Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following is a constituent of chewing gum ? |
|
Answer» `CaCO_(3)` |
|
| 2. |
Total number ofmolecular orbitals containing electrons present in O_(2)^(+) ion is |
|
Answer» |
|
| 3. |
Wavelengths of different radiations are given below : lamda (A) = 300nm, lamda (B) = 300mu m, lamda(C) = 3nm, lamda(D) = 30 Å Arrange these radiations in the increasing order of their energies. |
|
Answer» SOLUTION :`lamda (A) = 300NM = 300 xx 10^(-9) m = 3 xx 10^(-7) m, lamda (B) = 300 mu m = 300 xx 10^(-6) m = 3 xx 10^(-4) m lamda (C) = 3nm 3 xx 10^(-9)m, lamda (D) = 30 Å = 30 xx 10^(-10) m = 3 xx 10^(-9) m` `E = hv = h (c)/(lamda)`. THUS, `E prop (1)/(lamda)`. Hence, increasing order of energy is `B lt A lt C = D` |
|
| 4. |
What is wormery? |
| Answer» Solution :It is a special type of FUNCTIONAL isomerism in which single compound EXISTS in TWO readily interconvertible structures that differ markedly in the relative position of atleast ONE atomic hydrogen. The two different structures are known as TAUTOMERS. | |
| 5. |
Total number of 2^(o) carbon present in given compound is x, so the value of x-7 is |
|
Answer» 6 |
|
| 6. |
Which of the following metals are use in down cell as positive electrode and negative electrode respectively? |
|
Answer» GRAPHITE and steel |
|
| 7. |
What is the total number of orbitals associated with the principal quantum number n = 3? |
|
Answer» Solution :For n = 3, L = 0, 1, 2, i.e., there are three subshells DESIGNATED as 3s, 3p and 3d For 3s subshell, l = 0, `:.` m = 0 (i.e., one orbital), For 3p subshell, l = 1, `:.m = -1, 0 + 1` (i.e., 3 ORBITALS ) For 3d subshell l = 2, `m = -2, -1, 0 + 1, + 2` (i.e., 5 orbitals) `:.` TOTAL no. of orbitals PRESENT in the shell with n = 3 will be 1 + 3 + 5 = 9 Alternatively, no of orbitals presents in nth shell `= n^(2):.` No. of orbitals in the 3rd shell (n = 3) `= 3^(2) = 9` |
|
| 8. |
Which of the following statement is (are) correct in Bohr's if the mass of an electron because 10 timesits original mass ? |
|
Answer» VELOCITY of electron increases by `10 `TIMES |
|
| 9. |
The temperature at which the liquid and vapour phases are at equilibrium is called ………. |
| Answer» SOLUTION :CONDENSATION POINT | |
| 10. |
What causes hardness in water ? |
| Answer» Solution :WATER containing bicarbonates, sulphates and CHLORIDES of CALCIUM and MAG nesium is CALLED hard water. | |
| 11. |
What is covalent bond ? Explain by one example. |
|
Answer» Solution :Langmuir (1919) refined the Lewis postulations by abandoning the idea of the stationary cubical arrangement of the octet, and by introducing the term covalent bond. According to Lewis-Langmuir theory, atoms can combine by sharing of ELECTRONS between them. This type of bond is called covalent bond and each atom contributes equal number of electrons for sharing in order to acquire CONFIGURATION of nearest noble gas. Example : Covalent bond in dichlorine `(Cl_(2))` moecule. The Cl atom with electronic configuration [Ne]` 3s^(2)3p^(5)`, is one electron short of the argon noble gas. The FORMATION of the `Cl_(2)` MOLECULE can be understood in terms of the sharing of 9 pair of electrons between the two chlorine atoms, each chlorine atom contributing one electron to the shared pair.  The electron pair take part in octet of both d atom. So both chlorine atom get octet like Ar [Ne] `3s^(2) 3p^(6) `or [Ar] Note : .• and x. sysmbol used to REPRESENT electron of both chlorine atom. .-. used represent covalent bond between two Cl atom. |
|
| 12. |
Which of the following molecular orbitalshas maximum number of nodal planes ? |
|
Answer» `SIGMA^(**)1s` |
|
| 13. |
Which of the following is unacceptable resonating structure of buta-12,3-triene? |
|
Answer» `OVERSET(-)overset(•)(C)H_(2)-C-=C-overset(o+)(C)H_(2)` |
|
| 14. |
Using the equation of state pV=nRT, show that at a given temperature density of a gas is proportional to gas pressure p. |
|
Answer» Solution :According to EQ: 5.17, we have `M= (dRT)/P`or `d = M/(RT)*P` For a PARTICULAR gas, at CONSTANT TEMPERATURE, `M/(RT)` is constant `d prop P` i.e., at constant temperature, density of a gas is proportional to its pressure. |
|
| 15. |
When an alkali metal dissolves in liquid ammonia the solution can acquire different colours. Explain the reasons for this type of colour change. |
|
Answer» Solution : SOLUTIONS in liquid ammonia : The alkali metals dissolve in liquid ammonia giving deep blue solutions which are conducting in nature. `M + (x + y) NH_(3) to [M(NH_(3))_(x)]^(+) + [e(NH_(3))_(y)]^(-)` The blue COLOUR of the solution is due to the ammoniated electron which absorbs energy in the visible region of light and thus imparts blue colour to the solution. The solutions are paramagnetic and on standingslowly liberate hydrogen resulting in the formation of amide. `M_((am))^(+)+e^(-)+NH_(3(l)) to MNH_(2(am))+(1)/(2)H_(2(g))` (where .am. DENOTES solution in ammonia.) In concentrated solution, the blue colour changes to BRONZE colour and becomes diamagnetic. |
|
| 17. |
Whichof thefollowingspeciesdo NOTexist ? |
|
Answer» `BeF_(2)` |
|
| 18. |
What do you understand by the following statements ? enthalphy of formation of nitric oxide is +9.7 kJ. |
| Answer» Solution :When 1 MOLE of NO(g) is formed from its elements `N_(2)(g)` and `O_(2)(g)`, 90.7kJ of HEAT ABSORBED. | |
| 19. |
Which out of NaCl and CHCl_(3) reacts with AgNO_(3) solution to give a precipitate of AgCl ? |
| Answer» SOLUTION :NaCl (being an ELECTROVALENT compound, it GIVES `Cl^(-)`) | |
| 20. |
What is the action of ethene with bromine in carbon tetra chloride? |
| Answer» Solution :Ethene reacts with bromine in carbon tetrachloride to give 1,2 - dibromethane. Orange colour of bromine is discharged. `CH_(2)= CH_(2) + BR overset("CCl"_(4)"")(rarr)underset(Br)underset(|)(CH_(2)) -underset(Br)underset(|)(CH_(2)) ` | |
| 21. |
Which of the following alkaki metals is having the least melting point ? Na (b) K (c) Rb (d) Cs |
| Answer» SOLUTION :(d) Cesium (Cs) has the LEAST m.p. due to its MAXIMUM SIZE the LATTICE enthalpy. | |
| 22. |
Which of the following alkenes on ozonolysis gives a mixture of ketones only? |
|
Answer» `CH_(3)-CH=CH-CH_(3)` |
|
| 24. |
Which of the following reagents will distinguish between two fuctional isomers of formula C_(2)H_(4)O_(2) ? |
|
Answer» `NaHCO_(3)` |
|
| 25. |
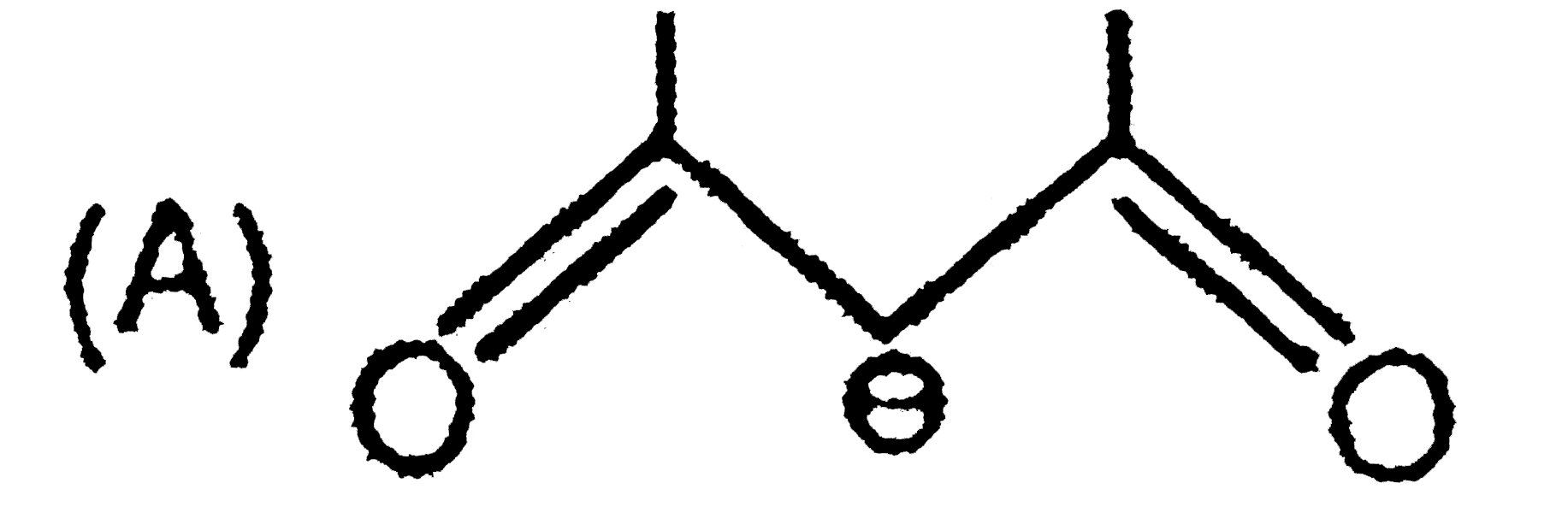
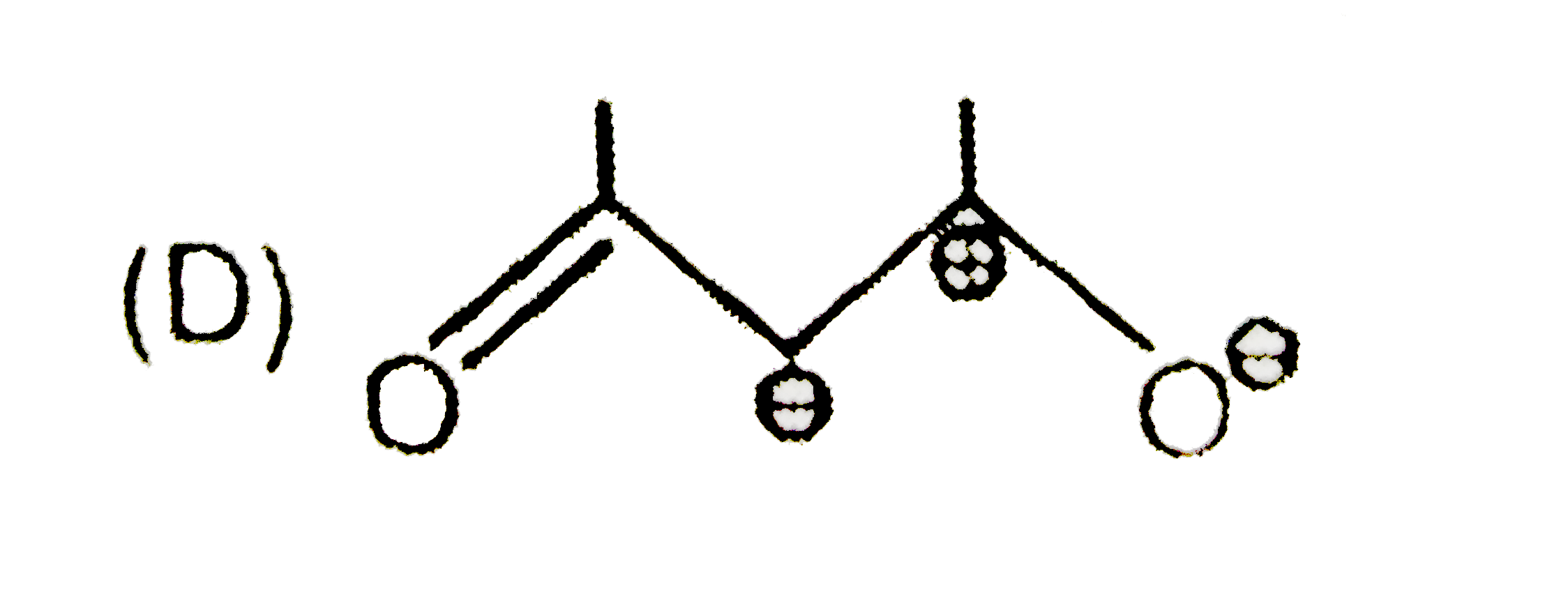
The skeletal structure of CH_(3)COOH as shown below is correct, but some of the bonds are shown incorrectly. Write the correct Lewis structure for acetic acid. |
Answer» SOLUTION :
|
|
| 27. |
Water decomposes by absorbing 286.2 kJ of electrical energy per mole. When H_(2) and O_(2) combine to form one mole of H_(2)O, 286.2kJ of heat is produced. Which law is proved ? What statement of the law followsfrom it? |
|
Answer» Solution :Law of conservation of energy ( or 1st law of thermodynamics) Def.Energy can neither be created or destroyed , ALTHOUGH it may be convertedfrom ONE formto another. |
|
| 28. |
When zinc pieces are added to concentrated NaOH solution, H_(2(g)) is formed along with |
|
Answer» `Na_3ZnO_2` |
|
| 29. |
Which of the following compounds will be optically active ? |
|
Answer» `(CH_(3))_(2) CHOH` |
|
| 30. |
What is the structural unit present in silicates ? |
| Answer» SOLUTION :`SiO_4^(-)` | |
| 31. |
What are the uses of solubility product ? |
|
Answer» Solution :(i) In CALCULATION SOLUBILITY of sparingly SOLUBLE salt. (ii) By mixing two solution `PP^+` will FORM or not can be predicted. |
|
| 32. |
The value of van der Waals constant 'a' for N_(2) and NH_(3) are 1.39 and 4.17 atm L^(2) mol^(-2) respectively. If these two gases have the same value of constant 'b' then under similar conditions : |
|
Answer» The PRESSURE exerted by `N_(2)` gas is more than that of `NH_(3)` |
|
| 33. |
Which of the following configurations represent the elemnet in the ground and which in the excited state ? Name the element in each case : (i) 1s^(2) 2s^(1) 2p^(1) (ii) 1s^(2) 2s^(2) 2p^(1) 2p^(1) (ii) 1s^(2) 2s^(2) 2p^(1) (iii) 1s^(2)2s^(1) 2p_(x)^(1) 2p_(y)^(1) 2p_(z)^(1) (iv) 1s^(2) 2s^(2) 2p^(6) 3s^(2) 3p_(x)^(1)3p_(y)^(1)3p_(z)^(1) 3d^(1) |
| Answer» Solution :(i) Be (II) B (iii) C (iv) S (All are in EXCITED state except (ii)) | |
| 34. |
Which of the following does not give benzoic acid on hydrolysis ? |
|
Answer» PHENYL cyanide |
|
| 35. |
which is not the correct statement for ionic solids in which positive and negtive lons are held by strong electrostatic attractive forces ? |
|
Answer» the RADIUS ratio `r_(+)//r_(-)`increases as COORDINATION number increases |
|
| 36. |
Which of the following statements is not correct regarding hydrogen: |
|
Answer» It resembles halogen in some of the properties |
|
| 37. |
This graph represents: |
|
Answer» FIRST ORDER reaction |
|
| 38. |
The second ionization enthalpy of which of the following alkaline earth metals is the highest ? |
| Answer» Solution :Due to small SIZE , Be has the HIGHEST 1st and 2 nd ionization energies amongst alkaline earth metals . | |
| 39. |
Which of the following molecule has no dative bond ? |
|
Answer» `CO` |
|
| 40. |
What is the type of hybridization in the following compounds (a) CH_(3)Cl,(b) (CH_(3))_(2)CO,(c) CH_(3)CN (d) HCONH_(2), (e) CH_(3)CH = CHCN |
|
Answer» SOLUTION :Write the partly condensed formulae of the above COMPOUNDS and indicate the type of hybridization on each carbon. (a) `overset(sp^(3))(CH_(3))-Cl`(B) `overset(sp^(3))(CH_(3))-UNDERSET(O)underset(||)overset(sp^(2))(C)-overset(sp^(3))(CH_(3))`(c) `overset(sp^(3))(CH_(3))-overset(sp)(C)-=N` (d) `H-underset(O)underset(||)overset(sp^(2))(C)-NH_(2)`(e) `overset(sp^(3))(CH_(3))-overset(sp^(2))(CH)=overset(sp^(2))(CH)-overset(sp)(C)-= N` |
|
| 42. |
What indicate if Q_(sp) ne K_(sp) ? |
|
Answer» SOLUTION :If `Q_(sp) ne K_(sp)` than the solubility of SPECIES is DIFFERENT than equilibrium concentration If `Q_(sp) ne K_(sp)` means precipitation take place and dissolution SALT take place. It indicates its direction. `Q_(sp) ge K_(sp)`precipitation take place `Q_(sp) lt K_(sp)` precipitation not take place |
|
| 43. |
What is the pecularity of group14 elements in respect of electronegativity? |
| Answer» SOLUTION :Except for carbon, ELECTRONEGATIVITY of REMAINING ELEMENTS is almost EQUAL. | |
| 44. |
Which one of the following is reduced with zinc and hydrochloric acid to give the corresponding hydrocarbon ? |
|
Answer» ETHYL acetate |
|
| 45. |
What is the volume of oxygen liberated at S.T.P When 3.4g of H_(2)O_(2) is heated? |
|
Answer» Solution :`2H_(2)O_(2)to2H_(2)O+O_(2)` `2"moles" ""1 "MOLE"` `2xx34 = 68gm`22.4lt as S.T.P From the blanced equation , we can SAY that 68g of `H_(2)O_(2)` on complete decomposition LIBERATES `(22.4)/(68)xx3.4 =1.12 LIT`. |
|
| 46. |
Which air pollutant gas reduces the rate of photosynthesis ? |
|
Answer» `SO_2` |
|
| 47. |
When a system is taken from state B to state A along path BDA as shown in figure below, 60 J of heat flows out of the system and 10J of work is doen on path ACB , then the heat corresponding to the processes AC and BC is respectively. |
|
Answer» `q_(AC)=-20J` & `q_(BC)=-50J` So, `""DeltaE_(BC)=q_(ac)+W_(ac)` `-30=q_(BC)+(20)` `q_(BC)=-50J` Now, `""q_(AB)+q_(AC)+q_(CB)` `70=q_(AC)+50(` since for path `BDA,E_(A)-E_(B)+Q+W=-60+10=-50` J and `W_(ACB)=-20J,` So, `q_(AB)=DeltaE_(AB)-W_(ACB)=50+20=70J)` `q_(AC)=20J` |
|
| 49. |
What is the difference between 2-butyne and 1-butyne ? By which test we can distinguish between these two ? |
|
Answer» SOLUTION :(a) 1-butyne and 2-butyne `(C_(4)H_(6))` are POSITION isomers of each other. `underset("1-butyne")(CH_(3)CH_(2)-C-=CH)""underset("2-butyne")(CH_(3)C-=C CH_(3))` (b) In 1-butyne have acidic HYDROGEN which ATTACH with carbon having triple bond. So it react with `NaNH_(2), AgNO_(3)//(NH_(4)OH)` and `CuSO_(4)//(NH_(4)OH)`, while in 2-butyne acidic hydrogen is absent so it does not give such acidic reaction of hydrogen. |
|
| 50. |
Which of the following statement(s) is/are not true about the following decomposition reaction ? 2KClO_(3)to2KCl+3O_(2) |
|
Answer» POTASSIUM is undergoing OXIDATION. (A) Oxidation number of K is not change. (B) Cl form reduction. ( C) Oxygen formed oxidation reaction. (D) Oxygen and Chlorine forms oxidation and reduction REACTIONS. |
|