OXIDATION of Cu |
|
| 47. |
The volume of HCI gas weighing 73 g at STP is |
|
Answer» 2.24 x 10-2m3 36.5 g of HCl occupies `2.24 xx 10^(-2) m^(3)` . `:.` 73 g of HCI at STP will occupy `(2.24xx10^(-2))/CANCEL(36.5) xx cancel(73^(2))=4.48xx10^(-2)m^(3)`. |
|
| 48. |
Which of the following is used as piezoelectric material ? |
| Answer» Answer :A | |
| 49. |
Which one the following is true ? |
|
Answer» LITHIUM on direct combination with NITROGEN from `Li_(3)N` |
|
| 50. |
What is responsible for the high melting and boiling points of water? |
|
Answer» HYDROGEN bonding |
|