Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What are diodes and transistors ? For what purpose are they generally used ? |
| Answer» SOLUTION :A diode is a combination of p- and n- type SEMICONDUCTORS which is used as a RECTIFIER. TRANSISTORS are sandwichsemiconductors of the type pnp or npn which are used to detect or amplify radio or AUDIO signals. | |
| 2. |
Write the resonance structure of CO_(3)^(2-) |
Answer» SOLUTION :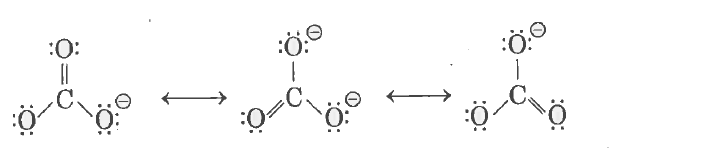
|
|
| 3. |
The rms speed of the molecules of a gas of density 4 kg m^(-3) and pressure 1.2xx10^(5) N m^(-2) is: |
|
Answer» `120m s^(-1)` |
|
| 4. |
Writethe mechanism of chlorinaltinof benezene |
|
Answer» Solution :Step: 1 Generationof `Ci^(@)` ELECTROPHILE `AICI_(3)+ CI_(3) to CI^(@) + AiCI_(4)` Step :2attackof theelectrophilicon the BENZENERING toformareniumion  Step :3rearomatisation ofarenium ion 
|
|
| 5. |
Why are the electron affinities of alkaline earth metals nearly zero? |
| Answer» Solution :Alkaline earth metals have stable `ns^2` configuration & are reluctant to ACCEPT electrons. Thus, their ELECTRON affinity values are nearly zero. These are metallic by nature and highly electropositive also, that is, they as it is have a poor TENDENCY to accept electrons. The negative ANIONS of group-2 metals are not known. | |
| 6. |
What is the difference between unit of K_p and K_c of any one reaction equilibrium ? |
|
Answer» SOLUTION :The value of `K_p` is bar or `P_a` or ATM whereas value of `K_c` is M or MOL `L^(-1)` . `K_p` is only for GAS phase whereas `K_c` for gas phase as well as solution also. |
|
| 7. |
Whichtwodevelopmentremovethe drawbacksof Bohr.smodel ? |
|
Answer» Solution :(i) Dualbehaviour of MATTER (DE - Brodliethory) (ii)HEISENBERG uncertaintyprincipletheory |
|
| 8. |
Which of the following is the most basic oxide ? |
|
Answer» `SeO_(2)` |
|
| 10. |
Which of the following has largest size |
|
Answer» Al |
|
| 11. |
Which of the following is incorrect for d-subshell (or) orbital |
|
Answer» d-orbital CONTAINS maximum of two electrons with opposite spins |
|
| 12. |
What is the difference in Mo energy level in Li_(2) to N_(2) and O_(2)to Ne_(2) ? |
| Answer» Solution :In `Li_(2) " to " N_(2) sigma_(2p_(z)) `has more energy than `pi` MO While in `O_(2) , F_(2) , Ne_(2). Sigma_(2p_(z)) ` has LESS energy than `pi` MO. | |
| 13. |
What is Matter ? |
|
Answer» All substances |
|
| 14. |
The RMS speed of heliu, in ms^(-1) (atomic mass= 4.0 gmol^(-1)) at 400 K is |
|
Answer» 16.8 |
|
| 15. |
Which of the following statements is/are correct . |
|
Answer» `TL^(3+)` salts are oxidising AGENTS |
|
| 16. |
Write the formula of plaster of pairs. |
| Answer» SOLUTION :`CaSO_(4). (1)/(4) H_(2) O` or `2CaSO_(4). H_(2) O` | |
| 17. |
whichof the following shows the maximum hydrophobic begaviour ? |
|
Answer» adenine |
|
| 18. |
Write the structure of the following compounds: (i) 1-Bromo-4-ethylcyclo hexane (ii) 1,4-Dichlorobut-2-ene (iii) 2 Chloro-3- methyl pentane |
Answer» SOLUTION :
|
|
| 19. |
Which lithium halide is soluble in ethanol, acetone, ethylacetate as well as pyridine? |
|
Answer» LiF |
|
| 20. |
Which of the following indicators is best suited in the titration of a weak acid versus a strong base?(PK_(10 ) values are given in bracket) |
|
Answer» PHENOLPHTHALEIN (8,3-10.0) ` rArr ` PH at neutralization ` gt 7` ` therefore ` phenolphthalein |
|
| 21. |
Which of the following order of acidic strengths is incorrect ? |
|
Answer» `H_(3)PO_(4) LT HNO_(3)` |
|
| 22. |
When CaO is heated in an oxy-hydrogen flame, |
|
Answer» it DECOMPOSES to give oxygen |
|
| 23. |
What isblacksubstance ? |
| Answer» SOLUTION :Idealblacksubstancemeanswhichabsorband emitradiationwith allfrequency . | |
| 24. |
Why benzene undergoes electrophillic substitution reaction where as alpenses undergoes addition reaction? |
|
Answer» SOLUTION :`(i)` BENZENE posses an in hybridised `p`- orbital containing one electron. The latral overlap of their `p`-ORBITALS produces `3pi`-bond `(ii)` The six-electron of the `p`-orbitals over all the six carbon atoms and are said to be delocated. `(iii)` Due to delocalisation strong `pi`-bond is formed which makes the molecule STABLE. `:.` benzene undergoes electrophilic substiution reaction. |
|
| 25. |
Which type of forces are present in given molecules ? |
|
Answer» Solution :`HF - HF, O_(2)-O_(2), CH_(4)-CH_(4), HF-H_(2), CO, NO, H_(2)O-H_(2)O` London force : `H_(2)-H_(2), O_(2)-O_(2),CH_(4)-CH_(4)` Dipole - Dipole force : `HF - HF, CO, NO, H_(2)O` Dipole - Dipole force (H - bond) : `HF - HF, H_(2)O-H_(2)O` Dipole - INDUCE Dipole FORCES : `HF-H_(2)` |
|
| 26. |
Write following conversions. 1 km = ...........nm= ...................dm 1 ml = ............. L = .............dm^(3) |
|
Answer» `1ml=0.001 L = 0.001dm^(3)` |
|
| 27. |
Which of the following are correct statements a) 760 torr is equal to 1 atmosphere b) 10^6 "dynes/cm"^2 is called 1 Bar c) 10^5 "Newtons/m"^2 is pascal d) 1 atmosphere is 1.013 xx 10^5 "dynes/m"^2 |
|
Answer» a,c |
|
| 28. |
What is the equilibrium concentration of each of the substances in the equilibrium when the intial concentration of ICI was 0*78 M ? |
|
Answer» Solution :Suppose at EQUILIBRIUM , `[I_(2)] = [Cl_(2)] = x "mol" L^(-1) .`Then `{:(,2I Cl ,hArr,I_(2)(g),+,Cl_(2)(g)), ("Intial conc." ,0*78 "M",,0,,0), ("At eqm.",0*78 - 2 x,,x,,x):}` ` K_(c) = ([I_(2)][Cl_(2)])/([ICl]^(2)) :. 0*14 = (x xx x )/(0*78 - 2X)^(2) ` or ` x^(2) = 0* 14 (0* 78 - 2x)^(2) or x/(0*78-2x) = sqrt(0*14) = 0*374 ` or `x = 0*292 - 0*748or 1*748 x = 0* 291 or x = 0* 167` Hence, at equilibrium , ` [I_(2) ] = [Cl_(2)] = 0*167 "M", [ICl ] =0* 78 - 2 xx 0* 167 "M" = 0*446 "M"` |
|
| 29. |
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group-13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also. TlCl_3 , TlCl |
| Answer» SOLUTION :TlCl is more stable than `TlCl_3` because moving down the GROUP lower oxidation state is more stable due to INERT PAIR EFFECT. | |
| 30. |
Write the decreasing order of (a) carbon-carbon and (b) carbon- hydrogen bond lengths in ethane, ethylene and acetylene molecules. |
|
Answer» Solution :Carbon carbon bond LENGTHS are: `C_(2)H_(6)gtH_(2)H_(4)gtH_(2)H_(2)` `1.54"pm"""134"pm"""120"pm"` Carbon hydrogen bond lengths are: `C_(2)H_(6)gtC_(2)H_(4)gtC_(2)H_(4)` `110"pm"""109"pm"""106"pm"` |
|
| 31. |
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group-13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also. AlCl_3 , AlCl |
| Answer» Solution :`AlCl_3`is more STABLE because it does not show inert pair effect. It is a COVALENT compound and acts as a LEWIS ACID | |
| 32. |
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group-13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also. InCl_3 , InCl |
|
Answer» Solution :InCl is more stable due to inert PAIR EFFECT and lower oxidation STATE +1 is more stable. In SHOWS both the oxidation states +3 and +1. `3InCl_((aq)) to 2In_((s)) + In_((aq))^(3+) + 3Cl_((aq))^(-)` |
|
| 33. |
Write the important conditions required for the linear combination of atomic orbitals to form molecular orbitals. |
|
Answer» Solution :(i) The combining ATOMIC orbitals should have comparable energies. For example, ls orbital of one atom can combine with ls atomic orbital of another atom, 2s orbital can combine with 2s orbital and so on (ii) The combining atomic orbitals must have proper orientations so that they are able to OVERLAP to a CONSIDERABLE extent. (III) The extent of overlapping should be large. |
|
| 34. |
Calcium nitrate decomposes on heating according to the equation : 2Ca(NO_3)_(2)to2CaO+4NO_(2)+O_(2) The relative molecular mass of calcium nitrate is 164. Calculate : the weight of calcium oxide obtained when 16.4 g of calcium nitrate is heated to constant weight. (Ca = 40, O = 16, N = 14) |
| Answer» SOLUTION :`2Ca(NO_3)_2 (s) OVERSET(Delta)to 2CaO(s) + 4NO_2(G) + O_2(g)` | |
| 35. |
What would have been the shape of benzene molecule had there been no resonance ? |
Answer» SOLUTION :Due to resonance, delocalization of SIX `PI`-electrons takes place. As a result, all the carbon-carbon bonds in benzene are equal (139 pm) and the molecule is SYMMETRICAL. Had there been no resonance, benzene would be an unsymmetrical molecule consisting of three carbon-carbon single bonds (154 pm) and three carbon-carbon double bonds (134 pm) as SHOWN ? 
|
|
| 36. |
Which is correct for any kind of species? |
|
Answer» `(E_(2)-E_(1))GT (E_(3)-E_(2)) gt (E_(4)-E_(3))` |
|
| 37. |
When the ratio of the radius of cation and anion in the crystal lattice is 0.53, the co-ordination number in the lattice is |
| Answer» ANSWER :B | |
| 38. |
What will be the value of Delta G^(@). If equilibrium constant for a reaction is 10. |
|
Answer» `-50.44 kJ MOL^(-1)` |
|
| 39. |
Which one of the following will have largest number of atoms?(i) 1 g of Au (s) (ii) 1 g of Na (s) (iii) 1 g of Li (s) (iv)1g Cl_(2) (g) |
|
Answer» Solution :(i) Molar mass of Au = 197 g `mol^(-1)` No. of atoms in 1 g of Au = `1/197 XX 6.023 xx 10^(23)` (II) Molar mass of NA = 23 g `mol^(-1)` No. of atoms in 1 g of Na = `1/23 xx 6.023 xx 10^(23)` (iii) Molar mass of Li = 7 g `mol^(-1)` No. of atoms in 1 g of Li =` 1/7 xx 6.023 X 10^(23)` (iv) Molar mass of CL, = 71 g `mol^(-1)` No. of atoms in 1 g of `Cl_(2)= 1/71 xx 6.023 x 10^(23)` Comparing the number of atoms, the LARGEST number of atoms will be present in 1 g of Li. Since the mass is same in each case, the element with the lowest molar mass would have the largest number of atoms. `:.` Li with lowest molar mass would have the largest number of atoms. |
|
| 40. |
What is the crystal structure of cesium chloride ? |
|
Answer» Body CENTERED cubic |
|
| 41. |
What is the nitrogen-oxygen bond order in NO_(3)^(-) ion? |
|
Answer» SOLUTION :`NO_(3)^(-)` The structure of `NO_(3)^(-)` ion is : `[O=underset(O)underset(darr)(N)-O]^(-)` it has four bonds and three CANONICAL STRUCTURES. BOND order -1.33. |
|
| 42. |
Which of the following is the uppermost region of the atmosphere ? |
|
Answer» Stratosphere |
|
| 43. |
Thermodynamics is applicable to____ system only |
| Answer» SOLUTION :MACROSCOPIC | |
| 44. |
What is the general electronic configuration of p - block elements ? |
| Answer» SOLUTION :[ NOBLE gas ] `NS^(2) np^(1-6)` | |
| 45. |
Which among the following equilibrium, K_(P) does not depend upon the initial pressure of ractants? |
|
Answer» `H_2(G)+I_2(s)hArr2HI(g)` |
|
| 46. |
Which of the following pair contains 2 lone pairs of electrons on the central atom? |
|
Answer» `I_(2), H_(2)O` |
|
| 48. |
When slaked lime is treated with chlorine, the product obtained is(a)CaCl_2(b)Ca(ClO)_2(c)Ca(ClO_3)_2(d) CaOCl_2 |
|
Answer» `CaCl_2` |
|
| 49. |
Which of the following is not a conjugate acid base pair? |
|
Answer» `HPO_4^(2-), PO_4^(3-)` |
|



